Abstract
Dyspepsia is a very common gastrointestinal (GI) condition worldwide. We critically examine the recommendations of recently published guidelines for the management of dyspepsia, including those produced jointly by the American College of Gastroenterology and the Canadian Association of Gastroenterology, and those published by the UK National Institute for Health and Care Excellence. Dyspepsia is a symptom complex, characterized by a range of upper GI symptoms including epigastric pain or burning, early satiety, and post-prandial fullness. Although alarm features are used to help prioritize access to upper GI endoscopy, they are of limited utility in predicting endoscopic findings, and the majority of patients with dyspepsia will have no organic pathology identified at upper GI endoscopy. These patients are labelled as having functional dyspepsia (FD). The Rome IV criteria, which are used to define FD, further subclassify patients with FD as having either epigastric pain syndrome or post-prandial distress syndrome, depending on their predominant symptoms. Unfortunately, the Rome criteria perform poorly at identifying FD without the need for upper GI endoscopy. This has led to the investigation of alternative diagnostic approaches, including whether a capsaicin pill or combined serum biomarkers can accurately identify patients with FD. However, there is insufficient evidence to support either of these approaches at the present time. Patients with FD should be tested for H. pylori infection and be prescribed eradication therapy if they test positive. If they continue to have symptoms following this, then a trial of treatment with a proton pump inhibitor (PPI) should be given for up to 8 weeks. In cases where symptoms fail to adequately respond to PPI treatment, a tricyclic antidepressant may be of benefit, and should be continued for 6 to 12 months in patients who respond. Prokinetics demonstrate limited efficacy for treating FD, but could be considered if other strategies have failed. However, there are practical difficulties due to their limited availability in some countries and the risk of serious side effects. Patients with FD who fail to respond to drug treatments should be offered psychological therapy, where available. Overall, with the exception of recommendations relating to H. pylori testing and the prescription of PPIs, which are made on the basis of high-quality evidence, the evidence underpinning other elements of dyspepsia management is largely of low-quality. Consequently, there are still many aspects of the evaluation and management of dyspepsia that require further research.
Introduction
Dyspepsia is a very common gastrointestinal (GI) complaint, with up to one in five individuals affected worldwide. 1 Of those with dyspepsia, around 40% will seek the advice of their primary care physician. Consultation behaviour is influenced, in part, by the frequency and severity of symptoms, and increasing age of the patient. 2 Almost 15% of patients with dyspepsia are referred to secondary care for further investigation and management. 3 The overall costs to the health service associated with managing dyspepsia are considerable, estimated to be over $18 billion per annum in the United States of America (USA). 4 Moreover, when one considers that dyspepsia impacts on physical, mental, and social aspects of health-related quality of life, 5 the true overall costs to society are likely to be far higher, and also encompass loss of economic productivity due to sickness-related absence from work. 6
Consequently, the scale of the clinical and economic problem guarantees firstly, that there is ongoing interest in the evaluation of novel approaches to the investigation and management of dyspepsia, and, secondly, that there is a need for comprehensive clinical guidelines to assist the physician in providing the best care to patients with dyspepsia; care that is both evidence-based and cost-effective.
This review aims to highlight the most important recent developments in both the investigation and management of dyspepsia, and to explore recommendations from the latest clinical guidelines. The most recent of these were published jointly by the American College of Gastroenterology (ACG) and the Canadian Association of Gastroenterology (CAG) in 2017. 7 These guidelines include the results of a number of updated meta-analyses that evaluate the efficacy of a range of treatments for dyspepsia. United Kingdom (UK) guidelines from the National Institute for Health and Care Excellence (NICE), which are also those recommended by United European Gastro-enterology, in lieu of producing their own guidelines, 8 were last updated in 2015.9,10 To provide a broader, global, perspective we also consider the Asian Consensus Report on Functional Dyspepsia (ACFD) from 2012, produced jointly by the Asian Neurogastroenterology and Motility Association and the Asian Pacific Association of Gastroenterology. 11 Finally, we summarize the findings of a number of relevant updates to Cochrane Collaboration systematic reviews that have been published over the last 2 years.
Evaluation
Diagnosing dyspepsia
Dyspepsia refers to a range of upper GI symptoms, including epigastric pain or burning, early satiety, and postprandial fullness. Patients presenting with these symptoms can be broadly thought of as belonging to one of three groups: those with uninvestigated dyspepsia, in whom investigation has yet to be performed in order to reveal the underlying diagnosis, those in whom investigation has revealed a specific organic or structural cause for their symptoms, and those where no organic cause is found and who are deemed to have functional dyspepsia (FD; Figure 1). Organic or structural causes of dyspeptic symptoms include chronic Helicobacter pylori (H. pylori) infection and peptic ulcer disease, upper GI cancer, biliary and gallstone disease, and pancreatic disease, including pancreatic cancer. In such cases, patient management is disease-specific. The difficulty lies in identifying which patients with uninvestigated dyspepsia need to undergo further tests, as well as the management of those patients where investigation fails to reveal an underlying organic diagnosis, and who are therefore labelled as having FD.
