Abstract
Background:
Accumulating evidence and conditional regional guidelines support early initiation of advanced therapy (AT) in Crohn’s disease (CD) and ulcerative colitis (UC). Nonetheless, AT utilization in real-world clinical practice remains limited.
Objectives:
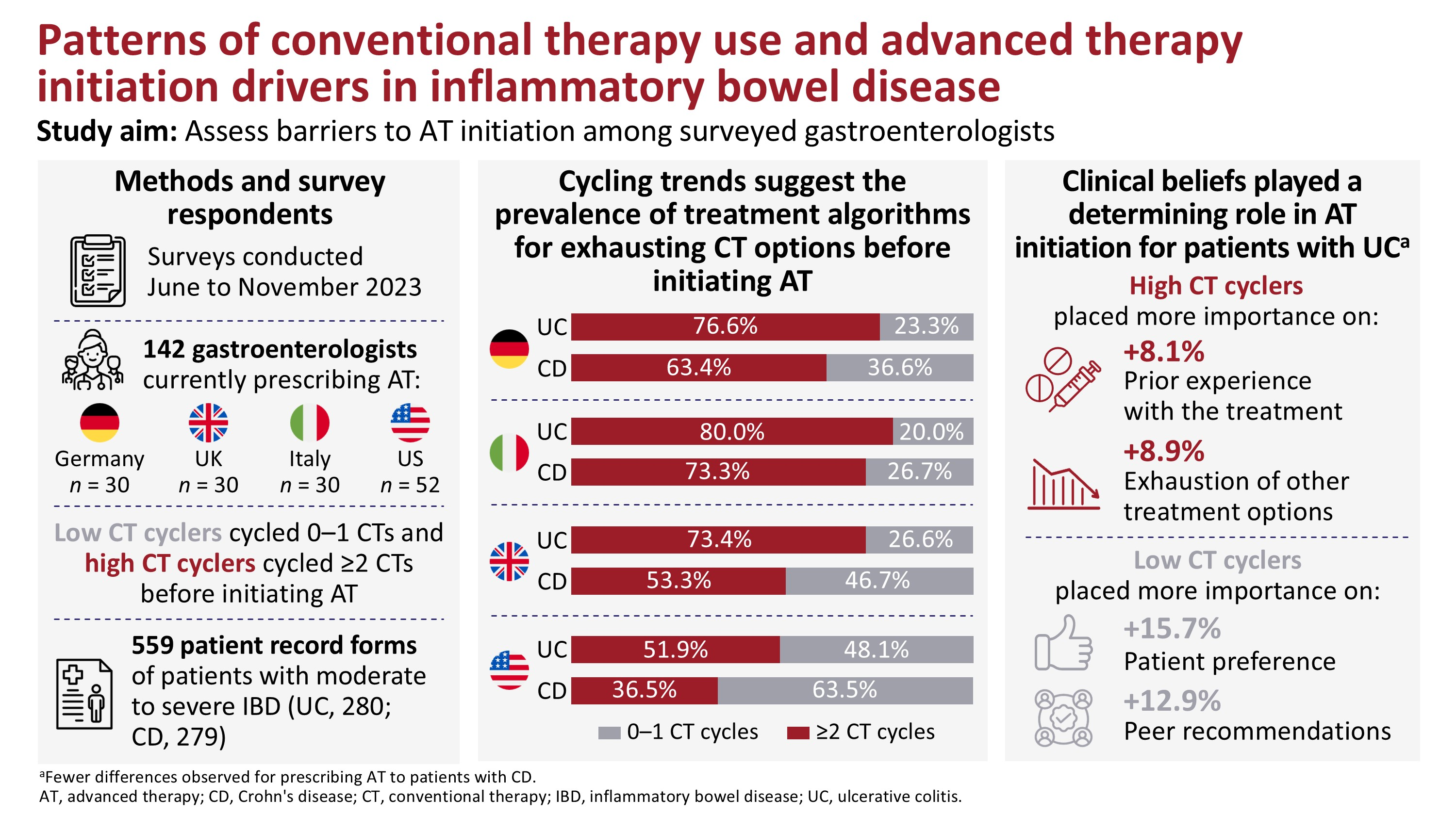
The aim of this study was to assess barriers for AT initiation among surveyed gastroenterologists.
Design:
Quantitative survey with a multimodal approach.
Methods:
In total, 142 gastroenterologists (30 each from Germany, Italy, and the United Kingdom; 52 from the United States) were surveyed from June to November 2023 to capture drivers of AT initiation. They were classified as low or high conventional therapy (CT) cyclers (using 0–1 or ⩾2 CT cycles before initiating AT, respectively). Behavioral analysis was performed employing a COM-B (Capability, Opportunity, Motivation, Behavior) model.
Results:
Across countries, 67.3%–90.0% of surveyed gastroenterologists agreed that AT initiation should be early enough to modify the trajectory of disease. Nonetheless, the majority (51.9%–80.0%) of surveyed gastroenterologists were high CT cyclers (except those in the United States treating CD (36.5%)). When surveyed about the importance of factors for initiating AT in patients with UC, high cyclers placed greater importance on treatment familiarity (+8.1%) than low cyclers and preferred to exhaust other treatment options first (+8.9%). Alternatively, low cyclers placed more importance on patient preference (+15.7%) and peer recommendations (+12.9%) than high cyclers. There were fewer differences in AT prescription drivers between low and high CT cyclers for CD than UC.
Conclusion:
High CT cyclers placed an overall lower value on AT, suggesting clinical beliefs played a determining role in AT initiation among surveyed gastroenterologists.
