Abstract
Cirrhotic cardiomyopathy (CCM) is a distinct form of cardiac dysfunction in patients with advanced chronic liver disease, defined by subclinical diastolic impairment and, less commonly, systolic dysfunction, in the absence of primary cardiac disease. The entity reflects a dynamic interplay between structural, functional, and biochemical alterations in the cirrhotic heart, compounded by the systemic effects of portal hypertension and inflammation. Recent advances in diagnostic criteria, most notably the 2020 CCM Consortium (CCC-2020) definitions, have refined diagnostics and aligned CCM with contemporary concepts of heart failure (HF). Nevertheless, conventional echocardiography provides only a static assessment, often underestimating cardiac vulnerability, whereas stress testing and novel imaging modalities highlight the central role of blunted cardiac reserve as a predictor of adverse outcomes. CCM is a stress-intolerant phenotype, with important implications in liver transplantation (LT), both peri- and post-operatively. Subclinical CCM is not considered a contraindication to LT, but accumulating evidence associates it with increased risk of post-LT HF and major adverse cardiac events, even though long-term survival is generally unaffected. Emerging data also challenge the long-held assumption of universal reversibility post-LT, suggesting variable trajectories of recovery that may extend over years. In the era of metabolic-associated steatotic liver disease, the intersection of persistent cardiac dysfunction with cardiometabolic multimorbidity further underscores the need for proactive surveillance post-LT and tailored management strategies. This review synthesizes current knowledge on CCM pathophysiology, assessment, and transplant implications, and highlights future research priorities, including risk prediction models, dynamic functional testing, and the potential role of HF therapies.
Plain language summary
Keywords
Introduction
Cirrhotic cardiomyopathy (CCM) is characterized by subclinical left ventricular (LV) diastolic dysfunction (DD), and less commonly systolic dysfunction (SD) in patients with advanced chronic liver disease (ACLD), in the absence of primary cardiac disease. Its pathophysiology is driven by impaired hepatic synthetic function and systemic inflammation, leading to structural and functional cardiac dysfunction. The 2020 CCM Consortium (CCC-2020) criteria have redefined diagnostic standards, 1 aligning with contemporary concepts of heart failure (HF) and allowing for a more consistent approach to assessment. However, traditional resting echocardiography only provides a static snapshot and may underestimate the degree of cardiac dysfunction.2,3 Patients may appear compensated at rest, but reveal critical limitations in contractile or diastolic reserve when challenged. With emerging tools, we can now phenotype cardiac reserve more precisely, linking blunted cardiac reserve (BCR) to clinical events such as peri- and post-liver transplantation (LT) cardiovascular disease and hepatorenal syndrome-acute kidney injury (HRS-AKI).3,4 At the same time, advances in imaging and molecular biology allow us to connect structural and metabolic signatures involving fibrosis, inflammation, and nicotinamide adenine dinucleotide (NAD+) depletion, 5 with functional reserve. This dual perspective reframes CCM as a dynamic, stress-intolerant state and opens the door to targeted interventions before and after LT.
In this review, we cover the current and central concepts (Figure 1) in CCM, in the context of LT. We detail the pathophysiology of CCM as it is currently understood, then outline the assessment of CCM pre-LT including echocardiography, other cardiac imaging and biomarkers, and the implications of CCM on mortality, perioperative complications, and cardiac outcomes after LT. Finally, we highlight the important considerations in post-LT monitoring and management of CCM. While there are several excellent recently published reviews on this topic, this review offers a detailed and structured approach to post-LT management.