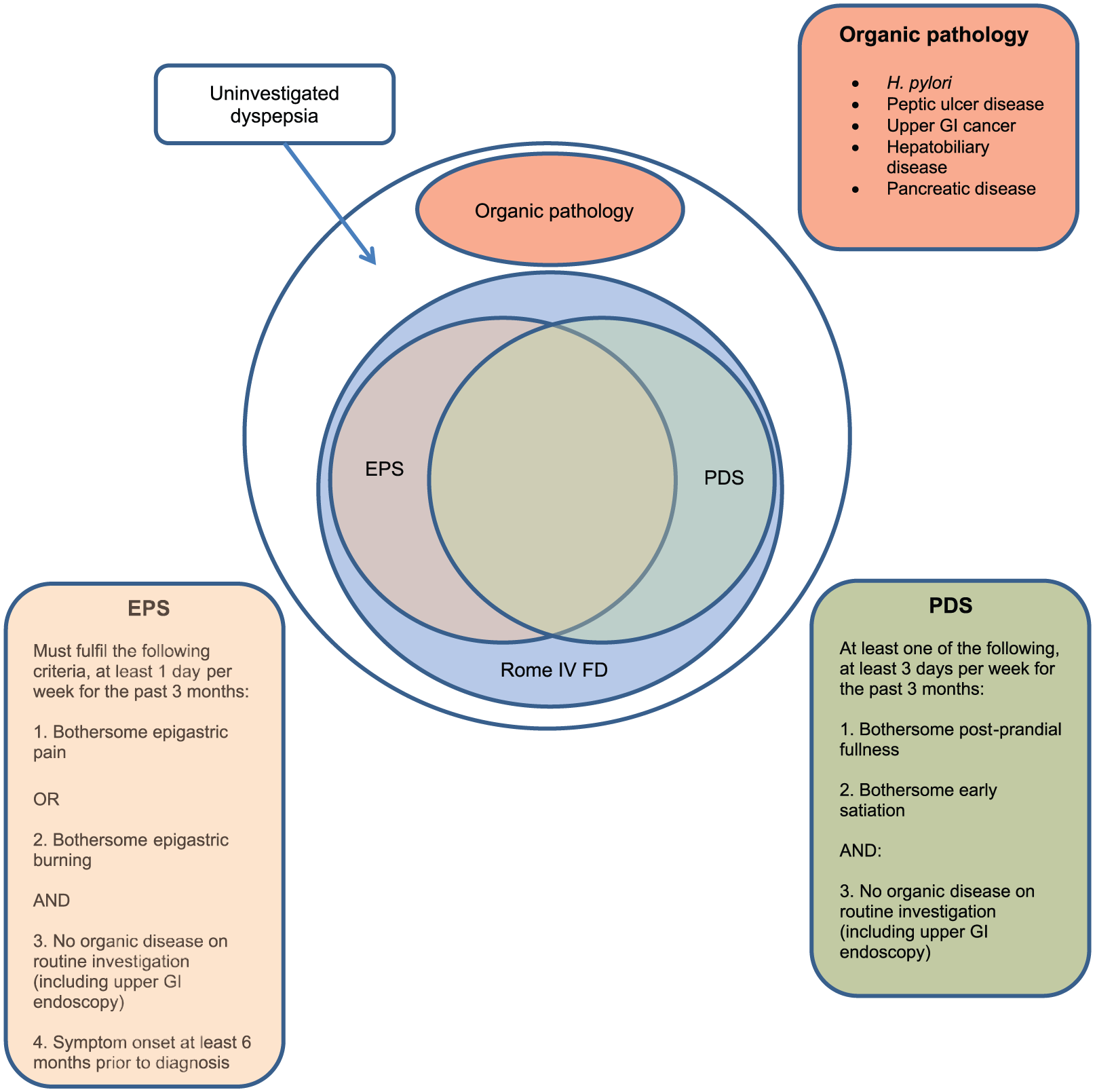
Pictorial representation of the classification of dyspepsia.
The Rome criteria
If symptoms were a good discriminator for the likelihood of a patient with dyspepsia having an underlying organic pathology, this would aid the physician greatly in their clinical assessment; however, unfortunately, this is not the case. A previous systematic review of diagnostic test studies demonstrated, convincingly, that a physician’s opinion, based on symptoms reported by the patient, had limited diagnostic ability to discriminate between organic causes of dyspepsia and FD. 12
Historically, FD was referred to as nonulcer dyspepsia, creating the impression that the diagnosis was based on the absence of specific underlying organic pathology, and therefore encouraging the use of investigations in all patients. Although more recently the terminology has changed, FD still remains a diagnosis of exclusion. The Rome Foundation, the expert group responsible for the classification of all functional GI disorders, defines FD using the Rome IV criteria as bothersome postprandial fullness, early satiety, epigastric pain, or epigastric burning, in the absence of structural disease, including at upper GI endoscopy, which would explain the symptoms. 13 According to these criteria the condition is subgrouped into two separate syndromes: the epigastric pain syndrome (EPS), and the postprandial distress syndrome (PDS). EPS consists of intermittent pain or burning in the epigastrium, occurring at least once per week. The pain may be either induced or relieved by eating but may also occur while fasting; however, it should not fulfil biliary pain criteria. Postprandial epigastric bloating, belching, and nausea can also be present, but are not specific to dyspepsia. PDS is defined as bothersome postprandial fullness after normal-sized meals, or else early satiety that prevents someone finishing a regular-sized meal, with such episodes occurring at least several times per week. Postprandial epigastric pain or burning, epigastric bloating, belching, or nausea may also be present. But are not specific to dyspepsia.. These definitions were determined based on a study of dyspeptic symptom groupings, 14 and also on the fact that up to 80% of patients with dyspepsia report that their symptoms are made worse by eating. 15
In the case of both EPS and PDS, although heartburn is not a dyspeptic symptom it may often coexist, but symptoms that are relieved by the passage of gas or faeces should generally not be considered to be part of dyspepsia. 13 Persistent vomiting should prompt consideration of an alternative diagnosis. 13 Patients may have both EPS and PDS, and studies in secondary care have demonstrated overlap between the two disorders in up to one-third of patients with FD. 16
Patients may struggle to interpret what is meant by verbal descriptions of dyspepsia symptoms. There is some evidence that representing symptoms using pictograms (Figure 2), can improve the concordance of symptom evaluation between patients and physicians, and may therefore be a useful tool in the assessment of the dyspeptic patient. 17

Pictograms showing the cardinal symptoms of functional dyspepsia (adapted from Tack and colleagues 17 ).