Plain language summary
Keywords
Introduction
Treatment approaches for inflammatory bowel disease (IBD; ulcerative colitis (UC) and Crohn’s disease (CD)) are rapidly evolving, with multiple options available. 1 Conventional therapy (CT) for IBD includes 5-aminosalicylates (5-ASA), corticosteroids, and immunomodulators (e.g., thiopurines and methotrexate). While CTs can be effective for induction therapy, corticosteroids have limited efficacy and no place for the maintenance of IBD remission; moreover, both corticosteroids and immunomodulators are associated with adverse safety profiles when used as long-term therapies. 2 Advanced therapy (AT) for IBD encompasses targeted biologic agents (e.g., monoclonal antibodies) and various oral small-molecule agents that ameliorate inflammation of the gastrointestinal tract by limiting the migration of pro-inflammatory leukocytes (e.g., vedolizumab, ozanimod, etrasimod) or suppressing inflammatory cytokine signaling pathways (e.g., adalimumab, certolizumab pegol, filgotinib, golimumab, guselkumab, infliximab, mirikizumab, risankizumab, tofacitinib (UC only), upadacitinib, and ustekinumab).2–4 Induction and/or maintenance therapy with ATs generally leads to improved rates of clinical response and remission versus placebo, and their safety profile is supported by data illustrating similar proportions of patients who experience adverse events compared with placebo.2,3
There is accumulating evidence supporting earlier and broader use of AT for the treatment of patients with IBD, especially those with CD.5–13 For example, the PROFILE study evaluating step-up (progression from CT to AT) versus top-down (AT included for induction) treatment approaches found that earlier intervention with AT led to significantly more patients with CD achieving week 48 surgery-free and steroid-free clinical remission. 7 Multiple other studies have also observed improved clinical outcomes in patients with CD given AT earlier in the course of disease.5,6,13 Similarly, the REACT trial found that earlier intervention with an AT led to a lower long-term risk of major adverse events in patients with CD. 14 For patients with UC, there is less evidence supporting the advantages of early AT intervention.11,15
Following clinical study data, recent treatment guidelines in Europe and the United States show gradual trends shifting toward recommending AT use for early treatment of IBD. Initially, the European Crohn’s and Colitis Organization (ECCO) 2022 guidelines for UC and 2020 guidelines for CD recommended AT only after inadequate response or intolerance to CT.3,16 However, the 2024 update to the ECCO guidelines for the medical treatment of CD no longer supports treatment sequencing from CT to AT, instead giving strong recommendations for the use of AT in induction therapy, including antitumor necrosis factor agents, interleukin-12/23 and interleukin-23 inhibitors, α4β7 integrin inhibitors, and one Janus kinase inhibitor. 2 Similarly, the American Gastroenterology Association (AGA) 2021 guidelines for CD and 2024 guidelines for UC provide conditional recommendations for early AT initiation, indicating that the majority of patients would benefit from AT, but commenting that patients with less severe disease may reasonably prefer CT.17,18
Despite accumulating evidence and clinical guidelines indicating the benefits of early AT, real-world data show limited AT use, suggesting the existence of barriers to AT implementation.19–21 It has also been reported that AT utilization can vary not only between different countries but also between different regions within the same country.22,23 Additionally, recent data highlight that not all AT-eligible patients are receiving AT, suggesting suboptimal disease management and undertreatment with AT in clinical practice.6,20,24 Here, we aimed to evaluate current prescribing trends and assess the barriers to AT use in patients with UC and CD using a multimodal approach.
Methods
Objectives
The first objective of the quantitative survey was to identify current trends for CT and AT prescription, as well as triggers for AT initiation. The second objective was to investigate perceptions of early AT initiation and drivers motivating early AT use based on behavioral analysis. The reporting of this study conforms to the Checklist for Reporting Results of Internet E-Surveys (CHERRIES; Supplemental Table 1). 25
Survey participants
Qualitative surveys were administered to healthcare professionals, patients, and patient organization representatives, the results of which were used to inform the development of questions for the quantitative survey used in this study. To participate in the qualitative survey, gastroenterology nurses and gastroenterologists were required to have no pharmaceutical affiliation, have >5 years of experience in gastroenterology, spend ⩾70% of their time in direct patient care as opposed to teaching or research, and be comfortable conversing in English. To identify patients for participation in the qualitative survey, quotas were utilized to ensure representative sampling across gender, race, education, employment status, and income range. To participate in the survey, patients had to be ⩾18 years old, not pregnant, have a confirmed diagnosis with UC or CD, and be currently treated with AT after receiving prior CT.
The quantitative surveys targeted healthcare professionals experienced in the treatment of IBD who mainly focused on patients with UC and CD, and who worked in a range of institutions (from small community hospitals to academic/specialist IBD centers). Specific quotas were used in the identification of eligible gastroenterologists for representative sampling in the quantitative survey, including for country of origin, years of practice, practice setting, and average patient income range. To participate in the quantitative survey, gastroenterologists were required to have 3–35 years of experience, spend ⩾70% of their time in direct patient care as opposed to teaching or research, be personally responsible for the decisions to initiate/repeat/switch prescriptions for IBD, and have ⩾10 patients with moderate-to-severe UC and moderate-to-severe CD each, of whom at least 4 per therapy area were currently treated with AT. Additionally, the survey was designed to include gastroenterologists from specialist centers versus the community in a 2:3 ratio. Gastroenterologist eligibility to participate in the quantitative survey was identified by a screening questionnaire, with only gastroenterologists meeting the criteria above proceeding to the main survey.
