Abstract
Acute esophagogastric variceal bleeding (AEGVB) represents one of the most critical complications encountered in patients with decompensated liver cirrhosis. Endoscopic techniques, characterized by their dual benefits of direct diagnosis and immediate therapeutic intervention, have emerged as the primary modalities for the diagnosis and management of AEGVB. These endoscopic interventions are effective in controlling acute hemorrhage and in reducing or obliterating varices. Recent advancements in endoscopic therapy have increasingly focused on precision and the integration of intelligent technologies. This article aims to review the current status and advancements in endoscopic treatments for AEGVB in the context of liver cirrhosis, assist endoscopists in developing individualized endoscopic intervention strategies for patients with AEGVB, ultimately optimizing therapeutic outcomes and enhancing patient prognosis.
Plain language summary
Acute esophagogastric variceal bleeding (AEGVB) represents one of the most critical complications encountered in patients with decompensated liver cirrhosis. Endoscopic techniques, characterized by their dual benefits of direct diagnosis and immediate therapeutic intervention, have emerged as the primary modalities for the diagnosis and management of AEGVB. These endoscopic interventions are effective in controlling acute hemorrhage and in reducing or obliterating varices. Recent advancements in endoscopic therapy have increasingly focused on precision and the integration of intelligent technologies. This article aims to review the current status and advancements in endoscopic treatments for AEGVB in the context of liver cirrhosis, assist endoscopists in developing individualized endoscopic intervention strategies for patients with AEGVB, ultimately optimizing therapeutic outcomes and enhancing patient prognosis.
Keywords
Introduction
In cases of decompensated cirrhosis, portal hypertension emerges as a significant complication. A hepatic venous pressure gradient (HVPG) of ⩾10 mmHg is correlated with clinical manifestations including ascites, gastroesophageal varices (GOV), and hepatic encephalopathy (HE). 1 When the HVPG surpasses the critical threshold of 12 mmHg, there is a marked increase in the risk of esophagogastric variceal bleeding (EGVB). 2 Acute esophagogastric variceal bleeding (AEGVB) is defined by bleeding episodes occurring within the preceding 120 h (5 days). 3 The annual incidence of AEGVB ranges from 25% to 40%, with a 6-week mortality rate reaching 15%–20%.4–8
The risk of AEGVB is positively correlated with the severity of liver disease, the diameter of varices, the Child–Pugh classification, and the presence of red signs on variceal walls. The classification of esophageal varices is primarily determined by their shape and size, using a three-grade system (Grade 1–3), where a higher grade signifies a larger diameter and greater severity. 9 In the context of gastric varices, the Sarin classification is predominantly utilized, categorizing varices into four types: gastroesophageal varices type 1 (GOV-1), gastroesophageal varices type 2 (GOV-2), isolated gastric varices type 1 (IGV-1), and isolated gastric varices type 2 (IGV-2). 5 The annual risk of bleeding is approximately 5% for small varices and 15% for large varices. Treatment modalities primarily encompass pharmacological therapy, endoscopic intervention, interventional radiological procedures, and surgical approaches.10–12 Endoscopy is considered the gold standard for diagnosing AEGVB. The diagnosis of variceal bleeding is confirmed endoscopically by directly visualizing active hemorrhage or stigmata such as a white nipple sign or clot. The diagnosis is also supported by the presence of variceal red signs along with blood in the stomach, after excluding other potential sources of bleeding. 1 Endoscopic treatment is highly effective in controlling acute bleeding and preventing early re-bleeding, with most clinical guidelines advocating for endoscopic intervention within 12 h of an acute AEGVB episode.1,7,13,14 Endoscopic treatment not only prevents initial episodes of EGVB but also manages AEGVB cases and reduces the risk of rebleeding. Repeated endoscopic interventions may lead to a reduction or elimination of venous distension through vascular occlusion.15,16
The primary techniques employed include endoscopic injection sclerotherapy (EIS), endoscopic variceal ligation (EVL), and endoscopic cyanoacrylate injection (ECI), among others. Identifying the source of bleeding during endoscopic examination can be challenging in the absence of active bleeding. Conversely, during active bleeding episodes, large blood clots may compromise endoscopic visibility. However, endoscopic ultrasound-guided selective varices devascularization facilitates precise identification of varices, thereby enabling targeted treatment.13,17 Despite its benefits, endoscopy is associated with certain invasive procedures that carry risks such as infection, perforation, bleeding, pain, and post-treatment ulcers. 18 This article seeks to review the current status and advancements in endoscopic treatments for AEGVB in the context of liver cirrhosis. It aims to guide endoscopists in developing individualized endoscopic intervention strategies for patients with AEGVB, with the objective of optimizing therapeutic outcomes and enhancing patient prognosis.