In a large validation study of the previous iteration, the Rome III criteria, 18 in secondary care 1452 consecutive adult patients with undifferentiated GI symptoms underwent a complete upper GI endoscopy. 19 The reference standard used to define the presence of true FD was epigastric pain, early satiety, or postprandial fullness, and no organic disease at endoscopy. Overall, 50% of patients met the Rome III criteria for FD, of whom 24% were found to have organic pathology detected at endoscopy. The Rome criteria therefore demonstrated a disappointing performance, in terms of their ability to identify patients with FD prior to performing an upper GI endoscopy, and therefore avoid investigation. Sensitivity was 60.7%, specificity was 68.7%, and positive and negative likelihood ratios (LRs) were 1.94 [95% confidence interval (CI) 1.69–2.22], and 0.57 (95% CI 0.52–0.63) respectively. Another study conducted in China found similar results; of 1655 patients with dyspepsia, 771 (47%) met the Rome III criteria for FD, but of these 280 (36%) had an organic cause identified at upper GI endoscopy. 16
The recent ACG/CAG guideline proposes a more pragmatic clinical definition of dyspepsia; predominant epigastric pain lasting at least 1 month, which may be associated with other upper GI symptoms such as epigastric fullness, nausea, vomiting, or heartburn, provided that epigastric pain is the patient’s primary concern. 7 However, this definition has not been validated in a clinical study, and it places the emphasis on the presence of epigastric pain, despite the extremely common reporting of postprandial symptoms by patients with dyspepsia. 15
The role of upper GI endoscopy
Any uncertainty over whether a patient’s dyspeptic symptoms have an organic or functional cause can, for the most part, be addressed by undertaking upper GI endoscopy, although in the case of suspected biliary or pancreatic disease, an abdominal ultrasound scan might also be necessary. However, a previous systematic review and meta-analysis has shown that more than 70% of people will be labelled as having FD following upper GI endoscopy, with <10% having peptic ulcer disease, and <1% having upper GI cancer 20 (Figure 3). These low rates of organic pathology do not justify undertaking an upper GI endoscopy in all patients with dyspepsia, even though symptom-based diagnostic criteria alone are insufficient to exclude organic disease. 12 This is supported by the findings of a primary care-based study, which demonstrated that the cost of detecting each case of upper GI malignancy among patients with dyspepsia was >$80,000. 21 Instead, recent guidelines recommend the targeted use of endoscopic investigation in patients with dyspepsia who also report alarm features, including increasing age, unintentional weight loss, vomiting, or dysphagia (Table 1). However, these also perform poorly for predicting endoscopic findings, with limited predictive value for upper GI malignancy, 22 and therefore using alarm features alone to risk-stratify patients and determine who should be referred for urgent endoscopic investigation is problematic.

Pooled prevalence of underlying pathology identified at upper GI endoscopy in patients with dyspepsia (adapted from Ford and colleagues 20 ).
Alarm features in patients with dyspepsia (adapted from Talley and Ford 23 ).

Note that the ACG/CAG guidelines now recommend an age threshold of 60 years or older.
The risk of malignancy predominantly relates to increasing age, and so guidelines have previously recommended upper GI endoscopy to routinely investigate dyspepsia only when patients are aged 55 years and older. The ACG/CAG guideline has now raised this age threshold to 60 years. 7 This recommendation is made on the basis of a cost-effectiveness analysis showing that, at all ages, upper GI endoscopy for investigating dyspepsia was dominated by alternative management strategies and that, using a threshold of 55 years, the evidence that upper GI endoscopy was cost-effective was borderline. 24 Currently, the age threshold used in NICE guidance remains at 55 years old or over,9,10 but it is important to acknowledge that cost-effectiveness decisions may vary between countries due to differences between patient populations and healthcare systems.
In the ACG/CAG guideline, even investigation of patients aged <60 years presenting with alarm features is not recommended because, as already discussed, they are a poor predictor of malignancy. 7 In addition, as the authors acknowledge, the baseline risk of someone <60 years old having malignancy is so low that the effect of any potential increase in risk from reporting an alarm feature is likely to be negligible, and so this still does not justify endoscopic investigation.