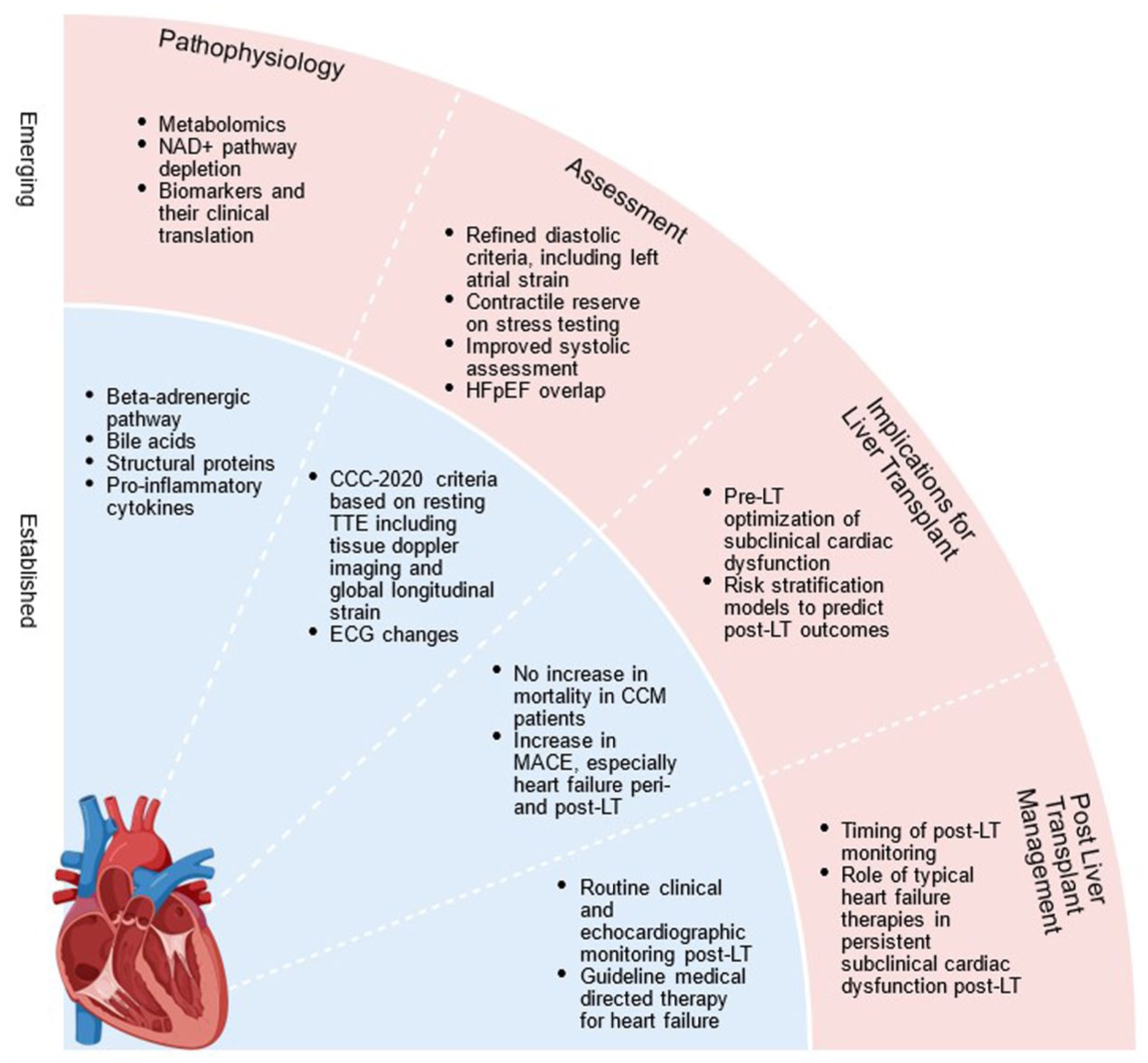
Central concepts of CCM in the context of LT.
Pathophysiology
The pathogenesis of CCM involves complex myocardial structural, functional, and biochemical perturbations at the cellular level, perpetuated by the hemodynamic alterations of ACLD and portal hypertension (Figure 2). Two main pathways underpin the development of CCM: (1) impaired hepatic synthesis and metabolism of lipids, carbohydrates, bile acids (BAs), and structural cardiac proteins and (2) portal hypertension and resultant gut bacterial translocation, endotoxemia, and systemic inflammation. Much of this mechanistic insight has been derived from animal models, with increasing translational work in human studies.

Pathophysiology of cirrhotic cardiomyopathy.
β-Adrenergic pathway dysfunction
Attenuated cardiac responsiveness to catecholamines has long been recognized in cirrhosis, 6 with impaired synthesis or dysfunction of the glycoprotein β-adrenergic receptor, and desensitization in the setting of chronic sympathetic nervous system (SNS) activation. 7 Early cirrhotic rat models demonstrated reduced density and selective downregulation of myocardial β-adrenergic receptor, 8 resulting in impaired inotropic responsiveness. In a more recent human study, 9 Ma et al. identified elevated levels of anti-β1-adrenergic receptor antibodies in patients with CCM, and this correlated with functional markers of cardiac impairment, suggesting an alternative mechanism for β-adrenergic dysfunction.
The hemodynamic implications of β-adrenergic dysfunction are highlighted by the “therapeutic window” for non-selective beta-blockers (NSBB) in ACLD and portal hypertension. In patients with refractory ascites, systemic vasodilation peaks and cardiac compensation rely on SNS overdrive. 10 When this is blunted by NSBB, cardiac output (CO) falls and systemic perfusion, particularly renal, suffers, negating the benefits of NSBB. β-Adrenergic function is crucial for cardiac responsiveness and compensation, and its downregulation in CCM has a significant impact on contractile reserve.
Cardiac structural proteins and lipid alterations
Myocardial contractility in CCM is further impaired by alterations in structural proteins, at the level of the cardiac myofilaments. Myosin heavy chains (MHC) play a central role in myocardial contraction, with the α-MHC isoform generating faster, stronger contractions than the β-MHC isoform. In cirrhotic rats, a shift from α to β-MHC has been observed, 11 possibly induced by BAs12,13 and contributes to SD. Alterations to titin, a giant sarcomeric protein involved in passive elastic recoil, may play a role in DD. Downregulation of protein kinase A, a titin regulator, along with disrupted collagen configuration, 14 has been observed in cirrhotic rat hearts.
The biophysical properties of the cardiac plasma membrane also altered in hepatic insufficiency. Reduced membrane fluidity, due to an increased cholesterol-to-phospholipid ratio, impairs β-adrenoreceptor function,15,16 thus decreasing cAMP production and blunting calcium release from the sarcoplasmic reticulum (SR). 17 Cannabinoid receptors, L-type calcium channels, 17 and Na+/K+ATPase15,16 are also affected and disturbance to their structure and function further contributes to electrophysiological and contractile dysfunction.
Bile acids
BAs, elevated in the cirrhotic state, exert multiple inhibitory effects on the myocardium. 18 Beyond inducing the α-to-β MHC switch, they disrupt calcium release from the SR, 19 switch myocardial metabolism from fatty acid oxidation to glucose utilization, 12 and upregulate selective cannabinoid-2 receptors, 20 resulting in negative inotropy. Human studies have linked elevated BAs to LV dysfunction and electrophysiological abnormalities. 21 Similarly, our group recently observed higher plasma levels of taurodeoxycholic acid, a secondary BA, in CCM patients 5 compared to those without.
