Abstract
Background:
Isolated gastric varices type 1 (IGV1) with spontaneous shunts carry a high risk of bleeding and ectopic embolization following cyanoacrylate injection.
Objectives:
This study aimed to compare the efficacy and safety of endoscopic ultrasonography-guided cyanoacrylate injection (EUS-CYA) versus direct endoscopic injection of cyanoacrylate (DEI-CYA) in patients with IGV1 and spontaneous shunts.
Design:
A prospective randomized controlled trial (RCT).
Methods:
This prospective RCT enrolled patients with cirrhosis complicated by IGV1 and spontaneous portosystemic shunts. Eligible patients were randomly assigned to either the EUS-CYA group or the DEI-CYA group. The primary outcome was the rebleeding rate, while the secondary outcomes included technical success rate, gastric varices occlusion rate, cumulative non-bleeding rate, mortality rate, and incidence of ectopic embolization.
Results:
A total of 96 patients were randomized to either the EUS-CYA (n = 47) or DEI-CYA (n = 49) group, both achieving 100% technical success. The rebleeding rate was significantly lower in the EUS-CYA group compared to the DEI-CYA group (19.15% vs 42.86%, p = 0.012), particularly for late rebleeding (17.02% vs 36.73%, p = 0.03). Kaplan–Meier analysis showed that the cumulative non-bleeding rates at 6, 12, 18, and 24 months were higher in the EUS-CYA group compared to the DEI-CYA group (p = 0.042). Mortality rates were similar between the groups (12.77% vs 16.33%, p = 0.621). No ectopic embolization occurred in the EUS-CYA group, while one case of pulmonary embolism was observed in the DEI-CYA group.
Conclusion:
EUS-CYA demonstrates a higher gastric varices occlusion rate and a lower rebleeding rate for IGV1 with spontaneous shunts compared to DEI-CYA.
Trial registration:
Chinese Clinical Trial Registry, ChiCTR2200058888.
Plain language summary
Isolated gastric varices type 1 (IGV1) are dilated submucosal veins in the stomach that can bleed heavily and cause serious health risks, especially in patients with cirrhosis. In some cases, these veins are connected to other blood vessels (spontaneous shunts), making treatment even more challenging. One common way to treat these veins is by injecting cyanoacrylate to block the veins. This procedure can be performed via two main techniques: direct endoscopic injection (DEI-CYA) or endoscopic ultrasound-guided injection (EUS-CYA). This study aimed to compare the safety and efficacy of these two methods. The findings revealed that while both methods were successful in achieving initial obliteration, the EUS-guided method (EUS-CYA) demonstrated superior outcomes.
Patients who received EUS-CYA had a lower rate of rebleeding (especially in the long term) and a higher chance of staying free from bleeding over time. No serious complications, such as embolism, were observed in the EUS-CYA group, while one patient in the DEI-CYA group experienced a pulmonary embolism. Overall, the study suggests that ultrasound-guided cyanoacrylate injection is a safer and more effective option for treating IGV1 with spontaneous shunts compared to the traditional direct injection method.