Survey design
The surveys were conducted from June through November 2023 in Germany, Italy, the United Kingdom, and the United States. Prior to launching the fieldwork, two pilot interviews were conducted—one in the US and one in the UK. Gastroenterologists completed the online survey while sharing their screens with a moderator. The questionnaire and survey link were adjusted according to the feedback provided. Translations and overlay commenced only after feedback from the pre-testing interviews had been incorporated and approved. Qualitative interviews with gastroenterologists, gastroenterology nurses, patients, and patient organization representatives were conducted to identify and develop relevant questions for the quantitative survey. The quantitative survey assessed prescribing trends among gastroenterologists currently prescribing AT to capture AT initiation drivers using a 45-min online survey (questionnaire excerpt summarizing key questions is provided in the Supplemental Appendix). Translations and back-translations were performed for non-English-speaking countries to ensure consistency of concepts. Additional details regarding the quantitative survey are provided in the CHERRIES checklist (Supplemental Table 1).
During the survey, gastroenterologists were also requested to complete four patient record forms from their recent patients (treated by the gastroenterologist in the past 6 months) who met the following criteria: aged ⩾18 years with moderate to severe disease and under the gastroenterologist’s management since treatment initiation, or for whom the gastroenterologist has a full treatment history for moderate to severe disease. Four patient record forms per gastroenterologist were requested, including two from patients with UC and two from patients with CD (including one patient per disease currently treated with CT but eligible for AT and the other one currently treated with AT). Moderate-to-severe UC was defined as five to eight bloody mucus or diarrhea events per day, fever, and active disease/feeling of illness. 26 Moderate-to-severe CD was defined as abdominal pain, diarrhea (more than three stools per day), weight loss, and fatigue. 27 Patients considered eligible for AT in the United States were those with moderate-to-severe UC/CD. In Europe, patients eligible for AT were those with moderate-to-severe UC/CD who had an inadequate response to prior CT.
For CT, treatment cycles were defined as periods of treatment with either a single drug or drug combination separated by a temporary cessation. Gastroenterologists cycling an average of 0–1 CTs before initiating AT were classified as low CT cyclers, and gastroenterologists cycling an average of ⩾2 CTs before initiating AT were classified as high CT cyclers.
Behavioral analysis was conducted using a Capability, Opportunity, Motivation, and Behavior (COM-B) model. 28 This model is widely used in behavioral sciences to understand the reasons behind certain behaviors. Briefly, capability (physical and psychological capacity to engage in a specific behavior) and opportunity (factors outside of the individual enabling or prompting the behavior) influence motivation (brain processes energizing and directing the behavior, including habitual processes, emotional responses, and analytical decision-making). 28 The interaction between capability, opportunity, and motivation generates behavior. In turn, enacting the behavior may further change capability, opportunity, and motivation. 28 In this project, the COM-B model was used to model behaviors related to the prescribing of medications.
Statistical analysis
Statistical analyses were conducted using SPSS v29 (Chicago, IL, USA). Where reported, statistically significant differences between survey responses are based on 95% confidence intervals (p < 0.05).
Results
Description of survey responders and patient record forms
The overall study design is shown in Supplemental Figure 1. A total of 62 participants responded to the qualitative interviews, including 28 patients (10 from the United States and 6 each from Germany, Italy, and the United Kingdom), 24 gastroenterologists (15 from the United States and 3 each Germany, Italy, and the United Kingdom), 7 gastroenterology nurses (4 from the United States and 1 each from Germany, Italy, and the United Kingdom), and 3 patient organization representatives (1 each from Germany, the United Kingdom, and the United States). Results from the qualitative survey were used to identify and develop relevant questions for the quantitative survey.
The quantitative survey included responses from 142 gastroenterologists from Europe and North America who were currently prescribing AT (52 from the United States and 30 each from Germany, Italy, and the United Kingdom). Gastroenterologist characteristics are summarized in Table 1. A total of 559 patient record forms were collected (280 from patients with UC and 279 from patients with CD). Patient characteristics by IBD type (UC or CD) and AT status are summarized in Table 2. In both UC and CD, patients currently receiving CT but eligible for AT and patients currently receiving AT were similar in their clinical characteristics (disease symptoms, disease severity, and impact on patients’ quality of life) overall. The key difference between patients currently receiving CT and AT was the time since diagnosis of moderate-to-severe UC or CD, which tended to be higher in patients currently receiving AT.
Gastroenterologist characteristics.