A positive association has also been shown between BAs and transthoracic echocardiography (TTE) indices such as LV volumes 22 and left atrial volume index (LAVI),22,23 and abnormal myocardial extracellular volume (ECV) on cardiac magnetic resonance imaging (CMR), an indicator of diffuse myocardial fibrosis (DMF). 24 In children with biliary atresia, BA levels >152 µmol/L conferred an eight-fold increased odds of abnormal LV geometry on TTE. 21 While there is clearly an interplay between BAs and cardiac function, their precise role in CCM pathogenesis remains to be fully defined, as well as the potential role of BA sequestrants as a therapeutic agent.
Systemic inflammation
Portal hypertension leads to splanchnic congestion, bacterial translocation, and endotoxemia, triggering a systemic proinflammatory state with TNFα, IL-β1, and IL-6 predominating. Increased levels of these cytokines have been shown in cirrhotic rat myocardium.25,26 IL-6 has been linked to decreased expression and function of calcium handling proteins, 27 and TNFα has been observed to depress contractility 26 through alterations in L-type calcium channels. In humans, IL-1β has been associated with impaired LV systolic response to stress. 28
Nitric oxide (NO), endocannabinoids, and galectin-3 are other inflammation-related mediators implicated in CCM. Proinflammatory cytokines also induce NO overproduction. Elevated NO has been reported in animal models of CCM, 29 inhibiting cardiac contraction via second messenger cGMP.25,28 Cytokines also drive the production of endocannabinoids, which act on cannabinoid receptors on cardiomyocytes26,30,31 to depress contractility. The tachycardia-stress-induced release of endocannabinoids may, in part, explain the impaired contractility with stress in CCM. 31 Galectin-3 is an important biomarker in HF of any etiology. 32 It is derived from macrophages, which are increased in cirrhotic hearts, and stimulates TNFα production,33,34 a known driver of contractile dysfunction in CCM.25,26,34,35 Galectin-3 also upregulates collagen I and increases collagen I/III ratio, 36 contributing to DD.14,34
Metabolic alterations
There is increasing interest in metabolomics in HF with preserved ejection fraction (HFpEF), a phenotypically similar entity to CCM. Metabolomic profiling of the HFpEF heart has revealed reduced free fatty acid use, increased complex lipid generation, and protein catabolism. 37 Others have defined the role of metabolic inflammation 38 and distinguished between HFpEF and other forms of HF on metabolomic profiling.38–41 Depletion of myocardial NAD+, a critical driver of cellular metabolism, has been demonstrated in human and murine models of HFpEF.42,43 Improvement in cardiac function has been proven with repletion of NAD+ precursors.44–47 Our group recently linked plasma NAD+ depletion to DD in patients with ACLD, 5 highlighting a novel and potentially targetable metabolic pathway in CCM.
Altered hemodynamics
Portal hypertension is a state of splanchnic vasodilation, driven by the overproduction of aforementioned vasodilatory mediators such as NO, carbon monoxide, and endocannabinoids. These substances, shunted into the systemic circulation via portosystemic collaterals and intrahepatic shunting, 48 contribute to systemic vasodilation and reduced peripheral vascular resistance. The resulting decrease in effective circulating volume due to splanchnic pooling activates compensatory mechanisms, including the renin-angiotensin-aldosterone system, SNS activation, and sodium and water retention, leading to a hyperdynamic circulatory state. Initially, CO increases to maintain arterial pressure. However, with disease progression, this compensatory mechanism becomes insufficient. This hemodynamic tipping point is believed to play a pivotal role in the pathogenesis of HRS-AKI.49,50 However, this paradigm has recently been challenged. 51 A study using MRI demonstrated elevated CO in patients with HRS-AKI 52 and another, using dobutamine stress echocardiography (DSE), suggested that it is not the resting CO, but the inability to increment this during physiological stress, that predisposes to HRS-AKI. 4 In CCM, reduced contractility further impairs this response to stress. Again, our group demonstrated that patients with BCR (an increase of <25% above baseline) had more severe liver disease and higher baseline CO. These findings suggest that patients with high resting CO may already be operating near their maximal cardiac reserve, and this may serve as a surrogate marker for CCM.3,51,53
Supporting this concept, a recent study utilizing point of care echocardiography (POC-Echo) in patient with acute-on-chronic liver failure 54 found that a high cardiac index (CI) at baseline with a lower CI after 72 h, so an inability to sustain an augmented CO due to poor cardiac reserve, was predictive of mortality. CCM, in this cohort, was associated with circulatory failure. Similarly, POC-Echo has been used in patients with HRS-AKI to predict terlipressin non-response over 72 h, which occurred more commonly in CCM patients with poor cardiac reserve. 55 Interestingly, 12 weeks of continuous long-term terlipressin infusion has been shown to improve contractile reserve, probably by increasing preload and optimizing the Starling curve, 56 providing evidence of reversibility of one of the functional components of cardiac dysfunction in cirrhosis. Diastolic indices, on the other hand, did not reverse with terlipressin.
Pre-LT assessment
The first diagnostic criteria for CCM were established in 2005 following the World Congress of Gastroenterology (WCOG) in Montreal. In 2020, the Cirrhotic Cardiomyopathy Consortium (CCC) updated these definitions to incorporate advancements in TTE, including speckle-tracking strain imaging for systolic assessment, and tissue Doppler imaging (TDI) for diastolic evaluation. These revisions reflect modern concepts of HF, including the distinction between reduced and preserved left ventricular ejection fraction (LVEF). Using the CCC-2020 criteria, reported CCM prevalence ranges between 15% and 30%, 57 with variability reflecting which grades of DD were included and availability of global longitudinal strain (GLS). TTE remains the sole modality used to diagnose CCM, while other techniques such as stress testing, electrophysiological studies, cardiopulmonary exercise testing (CPET), CMR, and biomarkers require future validation.