The recommendations concerning the role of upper GI endoscopy are qualified by highlighting that the quality of evidence is only moderate, and that current data have not adequately evaluated the importance of either the severity of, or combinations of, alarm symptoms. Therefore, some patients aged <60 years with alarm symptoms would warrant endoscopic investigation; for example, patients with significant weight loss, or rapidly progressive dysphagia. Similarly, patients with a family history of upper GI malignancy, or people from countries with a higher incidence of gastric cancer, such as Japan or China, may require upper GI endoscopy below this age threshold. There is also evidence to suggest that Latin Americans may have a higher incidence of gastric cancer. 25 Clinical judgement must therefore still be exercised judiciously.
Motility studies
Abnormalities of gastric motility and fundal accommodation have been observed in patients with FD. 26 However, clinically, the symptoms reported by a patient with FD and abnormal motility may be indistinguishable from those experienced by a patient with FD and normal motility, or indeed from those of someone with confirmed gastroparesis. 27 Fundal accommodation can be quantified using a gastric barostat 28 or single photo-emission computed tomography (CT), 29 and delayed gastric emptying can be investigated using scintigraphy, 30 breath tests, 30 or wireless motility capsule. 31 However, these tests are not widely available, and some of them can be unpleasant for patients. The satiety drinking test has been proposed as a simple and noninvasive means of evaluating gastric accommodation and early satiety, 32 although there is some evidence to suggest that it cannot replace the other modalities already discussed. 33 Also, since a definitive relationship between FD symptoms and delayed gastric emptying has not been demonstrated in studies, the ACG/CAG guidelines recommend that motility studies should not be performed routinely for patients with FD. 7 However, patients who have predominant symptoms of severe nausea and frequent vomiting may have gastroparesis, and should be investigated using a 4-hour solid phase gastric emptying scan, as per current guidelines for the management of gastroparesis, 34 if they fail to respond to empirical therapy. 7
Alternative investigations
As has been discussed above, symptoms cannot distinguish reliably between organic and functional causes of dyspepsia but also, for patients presenting with undifferentiated dyspeptic symptoms, the yield of upper GI endoscopy in revealing underlying organic pathology is low. Therefore, a means of screening patients with dyspepsia to identify those most likely to have organic disease, and therefore prioritize access to endoscopic investigation, would undoubtedly be very helpful.
Capsaicin
Capsaicin is the compound found in chillies and peppers that is responsible for causing sensations of heat and spice when they are eaten. 35 These sensations are mediated by the transient receptor potential vanilloid-1 receptor (TRPV1), which is part of a family of ion channel receptors responsible for sensing heat, and other noxious stimuli, including alcohol. 36 In FD, it has been demonstrated that patients can have visceral chemohypersensitivity involving the TRPV1 pathway. 37 However, those who are homozygous for the TRPV1 315CC genotype, and who therefore have higher expression of the TRPV1 receptor, have been shown to be less susceptible to FD. 38 The role TRPV1 might play in the pathophysiology of FD therefore requires further clarification. In addition, in a Japanese study of patients with FD, upper GI symptoms were commoner in those with higher consumption of spicy foods, irrespective of TRPV1 genotype. 39 Nonetheless, ingestion of a capsaicin pill appears to result in an increase in dyspeptic symptoms.40,41
Consequently, in a recent study, interest has focussed on a potential role for capsaicin as a diagnostic test for FD. In this single-centre study, 224 patients who were referred for evaluation of GI symptoms swallowed a capsule containing 0.75 mg of capsaicin. 42 They were asked to complete a questionnaire about symptom severity, both before and after taking the capsule. A diagnosis of FD was made if the patient’s symptoms met the Rome III criteria and if upper GI endoscopy was normal. Following investigation and assessment, only 72 patients were deemed to have FD, although these patients included some with FD and coexisting irritable bowel syndrome (IBS). The author reported a sensitivity of 51%, a specificity of 87%, and positive and negative LRs of 4.08 and 0.56 respectively for this oral capsaicin test in diagnosing FD.
It is therefore unlikely that the test will facilitate a diagnosis of FD without the need for upper GI endoscopy as, generally speaking, a positive LR of >10 and a negative LR of <0.1 are the minimum required for a test to be useful for ruling in or ruling out a disease respectively. 43 Moreover, the test was conducted in a mixed patient group comprising those with upper GI symptoms, as well as patients with known lower GI disorders, who may therefore have been less likely to have a positive test; these included 36 participants with IBS, and 26 with inflammatory bowel disease, potentially exaggerating the performance of the test. 44 It would have been better for the study to have been conducted in an unselected group of patients undergoing upper GI endoscopy for dyspeptic symptoms. Therefore, although the results of this study show promise, further studies in larger patient cohorts are needed before an oral capsaicin test can be recommended for the diagnosis of FD.