Common endoscopic therapies for AEGVB
EVL
EVL, introduced by Van Stiegmann in 1986, represents a significant advancement in the treatment of esophageal varices. The mechanism of action involves mechanical strangulation, ischemia, ulceration, and scar formation. 19 According to the guidelines set forth by the American Association for the Study of Liver Diseases (AASLD) and the European Association for the Study of the Liver, EVL is recommended as a primary approach for managing esophagogastric varices in three specific scenarios: (1) preventing bleeding in high-risk moderate-to-severe varices, such as those classified as Child-Pugh class B/C or exhibiting positive red signs; (2) providing emergency hemostasis for AEGVB; and (3) reducing the risk of rebleeding in secondary prevention when used in conjunction with nonselective β-blockers.1,13 Clinical studies have demonstrated that EVL is both highly effective and safe for managing GOV1,20,21 achieving an immediate hemostatic success rate exceeding 90% in cases of AEGVB (Table 1). 22 It is the preferred treatment modality for both hemorrhagic and nonhemorrhagic esophageal varices, including those associated with hepatocellular carcinoma and portal vein tumor thrombosis.1,13 Urgent EVL has been shown to reduce 30-day mortality and improve overall survival in certain patient populations.23,24
Effectiveness of different endoscopic therapies for EGVB.

ECI, endoscopic cyanoacrylate injection; EGVB, esophagogastric variceal bleeding; EIS, endoscopic injection sclerotherapy; EVB, esophageal variceal bleeding; EVL, endoscopic variceal ligation; GVB, gastric variceal bleeding.
The ligation procedure is performed in a systematic spiral pattern from distal to proximal with closely spaced bands, thereby achieving vascular occlusion through mechanical obstruction and venous fibrosis.22,41,42 The endoscopic severity of varices determines the appropriate number of ligation bands to be utilized. Although there is no consensus regarding the efficacy-risk balance of intensive band ligation, some studies indicate that it may increase the incidence of ulcers and the risk of early postoperative rebleeding. 43 Conversely, other studies suggest that it is more effective than conventional methods for AEGVB, as it achieves faster hemostasis and promotes more extensive mucosal fibrosis, thereby reducing variceal recurrence.44–46 Intensive band ligation is a technique that has been extensively developed and refined at our center (Figure 1), demonstrating significant efficacy in achieving hemostasis for AEGVB (Figure 2). According to the European Society of Gastrointestinal Endoscopy (ESGE) guidelines, postoperative follow-up for EVL should include sequential treatments 1–4 weeks after the initial EVL for AEGVB until the varices are eradicated, followed by endoscopic evaluations every 3–6 months during the first year and annually thereafter. 13
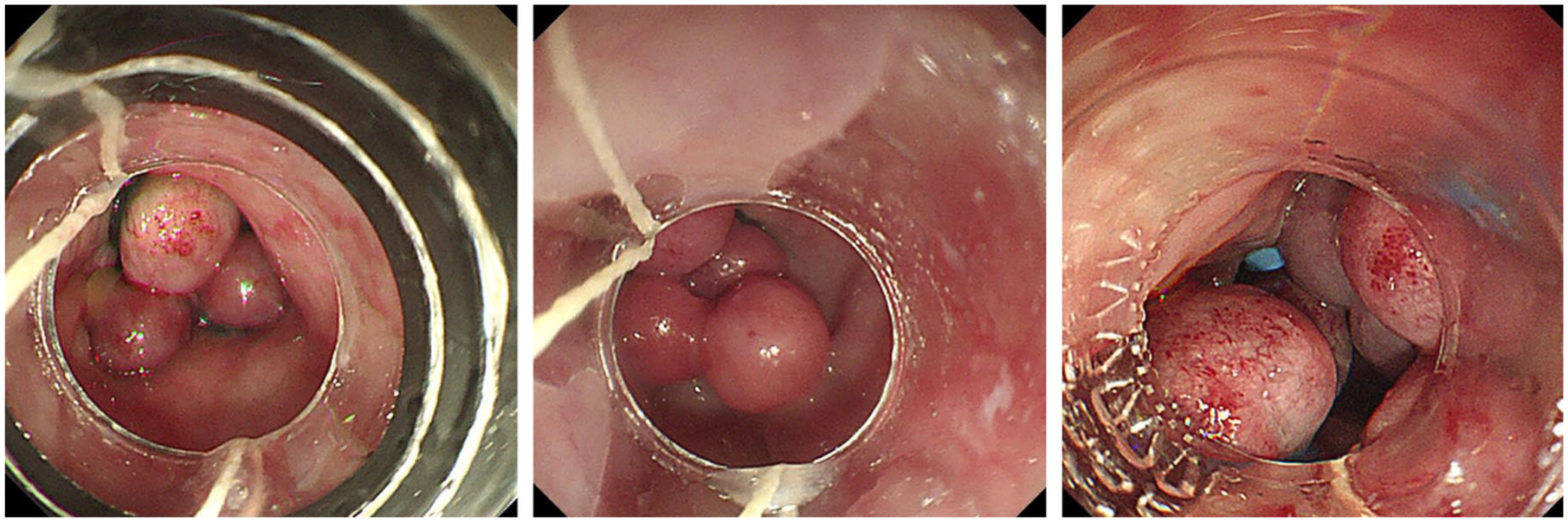
Endoscopic intensive band ligation in three cases.