Introduction
Gastric varices (GVs) are part of the pathological collateral circulation between the portal venous system and the systemic circulation. 1 Although the bleeding rate is lower than that of esophageal varices, GV presents with a larger volume of bleeding, necessitates higher transfusion rates, and is associated with a greater risk of rebleeding 2 (34%–89%) and mortality 3 (up to 30%). In the Sarin classification of GV, the bleeding incidence is highest for isolated gastric varices type 1 (IGV1), reaching 80%. 4 The Baveno VII consensus recommends direct endoscopic injection of cyanoacrylate (DEI-CYA) as the first-line treatment for IGV1. 5 However, the risk of rebleeding after cyanoacrylate injection is as high as 25%–50%, 6 and there is also a risk of ectopic embolization. 7 Endoscopic IGV1 appears as tumor-like varices, often accompanied by shunt vessels, which increases the likelihood of cyanoacrylate entering the systemic circulation, significantly elevating the risk of ectopic embolization. 7
In recent years, with a deeper understanding of the anatomy and hemodynamics of the collateral pathways associated with the GV system, optimal treatment strategies for gastric variceal bleeding have been continuously evolving. Endoscopic ultrasonography-guided cyanoacrylate injection (EUS-CYA) allows for accurate identification of varices and their feeding vessels, enabling precise intravascular injection that induces targeted vessel occlusion while reducing the amount of adhesive used and lowering the risk of embolization. 8 Some studies suggest that EUS-CYA is superior to DEI-CYA for GVs.9–12 However, for patients with IGV1 combined with spontaneous shunts, the risk of ectopic embolism raises questions about whether EUS-CYA is safer than DEI-CYA, as there is currently no research on this matter. Therefore, we conducted a prospective trial to compare the efficacy and safety of EUS-CYA versus DEI-CYA in treating IGV1 with spontaneous shunts.
Methods
Study design and ethics
This was a prospective study of patients admitted to the Department of Gastroenterology at the First Affiliated Hospital of Anhui Medical University from July 1, 2020, to December 31, 2023, for IGV1 bleeding due to cirrhosis. The patients were randomly divided into two groups: one group received EUS-CYA, while the other group received DEI-CYA. The study was approved by the Ethics Committee of the First Affiliated Hospital of Anhui Medical University (Approval No. PJ-2020-16-21), and informed consent was obtained from both the patients and their families. This study was conducted in accordance with the Declaration of Helsinki (2013), and the trial has been registered in the clinical trial database with the registration number ChiCTR2200058888. All patient information in text, tables, and figures was fully de-identified to ensure confidentiality.
Inclusion criteria: (1) age between 18 and 80 years; (2) diagnosis of liver cirrhosis according to treatment guidelines; (3) endoscopically confirmed active bleeding from IGV1 or a history of bleeding within the past 3 months; (4) confirmation of spontaneous portosystemic shunt via abdominal CT angiography.
Exclusion criteria: (1) the bleeding source was identified endoscopically as GOV1, GOV2, or IGV2; (2) presence of hepatocellular carcinoma or other malignancies at other sites; (3) severe cardiopulmonary diseases preventing surgical tolerance; (4) hepatic encephalopathy, hepatorenal syndrome, hepatopulmonary syndrome, or multiple organ failure; (5) previous endoscopic treatment, Transjugular intrahepatic portosystemic shunt (TIPS), or surgical procedures; (6) severe coagulopathy; (7) pregnancy.
The primary endpoint was the rebleeding rate, including both early (5 days to 6 weeks) and late (>6 weeks) postoperative rebleeding. Secondary endpoints included the technical success rate, gastric variceal occlusion rate, cumulative non-bleeding rate, mortality, incidence of ectopic embolization, and other procedure-related complications.
Randomization
This randomized controlled trial (RCT) was conducted in accordance with the SPIRIT guidelines and reported following the CONSORT 2025 standards.13,14 The patients who met the inclusion criteria were continuously randomized in a 1:1 ratio to either undergo EUS-CYA or DEI-CYA, utilizing the web-based randomization system Research Randomizer. Following eligibility screening, a research coordinator, who was not involved in data collection or analysis, entered the baseline data of the patients into the allocation system. The system generated a unique study identification number along with a corresponding treatment number for either EUS-CYA or DEI-CYA. This study was conducted using a single-blind design. Due to the nature of the interventions, both clinicians and patients were aware of the treatment allocation. However, individuals involved in data analysis remained blinded to the randomization list and did not participate in the treatment, thus ensuring blinding for treatment allocation.
Sample size calculation was conducted using PASS 15.0 software (NCSS, LLC,Kaysville, Utah, USA). To achieve 80% statistical power at a two-sided 5% significance level, and based on results from previous studies,11,12,15 it was assumed that the rebleeding rate was 8.8% for the EUS-CYA group and 32% for the DEI-CYA group. The sample size calculated by the software was 44 patients per group. Considering an estimated 10% loss rate, a minimum of 48 patients per group was required. Therefore, our target was to enroll a total of 96 patients.
