Abstract
Background:
Esophageal-gastric variceal bleeding (EGVB) is a serious complication in patients with liver cirrhosis, characterized by high mortality and rebleeding rates. The effect of sequential endoscopic therapy on patient mortality and rebleeding rates remains unclear.
Objectives:
This study aimed to evaluate the effects of sequential endoscopic therapy on mortality and rebleeding rates in patients with EGVB.
Design:
In this single-center retrospective study, 373 hospitalized cases of EGVB caused by liver cirrhosis, collected between November 2019 and November 2023, were divided into four groups according to different treatment methods: a sequential endoscopy group, emergency endoscopy group, emergency endoscopy plus transjugular intrahepatic portosystemic shunt (TIPS) group and control group.
Methods:
Mortality and rebleeding rates were compared among the four groups using statistical analyses.
Results:
The mortality and rebleeding rates of the sequential endoscopy group (3.7% and 19%, respectively) were significantly lower than those of the emergency endoscopy (22% and 36%, respectively), emergency endoscopy plus TIPS (33% and 28%, respectively), and control groups (33% and 51%, respectively) (p = 0.013 and p = 0.013, respectively).
Conclusion:
Sequential endoscopic therapy may significantly reduce the mortality and rebleeding rates of patients with EGVB compared to other conventional treatment strategies. The findings of the study could help develop approaches benefiting EGVB treatment.
Introduction
Esophageal-gastric variceal bleeding (EGVB) is one of the most common and serious complications of liver cirrhosis. Despite advances in diagnostic and treatment methods, the mortality rate of this condition remains high ranging from 15 to 20%.1 –3 In addition, recurrent bleeding is highly likely after management of EGVB, with a rebleeding rate of up to 17% within six weeks.2,4,5 Therefore, appropriate treatment of acute EGVB and prevention of variceal rebleeding are crucial for patients with liver cirrhosis. 6 In the past few decades, significant progress has been made in regard to treatment strategies for EGVB. Traditional treatment methods include drug therapy, endoscopic hemostasis, interventional radiology, and surgical treatment. The current first-line treatment modalities for acute variceal bleeding include nonselective beta-blockers, vasoactive agents, antibiotics, and endoscopic therapies.7 –9 In particular, endoscopic treatment is currently the preferred approach for EGVB and is widely favored by patients owing to its minimally invasive nature.10,11 For persistent esophageal variceal bleeding that continues despite medical or endoscopic therapy, urgent rescue treatment with a transjugular intrahepatic portosystemic shunt (TIPS) should be considered. 12 Although studies have demonstrated that TIPS is effective in preventing rebleeding, it has a higher incidence of postoperative hepatic encephalopathy, liver failure, and stent stenosis than that of other treatment methods, with no significant benefit in improving patient survival.13 –15 Many studies have compared different treatment methods for acute variceal bleeding. For example, in a randomized controlled study (RCT) by Garg et al., endoscopic band ligation plus low-dose sclerotherapy significantly reduced complications and rebleeding rates compared with endoscopic variceal sclerotherapy alone (3% vs 20% and 3% vs 16%, respectively). 16 In another RCT conducted by Liu et al., emergency endoscopic ligation combined with octreotide had lower costs, higher efficacy, and higher safety than the use of octreotide alone for treating acute esophageal variceal bleeding. 17 These studies aimed to investigate emergency hemostasis treatment after EGVB; however, owing to the persistent presence of portal hypertension, varicose veins can recur, leading to a high risk of rebleeding and compromising long-term treatment effectiveness. 18 Consequently, sequential endoscopic therapy has attracted much attention in the search for more effective treatment options.
Sequential endoscopic treatment includes two stages: emergency endoscopic hemostasis and prophylactic endoscopic treatment. One major advantage of gastroscopy is its reproducibility, such that multiple examinations and treatments under gastroscopy can gradually reduce the degree of varices. Preventive endoscopic treatment involves using various endoscopic methods to reduce the severity of esophageal-gastric varices (EGV), aiming to prevent rebleeding and thereby lowering patient mortality rates. The standard treatment for recurrent variceal bleeding as per the current guidelines is a combination of a nonselective beta-blocker and repeated endoscopic variceal ligation.19,20 However, clinical research data demonstrating the actual impact of repeated endoscopic therapy on patient mortality and rebleeding rates remains lacking. Therefore, this study aimed to review the application of sequential endoscopic therapy in patients with EGVB and compare its effectiveness in reducing patient mortality and rebleeding rates with that of conventional treatment strategies. Through a comprehensive evaluation of sequential endoscopic therapy, this study may provide more reliable treatment guidelines for clinical practice to improve patient prognosis.