Endoscopic intensive band ligation procedures in two cases. (a) Active bleeding esophageal varices. (b) Hemostasis by intensive band ligation therapy. (c) Intensive banding therapy. (d) Active bleeding esophageal varices. (e) Hemostasis achieved after intensive band ligation therapy. (f) Intensive banding therapy.
However, EVL has several limitations. First, EVL is constrained by the diameter of the varices, being recommended for those measuring 0.5–2 cm. Larger varices may result in incomplete ligation and substantial hemorrhage, whereas smaller varices increase the risk of excessive suction depth, potentially exacerbating pain and scarring.12,46,47 Second, the effectiveness of EVL can be influenced by the condition of the esophageal mucosa. Repeated ligations at the same site can lead to mucosal scarring and reduced elasticity, thereby increasing the risk of band slippage and ulcer formation. Severe mucosal erosion may be aggravated by excessive suction, potentially resulting in further bleeding episodes.26,48,49 Third, the systemic conditions and severity of bleeding in patients significantly affect EVL outcomes. Substantial blood loss during AEGVB can obscure visibility, complicating the placement of bands. Patients experiencing hemodynamic instability, severe HE, or liver–kidney dysfunction are generally in poor health and face higher anesthetic risks, necessitating fluid resuscitation and medication prior to endoscopy.12,50
Postoperative complications of EVL may occur, with an overall incidence ranging from 2% to 20%. Common complications include transient dysphagia and retrosternal pain, while other complications may encompass esophageal stricture, ulceration, perforation, infection, and hemorrhage. The period between postoperative days 5–10 is identified as the sloughing phase. During this phase, the ligation band may detach, potentially leading to ulcer formation. Ulcer-related bleeding is a serious complication associated with EVL, affecting approximately 2.3%–15% of patients due to ulcer detachment. This condition poses significant risks and can result in mortality.22,51–53
Endoscopic direct injection therapies for AEGVB
EIS
EIS was first introduced clinically by Crafoord and Freckner in 1939 for the treatment of esophageal varices and has demonstrated efficacy in managing AEGVB and improving patient outcomes (Table 1). Since the mid-1970s, EIS has undergone continuous refinement.54–56 It is particularly useful when EVL is challenging due to compromised visibility from AEGVB, extensive mucosal scarring, or mechanical obstructions, and when patients require stabilization prior to EVL.8,22,57 However, EIS is not recommended for patients with end-stage liver disease as it may exacerbate hepatic ischemia and increase the risk of liver failure. 58
In China, polidocanol is the preferred sclerosing agent due to its safety profile. The EIS technique involves: (1) Prioritizing intravenous injection in proximity to active bleeding sites to achieve immediate hemostasis through thrombus formation and vessel occlusion, ultimately leading to vessel obliteration via fibrosis. In cases where the bleeding source is not clearly identified, an injection administered 2 cm above the EGJ can effectively obstruct portal collateral blood flow. (2) Paravenous injection into the submucosal layer surrounding the bleeding vessel induces swelling, which compresses the vein and rapidly controls bleeding. 59
Some studies have indicated that EIS and EVL are comparably effective in controlling bleeding and exhibit a lower rate of rebleeding, although the rate of variceal eradication remains low (Table 2).60,61 EIS is particularly advantageous when varices are too large for ligation, rendering it a safe rescue option. However, EIS may lead to complications such as fever, pain, ulcers, and other adverse effects due to inflammatory responses and leakage of the sclerosing agent.62–65 Consequently, current guidelines advise against the use of EIS for primary prevention of AEGVB.66,67
Comparison of different endoscopic therapies for EGVB.