Procedures
All procedures were performed by two experienced endoscopists proficient in both EUS-guided and conventional endoscopic cyanoacrylate injection techniques. Each patient was treated by one of these operators to ensure consistency across both the EUS-CYA and DEI-CYA groups. Operator assignment was balanced and independent of treatment allocation.
Endoscopic ultrasound-CYA
Prior to treatment, gastroscopy and imaging results were reviewed. Endoscopic ultrasound (EUS) was used to identify GVs, assessing their size and relationship to surrounding structures. EUS revealed multiple anechoic cavities in the greater curvature of the stomach with extra-wall penetrating branches exhibiting blood flow signals. The blood flow spectrum was confirmed as venous flow. The “modified sandwich technique” was applied using lauromacrogol-cyanoacrylate injections, adjusted based on varix size. The cyanoacrylate dosage was based on varix size: 1 mL for varices 1 cm in diameter, and 1 mL/cm for larger varices. Ultrasound showed immediate cessation of blood flow post-injection, with additional injections as needed. A second injection could be given 2–3 weeks later if necessary (Figure 1(a)).
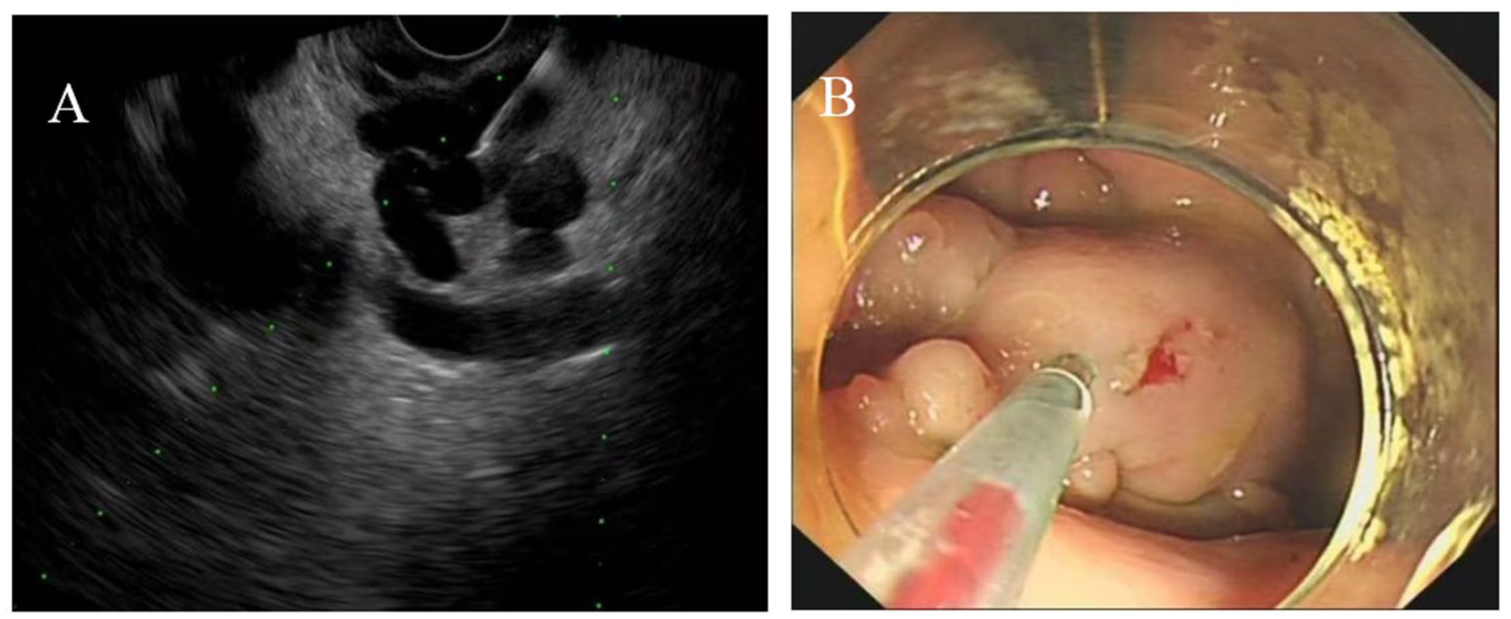
(a) EUS-CYA: The endoscopic ultrasound showed varicose veins within the gastric wall, with a puncture needle being guided toward the target blood vessel for the injection of cyanoacrylate. (b) DEI-CYA: Cyanoacrylate was injected into the varix using the “modified sandwich method.”