It is important to acknowledge the phenotypic and diagnostic overlap between CCM and HFpEF. Their diagnostic criteria are nearly identical, and with the rise of metabolic dysfunction-associated steatotic liver disease (MASLD), patients with cirrhosis now exhibit overlapping clinical features with HFpEF patients. 58 CCM and obesity-related HFpEF share hemodynamic similarities, both being high-output states. There is debate about whether CCM represents a distinct pathophysiological entity, or represents a HFpEF phenotype. 59 Interestingly, in our recent large retrospective study, we did not find associations between CCM and traditional HFpEF risk factors such as obesity, diabetes mellitus, and hypertension. 60 However, our understanding of the impact of metabolic comorbidities on CCM phenotypes may evolve in the future. The validity of HFpEF diagnostic tools in CCM requires further exploration, as well as the utility of HFpEF therapies in this population.
Transthoracic echocardiography—Systolic assessment
Historically, LV systolic function has been assessed by LVEF. However, LVEF ⩽50% is rare in pre-LT patients61–65 and cirrhotic patients,66–68 because it is overestimated by the hyperdynamic circulation 28 of ACLD. In fact, it may be normal or even increased, even when cardiac dysfunction is present. The LVEF threshold for diagnosing SD in CCM was reduced from 55% in the 2005 WCOG Criteria to 50% in the CCC-2020 criteria. However, these thresholds may not assure normal systolic function in patients with decompensated liver disease. In their large cohort of 2799 LT patients, Kwon et al. 61 found that LVEF ⩽60% is strongly associated with higher post-LT mortality in the Model for End-Stage Liver Disease (MELD) ⩾20 group. Thus, the optimal LVEF thresholds to diagnose CCM and prognosticate are ill-defined, and the definition of a “normal” LVEF in advanced liver disease patients requires reappraisal.
To address this limitation, the CCC-2020 criteria introduced GLS as a novel metric of systolic function. During systole, myocardial fibers shorten, and GLS quantifies this shortening as a negative percentage, representing the relative change in length from diastole. An absolute GLS <18% is considered abnormal. The data on strain in CCM vary, 1 and in retrospective studies of LT populations, GLS is often unavailable.65,69 In one of the few studies reporting GLS in pre-LT patients, 70 only 1.2% of patients met this criterion for SD. This is likely reflective of the predominance of DD in CCM. It may also be attributed to the progressive hyperdynamic circulation and increased LV loading that occurs as liver disease advances. When preload increases, sarcomeres are longer at the start of systole, which can increase strain values regardless of contractility. Additionally, when afterload is low in the setting of systemic vasodilation, the LV shortens more easily against lower resistance. In ACLD, these factors may mask LV dysfunction when using GLS. 71 Meta-analyses have shown reduced GLS in the cirrhotic population,72,73 but its accuracy in the presence of a hyperdynamic state needs validation. Its potential role in monitoring post-LT when hemodynamics has normalized needs prospective evaluation.
Transthoracic echocardiography—Diastolic assessment
The CCC-2020 criteria for DD (Figure 3) parallel the 2016 American Society of Echocardiography (ASE) update 74 and are less reliant on volume-dependent measures such as the ratio of early (E) to late (A) ventricular filling velocity. TDI measures myocardial tissue velocity rather than blood flow velocity, so it is less influenced by volume status. Early mitral annulus diastolic velocity (e′) is one of the most reliable measures of diastolic function, with lower values indicative of impaired LV relaxation. The ratio of mitral inflow diastolic velocity (E) to e′ correlates with pulmonary capillary wedge pressure and is accepted as a surrogate for LV filling pressure, with values increasing with DD. Left atrial (LA) dilation is a measure of both chronicity and severity of DD, with LAVI of >34 mL/m2 considered abnormal. Tricuspid regurgitant (TR) peak velocity reflects left-sided filling pressures but should be interpreted with caution in patients with possible portopulmonary hypertension, 1 and in isolation is non-specific.