Methods
Study design
The reporting of this study conforms to the guidelines set forth in the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 21 In this retrospective study, we enrolled 373 consecutive patients treated at Nanchong Central Hospital (The Second Clinical Medical College, North Sichuan Medical College) from November 2019 to November 2023. Only patients with liver cirrhosis who suffered from EGVB were eligible for inclusion in the study. The diagnosis of liver cirrhosis was based on a previous diagnosis of liver cirrhosis or on the results of a liver biopsy; otherwise, it was determined using a combination of clinical manifestations and laboratory and imaging examination results. In addition, the diagnosis of EGVB was based on preoperative endoscopic examination, intraoperative endoscopic hemostasis, or a comprehensive consideration of the medical history, clinical manifestations, and relevant auxiliary examinations before surgery. Patients who met the following criteria were excluded from the study: (1) less than 18 years old or older than 80 years old; (2) preoperative comorbidities, including significant heart failure (New York Heart Association cardiac function grades III and IV), sepsis, and renal failure; (3) concomitant hepatocellular carcinoma; (4) previously underwent TIPS in an external hospital; (5) lost to follow-up, refused the treatment plan determined by the doctors during hospitalization, or lacked relevant auxiliary inspection data.
Figure 1 shows a detailed research flowchart, wherein all data was obtained through the electronic medical record data management system of Nanchong Central Hospital. Any discrepancy in the data was corrected by retrieving hospital medical records. Before surgery, the following patient data were collected: age, sex, cause of cirrhosis, albumin (ALB) levels, prothrombin time (PT), total bilirubin (TBIL) levels, hemoglobin (HB) levels, white blood cell (WBC) count, neutrophil count, diabetes, hepatic encephalopathy (HE), history of splenectomy, location of varices, and initial endoscopic treatment. The severity of preoperative liver disease was also evaluated using the Child–Pugh grading system and Model for End-Stage Liver Disease (MELD) scoring criteria.20,22 The patients were divided into four groups according to different treatment methods. (1) Patients who underwent endoscopic hemostasis treatment after the first episode of acute EGVB and subsequently underwent gastroscopy every 1–3 months. In the absence of bleeding, treatment using endoscopic ligation, tissue glue, or sclerotherapy injections was performed to reduce the degree of EGV until gastric varices were completely eradicated and only small esophageal varices (less than 0.6 centimeters in diameter) that required no further treatment remained. 23 (2) The emergency endoscopy group was defined as patients with acute EGVB who underwent endoscopic hemostasis. (3) The emergency endoscopy plus TIPS group included patients with acute EGVB who underwent TIPS for recurrent bleeding or excessive ascites during hospitalization after having undergone emergency endoscopic hemostasis. (4) The control group was defined as patients with acute EGVB who did not undergo endoscopic hemostasis or TIPS and instead only received fluid resuscitation, medication (including vasoactive drugs, antibiotics, hemostatic drugs, and others), and red blood cell transfusion. The diagnostic criteria for EGVB were consistent for each group. In the absence of endoscopy in the control group, several methods were employed to preliminarily assess whether upper gastrointestinal bleeding had stopped. The bleeding was considered to have stopped when hematemesis, melena, and abdominal pain were significantly reduced or disappeared. In addition, the bleeding was considered under control when the physiological indicators of the patient such as blood pressure and heart rate began to stabilize. Similarly, the bleeding was considered to have stopped when the hemoglobin levels of the patients began to rise. Finally, the bleeding status was assessed clinically based on the general condition, skin color, and mucosal wetness of the patient.
