Abstract
Background: Cluster headache (CH) is the most painful and debilitating primary headache syndrome. Conventional treatment combines acute and prophylactic drugs. Also with maximal therapy a substantial proportion of patients do not experience a meaningful prevention or pain relief. Recent case series and early trials have suggested that occipital nerve stimulation can be very effective in the management of intractable CH.
Methods: Seven patients with medically intractable chronic cluster headache were implanted with high cervical epidural electrodes. After a median test phase of 10 days (range 4–19 days) an impulse generator was implanted subcutaneously. Mean follow up was 23 months (median 12 months, range 3–78 months).
Results: All patients showed significant treatment effects. In all patients, improvement occurred immediately after electrode implantation. The mean attack frequency decreased, as well as the mean duration and intensity of attacks. Also, depression, anxiety, and pain-related impairment scores decreased and medication intake was markedly reduced.
Conclusions: In this prospective series, high cervical spinal cord stimulation shows an effect size equal or larger than occipital nerve stimulation with immediate onset after surgery and may serve as a valuable additional treatment option of intractable cluster headache in the future.
Introduction
Cluster headache (CH) is an extremely severe and debilitating trigeminoautonomic pain syndrome. Clinically it is characterized by attacks of severe pain localized orbitally, suprorbitally, or temporally, lasting for 15–180 min and occurring from once every other day to eight times a day. The attacks are associated with one or more of the following: conjunctival injection, lacrimation, nasal congestion, rhinorrhoea, forehead and facial sweating, miosis, ptosis, and eyelid oedema (1). Attacks occur in series (cluster) that are usually separated by remission periods lasting for months or years.
In chronic cluster headache (cCH) no remissions occur. CH has a 1-year prevalence of 0.1% (2), and about 10% of this group have cCH (3). Conventional medical treatment comprises acute, intermediate, and prophylactic drugs. Also with maximal therapy a substantial proportion of patients do not experience a meaningful relief. Occipital nerve stimulation (ONS) has been used in migraine and hemicrania continua (4,5). Recent case series have suggested that ONS (6–8) can also be effective in the management of intractable cCH. After the encouraging outcome in a single patient with high cervical spinal cord stimulation (hcSCS) as a compassionate treatment for medically intractable cCH (9), we decided to offer hcSCS to further patients with refractory cCH. We applied recently proposed operational criteria to define pharmacologically intractable headache (10).
Materials and methods
Selection of patients
Patients with medically intractable cCH were recruited from the outpatient department of the interdisciplinary pain centre. Patients were referred from pain therapists and neurologists in southern Germany and were treated for several months before considering neuromodulation. Patients were thoroughly informed about the disease and available treatment options including neuromodulation, in particular ONS and hcSCS. All patients opted for hcSCS.
Patients’ demographics

F, female; M, male. aValues for first electrode implantation (bilateral electrodes).
Ethics committee approval
The Hospital Trust ethics committee agreed to offer hcSCS implants as compassionate treatment and to collect clinical data in these patients prospectively.
Operative technique
All patients gave written consent to the operations and (with exception of the first patient) to the participation in the prospective follow up. Implantation of the hcSCS electrode followed standard procedure for high cervical epidural neurostimulation. After local anaesthesia (10 ml scandicaine 1%) a 14 gauge Touhy needle was inserted at level Th 2/3. A saline probe with loss of resistance and fluoroscopic guidance in two planes ensured intraspinal placement of the needle tip. A 33 cm quadripolar electrode with 30 mm electrode distance, 6 mm electrode spacing, and 1.3 mm diameter (patients 1 and 2) or an eight pole electrode with 6 mm electrode spacing and 1.3 mm diameter (patients 3–7) were used. The electrode was slowly directed cranially to the upper cervical region until further movement was stopped by the inferior margin of the occipital bone or by the posterior arch of atlas. Final lead position was slightly paramedian (Figure 1). A test stimulation was performed in the operating room which induced ipsilateral paraesthesia covering the neck, the occipital, parietal and frontal skull, and the face (encompassing the sensory supply of the root C2 and trigeminal division V1 and to a lesser extent V2). In all patients, first electrode implantation was ipsilateral to the unilateral cCH. In two patients (patients 1 and 2), contralateral electrodes were implanted because of shift of cCH after 6 months and 7 years, respectively. In some cases the paraesthesia excluded the parietal and frontal/orbital region, which however did not influence the analgesic effect. Often during test stimulation also paraesthesia of the ipsilateral upper extremity was encountered. With a change of stimulation settings (particularly electrode polarity and impulse duration) unintended paraesthesia of the upper limb could be abolished or reduced to a minimum in most cases.