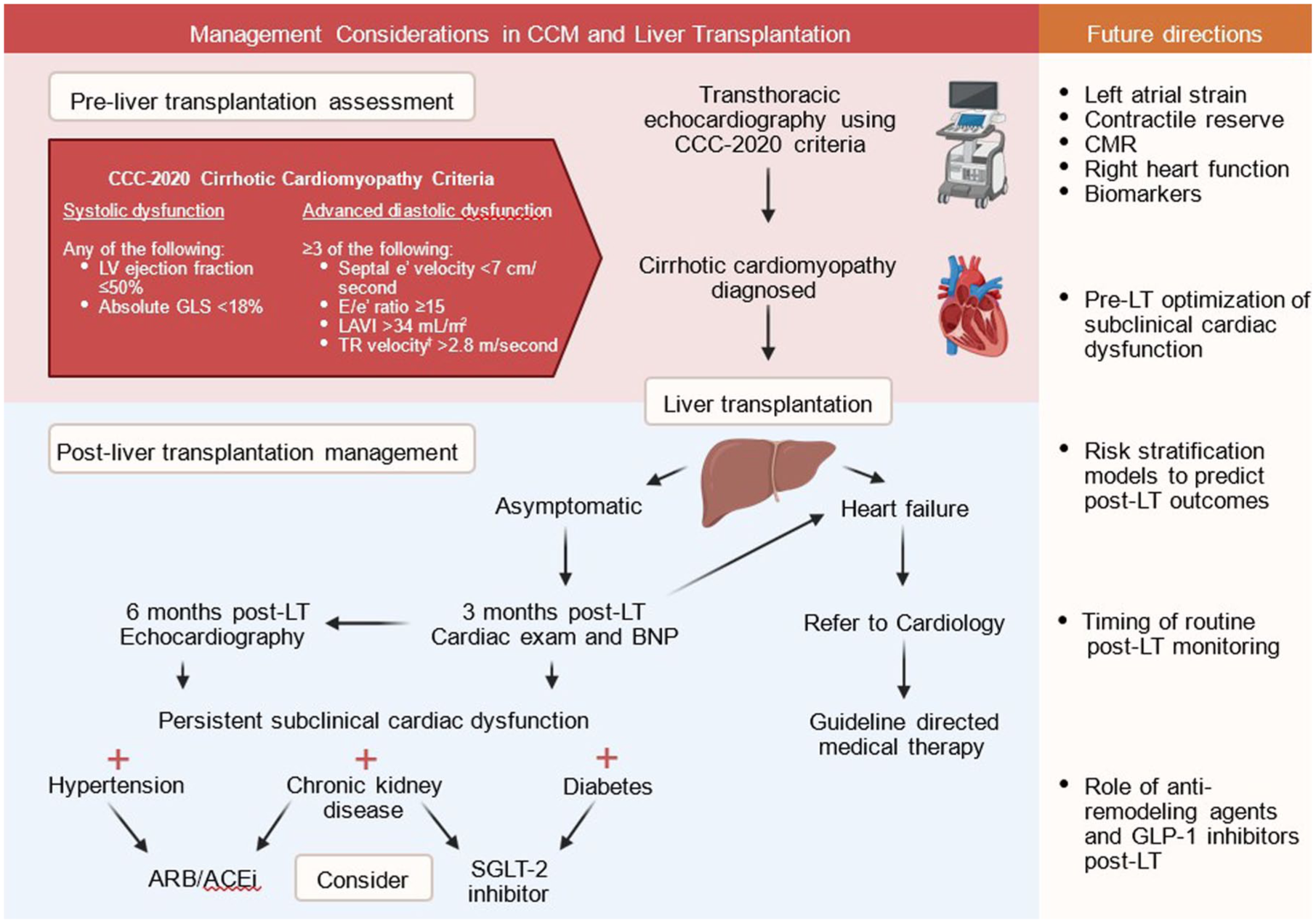
Management considerations pre- and post-LT in patients with CCM.
It should be noted that the ASE updated their recommendations for the evaluation of LV diastolic function 75 in 2025, and there are now some distinctions from the CCM criteria. The ASE 2025 diagnostic algorithm no longer includes TR peak velocity (although this is still used for DD grading), and considers e′ velocity as the first index in the algorithm given its direct dependence on LV relaxation. Age-specific ranges are also offered for e′ velocity. This new update allows for a diagnosis of DD at earlier stages of impairment, while the diagnostic criteria for CCM reflect only advanced DD. The application of this revised approach to diastolic assessment will need evaluation in CCM, acknowledging that the CCM criteria should be dynamic.
There is also growing interest in the additive value of LA strain in diastolic assessment. Meucci et al. 76 found LA strain to be independently associated with post-transjugular intrahepatic portosystemic shunt mortality and provided incremental prognostic value when compared to models using the existing criteria. Of the patients with indeterminate diastolic function, 30% were reclassified to advanced DD using LA strain. Skouloudi et al.77,78 linked LA strain to an increased likelihood of death or LT, and suggest it may be a novel sensitive marker of DD in CCM. Additionally, right heart function is affected by chronically elevated left-sided filling pressures, and the utility of right heart parameters on TTE in CCM is a potential area for future research. 79
Transthoracic echocardiography—Stress testing
BCR on stress testing was included as a feature of SD in the WCOG 2005 criteria. However, due to the lack of universal definition, its clinical utility was limited, and it was omitted from the CCC-2020 revision. 1 It is well established that CCM is often subclinical at rest, and may only become apparent during physiologic stress due to the inability to augment CO. 2 Recently, BCR on DSE was shown to be the strongest independent risk factor for post-operative major adverse cardiac events (MACE), HF exacerbations, and 30-day mortality post-LT 3 in a large study of 481 LT recipients. Notably, the addition of BCR to the CCC-2020 criteria improved MACE prediction. Furthermore, in patients not fulfilling CCM-2020 criteria, BCR alone conferred a five-fold increased risk of post-operative MACE. It is acknowledged that the current CCC-2020 criteria are not definitive. 80 CCM is clearly a dynamic, stress-intolerant phenotype of HF, defined by impaired cardiac reserve. Resting TTE alone may fail to identify CCM, or risk stratify accurately in the LT context. Stress testing, including contractile and diastolic reserve evaluation, is a crucial part of assessment for CCM, and may reveal cardiac vulnerability when resting indices have been normal or even supranormal. Additionally, abnormal diastolic reserve on exercise testing can be identified by an increasing E/e′, indicating raised left-sided filling pressure. 74 This is seen in patients with HFpEF, 81 and may prove to be a useful marker of subclinical DD in CCM. 1
Electrocardiography
Electrocardiography (ECG) abnormalities are frequently observed in patients with cirrhosis 82 but are of limited value in CCM. QTc prolongation (⩾440 milliseconds (ms)) occurs in over 50% of patients with cirrhosis,83,84 tends to worsen with advancing liver disease85,86 and reverses in up to 73% of patients after LT.84,86 Prolonged QTc has consistently been identified as an independent predictor of mortality in end-stage liver disease.85,87,88 QTc ⩾480 ms was associated with a five-fold increase in the risk of post-LT cardiac arrest and ventricular arrhythmia, 89 and the cardiac arrest risk index (CARI) risk score was developed, including QTc, age, sex, and MELD, to identify patients at risk. However, QTc does not correlate with post-LT mortality. 84 While it has long been considered the electrophysiological hallmark of CCM, in a study of 439 patients with cirrhosis, 65% had prolonged QT and yet no differences in LV or atrial remodeling, or resting systolic and diastolic function were observed across QTc groups. 83 These findings suggest that CCM and QT prolongation may represent distinct clinical phenomena, with different underlying pathophysiological mechanisms. As such, its place in the CCM paradigm is the subject of ongoing debate.