Combined serum biomarkers
The GastroPanel is a commercially available tool for measuring four blood biomarkers, namely basal gastrin-17 (G17), pepsinogen I and II (PGI and PGII), and H. pylori antibodies. 45 The design of the test is based on the Correa model for the development of gastric cancer. This hypothesizes that gastric adenocarcinoma is the end result of a pathway that starts with an environmental trigger in early life, now known to be H. pylori infection, 46 and progresses sequentially from superficial gastritis, to chronic nonatrophic gastritis, and then to chronic atrophic gastritis and achlorhydria. 47 Patients with atrophic gastritis are then at increased risk of developing intestinal metaplasia, which may progress to dysplasia, and then subsequently to adenocarcinoma of the stomach. 47
G17 is secreted only by the G cells of the gastric antral mucosa, PGI is only secreted by the oxyntic glands of the corpus mucosa, but PGII is produced in the duodenum as well as the gastric antrum. 48 Atrophy of the gastric mucosa leads to a reduction in glandular tissue. When this occurs in the antrum, levels of G17 may be reduced and, in combination with positive H. pylori serology, could therefore be indicative of the presence of atrophic gastritis. 49 Alternatively, atrophy affecting the gastric body could reduce secretion of PGI, or reduce the PGI/PGII ratio, either of which might also be suggestive of the presence of atrophic gastritis. 49 Hence these biomarkers have been chosen for their potential ability to detect atrophic gastritis, without the need for obtaining a biopsy at upper GI endoscopy.
Theoretically, therefore, the GastroPanel could be used as a noninvasive test to screen patients presenting with dyspepsia, in order to identify those who are at increased risk of gastric cancer, which in turn could be used to prioritize access to upper GI endoscopy. Those patients who test positive would undergo upper GI endoscopy to rule out gastric cancer, or else hopefully detect it at an early more treatable, or preferably even a premalignant, stage. Conversely, patients who tested negative would be presumed to have FD and be managed accordingly.
Whether such an approach is viable depends both on the performance of the test, and whether it is cost-effective. A recent systematic review and meta-analysis of 20 studies using biomarker panels for detecting H. pylori and atrophic gastritis, 19 of which used the GastroPanel, deemed the test to be reliable for the diagnosis of atrophic gastritis. 50 The authors reported a pooled sensitivity of 74.7% (95% CI 62.0%–84.3%), and a pooled specificity of 95.6% (95% CI 92.6%–97.4%). The median prevalence of atrophic gastritis across these studies was 27%, and the negative predictive value was 91%. In other words, 91 of 100 participants with a negative test would not have atrophic gastritis. However, if a patient is taking a proton pump inhibitor (PPI) caution is advised, as PPI use increases the probability of a false negative test. As the authors acknowledged, the meta-analysis was based on studies of low methodological quality, with the potential for various sources of bias, and there remains a need for well-designed studies, with large sample sizes, to adequately evaluate the performance of the GastroPanel.
It is also unclear whether use of the GastroPanel is cost-effective in the management of dyspepsia. Although cost-effectiveness analyses of serum biomarkers have been conducted, the majority of these were in very specific populations at high risk of gastric cancer, identified for screening based on their age, rather than upper GI symptoms,51–54 which does not help in determining whether this test has a role in the management of undiagnosed dyspepsia. Moreover, all of these studies only considered pepsinogen testing alone, rather than as part of a combined panel of biomarkers. In summary, therefore, there is currently insufficient evidence to recommend the use of combined serum biomarkers, such as GastroPanel, in the investigation of dyspepsia.
Management
An algorithm for the management of dyspepsia is summarized in Figure 4, with the evidence underpinning this described in more detail below.

Suggested management algorithm for patients with dyspepsia.
H. pylori testing and treatment
Both the ACG/CAG and NICE guidelines and the ACFD statement recommend that patients with either uninvestigated dyspepsia or FD should have a noninvasive test for H. pylori and receive eradication therapy if they test positive.7,9 NICE specifically recommend the use of either a breath test or a stool antigen test, rather than serology, after allowing a 2-week washout period for those taking PPIs. 10 A recent indirect test comparison of data from 99 studies concluded that there was a statistically significance difference in diagnostic accuracy between urea breath test-13C, urea breath test-14C, serology, and stool antigen testing (p = 0.024). 55 It was recommended that, in those patients who had not recently taken antibiotics or PPIs, and who had not had a gastrectomy, urea breath tests had higher diagnostic accuracy than either stool antigen or serological testing. These guidelines do not discuss the role of routine retesting to confirm successful eradication in H. pylori-positive patients with either uninvestigated dyspepsia or FD,7,9 although the Maastricht V consensus recommends retesting such patients using a urea breath test or a stool antigen test, at least 4 weeks after treatment, and having discontinued PPI therapy for at least 2 weeks, to confirm successful eradication. 56
The ACG/CAG guideline updated a previous Cochrane Collaboration systematic review and meta-analysis 57 comparing H. pylori test and treat with prompt upper GI endoscopy for the management of dyspepsia. This pooled data from six randomised controlled trials (RCTs), involving 2399 patients with uninvestigated dyspepsia. 7 There was no difference in global dyspepsia symptoms between groups [relative risk (RR) of remaining dyspeptic 0.94 (95% CI 0.84–1.04)], but only 25% of patients in the test and treat arms of the trials underwent an upper GI endoscopy over a 12-month period, in contrast with nearly all of the patients in the prompt upper GI endoscopy arms. The test and treat strategy was therefore associated with significant cost savings over endoscopic investigation. In terms of other management strategies for uninvestigated dyspepsia, analysis of data from four RCTs investigating test and treat versus empirical PPI therapy found no statistically significant difference between these two approaches (RR 0.89, 95% CI 0.77–1.04). Nonetheless, on the basis of a trend towards test and treat for both cost and clinical benefit from a previous individual patient data meta-analysis, 58 this was the preferred first-line approach, with empirical PPI recommended for H. pylori-negative individuals, or those in whom eradication therapy was not of symptomatic benefit.