X-ray showing the optimal lead location in anteroposterior and lateral view.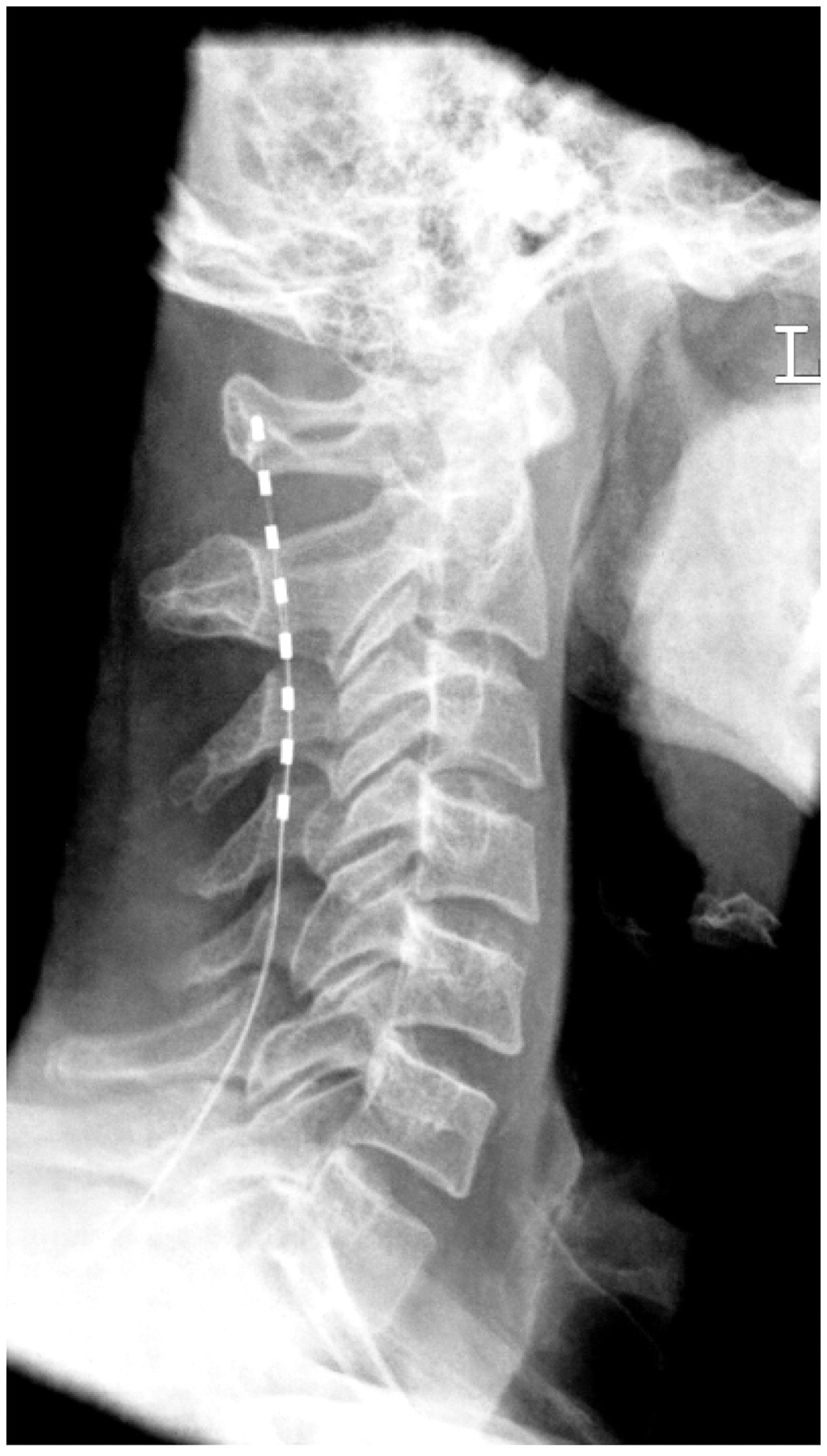
Settings of neurostimulator
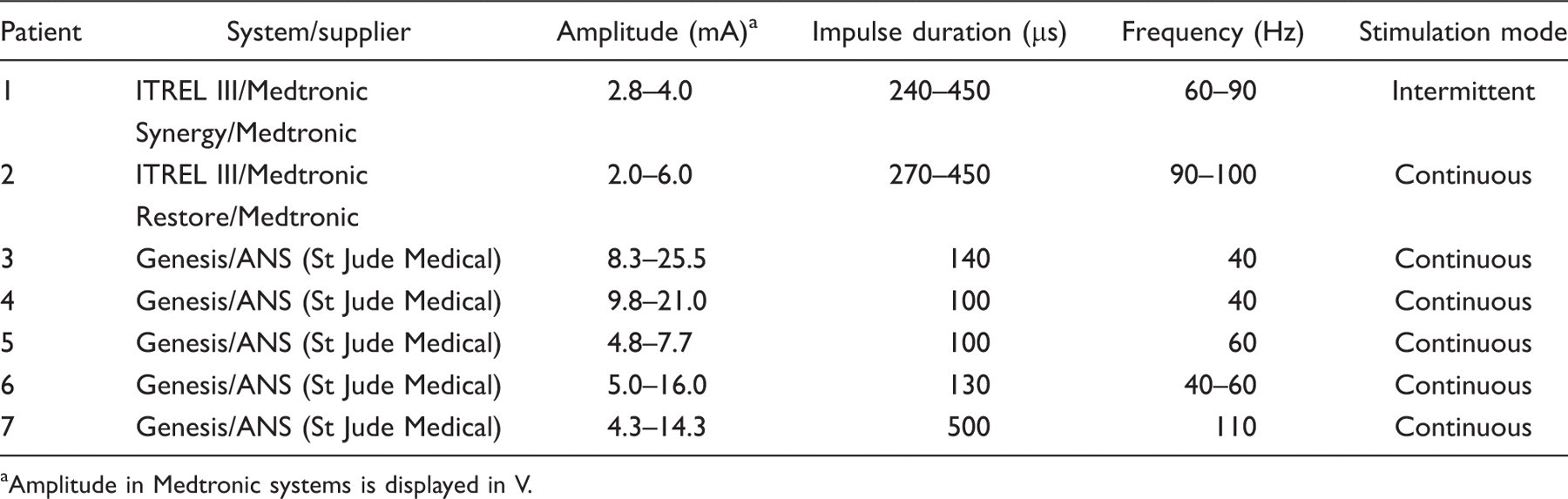
Amplitude in Medtronic systems is displayed in V.
Follow up and data collection
Apart from patient 1 who had been implanted 4 years earlier with good success, all patients were followed up prospectively. Mean follow up was 23 months (median 12 months, range 3–78 months). Scheduled follow-up visits were at 3, 6, 12, and 24 months.
All patients filled in pain diaries from 4 weeks prior to implantation until up to 2 years post implantation. Severity of CH attacks was assessed with pain diaries to document information of each individual attack with regard to time of onset, attack duration, pain intensity on the 11-point numeric analogue scale (NAS), and comments on acute treatments. For two patients (patients 1 and 3) we could not use pain diaries. Patient 1 had been operated several years ago, and being free of attacks he had stopped documenting pain intensity. Patient 3 had filled in a pain diary during his in-patient time, but because of his depression and anxiety disorder did not comply with continuous diary documentation. On outpatient presentations, both patients, however, were able to make clear statements regarding average attack frequency and attack duration in the preceding weeks. All other patients continued their pain diaries unless they were not entirely pain free.
Preventives, transitional treatments, and abortive drugs tried before implantation


CBZ, carbamazepine; GPT, gabapentine; PGB, pregabaline; TPM, topiramate; VPA, valproic acid.
DHE, dihydroergotamine infusion (given over 3–4 days).
GLOA, ganglionary local opioid application; GON, greater occipital nerve injection with lignocaine and steroid.