Cardiopulmonary exercise testing
CPET can be used in cardiovascular assessment for LT to assess functional capacity. 90 However, access is limited due to the requirement of complex infrastructure, and it can be practically challenging in decompensated patients. Peak oxygen consumption (pVO2) has been shown to predict pre- and post-LT mortality, 91 but its role in CCM specifically has not been evaluated.
CMR imaging
CMR offers advanced myocardial tissue characterization, using late gadolinium enhancement (LGE) and ECV quantification. LGE identifies advanced myocardial scarring, while ECV represents DMF at earlier stages. Myocardial fibrosis impairs LV compliance and contributes to DD. Very early autopsy studies identified DMF in the hearts of patients who had died from advanced liver disease. 92 Higher ECV on CMR has been observed in patients with cirrhosis, correlating with increasing Child-Pugh scores. 24 In a small study of 33 patients undergoing CMR before and after LT, 93 ECV was elevated in two-thirds of patients, while LGE was present in only one patient. Importantly, ECV significantly decreased 1-year post-LT, suggesting there is potential reversibility of this structural component of myocardial dysfunction. While promising, prospective trials are needed to clarify the role of CMR in CCM diagnosis, cardiovascular assessment pre-LT, and predicting reversibility post-LT.
Blood-based biomarkers
B-type natriuretic peptide (BNP) and N-terminal prohormone BNP (NT-proBNP) are firmly established biomarkers in HF, released by cardiomyocytes in response to strain. BNP is correlated with disease severity and portal hypertension, 94 so it is relatively non-specific in decompensated liver disease, as it can be elevated even when cardiac function is normal. Elevated BNP has been linked to an increased risk of post-LT mortality when measured in the first 3–7 days post-operatively,94,95 however, this is confounded by its association with liver disease severity, fluid overload, and renal failure. Experimentally, galectin-3 and soluble suppression of tumorgenicity-2 may reflect cardiac remodeling but are also elevated in liver inflammation and fibrosis so again are non-specific.1,96 There are currently no blood-based biomarkers validated for use in CCM diagnosis.
Implications for LT
When subclinical, CCM is not considered a barrier to LT. When overt HF is present, a LVEF ⩽40% is generally considered a contraindication to LT, 97 however, the scarcity of patients with LVEF ⩽50% in the LT literature suggests that this threshold is even more conservative in clinical practice. There are currently no specific disease-modifying therapies for CCM, and no studies investigating whether pre-LT optimization of subclinical CCM impacts post-LT outcomes. However, the impact of pre-LT CCM on perioperative complications, cardiovascular events, and mortality has been widely studied and is summarized below and in Table 1.
Prevalence of CCM and its association with mortality and major adverse cardiac events in cohorts of LT recipients.

No GLS available.
DD diagnosed if 2/4 criteria met.
BCR defined as an increase in cardiac output <25% above baseline in response to low-dose dobutamine infusion.
Increased risk of 30-day death (p = 0.49) in patients with BCR, but not at 90 or 180 days.
All-cause mortality assessed as part of composite endpoint.
ACS, acute coronary syndrome; ASE, American Society of Echocardiography; BCR, blunted cardiac reserve; CCC, Cirrhotic Cardiomyopathy Consortium; CCM, cirrhotic cardiomyopathy; DD, diastolic dysfunction; EACVI, European Association of Cardiovascular Imaging; GLS, global longitudinal strain; HF, heart failure; IHD, ischemic heart disease; LT, liver transplantation; MACE, major adverse cardiovascular events; MI, myocardial infarction.
Perioperative complications
LT results in massive hemodynamic stress. The clamping of the inferior vena cava and portal vein, blood loss, and reperfusion all cause rapid fluctuations in preload and afterload that challenge cardiac reserve and responsiveness. Despite this, major perioperative outcomes do not appear significantly affected by CCM. Observational studies have shown that the duration of vasopressor therapy and mechanical ventilation is unchanged in patients with CCM.63,98,99 Intensive care unit length of stay may be slightly longer in CCM patients60,100,101 although overall length of stay appears unaffected.60,98,100 Mohammadi et al. 102 reported that cardiac dysfunction was predictive of increased perioperative morbidity; however, their definition of morbidity and the criteria used to define DD were broad. 102 In a recent multicenter study of 308 LT recipients, Liu et al. 99 found no association between CCM and reperfusion syndrome, nor any other MACE, including HF, arrhythmia, cardiac arrest, or cardiac death, within 30 days of LT. In our large Australian cohort, 60 patients with CCM had a higher incidence of perioperative MACE (HF, arrhythmia, cardiac death, and/or intraoperative arrest) within 30 days of LT. However, this was driven exclusively by cases of HF, all of which resolved with standard therapy and did not require long-term follow-up. Given the massive hemodynamic stress of LT, these findings are reassuring, suggesting subclinical CCM does not confer an increased risk of major perioperative morbidity at the time of LT. It is important to note that patients with either HF with reduced ejection fraction (HFrEF), or LVEF ⩽50%, are often excluded from LT in both study populations and clinical practice. As such, these data reflect outcomes in patients with DD rather than overt SD.