In FD, a previous systematic review suggested that H. pylori eradication therapy had a small, but statistically significant, effect on dyspepsia symptoms, and was probably cost-effective, but that more research was needed. 59 Accordingly, the ACG/CAG guidelines have updated this systematic review and meta-analysis, evaluating a total of 4896 H. pylori-positive patients with FD from 22 RCTs comparing eradication therapy with placebo. 7 The beneficial effect of H. pylori eradication on the risk of remaining dyspeptic was statistically significant, with a RR of 0.91 (95% CI 0.88–0.94) and a number needed to treat (NNT) of 12.5. In FD, H. pylori-positive patients will, by definition, have no evidence of peptic ulcer disease or malignancy at upper GI endoscopy and the effects of H. pylori eradication therapy are modest. However, as the ACG/CAG guidelines highlight, the potential to reduce the risk of gastric cancer and peptic ulcer disease in the future, with little risk to the patient from treatment, justifies this approach. 7 It is also worth noting that, in contrast to the ACG/CAG guidelines, both the Kyoto and the Maastricht V consensus reports state that H. pylori infection must be excluded, or else successfully eradicated, in anyone complaining of dyspeptic symptoms before a diagnosis of FD can be made.56,60 This recommendation is based on the observation that H. pylori infection is associated with chronic active gastritis to a varying extent in all infected individuals, 61 and should therefore be considered as an organic disease. However, it must be emphasized that, at the present time, it is not universally agreed that dyspepsia can only be classified as being ‘functional’ when H. pylori infection is absent.
Acid suppression
NICE guidelines recommend that all patients with uninvestigated dyspepsia should be offered treatment with a PPI for 4 weeks, and subsequent treatment with a H2-receptor antagonist (H2RA) if there is an inadequate response. 9 For patients with FD, they recommend either PPI or H2RA treatment when symptoms persist following H. pylori eradication treatment, or for patients remaining symptomatic following a negative test for H. pylori infection. The ACG/CAG guidance differs in that, for both uninvestigated dyspepsia and FD, it recommends PPI therapy only for patients who remain symptomatic following H. pylori eradication therapy, or who are H. pylori-negative. 7 They do not recommend H2RAs in uninvestigated dyspepsia, as their analysis showed a significant benefit of PPI over H2RA for this indication (RR of remaining symptomatic = 0.81; 95% CI 0.72–0.91). 7 Although NICE guidelines advocate the use of H2RA in dyspepsia, there is no evidence to support this.
A recent Cochrane review of PPIs for the treatment of FD found them to be effective compared with placebo, independent of either the dose or duration of treatment, 62 a finding confirmed in the ACG/CAG meta-analysis, with a NNT of 10. 7 The Cochrane analysis suggested that PPIs may be equivalent to H2RAs in FD, but the ACG/CAG felt that there was insufficient evidence to be certain that H2RAs were not genuinely inferior to PPIs in FD. The ACG/CAG meta-analysis of six RCTs comparing PPIs with placebo or antacid therapy in 2709 patients with uninvestigated dyspepsia found a RR of remaining symptomatic of 0.75 (95% CI 0.64–0.88), with an NNT of six. 7
In FD, it has been suggested previously that PPIs are more effective for treating EPS symptoms rather than PDS symptoms. However, the recent Cochrane review suggested there was a trend towards a benefit of PPIs for those with PDS, but no benefit in EPS. 62 Indeed, in healthy volunteers, it has been shown that acid suppression with PPIs can reduce postprandial fullness which might partly explain any benefit in PDS. 63 Therefore, PPIs probably do have a role in treating patients with PDS, and in addition, the fact that PDS and EPS symptoms frequently overlap in FD 19 means PPIs are a justifiable treatment for many FD patients. The ACFD statement advises that PPIs should be used in the treatment of FD, but that evidence in Asian populations is lacking. 11
NICE advises against long-term use of PPIs, and suggests empowering patients to manage their own symptoms by taking them on an ‘as-needed’ basis. 9 ACG/CAG guidance advises stopping PPIs if there is no benefit after 8 weeks of therapy. They also point out that the majority of PPI trials employed once-daily dosing and that, in FD, the data do not support any benefit of doubling the dose of PPI therapy. 7
Neuromodulators
The concept of the brain–gut axis in functional GI disorders, 64 as well as their associations with mood disorders and somatisation, 65 has prompted interest in the use of neuromodulators, such as some antidepressants, as a potential treatment for FD. 66 One study demonstrated that anxiety, but not depression, was associated with an almost eight-fold increased risk of developing FD over the following 10 years. 67 It has also been demonstrated that antidepressants might improve fundic relaxation in FD 68 and can have analgesic effects. 69 However, there have been relatively few RCTs investigating the efficacy of neuromodulators, and the best evidence is provided by a previous systematic review and meta-analysis of the available data. 70 This considered all psychotropic drugs in FD and reported an NNT of six (95% CI 4–16). However, benefit was confined to tricyclic antidepressants (TCAs), such as amitriptyline and imipramine, and to antipsychotic drugs, such as sulpiride and levosulpiride.