ELE, eletriptan; FRO, frovatriptan; NAR, naratriptan; RIZ, rizatriptan; SUM, sumatriptan; ZOL, zolmitriptane.
Partial effect, attack not completely resolved but less intense.
Statistical analysis
From the pain diaries at each time point, a 2 week interval was analysed and mean attack frequency, intensity, and duration was determined. For statistical analysis, GraphPad Prism version 5.01 (GraphPad Software, La Jolla, USA) was used. A p-value less than 0.05 was considered to indicate a significant difference. A paired t-test was used for calculation of the statistical significance of the differences of mean attack frequency, attack duration and intensity, ‘pain load’, and HADS and PDI scores before and after treatment.
Results
Pain characteristics
At baseline, mean frequency of attacks was 6.0 attacks/day (range 1.7–10.0, SD 3.3, median 8.0), mean duration of attacks was 50 minutes (range 22–70 minutes, SD 16.9, median 60.0), and mean intensity was 7.4/10 on the NAS (range 4.3–10.0, SD 2.1, median 7.0). Postoperatively, mean attack frequency was reduced to 1.4 attacks/day (range 0–3.5, SD 1.2, median 1.3), mean duration of attacks to 23 minutes (range 0–56 min, SD 17.4, median 21.4), and attack intensity to 4.5 on the NAS (range 0–7.6, SD 2.6, median 5.2). These differences were statistically significant for attack frequency, attack duration, and attack intensity (p = 0.008, p = 0.006, and p = 0.013, respectively) (Table 4, Figures 2 and 3).
Course of the attack frequency over time. Course of the ‘pain load’ (attack frequency × attack duration × intensity of attacks; numeric analogue scale, NAS) over time. Synopsis of clinical data Pain load, mean attack frequency × mean attack intensity × mean attack duration. Assessment during the first 2 weeks after implantation of the hsSCS system. cCH, chronic cluster headache; hcSCS, high cervical spinal cord stimulation; NAS, numeric analogue scale.


Anxiety and depression, quality of life, and treatment satisfaction
At baseline, mean ratings for depressiveness were 14.0 (median 16.5, SD 5.9) and for anxiety 11.7 (median 12.0, SD 5.6) as measured by the HADS. Under hcSCS, these scores decreased to 9.8 (median 11.3, SD 3.9) for depressiveness and 9.4 (median 7.0, SD 4.8) for anxiety. These differences were not statistically significant.
The mean level of pain-related impairment as assessed by the PDI was reduced in six of seven items. For the item ‘professional activities’, patients rated their degree of physical impairment slightly higher postoperatively than at baseline. Mean ratings for all other items declined postoperatively. Only for the item ‘social activities’ this difference was statistically significant (p < 0.05).
All patients would recommend the treatment and six of seven patients would undergo the procedure again.
Stimulation characteristics
Stimulation produced a notable paraesthesia in the nuchal and occipital area in all patients. In five patients (1, 2, 4, 5, and 7), the paraesthesia also included the trigeminal region. Most patients used continuous stimulation, with the possibility to adjust the stimulation intensity (amplitude) according to their needs (Table 2).
Medication intake
Pain medication was markedly reduced after the operation in all patients. Triptan use was stopped in five patients (patients 1–5) and reduced in two patients (patient 6 and 7). Four patients could discontinue all prophylactic medication (patients 1, 2, 4, and 5), one patient still requires prevention (patient 5) and two patients (patients 3 and 7) have not yet tried to withdraw prophylactic medication. Patients who still used a prophylactic or acute medication postoperatively adhered to the medication used before the operation.
Complications
There were no major adverse events. No major procedure-related morbidity, like intraspinal bleeding or neurological deficit, occurred. A number of device-related complications, namely electrode fracture or, in two cases, lead migration, occurred, necessitating electrode revision in local anaesthesia. In three patients, the impulse generator had to be changed because of battery loss. There were no biological complications such as infection or wound breakdown. In none of the cases was there amplification of pain due to stimulation.
Patient 1 developed additional cCH on the contralateral side 7 years after the initial implant, requiring implantation of a second electrode. Since that time he has been free of attacks.
