Abstract
Background:
Functional dyspepsia (FD) is a prevalent condition characterized by symptoms such as postprandial fullness, early satiety, and epigastric pain, which significantly affect daily activities and quality of life.
Objectives:
This study aimed to assess the effectiveness of itopride hydrochloride, a prokinetic agent, in reducing the severity of these symptoms among patients with FD.
Design:
In this noninterventional, observational, post-authorization, prospective, multicenter study (without placebo or comparator) 339 patients with FD were treated with itopride and followed over a period of 12 weeks. The study clinical protocol ITO-NPS-01-PMCS-2021 was approved by the Ethical Committee of the University Hospital Brno and Faculty of Medicine, Masaryk University, Brno (ORG No: 100022981/LOC No:100031711), and reported to the Czech State Institute of Drug Control (identifier: 2110140000) following local legislation.
Methods:
The patients’ symptoms were quantified at baseline, 3–6 weeks, and 7–12 weeks using the modified “Leuven Postprandial Distress Scale.” Statistical analysis involved the Wilcoxon test, Mann–Whitney test, and Kruskal–Wallis test, focusing on changes in symptom scores and following potential adverse effects (AEs) over time.
Results:
Itopride hydrochloride treatment resulted in a statistically significant median reduction in symptom severity scores from baseline to the first and second follow-up assessments. The scores decreased by 5 points at the first follow-up and by 8 points at the second follow-up across all FD patient subgroups, regardless of age and gender. The study also observed minimal AEs, with only 2% of patients experiencing mild symptoms.
Conclusion:
Itopride appears to be a safe and effective treatment for reducing symptom severity in patients with FD. The findings support the use of itopride in clinical settings, providing evidence of its benefit in managing this challenging condition. This study brings additional clinical knowledge from the eastern European population (Czechs) to data published by the ROME IV foundation with various cohorts of patients with FD and its results further demonstrate the long-term benefits and safety profile of itopride with minimal non-AEs.
Plain language summary
Itopride is a safe and effective treatment for reducing symptoms in patients with functional dyspepsia. The results support its use in clinical practice and demonstrate its potential to help manage this condition. This study contributes new data from Eastern European patients (the Czech population) to the evidence already published by the Rome IV Foundation. The findings further indicate that itopride remains safe and beneficial over a 12-week period, with very few reported side effects.
Keywords
Introduction
Functional gastrointestinal disorders (FGIDs), such as irritable bowel syndrome, functional dyspepsia (FD), or functional constipation, although incompletely understood, and with a complex pathophysiology, account for at least a third of referrals to gastroenterology clinics. 1 Studies suggest a FGID prevalence rate of more than 40% within the general population at any given time, and FD was the most prevalent gastroduodenal disorder, with a pooled prevalence rate of 7.2% (7.1%–7.4%) for Internet and 4.8% (4.5%–5.1%) for household surveys. 2 These conditions frequently overlap and affect quality of life and healthcare use. More than two-thirds of FD patients see a doctor in the preceding 12 months, 40% use regular medication, and up to a third may undergo potentially unnecessary abdominal surgery for their symptoms. Because of the type of pathophysiology involved and the stigma associated with the term functional, these conditions have been redefined as disorders of gut–brain interaction, to better reflect their scientific basis. The fundamental gastrointestinal (GI) functions include motility, sensation, absorption, secretion, digestion, and intestinal barrier function. Symptoms of FGIDs (also called disorders of gut–brain interaction) are often triggered by meal intake, suggesting abnormalities in these physiological processes are involved in the generation of symptoms. 1
FD markedly affects the patient’s daily activities. According to the Rome IV criteria, FD is defined as the presence of at least one of the following symptoms: postprandial fullness, early satiation, epigastric pain, or burning, without evidence of structural disease to explain the symptoms fulfilling time criteria for the last 3 months with symptom onset at least 6 months before diagnosis and a frequency of at least 3 days per week. These symptoms should remain unexplained following a standard clinical assessment. 3
The ROME IV criteria subdivided FD into two syndromes: postprandial distress syndrome (PDS) and epigastric pain syndrome (EPS). PDS relates to all meal-associated symptoms that not only include the previously recognized postprandial fullness and early satiation but also symptoms, such as postprandial epigastric pain or burning, epigastric bloating, heightened belching, and nausea. 1 EPS retains its characterization, primarily centered around bothersome epigastric pain and/or burning sensations. Therefore, the ROME IV criteria offer a more nuanced approach aligning closely with clinical observations, facilitating a more detailed patient stratification, and enhancing therapeutic precision in everyday clinical settings.1,3,4