Direct endoscopic injection of cyanoacrylate
The patient’s medical history, physical examination, and pertinent diagnostic tests (gastroscopy and CT) were reviewed to assess varices. Following anesthesia, gastroscopy was performed to inspect the gastric mucosa, accurately identify the varices, and observe columnar reflux in the needle. The “modified sandwich technique” was employed for injection, with the amount of cyanoacrylate adjusted as previously described. After injection, the firmness of the vessel could be assessed by applying pressure with the needle sheath. If the varices still felt soft, additional glue injection was carried out until the entire varix was fully solidified. If necessary, a second injection could be performed 2–3 weeks after the initial procedure (Figure 1(b)).
Follow-up
No patients in this study received non-selective beta blockers (NSBBs) during the study period. EUS-CYA and DEI-CYA treatments were repeated every 2 weeks until GV eradication was confirmed. Rebleeding events were assessed within 6 weeks after the endoscopic intervention. Follow-up endoscopy or EUS was performed at 3 months (or earlier if rebleeding occurred) to evaluate variceal occlusion. Subsequent examinations were conducted every 6 months or upon rebleeding to monitor the resolution or recurrence of varices. Follow-up data included the occurrence, timing, and cause of rebleeding, survival status, and any complications. Follow-up concluded 6 months after the last patient was enrolled or at the time of death. For patients who experienced rebleeding, additional treatments—including EUS-CYA, DEI-CYA, Balloon-occluded retrograde transvenous obliteration (BRTO), TIPS, or surgery—were considered as appropriate.
Early rebleeding was defined as recurrent bleeding occurring between 5 days and 6 weeks after the procedure, whereas late rebleeding was defined as recurrence occurring more than 6 weeks post-procedure.
Statistical analysis
Data were analyzed using SPSS 26.0 (IBM, Armonk, New York, USA). Normally distributed continuous variables were expressed as χ ± s, with paired t tests for group comparisons. Non-normally distributed data were presented as median (Q1, Q3) with interquartile range, and compared using the Wilcoxon rank-sum test. Categorical data were shown as n (%), with comparisons made using the Chi-square or Fisher’s exact test for non-ordinal variables, and the Wilcoxon rank-sum test for ordinal variables. Kaplan–Meier curves were used to compare cumulative non-bleeding rates, with intergroup comparisons via the log-rank test. A p-value <0.05 was considered significant.
Results
Baseline characteristics
This study enrolled a total of 113 patients with bleeding due to cirrhosis and IGV1. After applying the inclusion and exclusion criteria, 17 patients were excluded, leaving 96 patients ultimately included in the study. The patients were randomly divided into two groups: one group received EUS-CYA, comprising 47 patients, while the other group received DEI-CYA, comprising 49 patients (Figure 2). There were no statistically significant differences in baseline characteristics between the two groups (p > 0.05; Table 1).

Flowchart of study profile.
Baseline characteristics of the study group.

ALB, albumin; ALP, alkaline phosphatase; ALT, alanine transaminase; AST, aspartate transaminase; BUN, blood urea nitrogen; DEI-CYA, direct endoscopic injection of cyanoacrylate; EUS-CYA, endoscopic ultrasonography-guided cyanoacrylate injection; GGT, gamma-glutamyl transpeptidase; GV, gastric varices; INR, international normalized ratio; PLT, platelet; PT, prothrombin time; TBIL, total bilirubin.