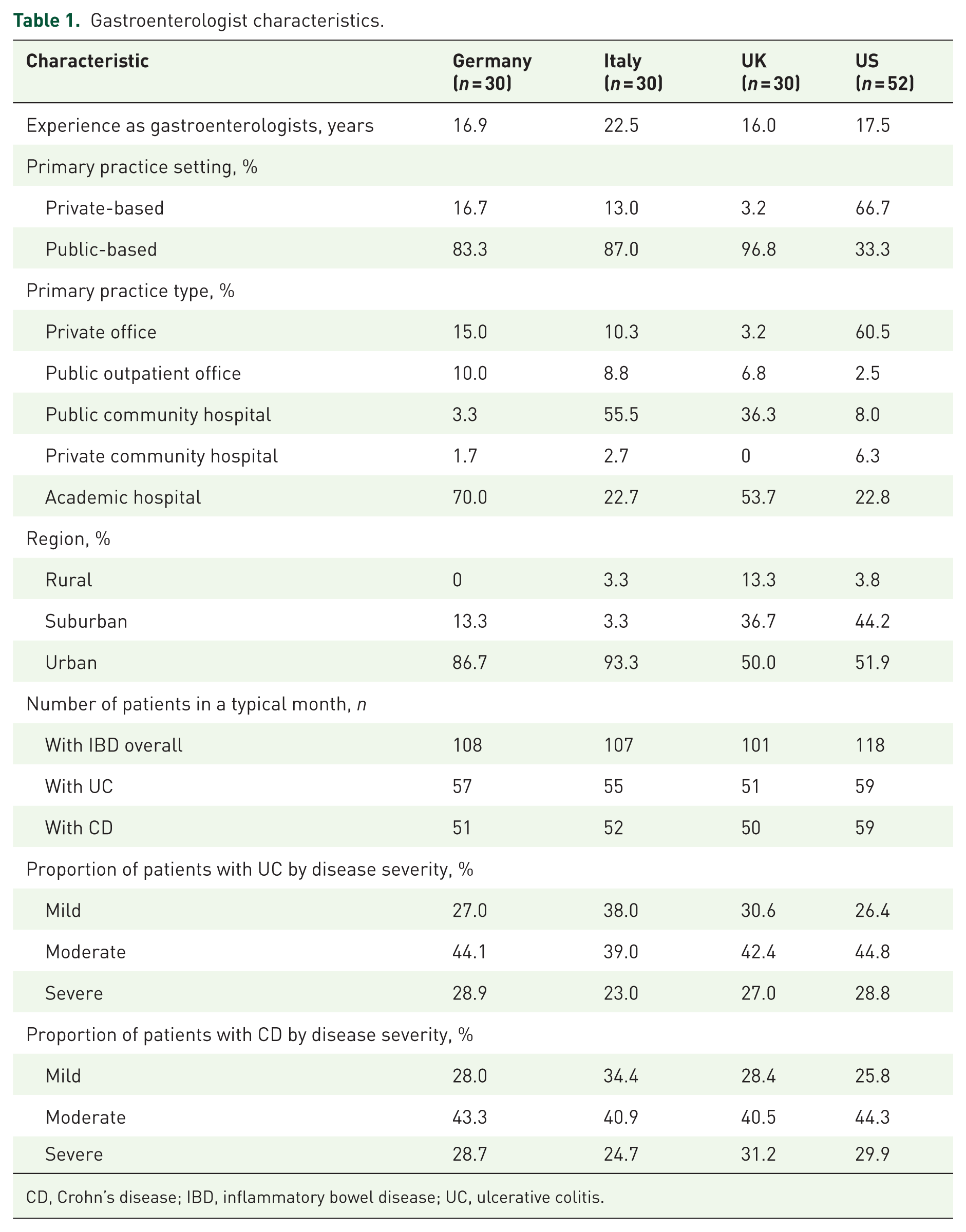
CD, Crohn’s disease; IBD, inflammatory bowel disease; UC, ulcerative colitis.
Characteristics of analyzed patients.

AT, advanced therapy; CD, Crohn’s disease; CT, conventional therapy; QoL, quality of life; UC, ulcerative colitis.
Current trends in CT and AT prescription
Gastroenterologists were less likely to administer AT treatment in adult patients with moderate-to-severe UC than in those with moderate-to-severe CD (Figure 1(a) and (b)). Furthermore, prescribed AT monotherapy in the United States was significantly higher and CT monotherapy significantly lower compared with Germany, Italy, and the United Kingdom for patients with UC and CD (Figure 1). CT + AT combination therapy was similar across countries as well as between UC and CD (range: UC, 24.8%–38.9%; CD, 22.1%–33.0%).
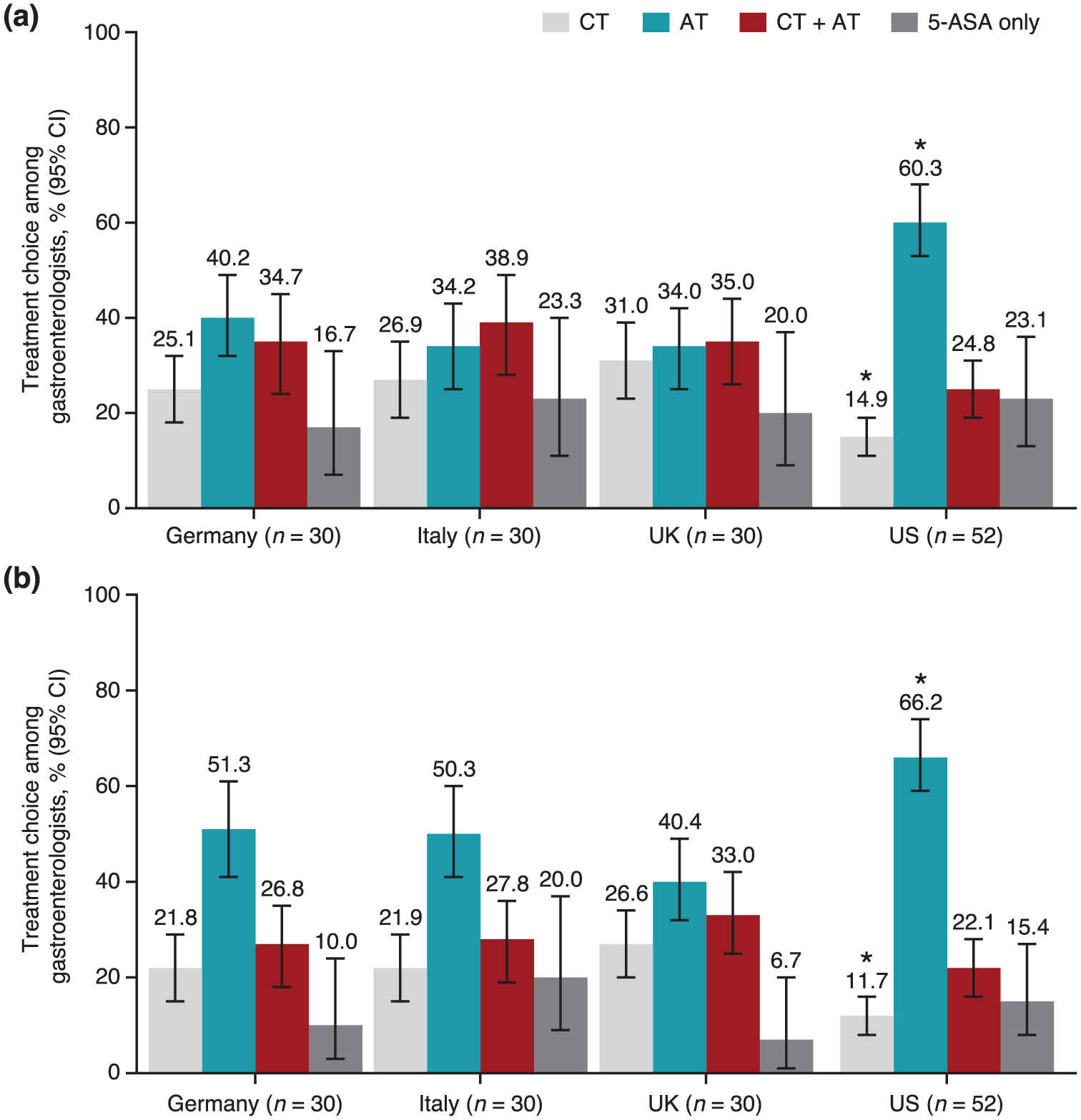
CT and AT prescribing trends. Surveyed gastroenterologists reported average prescription rates of CT, AT, or CT + AT as well as 5-ASA monotherapy when treating patients with (a) moderate-to-severe ulcerative colitis or (b) moderate-to-severe Crohn’s disease.