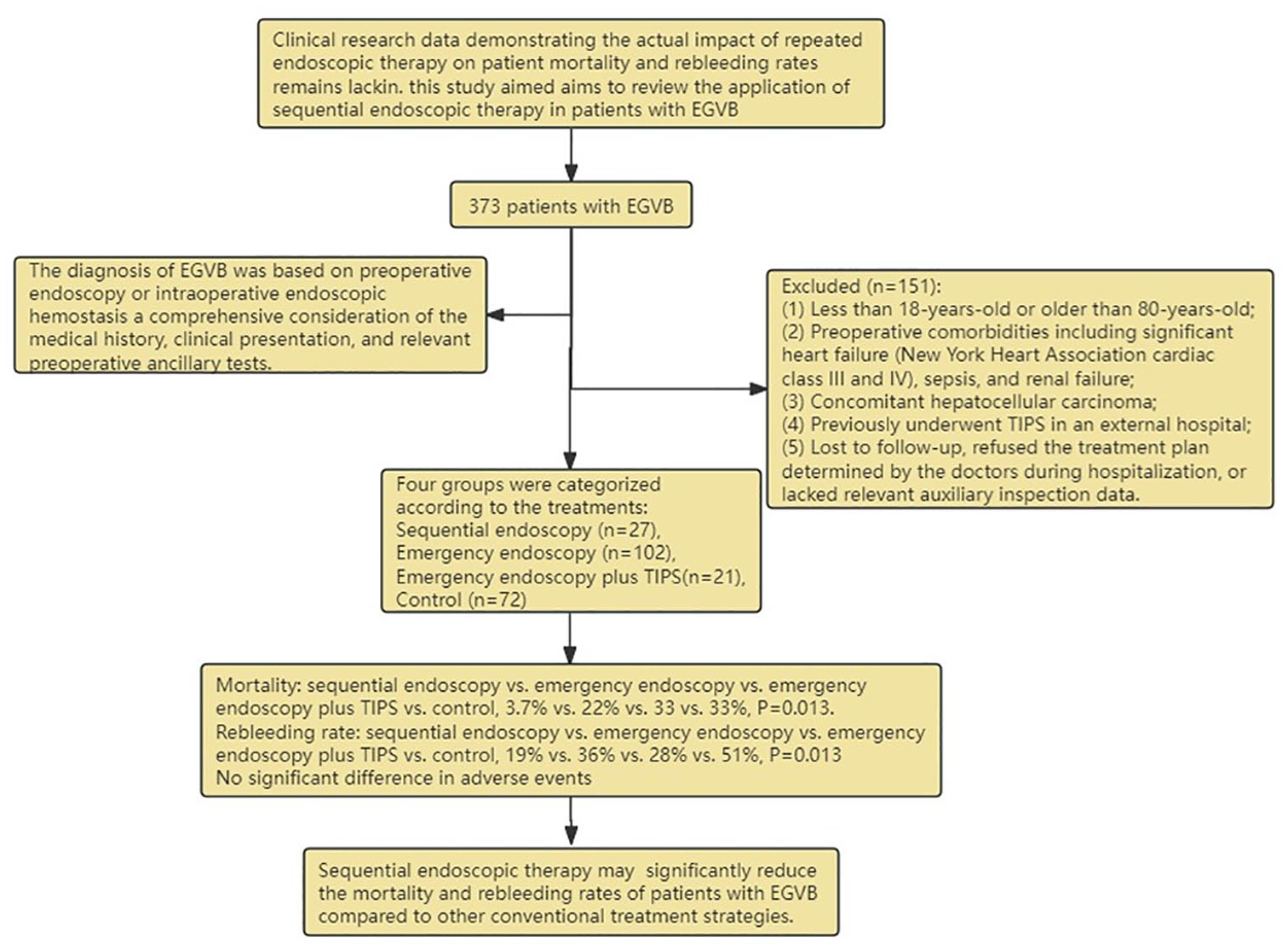
Detailed flowchart of the article.
Endoscopic treatment procedures
The treatment protocol for all patients upon admission included fluid resuscitation, drug therapy (including vasoactive drugs, antibiotics, hemostatic drugs, and others), and red blood cell transfusion. The non-control groups underwent additional endoscopic treatment or TIPS as recommended by international consensus guidelines. 20
Endoscopic treatment was performed by experienced endoscopic experts. The procedure included ligation, sclerotherapy, and tissue adhesive injection. Esophageal variceal bleeding was mainly treated using endoscopic variceal ligation (EVL). When ligation techniques were difficult or active variceal bleeding was observed during the endoscopic examination, sclerotherapy or a combination of sclerotherapy and EVL was considered. Gastric variceal bleeding was usually treated using a tissue adhesive (N-butyl cyanoacrylate). In addition, a combination of a sclerosing agent (lauromacrogol) and tissue glue could be injected into the active bleeding site. The size of the varicose vein determined the number of rubber rings required for ligation, as well as the required dosage of lauromacrogol and cyanoacrylate. Visual confirmation was obtained to ensure that active bleeding had stopped or that varicose veins had decreased to near-occlusion. Patients were usually advised to undergo endoscopic follow-ups at intervals of 1–3 months after the first endoscopic variceal treatment, with additional endoscopic varicose vein treatment performed if necessary. If endoscopic examination failed to control the bleeding, the patient would receive interventional treatment.