In clinical practice, antisecretory agents such as proton pump inhibitors, prokinetics, and Helicobacter pylori eradication are actively used to treat patients with FD. 5 Gastric motility is a key pathophysiologic factor in PDS, and prokinetic agents are often proposed as the treatment of choice for this subgroup. Pittayanon et al. reviewed clinical data and published a meta-analysis of the efficacy of prokinetics in FD based on 38 trials involving 12,095 patients. The combined analysis of this large data set showed a significant benefit of prokinetics over placebo in improving overall FD symptoms, with a number needed to treat of 7. 6
Meta-analysis data clearly indicate that prokinetic agents are significantly more effective than placebo in the treatment of FD. 7 Despite that, there is a lack of widely available agents of proven efficacy in a number of countries. 8 Moreover, some prokinetics which are widely prescribed (metoclopramide and domperidone), are associated with adverse effects (AEs) such as drug-induced movement disorders, cardiovascular side effects, galactorrhea, and hyperprolactinemia. 9 Other newer prokinetics (cisapride) have been withdrawn from the market after 2000 because of a risk of QT prolongation and life-threatening arrhythmias.8,10 Itopride, on the other hand, has good efficacy in terms of global patients assessment, postprandial fullness, and early satiety in the treatment of patients with FD, can greatly improve FD syndromes, and shows a low rate of adverse reactions. 11
The objective of this research was to prospectively evaluate consecutive patients diagnosed with FD according to ROME IV criteria undergoing treatment with itopride hydrochloride. Utilizing a questionnaire approach, we aimed to assess: (a) changes in the severity of eight distinct symptoms (postprandial fullness; early satiety; bloating; belching; nausea; epigastric burning; epigastric pain; heartburn) throughout the course of therapy; and (b) the potential emergence of adverse reactions.
Study design
This was a noninterventional, observational, postauthorization, prospective, multicenter study (without placebo or comparator) to evaluate the dynamics of symptom evolution in patients with FD (PDS, EPS, MIX) during treatment with itopride hydrochloride. The study’s endpoints were as follows:
To evaluate patients’ progression of the eight symptoms severity over the course of 12 weeks of itopride therapy (the start of therapy, 4 weeks of therapy, and after 12 weeks of therapy) by means of questionnaire method.
To evaluate potential occurrence of AEs.
Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (see Supplemental Materials). 12
Clinical and laboratory examinations remained the same as during standard of care check-ups.
The dosage of orally administered itopride hydrochloride (Itoprid PMCS; 50 MG TBL FLM 40 II/50 MG TBL FLM 100 II/50 MG TBL FLM 120 II; PRO.MED.CS, Praha a.s.) was determined according to the approved Summary of Product Characteristics and depended on the FD of the patient. The daily itopride dosage (150 mg per day, i.e., 3× 50 mg prior to meal) was entirely at the discretion of the examining physician. All subjects were recruited in the context of their standard medical care at a gastroenterology clinic for the diagnosis and treatment of FD. The final decision on treatment inclusion was made by the treating physician in collaboration with the patient, based on the patient’s status and standard of care. All the participants provided written informed consent. The study synopsis was approved by the Ethical Committee of the University Hospital Brno and Faculty of Medicine, Masaryk University, Brno (ORG No: 100022981/LOC No:100031711), and reported to the Czech State Institute of Drug Control (identifier: 2110140000) following local legislation.
Detailed information about study design and used materials and methods are described in Supplemental Data (Materials and Methods).
Results
Summary of the patient cohort—gender, age distribution, and classification of FD
The study cohort consisted of 339 patients who were consecutively recruited within one clinical center and evaluated for FD (Figure 1). The patients were enrolled into the study during the period of October 2021 and July 2022, followed by a biostatistical analysis. The patients’ age span was from a minimum of 20 years to a maximum of 86 years (the average and median ages being 54 years). Furthermore, a significant proportion of the patients were aged 50 years and above, comprising 61.4% of the cohort (Figure 2(a)). The gender distribution within this cohort indicates a slightly higher representation of females compared to males, with 54% (183 patients) being female and 42.8% (145 patients) being male. Additionally, a small fraction of the cohort, 3.2% (11 patients), did not have their gender stated (Figure 2(b)).