T-cell-mediated rejection (TCMR) is another important early complication after LT. Mittal et al. 103 linked worsening DD to TCMR risk in the first year after LT. Their proffered mechanism of proinflammatory cytokine upregulation in DD is intriguing, however, these findings have not been replicated. In unpublished data from our cohort, there were no differences in biopsy-proved rejection rates between patients with or without CCM.
Major adverse cardiac events
Several studies, summarized in Table 1, have demonstrated at least a two-fold increase in post-LT MACE among patients meeting the CCC-2020 criteria for CCM.60,65,70,99,104 However, comparisons between studies are limited by heterogeneity in endpoint definitions and follow-up durations. In contrast, Ali et al. 69 and Marella et al. 98 reported no increased MACE risk using CCC-2020 criteria, though Ali et al. included all-cause mortality as part of their MACE, while Marella et al. restricted their follow-up to 30 days. In our Australian cohort, CCM was a strong predictor of post-LT MACE, driven purely by HF. 60 In the older literature, DD had been linked to post-LT HF,62,105 although these used now-outdated diagnostic criteria. More recent studies have yielded mixed results. Spann et al. 65 found no significant association between CCM and HF individually, while Izzy et al. 70 reported a non-significant trend. Notably, these two studies pooled advanced DD with indeterminate grade, possibly diluting their HF incidence. Our inclusion of only advanced LVDD likely strengthened our association. Subgroup analysis further demonstrated that patients with indeterminate DD were not at increased risk of MACE or HF, which may narrow patient selection for routine post-LT monitoring. Mechanistically, the LA dilation accompanying DD could predispose to atrial arrhythmias. While Liu et al. 99 found an increased risk of atrial fibrillation with increasing LAVI, |our data did not support this association. 60 Preoperative recognition of CCM may enhance the uptake of targeted surveillance post-LT and early detection of post-LT HF, as discussed below.
Predictive value of individual echocardiographic parameters
The prognostic value of individual echocardiographic parameters within the CCC-2020 criteria has been evaluated in several studies. Among these, a septal e′ velocity <7 cm/s has been shown as the most robust predictor of MACE 65 and has been associated with a three or fourfold increased risk of HF.60,65,99 Reduced e′ velocity has also been linked to increasing all-cause mortality,65,101,104 however, e′ velocity reduces with age and not all studies adjusted for this as a confounder. An elevated E/e′ has similarly been shown to predict HF,60,62,65 while increased LAVI has been associated with MACE, arrhythmia, 99 HF,62,70 and death within first year of LT. 70 GLS, though not extensively studied in this context yet, was shown by Izzy et al. 70 to predict HF when the absolute GLS was <20.6%. However, there is a lack of evidence for its prognostic utility, particularly compared to diastolic parameters.
Mortality after LT
Most studies have found no difference in post-LT survival63,65,69,70,98,100,102,104 (Table 1) between patients with and without CCM. However, as noted earlier, patients with clinically overt HF usually do not proceed to LT, so these data pertain to subclinical CCM. One earlier study reported a significantly increased all-cause mortality and graft failure in patients with Grade 2 or Grade 3 DD, 103 however, this has not been consistently replicated. Despite associations between CCM and MACE, the absence of a long-term survival difference suggests that functional recovery or reversibility may be occurring in the long term. In a large cohort of 3244 patients with end-stage liver disease, Shin et al. 106 used the Heart Failure Association-PEFF (HFA-PEFF) score to identify patients with a high probability of HFpEF. This group demonstrated reduced survival at 1- and 12-year following LT. Although the HFA-PEFF score does not specifically assess for the presence CCM, it incorporates several risk factors such as female sex, age, hypertension, and dyslipidemia in addition to echocardiographic findings, and serves as a valuable prognostic tool. Its relevance may increase as the population of LT patients becomes older and more comorbid with the increasing incidence of LT for MASLD. 58
Effect of immunosuppression
Calcineurin inhibitors (CNIs) are the cornerstone of most immunosuppression regimens post-LT. In murine models, genetic inhibition of calcineurin has been shown to induce DD. 107 In human renal transplant recipients, discontinuation of CNIs has been associated with stabilization of DD. 108 In pediatric LT recipients, tacrolimus has implicated in the development of hypertrophic cardiomyopathy, 109 however, this association has not been observed in adults, 110 aside from isolated cases reports of tacrolimus-induced acute HF. 111 In a randomized controlled trial by Therapondos et al. 112 40 adult LT recipients were assigned to receive tacrolimus or cyclosporin-based immunosuppression. LV diastolic function deteriorated over the first 3 months following LT, but no significant difference in cardiac function was observed. To date, there is no clear evidence to suggest that CNIs or other immunosuppression agents influence the natural history of post-LT cardiac dysfunction. Accordingly, no specific immunosuppressive regimen is necessary in CCM patients.