The TIPS procedure was performed by experienced interventional radiologists. Under local anesthesia, the right internal jugular vein was percutaneously punctured, and a Rups-100 sheath was introduced into the hepatic vein. Contrast imaging was employed to visualize the hepatic vein and its branches. Based on abdominal CT images of the portal vein trajectory, the Rups-100 puncture needle successfully accessed the main trunk of the left or right branch of the portal vein, with the guidewire advanced into the catheter to the distal end of the superior mesenteric vein or the splenic vein. The Rups-100 sheath was then advanced to the main trunk of the portal vein, with contrast imaging revealing dilated and tortuous esophageal or gastric varices. A 5F Cobra catheter and a 3F microcatheter were selectively inserted into the varices, followed by balloon dilation of the puncture tract using a balloon dilation catheter (Cook Medical, USA, 80 × 6 mm). Subsequently, a Viatorr-covered stent (Gore Medical, USA, 8 × 60 mm) was deployed. Fluoroscopy confirmed stent expansion, and a follow-up portal venography was performed. A Doppler ultrasound of the portal vein was recommended 1–2 months later to assess the condition of the portal vein stent.
Clinical assessment and follow-up
Before November 30, 2023, patients and their families were contacted by telephone to collect data on their rebleeding and survival status. A total of eight patients were lost to follow-up in the sequential endoscopy group, 29 in the emergency endoscopy group, zero in the emergency endoscopy plus TIPS group, and 36 in the control group (Figure 2). The main indicators observed in this study was the all-cause mortality rate, whereas the secondary indicators included the rebleeding, recent rebleeding, and long-term bleeding rates. In accordance with the Baveno V consensus, rebleeding was defined as recurrent black stools or hematemesis resulting in hospitalization, transfusion, a decrease of 3 g/L in hemoglobin level, or death within 6 weeks. 24 Recent rebleeding referred to rebleeding that occurred within 6 weeks after the initial control of bleeding, whereas long-term rebleeding referred to rebleeding that occurred more than 6 weeks after the initial control of bleeding. 25

Flowchart of patient inclusion and exclusion criteria.
Statistical analysis
The continuous variables were expressed as means ± standard deviations or medians with interquartile ranges (IQR) and were analyzed using analysis of variance or Mann–Whitney non-parametric tests. The categorical variables were expressed in numbers and percentages and analyzed using the Chi-squared test or Fisher’s exact test. A Kaplan–Meier analysis was performed to study the time to rebleeding and time to death, and a log-rank test was applied to compare differences between groups. Univariate and multivariate analyses were conducted using differential analysis and logistic regression analysis respectively to assess potential risk factors for all-cause mortality. A two-sided p-value < 0.05 was considered statistically significant. Statistical analyses were conducted using version 27 of the IBM SPSS Statistics software.