Patient 2 had an immediate good response with reduction of attack rate to one attack per day. Attack frequency rose slightly after lead revision because of dislocation after 3 months and after implantation of a contralateral electrode because of shift of cCH after 6 months. Since that time attack frequency, as well as intensity and duration of attacks, has decreased.
Before the operation, patient 3 had a very high attack frequency and ‘pain load’. He was severely depressed and anxious and suffered from social phobias because of his attacks. He had an uneventful implantation and clinical course. cCH burden after the operation was drastically reduced, but the depression and anxiety disorder improved only slowly and the social phobias are still present.
Patient 4 had an uneventful course except for a lead revision because of breakage after 6 weeks. Directly from the time of implantation up to now he has been free of attacks.
Also patient 5 was free of attacks in the postoperative course. Unfortunately he repeatedly needed revisions because of lead dislocation and breakage as well as battery depletion. Currently, after implantation of a paddle-type electrode, he is free of attacks.
In patient 6 we found it extremely difficult to achieve paraesthesia spreading beyond the nuchal area. She had an initial reduction of attacks from 8 to 3.5 attacks/day. In the last 12 months, attacks showed a slow progression despite a further lead revision after 7 months. On the last follow up, lead failure became evident and revision surgery was scheduled.
Patient 7 had a comparably modest reduction of attack frequency, but the ‘pain load’ has decreased considerably and he is highly satisfied with treatment.
Discussion
Our study hints at a promising role for high cervical epidural neurostimulation in medically intractable cCH, which warrants further clinical study in the future.
Spinal cord stimulation (SCS) has been successfully employed for a variety of chronic pain conditions in the past 30 years, as meta-analyses have shown (11). In a recent retrospective single-centre study a high maintenance of efficacy over the years was found (12). Moreover, lately a randomized controlled multicentre study showed improved pain relief, quality of life, and functional capacity besides greater treatment satisfaction in patients treated with SCS compared to patients treated with conventional medical treatment for neuropathic pain (13).
SCS is believed to act via multiple mechanisms acting at the segmental level as preclinical animal models indicate. Under SCS, attenuation of the hyperexcitable multimodal wide dynamic range cells (WDR neurons) in the dorsal horn (14) was observed and increased γ-aminobutyric acid (GABA) release (15,16) from inhibitory interneurons was demonstrated to account for that effect, particularly by activation of the GABAB receptor (17). Also the cholinergic system seems to contribute to SCS effects, as a release of acetylcholine in the dorsal horn under SCS was observed and later attributed to activation of the muscarinic M4 receptor (18). Moreover, activation of supraspinal pain-modulating centres via rostral projections of the dorsal column nuclei has been observed (19). In a recent study, unlike dorsal column stimulation, dorsal root stimulation failed to inhibit windup in WDR neurons in neuropathic conditions. (20). This may point towards a neurobiological difference between the mechanisms of SCS and peripheral nerve stimulation as in ONS.
Cervical SCS, in particular, has been described as efficacious in complex regional pain syndrome, brachial plexus injury, Raynaud’s syndrome, and neck and upper limb pain after surgery (21). The technique is relatively simple and carries a very low risk of procedural morbidity. We used a technique similar to the one used in previous reports on cervical epidural neurostimulation (21) except that in our patients the electrode was moved 3–4 cm further cranially than in SCS for upper limb pain.
Our study included a limited number of cases but it has a relatively long follow up (3–78 months). All patients had a long lasting history of cCH and a variety of medications had been used without sufficient pain relief prior to implantation (Table 3). In more than half of the patients, medication was stopped shortly after the implantation, and in the other patients, medication intake, in particular triptans, was significantly reduced. When technical problems with the stimulation occurred, cluster headache returned within few hours to days. This makes a solely placebo mode of action unlikely, although placebo responses in cluster headache have been described (22). To current knowledge, for neuropathic pain, successful stimulation requires paraesthesia (23) within the painful region, whereas this may not be necessary in the treatment of refractory angina pectoris, as a recent study has shown therapeutic effects with subthreshold stimulation (24). Similar cross-over study designs are conceivable for headache patients but require an initial good response to treatment with SCS. Our data suggest a robust and long-lasting effect of hcSCS in cCH as prerequisite for future controlled study designs.