The number of CT cycles employed before advancing patients to AT is shown in Figure 2(a). At least half of all gastroenterologists reported using ⩾2 CT cycles before transitioning patients to AT, except for US gastroenterologists treating patients with CD, where only 36.5% reported using ⩾2 CT cycles (Figure 2(b)). Additionally, gastroenterologists in all countries were more likely to utilize ⩾2 CT cycles before initiating AT when treating patients with UC than those with CD. Overall, 46 gastroenterologists were classified as low CT cyclers (0–1 CT cycles) and 96 as high CT cyclers (⩾2 CT cycles) for the treatment of patients with UC. For treating patients with CD, 66 gastroenterologists were classified as low and 76 as high CT cyclers.

CT cycling behavior of surveyed gastroenterologists. (a) Surveyed gastroenterologists reported the number of CT cycles used when treating patients with moderate-to-severe ulcerative colitis or Crohn’s disease before initiating advanced therapy. (b) Gastroenterologists were categorized as low CT cyclers (0–1 CT cycles) or high CT cyclers (>2 CT cycles).
The reported duration of the first CT cycle ranged from 3.3 to 5.0 months in UC and from 3.3 to 4.5 months in CD (Figure 3). Across countries, the second CT cycle was typically longer than the first CT cycle (range: UC, 3.4–5.7 months; CD, 2.9–6.8 months). A minority of gastroenterologists prescribed a third CT cycle (range: UC, 2.5–4.9 months; CD, 2.5–3.9 months). The cumulative duration of CT cycles before patients advanced to AT was also examined. The cumulative duration of two CT cycles was similar for patients with UC and CD (range: UC, 6.7–10.7 months; CD, 6.2–11.3 months). When including a third CT cycle, the cumulative duration of CT cycles ranged from 11.3 to 13.6 months for UC and 8.7 to 15.2 months for CD.

Average CT cycle duration. Gastroenterologists reported the average duration of CT cycles before initiating AT for patients with moderate-to-severe UC or CD. Individual cycles were summed to determine cumulative CT cycle duration.
Gastroenterologists’ preferred first- and second-line CT choices were also queried (Figure 4). There was no clear standard choice for first- and second-line CT. Across countries, 28.8%–50.0% of patients with UC were prescribed 5-ASA + corticosteroids and 16.7%–23.3% were prescribed 5-ASA alone as first-line CT. In total, 63.4%–86.6% of gastroenterologists reported using 5-ASA either alone or in combination as first-line therapy for UC. On the other hand, 5-ASA was reportedly utilized in 23.3%–66.6% of first-line therapies (alone or in combination) for patients with CD, despite the AGA and ECCO guidelines’ recommendations against the use of 5-ASA for induction and maintenance of remission in CD due to a consistent lack of evidence of efficacy.2,17 Total 5-ASA use as a first-line therapy for CD was highest among gastroenterologists from Italy (66.6%) and Germany (63.3%) versus the United States (36.5%) or United Kingdom (23.3%).

CT prescribing trends for first- and second-line therapy. Gastroenterologists reported the most frequently prescribed (a) first-line CT in patients with UC, (b) second-line CT in patients with UC, (c) first-line CT in patients with CD, and (d) second-line CT in patients with CD.
Furthermore, there was a trend across all countries for the greater use of corticosteroids as first-line monotherapy in CD compared with UC (range: UC, 3.3%–10.0%; CD, 23.1%–30.0%). However, when given either as monotherapy or in combination, corticosteroids were preferred as first-line therapy by 49.9%–73.3% of gastroenterologists when treating patients with UC and by 53.8%–76.7% when treating patients with CD. CT combination therapy was more common than monotherapy as both first- and second-line treatments. A minority of the surveyed gastroenterologists from Germany, the United Kingdom, or the United States skipped CT entirely for first-line therapy (range: UC, 6.7%–15.4%; CD, 3.3%–25.0%). No gastroenterologists from Italy reported skipping CT.