ECI, endoscopic cyanoacrylate injection; EGVB, esophagogastric variceal bleeding; EIS, endoscopic injection sclerotherapy; EVB, esophageal variceal bleeding; EVL, endoscopic variceal ligation; GOV, gastroesophageal varices; GVB, gastric variceal bleeding.
Recent studies have introduced novel EIS techniques to address the limitations of traditional methods. One such technique, balloon compression EIS (bc-EIS), mechanically compresses varices and occludes blood flow, thereby prolonging sclerosant retention and preventing systemic dissemination. While bc-EIS is effective and safe for the treatment of EVs, it presents greater complexity and technical challenges compared to EVL or conventional EIS.64,80,81
ECI
In 1986, Soehendra introduced cyanoacrylate (CYA) as a treatment for large GOV. 82 According to the ESGE guidelines and Baveno VII consensus, tissue adhesives are recommended as the primary treatment for acute bleeding from IGV1 and GOV2. Notably, the Baveno VII consensus places particular emphasis on GOV2 that extends beyond the cardia.7,83 ECI should be repeated 2–4 weeks following the initial treatment to minimize the risk of rebleeding. 4 Studies have demonstrated the superior efficacy of ECI in treating gastric variceal bleeding (GVB) and acute bleeding compared to EVL or EIS (Table 2).36,73,76,84–87
Commonly used tissue adhesives include CYA, with N-butyl-2-cyanoacrylate being frequently utilized in China. Chinese studies report that gel expulsion after ECI occurs within 1–3 months, with expulsion rates of 86.10% at 1 month and approximately 28% at 3 and 6 months.88,89 Gel expulsion typically concludes within a year; however, incomplete vascular occlusion can lead to early bleeding due to premature gel expulsion. 36
Key complications associated with ECI include ulcer formation, rebleeding, and ectopic embolism. Ulcers can form due to local tissue ischemia caused by intravascular coagulation from tissue glue. Rebleeding might happen from ulcer formation at the injection site, ulcer detachment, nonintravascular injections, inadequate dosage, or bleeding after expulsion. Patients with portosystemic shunts, particularly gastrorenal shunts, are at a high risk of ectopic embolism in organs such as the lungs, brain, spleen, kidneys, and coronary arteries, with the risk increasing with larger glue doses. Preoperative CT venography screening for shunt vessels can help mitigate the risk of embolism. Other complications include fever, infection, and needle tip obstruction.89–92 ECI demands precise intravascular injection techniques, teamwork, and quick single-point administration.
Tissue adhesives, dosages, and formulations vary worldwide. The traditional “iodine oil + tissue adhesive + iodine oil” sandwich technique is sometimes replaced with a “50% glucose solution” to avoid allergic reactions to iodine. In China, a new method using “polidocanol-tissue adhesive-polidocanol” has been suggested, combining a sclerosing agent with tissue adhesive for improved safety and effectiveness. This technique, which involves the administration of a sclerosing agent prior to the application of tissue adhesive, facilitates thrombus formation, thereby ensuring vessel occlusion and mitigating bleeding risks. Additionally, it extends the adhesive’s efficacy and reduces the likelihood of ectopic embolism, permitting the use of a reduced dosage of tissue adhesive.93,94 The sandwich injection technique optimizes varicose vein treatment by integrating pharmacological agents to enhance vessel closure and provide effective hemostasis.94–96 Future investigation should aim to standardize dosing protocols and develop biodegradable adhesives to enhance long-term safety.
In conclusion, EVL is identified as the most effective therapeutic approach for esophageal variceal bleeding compared to EIS and ECI, while ECI is distinctly more efficacious for GVB. Combination endoscopic strategies generally result in superior clinical outcomes. Nonetheless, the final treatment decision should be individualized by an experienced endoscopist, taking into account the specific clinical circumstances of each patient (Table 2).