Design of the study, data validation, and patients’ set definition.
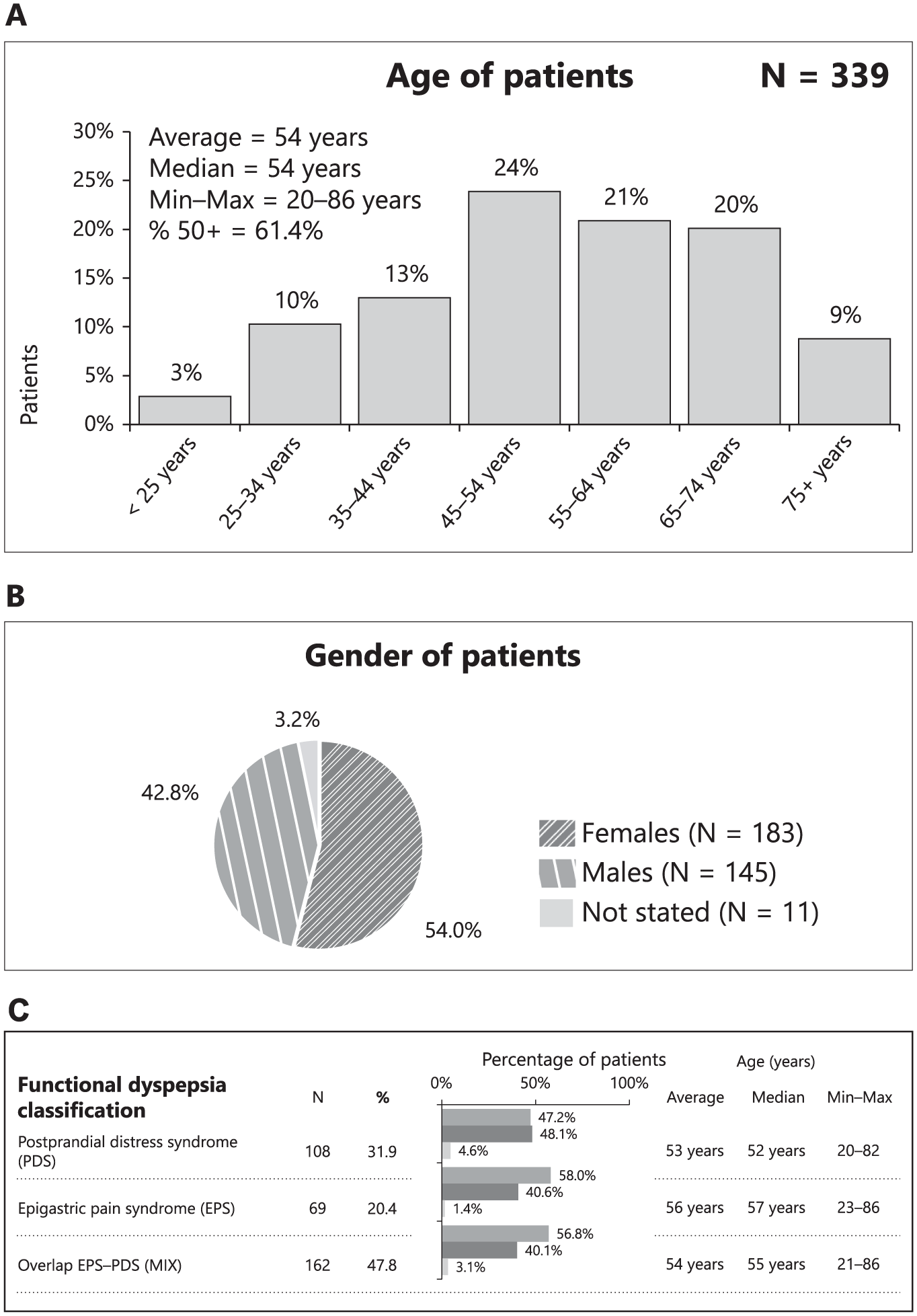
The summary of the patient’s enrollment into the study is presented in the figure. (a) Age of patients. (b) Gender of patients. (c) Type of FD.