Results
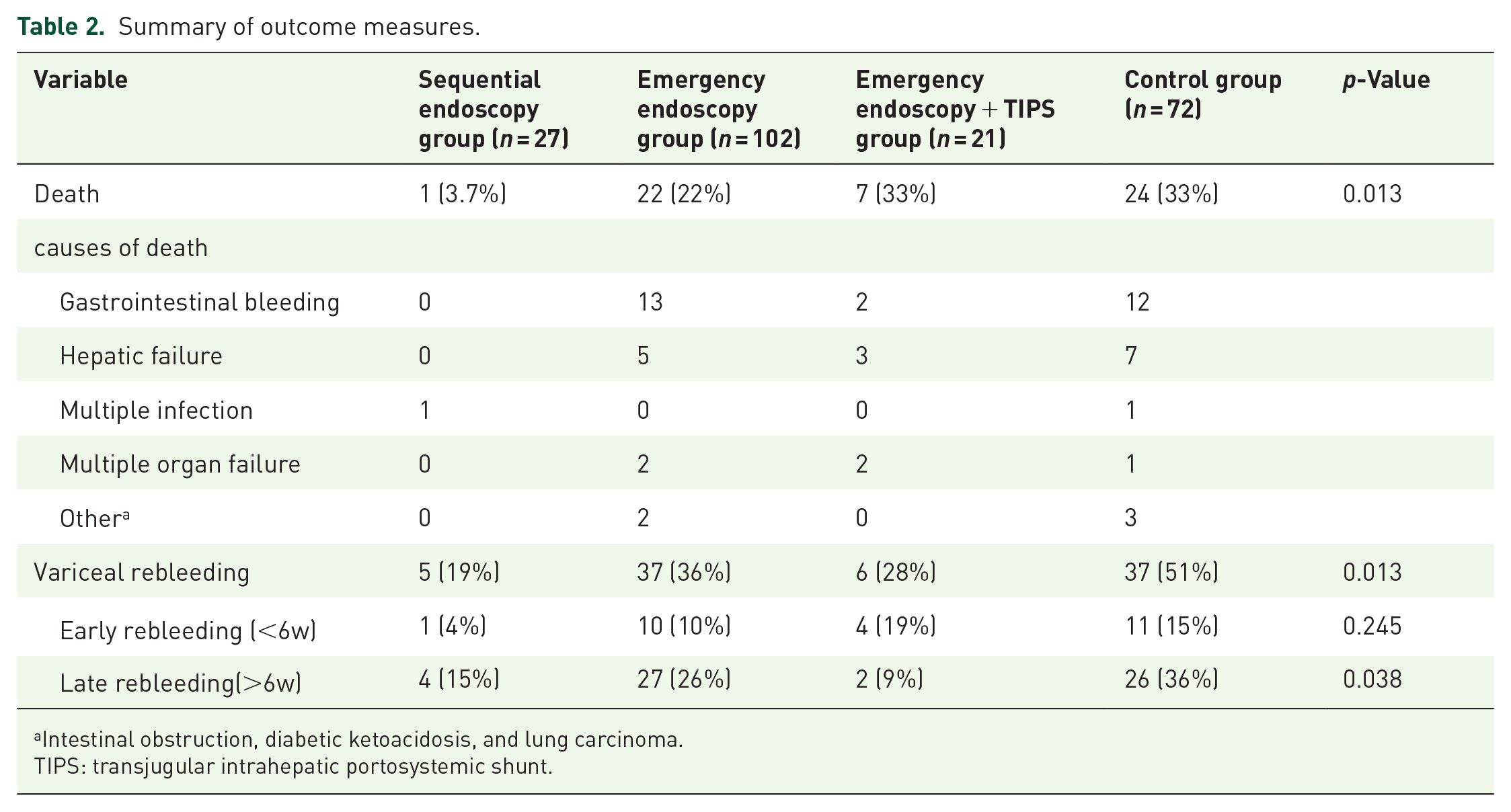
A total of 373 patients were included in this study. Figure 1 shows a detailed research flowchart. Ultimately, 27 cases were included in the sequential treatment group, 102 in the emergency endoscopy group, 21 in the emergency endoscopy plus TIPS group, and 72 in the control group. The median follow-up time was 11.4 months (IQR: 6.5–28) for the sequential treatment group, 15.7 months (IQR: 7.3–30.3) for the emergency endoscopy group, 14.7 months (IQR: 2.6–25.5) for the emergency endoscopy plus TIPS group, and 14.3 months (IQR: 3.2–28.1) for the control group. The demographic data recorded at admission included age, sex, cause of cirrhosis, ALB, PT, TBIL, HB, WBC, neutrophils, history of diabetes, history of HE, history of splenectomy, Child–Pugh classification, MELD score, location of varices, and initial endoscopic treatment. The baseline characteristics of the patients are shown in Table 1. The results and adverse events have been summarized in Tables 2 and 3, respectively. The process of sequential endoscopic treatment is shown in Figure 3.
Baseline characteristics of the patients.

Data are expressed as mean ± standard deviation or median (range); categorical data are presented as number or percentage of patients.
The Child–Pugh score ranges from 5 to 15 points, with Class A (5 to 6 points) indicating the mildest condition, Class B (7 to 9 points) indicating moderate severity, and Class C (10 to 15 points) indicating the most severe condition. Patients with a Child–Pugh score greater than 13 are not included in the study.
The MELD (Model for End-Stage Liver Disease) score ranges from 1 to 40, with higher scores indicating more severe illness.