Management following the failure of endoscopic treatment
Although the majority of patients with AEGVB achieve hemostasis following endoscopic intervention, approximately 20% experience rebleeding due to incomplete treatment or complications, which contributes to elevated mortality rates. 97 In the context of endoscopic salvage therapies, common interventions include repeat endoscopic procedures and the deployment of self-expanding metal stents (SEMS). Additionally, therapeutic modalities, such as pharmacotherapy, balloon tamponade, transjugular intrahepatic portosystemic shunt (TIPS), balloon retrograde transvenous obliteration, and surgical interventions, remain crucial components of the rescue strategy for refractory cases. The Baveno VII consensus recommends balloon tamponade or SEMS as bridging therapies, with SEMS being considered the safer option. For variceal bleeding that is unmanageable by medical and endoscopic means, salvage polytetrafluoroethylene-coated TIPS is regarded as the optimal approach (Table 3). 7
Hierarchical salvage therapy for AEGVB rebleeding.

AEGVB, acute esophagogastric variceal bleeding; HVPG, portal pressure gradient; MELD, model for end-stage liver disease; SEMS, self-expanding metal stents; TIPS, transjugular intrahepatic portosystemic shunt.
Endoscopic retreatment
According to the ESGE, repeat endoscopic therapy should be considered for recurrent AEGVB following initial endoscopic hemostasis, although no standardized protocol currently exists for secondary treatment post-AEGVB. 13 A study has demonstrated that secondary endoscopic hemostasis on residual esophageal varices using EVL, EIS, or ECI is feasible and can serve as a primary salvage therapy. 49 Furthermore, Tang et al. 103 successfully achieved hemostasis for recurrent bleeding from gastric varices through endoscopic ultrasonography (EUS)-guided coil and cyanoacrylate injection. Mangas-Sanjuan et al. 104 posited that the Over-the-scope clip is effective as a salvage therapy for achieving hemostasis in cases of acute bleeding from small varices following the failure of EVL. Additionally, a second endoscopic intervention has been associated with a decreased rate of early re-bleeding after EVL in patients with AEGVB. 105 However, several critical considerations must be addressed: (1) The available evidence is limited, with only a few published reports evaluating the efficacy of a second endoscopic treatment following the failure of initial endoscopy for AEGVB. There is a significant lack of high-quality randomized controlled trials assessing this intervention. (2) There is a need for well-defined criteria for selecting appropriate treatment techniques. Secondary ligation is recommended for residual venous columns exceeding 5 mm without active bleeding, whereas EIS/ECI is more suitable for diffuse oozing or small remnants, although these methods carry the risk of ectopic embolization. Combining techniques, such as ligation and sclerotherapy, may improve hemostasis, although further validation of their safety is necessary. Future research should prioritize multicenter studies aimed at refining stratification criteria for secondary endoscopic procedures, taking into account bleeding patterns and variceal classification. The identification of biomarkers, such as changes in HVPG, may enhance the understanding of the benefits associated with personalized second-line interventions.
SEMS
Over the past decades, SEMS have been increasingly utilized as a salvage therapy for refractory EGVB, particularly in cases where bleeding persists or recurs and alternative treatments, such as TIPS or surgical options, are not available.7,106,107 SEMS are endorsed by the ESGE, Baveno VII, and AASLD guidelines as a superior option to balloon tamponade due to their enhanced effectiveness as a temporary and remedial measure.1,7,13
The placement of SEMS is relatively straightforward and demonstrates a high success rate for stent deployment, making it particularly advantageous in instances of massive AEGVB or rebleeding, where blood may obscure endoscopic visualization. SEMS provide temporary hemostatic compression, maintain the esophageal lumen to facilitate early oral intake, and allow time for additional interventions such as TIPS. Compared to balloon tamponade, SEMS are less complex to manage and can remain in situ for up to 7 days, although they are associated with higher costs. Commonly utilized stents include the SX-Ella-Danis stent, which has demonstrated a bleeding control rate ranging from 90.6% to 96% and a rebleeding rate of 35% following removal.108,109 Some experts advocate for the use of metallic clips or the Overstitch system to secure the stent and mitigate the risk of migration.110,111
Other endoscopic interventions
Hemostatic clips placement
Hemoclips were initially employed in the treatment of variceal hemorrhage by Miyoshi in 1990. The mechanism of action involves the mechanical occlusion of the bleeding vessel and surrounding mucosal tissue, thereby achieving immediate hemostasis. Yol et al. 112 have advocated for the use of titanium clips as an adjunctive treatment for larger GOV situated deeper within the mucosa. 113 In most clinical settings, metal clips are employed to complement other endoscopic techniques for the management of AEGVB. For instance, metal clips can be used in conjunction with ECI to occlude veins and reduce or block blood flow prior to conventional ECI, thereby decreasing rebleeding rates and mitigating postembolization risks associated with ECI.114,115
Several studies have documented successful cases of using hemostatic clips for the treatment of EGVB.104,116 Hemostatic clips represent a straightforward and viable transitional method for emergency hemostasis, which can be effectively implemented in primary care hospitals. However, the use of over-the-scope clip is associated with high costs and potential risks, such as clip failure, delayed displacement, and ischemic necrosis of the mucosa. Consequently, further research is warranted to validate their efficacy in treating AEGVB.