FD among the patients was classified into three categories: PDS, EPS, and an overlap of both conditions (overlap). The PDS group comprised 108 patients, representing 31.9% of the cohort, with an age range of 20–82 years, a median age of 52 years, and an average age of 53 years. The EPS group included 69 patients, accounting for 20.4% of the cohort, with an age range of 23–86 years, a median age of 57 years, and an average age of 56 years. The largest group, with PDS-EPS overlap, consisted of 162 patients, making up 47.8% of the cohort, with an age range of 21–86 years, a median age of 55 years, and an average age of 54 years (Figure 2(c)).
Symptom development over time
Figure 3 provides a comprehensive analysis of symptom development over time for patients undergoing itopride therapy. The data are segmented to illustrate changes from the initial inclusion (D0) to the first control visit (D1) and further to the second control visit (D2). This analysis underscores the effectiveness of itopride therapy across various symptoms (S1–S8, see Figure 3 legend) and patient demographics (Figure 3(c)). A detailed progression of FD symptoms (S1–S8) are presented in Supplemental Figure 1(a) and (b).

Scores of all symptom development over time for all patients are categorized as follows: (a) Total score by symptoms. (b) Overall score changes between visits (D0–D1). (c) Detailed patients’ data and statistical evaluation. (d) Change from D0 to D1. (e) Change from D0 to D2. Statistical significance was determined using P values, with thresholds defined as follows: P < 0.05, P < 0.01, and P < 0.001. These levels are denoted by one *. ** and ***, respectively, corresponding to increasing levels of statistical confidence.
Symptom scores over time
For all patients, the mean symptom scores show a consistent decrease across all symptoms from D0 to D1 and to D2 (Figure 3(d) and (e)). Similarly, S2 shows a reduction from 1.3 at D0 to 0.7 at D1 and to 0.3 at D2. This pattern of decreasing symptom scores is consistent across all symptoms, with statistically significant reductions (p-values < 0.001).
Symptom score changes over time by various patient characteristics
Figure 3 also illustrates the percentage of patients experiencing changes in their total symptom score from study enrollment to the first follow-up (D0–D1, Figure 3(d)) and from study enrollment to the second follow-up (D0–D2, Figure 3(e)). A significant portion of patients (54%) experienced a decrease in their symptom scores between 1 and 5 points from D0 to D1, while 34% had a decrease of 6 and 10 points. By the second follow-up (D0–D2), the improvements were even more pronounced, with 47% of patients experiencing a decrease of 6–10 points and 24% showing a reduction of 1–5 points. The mean change in total symptom score was −5.9 from D0 to D1 and −8.7 from D0 to D2, with median changes of −5.0 and −8.0, respectively. These results indicate a substantial improvement in the overall symptom burden for most patients already at the first check-up point (Figure 3(b)–(d)) and a further drop in the FD score during the second follow-up evaluation (Figure 3(b)–(e)). Figure 4 presents a detailed analysis of the changes in symptom scores among patients undergoing itopride therapy. The data are divided into two main comparisons: changes from inclusion to the first control visit (D0–D1) and changes from inclusion to the second control visit (D0–D2). The figure is organized into multiple sections (A–H), each examining the scores for different symptom categories (S1–S8) across various patient demographics and characteristics.

Occurrence of AEs at one follow-up (A) or at both (B) follow-ups. See text for an explanation.
Overall patient population
For the overall patient population, the mean symptom score significantly decreased from 1.9 at inclusion (D0) to 1.0 at the first control visit (D1), with a median score changing from 2.0 to 1.0. This improvement continued, with the mean score further decreasing to 0.6 at the second control visit (D2), maintaining a median score of 1.0. These changes were highly significant, with p-values less than 0.001, indicating the effectiveness of the itopride therapy.
Age groups
When examining the data by age groups, patients younger than 50 years showed a mean score decrease from 1.9 to 1.0 (median from 2.0 to 1.0) between D0 and D1, and further to 0.6 (median 1.0) by D2. Similarly, patients aged 50 and older had a mean score decrease from 1.9 to 1.0 (median from 2.0 to 1.0) between D0 and D1, and then to 0.6 (median 1.0) by D2. The changes in both age groups were statistically significant, with p-values less than 0.001.
FD types
The analysis by FD types revealed significant improvements across all categories. Patients with PDS experienced a mean score decrease from 2.3 to 1.2 (median 2.0 to 1.0) between D0 and D1, and further to 0.7 (median 1.0) by D2. Those with EPS saw their mean score drop from 1.4 to 0.9 (median 1.0) between D0 and D1, and to 0.5 (median 0.0) by D2. Patients with overlap symptoms showed a mean score reduction from 1.8 to 1.0 (median 2.0 to 1.0) between D0 and D1, and to 0.5 (median 1.0) by D2. All these changes were significant with p-values less than 0.001.