Looking at TCAs specifically, data were available from three eligible RCTs involving 339 patients with FD. The RR of dyspeptic symptoms not improving with TCAs, compared with placebo, was 0.74 (95% CI 0.61–0.91). They may be more effective in treating EPS rather than PDS, as suggested by the results of one of the trials. 71 Conversely, selective serotonin reuptake inhibitors, which have been shown to be of benefit in other functional GI disorders such as IBS, 72 were not found to be effective in FD (RR 1.01; 95% CI 0.89–1.15) in two robust RCTs in almost 400 patients.
On the basis of this evidence, the ACG/CAG guidelines advocate the use of TCAs for treating FD in those patients who fail to respond to PPI and H. pylori eradication therapy. Interestingly, they also recommend their use in patients with uninvestigated dyspepsia, despite the fact there are no RCTs of TCAs in this group of patients, because the majority are likely to have FD. NICE guidelines do not recommend the use of TCAs, but identified a need for further research in this area, comparing TCAs with PPIs, and with outcomes focussed on quality of life measures. 9 The ACFD statement advocates the use of anti-depressant and anxiolytic agents for treating FD, but felt that the level of evidence in Asian populations was weak overall. 11
It is well recognized that there is overlap between different functional GI conditions and some patients with FD may have coexistent IBS. 73 Given that TCAs have been shown to be an effective treatment for IBS, these patients may derive dual benefit from TCA treatment; 72 however, TCAs can cause constipation, and patients should be warned about this.
Prokinetics
Patients with dyspepsia may have abnormalities of gastric emptying and gastric accommodation, and certainly many report symptoms of post-prandial fullness, providing a rationale for the use of prokinetic agents in the management of dyspepsia. However, in the case of uninvestigated dyspepsia, the ACG found no trials comparing prokinetics with placebo. Instead, there were a handful of trials comparing prokinetics with PPIs, with the meta-analysis performed to inform the guideline demonstrating a trend in favour of PPIs, although this did not reach statistical significance. 7
Conversely, in the treatment of FD, a previous meta-analysis of RCTs showed prokinetics to be more effective than placebo, 74 an effect which was confirmed in an updated meta-analysis for the ACG/CAG guidelines. 7 This showed a statistically significant, albeit modest, effect of prokinetic therapy in reducing global FD symptoms with a RR of remaining dyspeptic of 0.92 (95% CI 0.88–0.97). However, individually, the most effective drug was cisapride (pooled RR 0.74; 95% CI 0.62–0.89), which has been withdrawn because of an increased risk of cardiac events. 75 Other drugs, including tandospirone and itopride, were found to be no more effective than placebo, with pooled RRs for remaining symptomatic of 0.94 (95% CI 0.84–1.05) and 0.90 (95% CI 0.78–1.03), respectively.
Acotiamide is a novel prokinetic agent, which is an acetyl-cholinesterase inhibitor. The drug improves gastric accommodation and gastric emptying. It has been licensed for the treatment of PDS in Japan, on the basis of an RCT from 2012, which showed promising results in 897 patients. 76 Symptom improvement occurred in 52% of those on active treatment, compared with 35% in the placebo arm (p < 0.001) However, combining these results with earlier data from 2010, 77 the ACG/CAG meta-analysis confirmed only a very modest overall effect with a pooled RR of remaining symptomatic of 0.95 (95% CI 0.91–0.99). 7 A 52-week, phase III, open-label trial of acotiamide for the PDS subgroup of FD appeared to confirm the long-term safety of the drug, 78 and also suggested a clinically important effect on PDS symptoms. However, there was no control arm in this study, and the placebo response rate for drugs in functional GI disorders is usually high.
Regardless of these findings, none of the prokinetic drugs discussed so far is available in the UK, Europe, or the USA. Metoclopramide, although widely available, is not recommended as there are no clinical trial data in FD, and it has potentially serious side effects, including irreversible tardive dyskinesia. 23 Domperidone does have clinical trial data for its use in FD, and is readily available in the UK, Europe, and Canada, but not in the USA. The ACG/CAG guidelines included a meta-analysis of domperidone, which showed a statistically significant effect on symptoms with a pooled RR of 0.71 (95% CI 0.53–0.97), although all the trials were at unclear or high risk of bias, and indeed all the prokinetic data exhibited significant unexplained heterogeneity. 7 Concerns have also been raised about the safety of domperidone, as it may increase cardiac risk through QT prolongation. 79 Levosulpiride, although usually considered to be an antipsychotic medication, also has prokinetic properties and has been shown to be of benefit in FD.80,81
Overall, the ACG/CAG guidelines recommend prokinetics in FD only if TCAs fail to work, on the basis that TCAs have stronger evidence for efficacy, whereas in uninvestigated dyspepsia they advise that they are tried if H. pylori eradication and PPI therapy are unsuccessful, but before TCAs are tried. However, the evidence for prokinetics is poor, and their restricted availability and potentially serious side effects makes it difficult to prescribe them for patients with dyspepsia. In addition, when one considers that the majority of patients with uninvestigated dyspepsia probably have FD, it may be better to use TCAs first, given the putative role of the brain–gut axis in this condition. NICE guidance has removed the recommendation to try prokinetics in dyspepsia, as this was made originally on the basis of cisapride data, and the drug has been withdrawn. 9 The ACFD statements supports the use of prokinetics in FD, highlighting the results of the trial of acotiamide in Japanese patients. 11
Psychological therapies
The role of psychological therapies in dyspepsia remains an understudied area and, despite a number of new studies in recent years, only four trials have provided dichotomous outcome data suitable for meta-analysis. Such a meta-analysis was undertaken for the ACG/CAG guideline and, overall, this suggested a significant benefit of psychological therapies in reducing dyspepsia symptoms (RR 0.53; 95% CI 0.44–0.65) with an NNT of 3. 7 Overall, two of the studies looked at cognitive behavioural therapy (CBT), while the remaining two studies considered other forms of psychotherapy, including group therapy.