Argon plasma coagulation combined with EVL/EIS
Argon plasma coagulation (APC) achieves hemostasis by employing high-frequency electrical current to ionize argon gas, thereby generating a conductive plasma stream. This plasma induces superficial tissue coagulation through thermal protein denaturation without necessitating direct contact between the probe and the tissue. The technique effectively occludes microvessels with diameters of ⩽0.3 cm, facilitating extensive hemostasis in a short duration. However, there is currently a paucity of robust evidence-based medical data supporting the use of APC as a monotherapy for AEGVB. APC’s characteristics of shallow penetration and rapid coagulation minimize excessive damage to surrounding healthy tissues and reduce the incidence of complications. Consequently, APC is frequently used in conjunction with other endoscopic treatment modalities. 117 Some studies have indicated that APC, when used following EIS, is both safe and effective in preventing the recurrence of esophageal varices. Sequential treatment strategies that combine EVL, EIS, and APC have been shown to significantly decrease the rates of variceal recurrence and rebleeding.118–120
Thrombin injection
Additionally, thrombin injection has been employed in the management of gastric varices since 1947. This endoscopic technique involves the direct injection of thrombin into the bleeding varix using a sclerotherapy needle. Thrombin facilitates hemostasis by converting fibrinogen into a fibrin clot within the bloodstream. Presently, recombinant human thrombin is the most frequently utilized formulation. 121 The Baveno VII consensus acknowledges thrombin as a tissue adhesive and advocates for its application in AEGVB originating from IGV1 and GOV2 varices that extend beyond the cardia. 7 Similarly, guidelines in the United Kingdom also consider thrombin injection for these specific variceal types. 122 A notable advantage of thrombin injection is its favorable safety profile and low incidence of complications, which facilitate effective hemostasis in AEGVB. Research indicates that, in comparison to cyanoacrylate injection, human thrombin injection is associated with a lower incidence of ulceration, a reduced re-bleeding rate for GOV, and avoids severe complications such as ectopic embolism, which can occur with cyanoacrylate.123–125 Despite these advantages, the adoption of thrombin injection is constrained by certain limitations. Recombinant human thrombin is relatively costly, and as a result, most clinical guidelines have not unequivocally endorsed it as a first-line treatment for AEGVB. Moreover, the existing corpus of clinical research on thrombin injections limited, highlighting the necessity for additional randomized controlled trials to definitively establish its long-term efficacy and safety.
EUS-guided injection
During AEGVB, numerous blood clots may persist within the gastric cavity, obstructing the endoscopic view. EUS has the capability to penetrate blood clots and mucosal layers, differentiate between varices and gastric wall folds, and facilitate precise localization and targeted injection. Real-time monitoring of tissue adhesive diffusion patterns allows for immediate adjustments to the injection volume, thereby preventing extravasation beyond the vessels and enhancing injection efficiency while minimizing the risks of embolization.126,127
Currently, EUS intervention therapy involves the injection of tissue adhesives and the use of endoscopic coils for embolization, either independently or in combination. EUS-CYA injection provides rapid hemostasis, reduced tissue adhesion, and a lower rebleeding rate compared to traditional methods.128–131 Binmoeller et al. demonstrated that the combination of EUS-guided coil placement with CYA achieves a vessel occlusion rate of up to 96%. The coils function as scaffolds for glue retention, thereby reducing glue usage and minimizing the risks of ectopic embolization. This approach has proven effective in managing active bleeding from gastric varices and spontaneous portosystemic shunts.132–134 Evidence suggests that EUS-guided CYA injection combined with coil embolization significantly reduces rebleeding rates and adverse events compared to the use of CYA or coils alone, although the majority of studies are retrospective or nonrandomized.126,135–137
EUS-guided coil placement with CYA therapy presents certain limitations, such as the risks of coil displacement, bleeding, pain, fever, and embolism.138,139 Bazarbashi et al. 140 proposed the use of an absorbable gelatin sponge in conjunction with coils to mitigate the risk of embolism compared to CYA, although this approach is supported by limited studies.