Food-related symptoms
Patients with food-related symptoms had a mean score decrease from 2.0 to 1.1 (median 2.0 to 1.0) between D0 and D1, and to 0.6 (median 1.0) by D2. Patients without food-related symptoms saw their mean score drop from 1.5 to 0.9 (median 1.0) between D0 and D1, and to 0.5 (median 1.0) by D2. Both groups showed significant improvements with p-values less than 0.001.
Gender differences
Gender-based analysis showed that females had a mean score decrease from 1.9 to 1.1 (median 2.0 to 1.0) between D0 and D1, and to 0.6 (median 1.0) by D2. Males experienced a mean score reduction from 1.8 to 1.0 (median 2.0 to 1.0) between D0 and D1, and to 0.5 (median 1.0) by D2. The symptom score changes in both females and males were statistically significant with p-values less than 0.001.
Use of chronic medicines
The use of chronic medicines was another factor analyzed. Patients using chronic medicines showed a mean score decrease from 1.9 to 1.1 (median 2.0 to 1.0) between D0 and D1, and to 0.6 (median 1.0) by D2. Patients not using chronic medicines had a mean score reduction from 1.7 to 0.9 (median 2.0 to 1.0) between D0 and D1, and to 0.6 (median 1.0) by D2. Both groups demonstrated significant changes with p-values less than 0.001.
Use of dyspeptic medicines
Lastly, the analysis considered the use of dyspeptic medicines. Patients using dyspeptic medicines had a mean score decrease from 1.8 to 1.0 (median 2.0 to 1.0) between D0 and D1, and to 0.6 (median 1.0) by D2. Those not using dyspeptic medicines saw their mean score drop from 2.1 to 1.1 (median 2.0 to 1.0) between D0 and D1, and to 0.5 (median 1.0) by D2. Significant improvements were observed in both groups with p-values less than 0.001. Overall, Figure 3 demonstrates the significant reduction in symptom scores across various patient demographics and characteristics, indicating the broad effectiveness of itopride therapy in managing symptoms of FD.
Adverse effects and completion rates
In terms of safety and compliance, the data show that adverse effects (AEs) were minimal. Only 2% of patients reported at least one AE, while 96% reported no AEs, and 4% had insufficiently completed tests (Figure 4(a) and (b)). This low incidence of AEs, coupled with a high completion rate, suggests that itopride therapy is both safe and well-tolerated by patients. Among AEs observed in the patients during the study were diarrhea, headache, dry mouth, bloating, nausea, and feeling of fullness.
Discussion
The pathogenesis of FD is not fully understood, but GI motility and visceral sensitivity are key factors in FD symptom manifestation. 13 Several research meta-analyses highlighted the significant benefits of prokinetic agents for FD patients.11,14–16 Prokinetic agents are a heterogeneous class of medications that amplify and co-ordinate GI smooth muscle contractions to enhance gastric emptying and transit of intraluminal content. These agents are widely used to improve a wide range of GI motility disorders and related symptoms even though high-quality evidence is limited. 17 Given concerns regarding the safety and efficacy of older prokinetics, a safer and more effective agent is preferred. As research indicates,7,18 itopride could potentially serve as such an alternative. Its mechanism of dual action includes antagonism of dopamine receptors and inhibition of acetylcholinesterase, which promotes GI motility. Existing RCTs on itopride’s efficacy in FD treatment have shown varied results.19–26 Itopride demonstrated promise in a phase II study for FD symptom relief. 20 However, two later phase III trials produced contradictory outcomes. 24 The disparities in these results are thought to originate from variations in study populations. Specifically, the phase II trial included a higher number of subjects with heartburn, underscoring how specific study demographics can influence therapeutic responses. 27 The findings of the meta-analysis by Ford et al. 14 align with other systematic reviews comparing drug efficacy against placebos. Notably, in studies with a low risk of bias, while tricyclic antidepressants emerged as the top-performing drug, itopride was a significant runner-up, ranking second. A separate meta-analysis involving 2620 patients found that itopride had led to superior rates in the global assessment of dyspepsia symptoms. 11 A detailed expert review of clinical trials with various prokinetics has been recently published as a result of the cooperative initiative of leading experts from The European Society of Neurogastroenterology and Motility/The American Neurogastroenterology and Motility Society. 17