The effects observed in our patients – stable pain reduction, relief of medically intractable symptoms, and recurrence of pain with cessation of stimulation therapy (i.e. in case of battery failure) – are unlikely to be solely because of placebo effects. Nevertheless a placebo mode of action cannot entirely be ruled out.
Also a concomittant stimulation of the C2 nerve root cannot be ruled out, as the lead position relative to the spinal cord can not be determined fluoroscopically and a variability of the spinal cord position within the spinal canal is known (25). Moreover the lead position in cervical SCS appears to be somewhat variable because of the mobility of the cervical spine.
Our patients reported concurrent stimulation of the shoulder and parts of the upper limb, which mostly could be reversed with adjustment of the pulse width and poling of the electrode, but in most patients remained to a lesser extent. This makes an exclusive action through stimulation of the C2 nerve root improbable.
Therapy refractory cCH is a rare condition which makes it difficult to run studies with high patient numbers. Nevertheless, one can draw conclusions regarding the feasibility and the efficacy also from that limited number of patients.
Up to now, studies of interventions in cCH have focussed on attack frequency rather than pain intensity or attack duration. We believe that it is important to measure the overall time in which the patient succumbs to attacks and the pain intensity suffered during that time, which led us to decide on the construct of ‘pain load’. Under treatment, four patients described occasional attack-wise occurrence of parasympathetic symptoms without head pain (‘cold attacks’). For most patients with cCH, a reduction of attack frequency is of paramount importance but also the duration and the intensity of attacks play a major role in their suffering.
Interestingly, in two of our patients (after 18 months and after 6 years after implant), additional cCH occurred on the contralateral side. Both patients received a second, contralateral electrode. In both patients, attacks occurred at different times on both sides and could be influenced independently with the bilateral SCS system, which allows side-specific programming. This suggests that bilateral CH can be driven by two chronologically independent generators.
The effect size in hcSCS appears to be equal or larger compared to ONS, as the percentage of significant pain reductions in our data and the published data on ONS (6–8) indicate. Burns and colleagues published a pilot study of eight patients and a follow-up study of 14 patients (6,8). In the pilot study six of eight patients and in the follow-up study 10 of 14 patients experienced a reduction in attack frequency. The reduction of attack frequency was more than 40% in six of eight patients in the pilot study and six of 14 patients in the follow-up study. In the study by Magis and colleagues (7), seven of eight patients had a decrease in attack frequency of more than 40%. In our study, attack frequency was reduced by more than 40% in seven of seven patients. Mean attack frequency was decreased by 19, 29, and 80% in the two studies of the London group and the Belgian study, respectively. In the latter study on average, the ONS to baseline attack ratio per month was 0.65 during the whole follow up (mean 15.1 months). In our study, decrease of the mean attack frequency compared to baseline was 77% postoperatively, 74% at 3 months, 72% at 6 months, and 84% at 12 months (Figure 2). Attack intensity decreased by 23 and 6% in the two studies by Burns et al. and 44% in the study of Magis et al. In our study it was reduced by 39%. Mean baseline attack frequencies were 3.9 and 4.8 attacks/day respectively in the two studies by Burns and colleagues and 1.7 attacks/day in the Magis study while in our study was higher with 5.9 attacks/day.
One of the advantages of hcSCS is that it exerts an immediate and almost maximal effect after surgery. This allows an inpatient test phase prior to implantation of the impulse generator with the possibility of removing the electrode in case of insufficient pain relief. A technical challenge still consists in the high rate of battery failures and electrode breakages. So far, we have initially implanted non-rechargeable battery-powered impulse generators as the usage and energy consumption of the system in the long term was unknown. Now it appears that many patients prefer continuous stimulation. In the meantime three IPGs with battery failure were replaced by rechargeable systems. If battery failure within 2 years after implantation occurs in more patients, the use of rechargeable stimulators at first line may be advisable. Electrode breakage is a known technical complication (11,26) of SCS, in particular at the cervical spine (27), probably because of the higher mobility. All in all, up to now in hcSCS the amount of these device-related complications appears to be higher than in ONS (4,6–8,28), Usually the clinical symptoms caused by electrode breakage are benign and reversible.
More studies for hcSCS in cCH are warranted in order to define its future role as a reserve therapy in case of failure of ONS or as a first-line interventional therapy for medication refractory cCH.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