Triggers for AT initiation
There was no clear consensus between gastroenterologists from different countries on the optimal timing of AT initiation (Figure 5). Although most gastroenterologists (69.2%–90.0% in UC and 67.3%–90.0% in CD) agreed with the statement that AT should be started early enough to modify the course of disease, there was a lack of agreement regarding specific targets defining the optimal timing of AT initiation. In UC, 51.9%–93.3%, 50.0%–66.7%, and 50.0%–70.0% of gastroenterologists disagreed with statements that the optimal timing of AT initiation was as early as possible to minimize the use of CT, early enough to mitigate the need for surgery, and early enough to mitigate the risk of cancer, respectively. In CD, the proportions of gastroenterologists disagreeing with these statements were 44.2%–90.0% for as early as possible to minimize the use of CT, 46.7%–63.3% for early enough to mitigate the need for surgery, and 53.3%–76.7% for early enough to mitigate the risk of cancer.
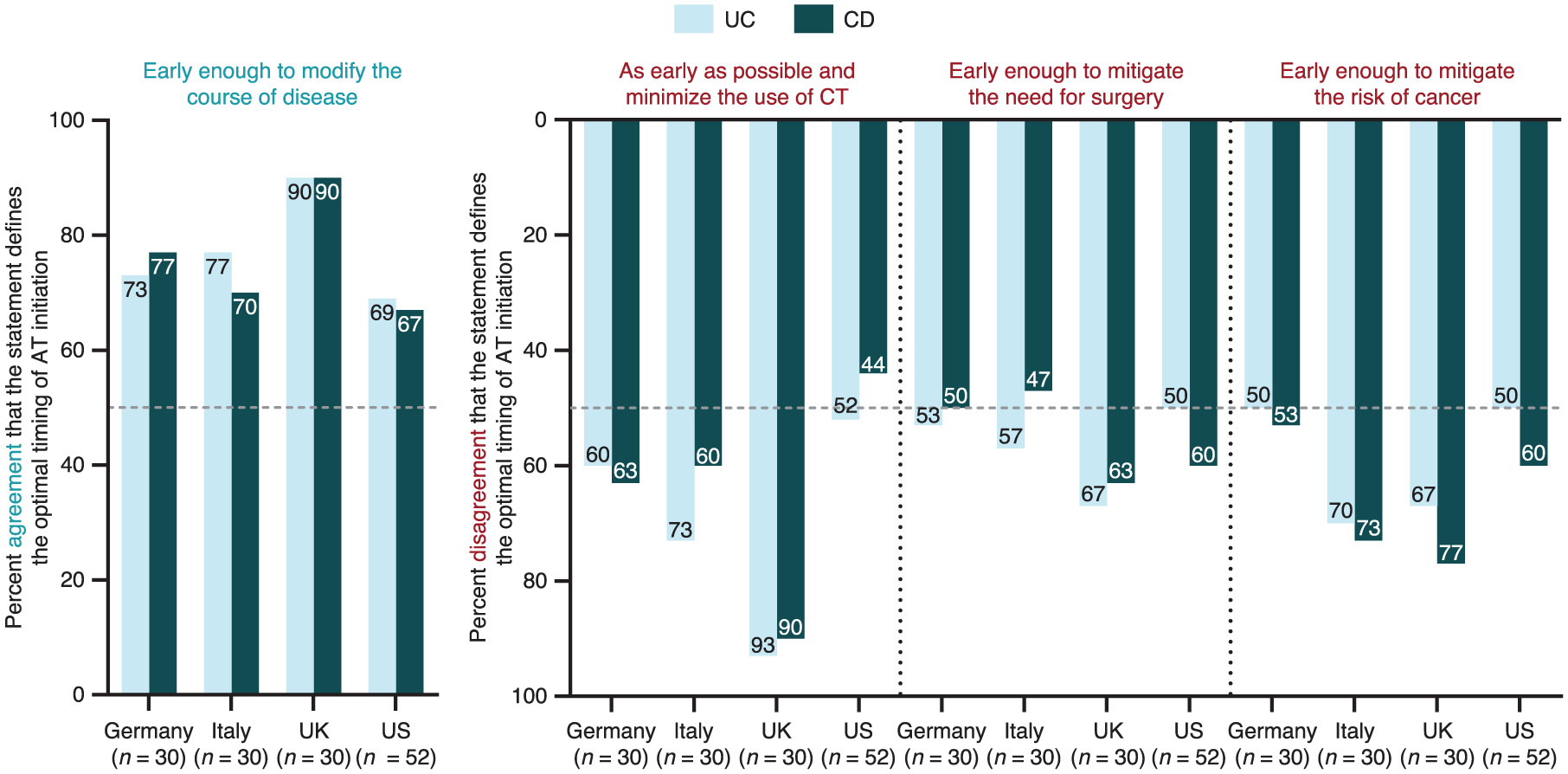
Gastroenterologist agreement or disagreement with specific statements defining the optimal timing of AT initiation.
Perceptions of early AT initiation and drivers motivating early AT use
High CT cyclers generally had a lower belief in the value of early AT in patients with UC (Supplemental Figure 2(a)) and the clinical value of AT in patients with CD (Supplemental Figure 2(b)) compared with low CT cyclers. For example, most low CT cyclers across countries reported strong agreement that there is clear evidence for a connection between optimal AT initiation and improved patient outcomes (range: UC, 50.0%–88.0%; CD, 50.0%–87.9%) compared with high CT cyclers (range: UC, 34.8%–50.0%; CD, 31.3%–54.5%). The exception was for low CT cyclers from Germany treating patients with UC (14.3%).