EGV, esophageal and gastric varices; EIS, endoscopic injection sclerotherapy; EV, esophageal varices; EVL, endoscopic variceal ligation; GV, gastric varices; HB, hemoglobin; HBV, hepatitis B virus; HCV, hepatitis C virus; HE, hepatic encephalopathy; INR, international normalized ratio; PT, prothrombin time; RBC, red blood cell; TBIL, total bilirubin; TIPS: transjugular intrahepatic portosystemic shunt; WBC, white blood cell; Other: including EIS, tissue glue, EVL + EIS and EVL + tissue glue.
Summary of outcome measures.
Intestinal obstruction, diabetic ketoacidosis, and lung carcinoma.
TIPS: transjugular intrahepatic portosystemic shunt.
Adverse events.
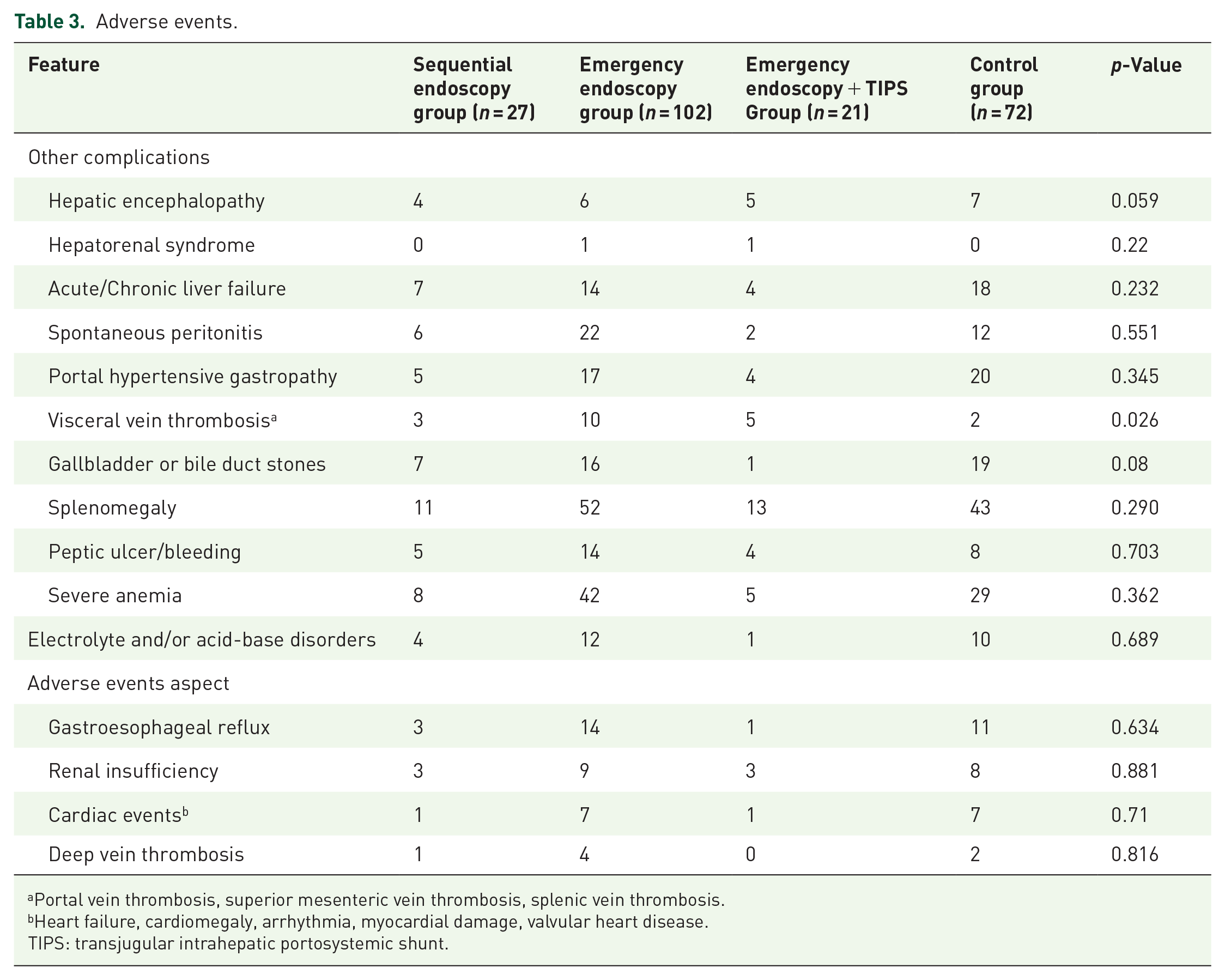
Portal vein thrombosis, superior mesenteric vein thrombosis, splenic vein thrombosis.