Our institution has successfully implemented EUS-guided coil placement with ECI for the treatment of gastric fundal varices (Figures 3–5). Nonetheless, the availability of EUS equipment is limited, and the cost for coils remains high. There is considerable variability in endoscopic techniques, including injection sites, needle sizes, coil specifications, and types of CYA used across different studies. Therefore, standardization of EUS-guided variceal injection methods and further research into their efficacy are imperative.

EUS-guided coil placement. (a) Gastric varices. (b) EUS showing varices (white arrow). (c) EUS-identified perforating branches originating from the left gastric vein (white arrow). (d) Visualization of gastric variceal clusters (white arrow) on postpuncture venography. (e) EUS-guided coil (white arrows) deployment. (f) Completion of coils (white arrows) deployment.

EUS-guided sclerotherapy of esophageal perforating vessels. (a) Incomplete resolution of varices. (b) EUS reveals esophageal veins and perforating veins. (c) Residual varicose veins. (d) Sclerotherapy of perforating vessels. (e) Disappearance of esophageal varices. (f) No vascular echoes within the esophageal wall.
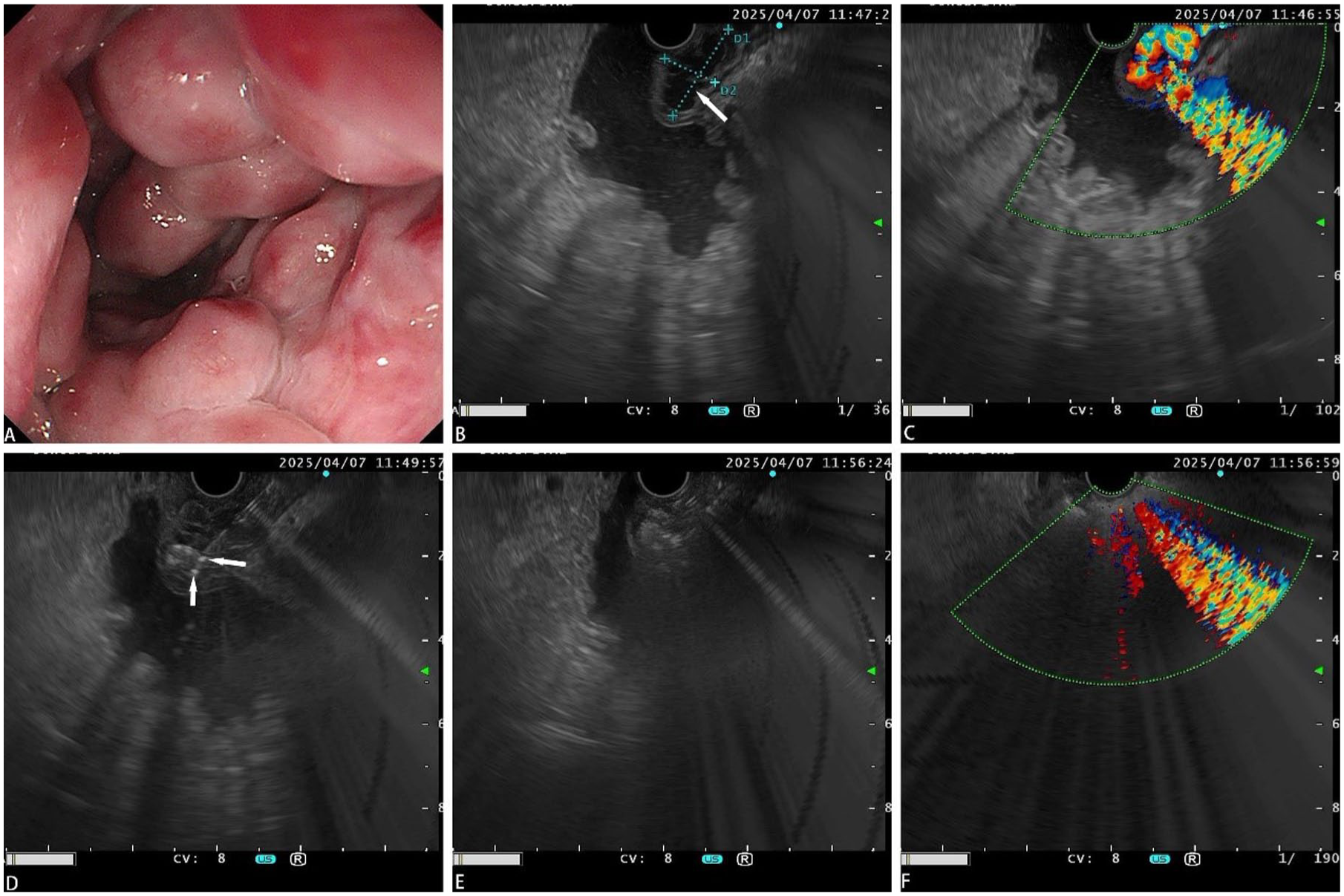
EUS-guided coil placement combined with tissue adhesive injection. (a) Gastric varices. (b) EUS measurement of variceal size (white arrow). (c) EUS confirming Doppler flow within varices. (d) EUS-guided coil deployment (white arrows). (e) EUS-guided tissue adhesive injection. (f) Diminished Doppler flow on EUS.
Hemostatic powder spray
Hemostatic powder, administered through endoscopic spraying, demonstrates efficacy in arresting hemorrhage by creating a physical barrier and promoting coagulation, even in patients receiving antiplatelet or anticoagulant therapy. This method is particularly advantageous in cases of diffuse bleeding or in environments with limited visibility, offering a high rate of immediate success. While the European guidelines acknowledge its utility as a salvage intervention, global clinical experience with hemostatic powder remains sparse. It can serve as a temporary solution prior to definitive treatment when conventional endoscopic methods are unsuccessful or when TIPS procedures are contraindicated, providing a therapeutic window of 48–72 h. Initiating a transition to TIPS or a secondary endoscopic intervention within 24 h following the application of the powder may mitigate the risk of rebleeding.13,141–143 However, its clinical use is constrained by the complexity of its delivery method and route.
Future directions