In the current study, the results based on itopride therapy provide encouraging real-world data on symptom reduction in FD patients. This is especially reflected in the improvement in symptom severity across various patient subgroups differentiated by FD type, age, and gender, whose scores decreased by 5 points at the first follow-up and by 8 points at the second follow-up across all FD patient subgroups. Such findings not only reinforce the clinical use of itopride but also contribute to the broader discourse on managing Disorders of Gut–Brain Interaction (DGBIs) more effectively. Most importantly, this is the first study performed in an eastern European population of patients with FD following ROME IV criteria thus broadening clinical knowledge on this topic. Similar findings, particularly a significant improvement in symptoms among patients with FD, were confirmed by other research 20 conducting a randomized control trial among 554 patients with FD. The most substantial enhancement in symptom scores was observed in the 100- and 200-mg itopride groups (−6.24 and −6.27, respectively, as opposed to −4.50 in the placebo group; p = 0.05). 20
In addition, the results of the present study report the minimal incidence of AEs, only 2%, and thus enhance the profile of itopride as a safe option for long-term management of FD. This aspect of drug safety is crucial, given the chronic nature of FD and the need for sustained management strategies. This is in line with the findings of other studies, which report that itopride therapy is well tolerated, safe, and results in good symptomatic relief.7,18,22,28 The safety findings of the current study are consistent with previously published long-term study in which itopride was administered for up to 12 months, demonstrating a favorable safety profile and sustained symptom improvement in patients with FD. 29 However, it should be noted that the primary focus of this study was on assessing the 12 weeks symptom benefits of itopride in FD, rather than long-term safety outcomes.
The detailed exploration of FD using the Rome IV criteria and its treatment with itopride provides valuable insights into the potential benefit of prokinetics (Itopride) in FD. Strengths of the study are its large patient population and the use of a validated assessment tool, the Leuven Postprandial Distress Scale and positive safety profile of the itopride therapy. Limitations are the lack of a placebo or other control arm and the variable follow-up times, with up to 3 weeks difference. The latter does reflect clinical reality where follow-up visits have an irregular pattern. The positive outcome should prompt a placebo-controlled trial of itopride in FD defined by the Rome IV criteria, to determine the margin of efficacy over placebo.
Conclusion
In this noninterventional, observational, postauthorization, prospective, multicenter study, treatment with itopride hydrochloride was associated with clinically meaningful improvements in FD symptoms, including postprandial fullness, early satiety, and epigastric pain, over a treatment period of up to 12 weeks. The therapy was well tolerated, with a low incidence of AEs, supporting its favorable safety profile in routine clinical practice. These findings suggest that itopride may provide symptomatic benefit for patients with FD in real-world settings, complementing evidence from controlled clinical trials.
Supplemental Material
sj-docx-3-tag-10.1177_17562848261417732 – Supplemental material for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study
Supplemental material, sj-docx-3-tag-10.1177_17562848261417732 for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study by Jiri Dolina, Jan Tack, Serhiy Forostyak, Jolien Schol, Radek Kroupa, Ilja Tachecí, Martin Bortlík, Tomáš Pavlík and Michal Novotny in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848261417732 – Supplemental material for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study
Supplemental material, sj-docx-4-tag-10.1177_17562848261417732 for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study by Jiri Dolina, Jan Tack, Serhiy Forostyak, Jolien Schol, Radek Kroupa, Ilja Tachecí, Martin Bortlík, Tomáš Pavlík and Michal Novotny in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-1-tag-10.1177_17562848261417732 – Supplemental material for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study
Supplemental material, sj-tif-1-tag-10.1177_17562848261417732 for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study by Jiri Dolina, Jan Tack, Serhiy Forostyak, Jolien Schol, Radek Kroupa, Ilja Tachecí, Martin Bortlík, Tomáš Pavlík and Michal Novotny in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-2-tag-10.1177_17562848261417732 – Supplemental material for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study
Supplemental material, sj-tif-2-tag-10.1177_17562848261417732 for Impact of itopride on functional dyspepsia symptom progression: a multicenter noninterventional postregistration study by Jiri Dolina, Jan Tack, Serhiy Forostyak, Jolien Schol, Radek Kroupa, Ilja Tachecí, Martin Bortlík, Tomáš Pavlík and Michal Novotny in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