On this basis, patients with FD who are still symptomatic despite drug treatments should be offered psychological therapy, although access to such services can be difficult. In addition, provision of psychological therapies requires a skilled practitioner, and the patient needs to be motivated to engage. A web-based form of CBT may help to make psychological treatment more readily available, and an RCT comparing therapist-delivered CBT to web-based CBT in IBS is currently underway. 82 Hypnotherapy has previously been suggested as an effective long-term management strategy for FD with the ability to reduce medication use and consultation rates, 83 but overall trial data remains limited, and more studies are needed to confirm these findings. More trials of psychological therapies in dyspepsia are needed, as current evidence is of poor quality overall, with an inherently high risk of bias due to the difficulties in blinding patients to treatment allocation in such studies.
Complementary and alternative therapies
A variety of complementary and alternative therapies have been suggested for the treatment of dyspepsia. One herbal remedy of interest, which has been evaluated in a single large RCT, is iberogast, also known as STW5. It is a mixture of nine different herbs, including milk thistle, chamomile, peppermint, and liquorice. In healthy volunteers, STW5 stimulated gastric relaxation and increased antral motility, which may account for any potential therapeutic effects in FD. 84 Indeed, in the trial comparing STW5 with placebo in 315 patients with FD, there was a statistically significant improvement in GI symptom scores in the STW5 arm, compared with placebo. 85 However, the ACG/CAG guidelines advise caution, as the effect was marginal and therefore, although it was statistically significant, the clinical significance may be debatable. 7 Rikkunshito, a Japanese herbal medicine with possible prokinetic properties, has been evaluated in 247 patients with FD in a multicentre double-blind RCT. 86 After 8 weeks of treatment, although there was a higher symptom response rate in the treatment arm, compared with placebo (33.6% versus 23.8%), this did not reach statistical significance.
Acupuncture has also been investigated for the treatment of FD. However, good quality trials are lacking and a Cochrane review concluded that there was insufficient evidence to be able to draw firm conclusions on its efficacy. 87 Chinese herbal remedies may be effective for treating FD, based on a meta-analysis of RCT data, but most studies are small, with a high risk of bias, and high clinical heterogeneity, meaning further rigorous studies are needed. 88 Similarly, there is little evidence for the use of either probiotics or homeopathy in dyspepsia.
Overall, complementary and alternative therapies are not recommended by the ACG/CAG dyspepsia guidelines, although it is accepted that patients may wish to try them, particularly if conventional treatments have proven ineffective. Nevertheless, it is important to emphasize that there is currently no clear evidence to support their use. NICE guidelines make no statement regarding their use in dyspepsia.
Conclusion
With the exception of recommendations related to H. pylori testing and the prescription of PPIs, which are made on the basis of high-quality evidence, all of the other recommendations made in current guidelines for the management of dyspepsia draw on evidence which is moderate at best, the majority being underpinned by low or very low-quality evidence. Consequently, there are still many elements of the evaluation and management of dyspepsia that require further research.
Future studies should focus on refining symptom-based definitions of FD to make them more specific and exploring whether certain clusters or patterns of red-flag symptoms are better able to predict the presence of organic disease, in order to prioritize access to endoscopic investigation. Oral capsaicin represents a potentially interesting strategy for diagnosing FD without upper GI endoscopy, but the current data are flawed, and further studies in larger populations of patients with undifferentiated upper GI symptoms are needed to validate this approach. Similarly, although combined serum biomarker testing can predict the presence of atrophic gastritis, and therefore potentially stratify gastric cancer risk, there are no data to show that adopting this test as part of a pathway for managing dyspepsia is either helpful or cost-effective.
It is also important to consider that, although treatments aim to target pathophysiological mechanisms believed to be responsible for causing dyspepsia, patients are always selected for trials on the basis of their symptoms, rather than pathophysiological abnormalities. Therefore, patient cohorts may theoretically exhibit significant heterogeneity regarding underlying pathophysiologies, which might limit the ability of trials to demonstrate treatment effects. Consideration should be given to conducting trials focussing on specific pathophysiological mechanisms.
More work is needed to explore the relationship between disordered gastric motility and symptoms in FD, in order to better understand the place of gastric physiology tests in dyspepsia management. When disordered motility is suspected or proven, current options for prokinetic therapy are limited, and this should be a focus for future drug development. TCAs seem to be beneficial for the management of FD, but larger trials are needed, which make comparisons with other available effective treatments, rather than placebo. Finally, psychological therapies may be valuable in the management of FD, but this remains an understudied area, and more trials are needed.
Footnotes
Acknowledgements
CJB, LAH, and ACF drafted and approved the article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