The primary endpoint of the study
Rebleeding
The mean follow-up duration was 24.17 ± 10.51 months for the EUS-CYA group and 24.69 ± 11.71 months for the DEI-CYA group, with no significant difference (p < 0.05). During follow-up, nine rebleeding cases (19.15%) occurred in the EUS-CYA group, including one early and eight late rebleeding cases. The DEI-CYA group had 21 cases (42.86%), with 3 early and 18 late rebleeding. The rebleeding rate in the EUS-CYA group was significantly lower than that in the DEI-CYA group (χ2 = 6.276, p = 0.012). The late rebleeding rate was also significantly lower in the EUS-CYA group (17.02%) than in the DEI-CYA group (36.73%; χ2 = 4.721, p = 0.03; Table 2).
Comparison of the primary study endpoint results between the two groups.

DEI-CYA, direct endoscopic injection of cyanoacrylate; EUS-CYA, endoscopic ultrasonography-guided cyanoacrylate injection.
The secondary endpoints of the study
Treatment outcomes
In the EUS-CYA group, 6 procedures were performed emergently and 41 electively, achieving a 100% technical success rate. In the DEI-CYA group, 9 procedures were emergent and 40 elective, also with a 100% success rate. In the EUS-CYA group, 38.30% (18/47) of patients had para-gastric veins, with an average vessel diameter of 7.20 ± 2.32 mm (3–12 mm). The cyanoacrylate volume used was 1.63 ± 1.08 mL in the EUS-CYA group and 2.59 ± 1.66 mL in the DEI-CYA group, with a significant difference (t = 3.385, p = 0.001). The volume of lauromacrogol was 4.99 ± 4.79 mL in the EUS-CYA group and 7.31 ± 5.32 mL in the DEI-CYA group (t = 2.196, p = 0.031; Table 3).
Comparison of the secondary study endpoint results between the two groups.

DEI-CYA, direct endoscopic injection of cyanoacrylate; EUS-CYA, endoscopic ultrasonography-guided cyanoacrylate injection.
In the EUS-CYA group, 37 patients required a single endoscopic procedure, 9 required two, and 1 required three, with a mean of 1.23 ± 0.48 procedures per patient. In the DEI-CYA group, 28 patients required 1 procedure, 14 required 2, and 7 required 3, with a mean of 1.57 ± 0.74 procedures per patient (t = 2.678, p = 0.009; Table 3).
GV occlusion rate
Three months post-surgery, all patients underwent endoscopic or EUS examination. In the EUS-CYA group, 42 cases exhibited complete occlusion of GV, while 5 cases showed partial occlusion. In the DEI-CYA group, 33 cases had complete occlusion, and 16 cases had partial occlusion. The difference between the two groups was statistically significant (χ2 = 6.803, p = 0.009; Table 3).
Cumulative rate of non-bleeding
Kaplan–Meier analysis indicated that the cumulative rate of non-bleeding at 6, 12, 18, and 24 months in the EUS-CYA group was 89.4%, 84.6%, 81.8%, and 76%, respectively. In the DEI-CYA group, the rates were 81.6%, 77.3%, 68.4%, and 63.2%, respectively. The difference between the two groups was statistically significant (log-rank test, χ2 = 4.154, p = 0.042; Figure 3).

Kaplan–Meier curve analysis of the cumulative incidence of non-bleeding.
Survival
During the follow-up period, six patients in the EUS-CYA group died. Of these, two died from liver cancer, three from liver failure, and one from bleeding. In the DEI-CYA group, eight patients died, with two succumbing to liver cancer, four to liver failure, and two to bleeding. The mortality rates in the two groups were 12.77% and 16.33%, respectively, with no statistically significant difference (χ2 = 0.244, p = 0.621; Table 3).