Drivers for AT initiation differed between gastroenterologists who reported high versus low CT cycling for patients with UC (Supplemental Figure 3(a)). Low CT cycling gastroenterologists assigned higher importance to patient preference (+15.7%) and peer recommendations (+12.9%) compared with high CT cyclers. By contrast, high CT cycling gastroenterologists assigned higher importance to prior experience with the treatment (+8.1%) and exhausting other treatment options (+8.9%) compared with low CT cyclers. Additional drivers that were more influential for gastroenterologists who reported high CT cycling were patient-reported quality of life, insurance coverage/co-pay requirement, and safety profile. Additional drivers that were more influential for gastroenterologists who reported low CT cycling were reimbursement, comorbidities/concomitant medications, treatment cost, and route of administration. In CD, there were fewer differences between high and low CT cyclers as compared with the differences between high and low CT cyclers in UC (Supplemental Figure 3(b)).
Discussion
Overall, the majority of surveyed gastroenterologists treating patients with UC or CD were high cyclers, preferring to prescribe at least two cycles of CT that could last from 6.2 to 15.2 months, depending on whether a third CT cycle was utilized or not. This suggests that, despite many patients with UC or CD ultimately receiving AT either as monotherapy or in combination with CT, the majority of these patients only received AT after a significant delay. Concordantly, patient record forms analyzed here indicated that those currently receiving AT tended to have a longer time from initial diagnosis compared with those receiving CT. In all countries, the results of the quantitative survey indicated that AT prescription was less likely for patients with UC versus CD. Taken together, these results among surveyed gastroenterologists from Germany, Italy, the United Kingdom, and the United States suggest the prevalence of treatment algorithms for exhausting CT options before initiating AT.
Clinical beliefs contribute to AT prescription behavior among gastroenterologists
Differences in CT and AT use observed within countries suggest that clinical beliefs are likely to play a determining role in prescribing behavior. In our analysis, high CT cyclers generally showed less agreement with the statement that there is clear evidence supporting the connection between optimal AT initiation and achievement of improved outcomes, both in UC and CD, compared with low CT cyclers. This suggests a potential unmet need for better dissemination of evidence supporting AT use. Unsurprisingly, in both UC and CD, high CT cyclers also generally showed a lower agreement than low CT cyclers with the statements about the importance of initiating AT immediately after diagnosis and initiating AT as early as possible to optimize treatment and achieve improved long-term patient outcomes. Furthermore, high CT cyclers reported a higher agreement with the statement about delaying initiation of innovative therapies until after other therapies have been delivered to the patient, compared with low CT cyclers, for the treatment of both UC and CD. Despite the emerging trends described previously, the differences between high and low CT cyclers varied based on country of origin and by IBD type (UC or CD). Overall, these findings highlight that prescribing behavior appears to be strongly motivated by clinical beliefs.
The actual drivers of AT patient initiation across this cohort of gastroenterologists were also evaluated. Among AT initiation drivers in UC, familiarity with treatment and exhausting other treatment options were more important for high CT cyclers than low CT cyclers, supporting the hypothesis that prescribing behavior in high CT cyclers is driven by the need to exhaust CT options before initiating AT. In contrast, patient preference and peer recommendations were more important for low CT cyclers than high CT cyclers in UC, supporting the finding that low CT cyclers are more familiar with shared decision-making and are more influenced by peer knowledge sharing networks, which may explain more optimal CT use in these gastroenterologists. For CD, AT initiation drivers were more similar between low and high CT cyclers compared with AT initiation drivers in UC. One potential explanation for this finding is that despite evidence illustrating the progressive nature of UC, physicians tend to perceive UC as being relatively more benign, easier to treat, and less progressive than CD.29–32
To date, there remains a lack of agreement regarding objective criteria defining optimal “early” AT initiation. Among the gastroenterologists surveyed here, there was no consensus on the ideal timing for AT initiation for UC and CD. One possible explanation is that optimal AT initiation depends on the treatment goal that physicians and patients are targeting. 2 Regardless, the existing evidence for the treatment of patients with CD, including clinical studies (e.g., PROFILE, REACT, CALM, LOVE-CD) and meta-analysis data, indicates superior patient outcomes when AT is initiated early in the disease course.7,13,14,33–35 The accumulating evidence for early AT initiation in patients with CD is reflected in the evolution of clinical practice guidelines. Specifically, the 2021 AGA guidelines on CD conditionally recommend early introduction of AT over delaying AT use until after the failure of 5-ASA and/or corticosteroids in moderate-to-severe CD. 17 Similarly, although the 2020 ECCO guidelines for treating CD recommended AT only after the failure of CT, the newest 2024 ECCO guidelines on CD recommend various ATs for both induction and maintenance therapy.2,16 Although the survey data collected here were from 2023 and likely reflect adherence to the 2020 ECCO and 2021 AGA CD guidelines, the stark shift under the 2024 ECCO guidelines provides an opportunity moving forward to highlight the value of early intervention with AT for optimal disease management in patients with CD.