Reversibility of CCM
CCM has long been considered reversible following LT, akin to other extrahepatic manifestations of cirrhosis such as autonomic and peripheral neuropathy, which often resolve with the restoration of liver function and the removal of the neurohormonal changes of portal hypertension. 113 One of the earliest studies examining reversibility demonstrated regression of ventricular wall thickness and improvements in both diastolic function and contractile reserve on stress testing in the 6–12 months after LT. 114 However, more recent data have challenged the assumption of universal reversibility. Studies using the contemporary diagnostic criteria vary in their reported reversibility. Sonny et al. reported worsening DD by ⩾1 grade in 40% of their 100 patients over a median follow-up of 3.4 years, however, patients with milder Grade 1 DD were included and TTE was only done in response to symptoms. Ali et al., 69 who repeated TTE at a median of 70 days post-LT in all 23 patients with DD (from a cohort of 131), observed recovery in 13 (57%). Izzy et al. 70 reported recovery in only 6 of 22 patients (27%) at a median of 1 year. Again, the follow-up TTEs in this study were performed only when clinically indicated, potentially underestimating the true rate of recovery. The only study with extended longitudinal data is that by Voet et al., 104 who performed repeat TTEs in 9 of 12 patients with DD at a median follow-up of 3.8 years post-LT. At that time point, only one had persistent DD. These findings suggest reversibility of DD may be delayed, occurring over several years rather than months. Data on systolic recovery is limited, as LVEF has consistently been shown to decline post-LT,62,100,114 reflecting the normalization of peripheral vascular resistance and blood volume. As GLS becomes routine for systolic assessment, more data may become available. As aforementioned, myocardial ECV on CMR correlates with reversible DMF and may represent one of the structural alterations in CCM. Kim et al. 93 performed CMR on 19 patients both before and after LT. Irreversible LGE was only observed in one patient, whereas ECV was significantly elevated in patients with cirrhosis and regressed at 12 months post-LT. This suggests that the fibrotic myocardial changes in CCM may, at least in part, be reversible, deepening our understanding of the structural remodeling in this condition. A current prospective study by our group (Australian Clinical Trials Registry Number 12624000321549) will address the question of reversibility using dynamic indices of cardiac function pre- and post-LT, including cardiac reserve on stress testing.
Monitoring after LT
As MASLD becomes as a leading indication for LT, 58 persistent cardiac dysfunction post-LT will intersect with the metabolic multimorbidity of these patients, underscoring the importance of timely recognition. Until recently, there were no consensus recommendations for the post-LT monitoring of patients with CCM. In their 2022 multidisciplinary, practice-based recommendations, 97 Izzy et al. advised a focused cardiac physical examination and BNP measurement for the first 3 months after LT in patients with pre-existing cardiac disease, including CCM. For those with subclinical cardiac dysfunction prior to LT, they recommend TTE every 6 months following LT, until resolution of dysfunction is confirmed. This interval was also recommended by the CCC, for up to 24 months post-LT. 1 Early identification of persistent or progressive cardiac dysfunction in the post-LT period is clinically important, as it allows for timely initiation of interventions such as blood pressure optimization and the use of guideline-directed medical therapy for HF.
Pharmacological management after LT
There are currently no disease-modifying therapies specific to CCM, and the effect of pre-LT optimization of subclinical CCM on pre- or post-LT outcomes has not been studied. Although several traditional HF therapies such as ivabradine 115 and beta-blockers 116 have been trialed in CCM patients in the pre-LT setting, there remains a paucity of evidence to support any specific pharmacological strategies in CCM patients post-LT. A management algorithm for CCM management post-LT is proposed in Figure 3. The management of overt HF post-LT follows the same principles as in non-cirrhotic HF, guided by whether LVEF is preserved or reduced. Early referral to cardiology is essential to facilitate the timely initiation of guideline-directed medical therapy.117,118
For patients with persistent subclinical cardiac dysfunction post-LT, addressing modifiable HF risk factors, such as obesity, hypertension, and diabetes mellitus, is critical. Identifying these patients in pre-LT assessment and monitoring them postoperatively allows for risk factor modification, which may help mitigate progression to overt disease. It may also inform therapeutic choices, leveraging agents that offer cardiovascular benefits when indicated for other common post-LT conditions. Hypertension is a common complication post-LT, usually related to CNI use or metabolic syndrome, and contributes to DD. 119 While amlodipine is often used as first-line antihypertensive in LT recipients, angiotensin-converting enzyme inhibitors (ACEi) or angiotensin receptor blockers (ARBs) may be preferred in patients with early SD due to their additional anti-remodeling benefits. Sodium-glucose co-transporter 2 (SGLT2) inhibitors, such as dapagliflozin or empagliflozin, are now recommended in patients with HFpEF or mild HFrEF, 118 regardless of diabetic status. In LT patients with concomitant diabetes mellitus or new-onset diabetes of transplant and evidence of subclinical cardiac dysfunction, SGLT-2 inhibitors may offer dual benefits. Similarly, SGLT-2120–122 inhibitors and ACEi 123 are indicated in chronic kidney disease with proteinuria, a frequent post-LT complication. The benefit of these agents in subclinical cardiac dysfunction or CCM has not been studied, but their use is reasonable in the presence of other indications. Statins may reduce HF hospitalizations, 124 and cardiovascular disease risk assessment should be routinely performed in LT patients, particularly those with MASLD, to determine treatment thresholds for initiating lipid-lowering therapy. Finally, there is emerging evidence for the use of glucagon-like peptide (GLP)-1 inhibitors in HFpEF with either obesity or diabetes. 125 Post-LT obesity is a major issue, and GLP-1 inhibitors are being used increasingly in this context. Their potential impact on post-LT cardiac dysfunction is an area for future research.
Conclusion
The clinical relevance of CCM is increasingly recognized in LT. While it does not appear to influence immediate peri-operative outcomes or long-term survival, patients with pre-LT CCM are at greater risk of developing overt HF post-LT. Routine post-LT monitoring may facilitate earlier detection and management of persistent or progressive cardiac dysfunction, integrating potential therapies into the management of other post-LT complications. Further research should focus on risk prediction models for waitlist and post-LT patients, the probability and timing of reversal after LT, and the role of HF therapies in CCM.