Complications
No ectopic embolism occurred in the EUS-CYA group. In the DEI-CYA group, one patient developed respiratory distress and chest pain on the first postoperative day. Endoscopy showed nodular GV (3.0 cm) and a gastric-renal shunt (9.1 mm). The patient received 4 mL of glue and 12 mL of polidocanol. A chest CT and CT pulmonary angiography revealed a pulmonary embolism. After treatment, symptoms improved, and no recurrence occurred during follow-up. There was no significant difference in complication rates between the groups (Table 3).
Discussion
This study primarily investigated the efficacy of EUS-CYA and DEI-CYA in patients with IGV1 and spontaneous shunt pathways. The results indicated EUS-CYA effectively detects the para-gastric veins, achieves a higher rate of vessel occlusion, utilizes less adhesive, and reduces the number of procedures required. While early postoperative rebleeding rates were similar, EUS-CYA had significantly lower rates of late rebleeding, overall rebleeding, and cumulative non-bleeding at 6, 12, 18, and 24 months (p < 0.05), suggesting its advantage in prolonging rebleeding-free periods and reducing long-term rebleeding risk. Both treatments had no impact on overall survival. One ectopic embolism occurred in the DEI-CYA group, while no systemic ectopic embolism was observed in the EUS-CYA group.
DEI-CYA is one of the effective treatments for gastric variceal bleeding. 16 However, the use of DEI-CYA for treating IGV1 is “blind,” as IGV1 involves a large network of para-gastric collateral circulation that cannot be identified endoscopically, particularly the complex inflow and outflow vessels located outside the gastric wall. 17 Research has shown that the risk of variceal recurrence and bleeding is associated with the number, diameter, and persistent blood flow of the varices. 18 Therefore, closing GV and para-gastric veins can effectively reduce rebleeding risk. 19 EUS, combining endoscopy and ultrasound, accurately detects IGV1, 15 identifies submucosal GV, dilated vessels, para-gastric vessels, and perforating veins, 20 improving injection accuracy and reducing adhesive use and ectopic embolism risks. 21 In this study, there was no difference in early rebleeding rates, but the EUS-CYA group had significantly lower rates of late rebleeding and overall rebleeding. EUS-CYA allowed for precise identification of GV and feeding veins, leading to better control of glue application, reducing glue leakage, and ensuring more reliable embolization, thus lowering the long-term rebleeding risk.
In this study, the EUS-CYA group used significantly less cyanoacrylate than the DEI-CYA group (1.63 ± 1.08 vs 2.59 ± 1.66 mL, p = 0.001), consistent with findings by Sabry et al. 22 and Bick et al. 11 The EUS-CYA group also required fewer treatment sessions, with 37 cases (78.72%) achieving occlusion after a single treatment, compared to 28 cases (57.14%) in the DEI-CYA group, consistent with the results of Sabry et al. 22 The EUS-CYA group had a lower rebleeding rate, less cyanoacrylate usage, and fewer injections. These results are due to EUS’s ability to precisely locate GV, target varicose veins, and confirm eradication via post-procedural Doppler examination. 11
In a study on EUS-CYA for the treatment of GV, all patients received cyanoacrylate injection targeting the perforating veins, which significantly reduced the amount of cyanoacrylate used and the risk of ectopic embolization. 23 The research by Romero-Castro et al. 24 also concluded that targeting gastric perforating veins or collateral veins is feasible and does not increase the incidence of complications compared to DEI-CYA. Additionally, Ma et al. 19 investigated the independent risk factors for rebleeding in the treatment of GV, and the results showed that the presence of para-gastric veins was a risk factor for rebleeding. In the EUS-CYA group of this study, 18 cases of perforating veins outside the gastric wall were identified, with a diameter of 7.20 ± 2.32 mm, all of which were successfully occluded. EUS-guided injection of cyanoacrylate into the perforating veins can achieve maximal blood flow obstruction while using a lower volume of cyanoacrylate. This approach helps avoid additional punctures or excessive adhesive filling of the varices, thus reducing the incidence of rebleeding and adverse reactions. 23 In this study, there was no statistically significant difference in the mortality between two groups. Previous research had shown that the mortality rate in patients with GV was related to Child-Pugh and Model for End-stage Liver Disease (MELD) score,25–28 suggesting that patient’s survival was primarily associated with the underlying liver disease.