In contrast, the efficacy of early AT initiation in UC remains a topic for debate.11,12,15,32,36 Nevertheless, the existing reports highlight the risk of UC complications associated with delays in diagnosis and receiving treatment.37,38 The 2020 and 2024 AGA guidelines for UC conditionally recommend the early use of AT over gradual step-up treatment after the failure of CT with 5-ASA in moderate-to-severe UC, although noting that step-up therapy with 5-ASA can be reasonably chosen in patients (especially those with less severe disease) who place higher value on the safety of 5-ASA and lower value on the efficacy of AT.10,18 Alternatively, the 2022 ECCO guidelines for UC recommend CT with systemic corticosteroids for induction of remission in moderate-to-severe UC (up to 3 months’ duration in a year). 3 ATs are strongly recommended for induction of remission only in patients who had an inadequate response or intolerance to CT. 3 These trends were reflected in the survey data collected in this study, where gastroenterologists reported greater prescription of AT in patients with CD versus those with UC. Ultimately, the vast amount of literature on AT treatment choices in IBD 39 and the limited data on comparative efficacy and safety are likely to lead to considerable practice variability.40,41
CT prescribing trends suggest suboptimal treatment for patients with UC and CD
In our survey, gastroenterologists did not report any clear choices for the first- and second-line CT. However, the prevalent use of 5-ASA in CD, including by 23.3%–66.6% of the gastroenterologists surveyed here, and despite clinical data and official guidelines recommending against it,2,17 illustrates persistent suboptimal disease management for patients with CD who may be eligible for AT. Similarly, corticosteroids have little confirmed effect on maintenance and endoscopic response in UC, and the side effects of corticosteroid use (especially in those receiving corticosteroids in high doses and/or for prolonged periods of time) are well known.3,42,43 Unsurprisingly, clinical guidelines caution against prolonged use of corticosteroids both in UC and CD,2,3,10,17 with the 2022 ECCO guidelines on UC specifying that corticosteroid courses should be restricted to a maximum of 3 months. 3 AT should be considered for any patient who requires more than a single course of systemic corticosteroids in a year.
Limitations of the study
This study had some limitations. Selection bias was inevitable. Only gastroenterologists who were willing and able to complete the survey were included. Additionally, the survey targeted gastroenterologists familiar with IBD care who had notable exposure to patients with IBD and were responsible for making treatment decisions, with the aim of including physicians who may not be IBD experts but nevertheless represent typical physicians in charge of management of patients with IBD. Furthermore, the survey was conducted among a small number of gastroenterologists and only those who had prior experience prescribing ATs, suggesting the results reported here may underestimate overall CT prescribing trends. Moreover, given the sample size of gastroenterologists surveyed in this study, these findings were useful for providing insight into the clinical beliefs of gastroenterologists who utilize low versus high CT cycling but may not accurately reflect the real-world practice of gastroenterologists overall. This is supported by the observation that AT use in the United States captured in our retrospective, self-reported survey was markedly higher than that reported in the claims analyses of patients with UC and CD in the United States (60.0% in UC and 66.0% in CD in our study vs 5.9% in UC and 14.4% in CD as reported by Siegel et al. 20 ). Additionally, while input from the qualitative survey informed the identification of relevant clinical beliefs related to CT and AT prescribing behaviors (Supplemental Appendix, quantitative survey, Q2a–b), we cannot rule out the possibility of other factors that were not surveyed here.
Another limitation is the dynamic IBD treatment landscape; a survey conducted in 2023 may no longer be wholly reflective of the current treatment landscape as new data in IBD continues to influence prescribing behaviors. On the other hand, the current survey also had major strengths. Although survey-based designs are categorized as lower-level evidence compared with interventional studies, they provide critical insights into real-world perceptions and practices, which cannot be captured through experimental designs. Comparisons between low and high CT cyclers within the sampled group are less likely to be influenced by the aforementioned selection bias, which contributes to the study’s strengths. Moreover, the survey included gastroenterologists from both North America and Europe, which increases the generalizability of the results. Finally, the survey employed a multimodal approach to assess AT initiation barriers that included a quantitative approach utilizing behavioral analysis.
Conclusion
The results of this survey of gastroenterologists from Germany, Italy, the United Kingdom, and the United States revealed no clear choice for first- or second-line CT and no consensus on the ideal timing of AT initiation. Additionally, behavioral analyses in surveyed gastroenterologists showed that clinical beliefs played a determining role in prescribing behaviors for AT versus CT. Overall, the survey results highlighted frequent suboptimal treatment of patients with IBD and confirmed that barriers to AT use persist, highlighting an unmet need for continued physician education and alignment with clinical guidelines, as well as the need for continued advocacy for AT in patients with IBD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848261428791 – Supplemental material for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease
Supplemental material, sj-docx-1-tag-10.1177_17562848261428791 for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease by Tim Raine, Christian Agboton, Alex Geddes, Tom Halmos, David Laharie and Bram Verstockt in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848261428791 – Supplemental material for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease
Supplemental material, sj-docx-2-tag-10.1177_17562848261428791 for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease by Tim Raine, Christian Agboton, Alex Geddes, Tom Halmos, David Laharie and Bram Verstockt in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-eps-3-tag-10.1177_17562848261428791 – Supplemental material for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease
Supplemental material, sj-eps-3-tag-10.1177_17562848261428791 for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease by Tim Raine, Christian Agboton, Alex Geddes, Tom Halmos, David Laharie and Bram Verstockt in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-eps-4-tag-10.1177_17562848261428791 – Supplemental material for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease
Supplemental material, sj-eps-4-tag-10.1177_17562848261428791 for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease by Tim Raine, Christian Agboton, Alex Geddes, Tom Halmos, David Laharie and Bram Verstockt in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-eps-5-tag-10.1177_17562848261428791 – Supplemental material for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease
Supplemental material, sj-eps-5-tag-10.1177_17562848261428791 for Patterns of conventional therapy use and advanced therapy initiation drivers in inflammatory bowel disease by Tim Raine, Christian Agboton, Alex Geddes, Tom Halmos, David Laharie and Bram Verstockt in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We thank the responders who participated in the survey and the study investigators. Special thanks to Rob Dubman, Market Insights Lead at Takeda, who contributed to the analysis and interpretation of the data. Medical writing support was provided by Claire Line, PhD, Milda Tyler, PhD, and Chase Hellmer, PhD, of Envision Catalyst, an Envision Medical Communications agency, a part of Envision Pharma Group, funded by Takeda Development Center Americas, Inc.
Declarations
Supplemental material
Supplemental material for this article is available online.