EUS-guided TIPS
The TIPS procedure poses challenges for patients with obstructions in the inferior vena cava or hepatic veins. Researchers propose the use of EUS-guided TIPS, which involves the puncturing of the hepatic and portal veins under EUS guidance, as an alternative to traditional imaging techniques. This method circumvents the need to access the right heart or inferior vena cava and eliminates radiation exposure for operators. It can also be integrated with other EUS-guided treatments to enhance efficiency. Although this method has been tested in animal models, successful clinical applications remain limited, necessitating further studies to validate its safety and practicality. 144
Conclusions and prospects
AEGVB is a prevalent and severe complication in patients with liver cirrhosis. In recent years, various treatments have been developed, each with distinct advantages and disadvantages. The current endoscopic treatment framework for AEGVB is structured around a three-tiered objective of “hemostasis-eradication.” (1) Acute hemostasis: EVL is the preferred initial treatment. EIS is indicated when EVL is unsuccessful or in cases of refractory bleeding. ECI and modified techniques are indispensable for managing gastric fundal variceal bleeding. Additionally, thrombin injection may be considered. Novel combined approaches, such as preplaced coils with low-dose tissue adhesive, can mitigate the risk of ectopic embolization. (2) Bridge therapy for refractory bleeding: SEMS may replace balloon tamponade as the primary bridging method. (3) Sequential eradication: To effectively eliminate residual varices and prevent early rebleeding, sequential eradication protocols incorporate supplementary EVL, EIS, or low-dose ECI, along with EUS-guided injection. It is recommended that endoscopic follow-up be conducted within 6–12 months, with new variceal vessels potentially managed using EUS guidance. The continuous advancement of EUS technology enhances its role as both a diagnostic and therapeutic tool in the management of GOV. The development of EUS-related techniques is progressing rapidly, showing promise as a primary method for precise targeted therapy. Various endoscopic techniques for AEGVB possess distinct indications and limitations; therefore, treatment should be tailored based on a comprehensive assessment of variceal type, bleeding activity, and liver function. The primary objectives are to achieve immediate hemostasis and implement sequential therapy to reduce the risk of rebleeding and accomplish variceal obliteration, ultimately improving long-term patient outcomes.