In our treatment process, the primary concern is the risk of ectopic embolism, which literature suggests ranges from 0% to 2% after cyanoacrylate injection. 29 This study included patients with IGV1 and spontaneous shunts, and the risk of ectopic embolization was significantly elevated postoperatively. In the DEI-CYA group, one patient with a varix diameter of 3.0 cm, exhibiting a tumor-like shape and a large shunt diameter, developed a pulmonary embolism after the procedure. This suggests that large IGV1 is frequently associated with spontaneous shunts, thus presenting a higher risk of ectopic embolization. Additionally, the volume of cyanoacrylate used also contributes to this risk.2,30 No ectopic embolization occurred in the EUS-CYA group. While EUS-CYA also carries some risk, it allows for precise blood flow assessment, controlled injection speed, and glue delivery, reducing the risk of excessive glue use. Additionally, EUS with Doppler ensures accurate vessel closure and minimizes ectopic embolization. 21 There was no statistically significant difference in the incidence of ectopic embolism between the two groups. As cyanoacrylate and polidocanol are not detectable by X-ray, this study could not detect asymptomatic embolism. Future research should use iodine oil into cyanoacrylate and routine CT scans to identify asymptomatic embolisms.
Several studies have reported that the EUS coil + glue combination achieves higher variceal obliteration rates and lower rebleeding rates compared with EUS-guided glue injection alone.31,32 However, coil-assisted therapy was not included as a study arm in our trial for several reasons. First, coil deployment was not routinely performed at our center during the study period due to limited device availability and variable operator experience. Second, the primary objective of our trial was to assess whether EUS guidance alone—independent of coil use—could reduce rebleeding and ectopic embolization in IGV1 patients with spontaneous shunts, a subgroup at particularly high risk when treated with direct endoscopic injection. Third, the coil + glue technique entails substantially higher costs and was not covered by local insurance at the time of patient recruitment, limiting its feasibility for inclusion. Future randomized studies comparing EUS-glue, EUS coil + glue, and DEI-CYA are warranted to further determine the optimal strategy for managing IGV1 with spontaneous shunts.
This study has several limitations. First, all surgeries were performed at the same tertiary hospital, potentially introducing regional bias and limiting generalizability. Second, all procedures were conducted by experienced endoscopists, which ensures precision but limits applicability to other practitioners with varying expertise. Third, the small sample size may impact the results’ stability. Fourth, NSBBs were not administered in this study, which may limit the generalizability of our findings to populations where NSBB use is standard practice. Future multi-center, large-scale RCTs are needed to confirm the clinical efficacy of EUS-CYA in IGV1 patients with spontaneous shunts, while also evaluating the combined effects of NSBB therapy and endoscopic interventions on rebleeding and overall clinical outcomes.
Conclusion
EUS-CYA demonstrates a high GV occlusion rate and a lower rebleeding rate for IGV1 with spontaneous shunts compared to DEI-CYA.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251413353 – Supplemental material for Endoscopic ultrasonography-guided cyanoacrylate injection versus endoscopic cyanoacrylate injection for treating isolated gastric varices type 1 bleeding with spontaneous shunts: a randomized controlled trial
Supplemental material, sj-docx-1-tag-10.1177_17562848251413353 for Endoscopic ultrasonography-guided cyanoacrylate injection versus endoscopic cyanoacrylate injection for treating isolated gastric varices type 1 bleeding with spontaneous shunts: a randomized controlled trial by Jing Li, Yaxian Kuai, Xuecan Mei, Zhihong Wang, Zhuang Zeng, Yingying He and Derun Kong in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