Heart failure, cardiomegaly, arrhythmia, myocardial damage, valvular heart disease.
TIPS: transjugular intrahepatic portosystemic shunt.

The process of sequential endoscopic treatment: (a) shows acute esophageal variceal hemorrhage. (b) An unclear fundus with large amounts of blood. (c) Typical endoscopic injection of a sclerosing agent. (d and e) Esophageal and gastric varices that did not bleed during the follow-up. (f) Endoscopic banding of varices. (g, h, i) Esophageal-gastric varices that disappeared at the last follow-up.
All-cause mortality
A statistically significant difference in the mortality rate was observed among the four groups. The mortality rate of the sequential endoscopy group was significantly lower than those of the other groups (sequential endoscopy vs emergency endoscopy vs emergency endoscopy plus TIPS vs control, 3.7% vs 22% vs 33% vs 33%, p = 0.013). The Kaplan–Meier analysis revealed a statistically significant difference in cumulative survival rates between the sequential endoscopy group and the other three groups, with the sequential endoscopy group exhibiting a higher rate than those of the other groups (Figure 4). In addition, univariate analysis revealed that the postoperative mortality was significantly associated with the postoperative Child–Pugh score and age. Furthermore, a multivariate analysis showed that the postoperative Child–Pugh score was an independent risk factor for increased mortality (odds ratio (OR): 2.59, 95% confidence interval (CI): 1.46–4.59). The specific causes of death for each group are shown in Table 2.

The cumulative survival rate of the sequential endoscopy group was higher than that of the other groups. (p < 0.05, log-rank test).
Rebleeding rate
All patients achieved hemostasis during hospitalization, and a statistically significant difference in rebleeding rates was observed among the four groups (sequential endoscopy vs emergency endoscopy vs emergency endoscopy plus TIPS vs control, 19% vs 36% vs 28% vs 51%, p = 0.013). A pairwise comparison showed that the rebleeding rate in the sequential endoscopy group was significantly lower than that in the control group (19% vs 51%, p = 0.003). Similarly, the Kaplan–Meier analysis showed that the cumulative rebleeding rate of the control group was significantly higher than that of the other groups (Figure 5). No significant difference in recent rebleeding rate was observed among the groups (sequential endoscopy vs emergency endoscopy vs emergency endoscopy plus TIPS vs control, 4% vs 10% vs 19% vs 15%, p = 0.245). In contrast, a statistically significant difference in the long-term rebleeding rates was observed among the four groups (sequential endoscopy vs emergency endoscopy vs emergency endoscopy plus TIPS vs control, 15% vs 26% vs 9% vs 36%, p = 0.038). A pairwise comparison showed that the rebleeding rate in the sequential endoscopy group was lower than that in the control group (15% vs 36%, p = 0.05).

The cumulative rebleeding rate of the control group was higher than those of the other groups. (p < 0.05, log-rank test).
Discussion
This retrospective study aimed to evaluate the impact of sequential endoscopic treatment for EGVB on patient mortality and rebleeding rates. Our findings highlight that sequential endoscopic therapy significantly reduces patient mortality and rebleeding rates compared with other conventional treatment strategies. This discovery may hold significant potential for guiding clinical practice and improving patient prognosis.
EGVB is a serious complication of liver cirrhosis. Despite the availability of various treatments, some patients still opted for conservative approaches. This preference was primarily a result of the underdeveloped economy in the region, as endoscopic treatments can incur high medical costs, particularly for those without insurance or adequate coverage. Previous studies have highlighted the effectiveness of endoscopic therapy in controlling bleeding and reducing mortality.10,11 However, our study further underscored the significance of sequential endoscopic therapy, which addresses both primary hemorrhagic lesions and potential causes of portal hypertension.
Endoscopic therapy can be implemented early after bleeding begins by quickly identifying the bleeding sites and employing various hemostatic techniques. These techniques may include endoscopic ligation, injection of tissue glue and sclerosing agents, or a combination of these methods to enhance the hemostatic effect. Timely hemostasis can prevent dangerous conditions such as massive blood loss and hypotensive shock. In addition, by decreasing the recurrence of acute bleeding events, the risk of additional bleeding and associated complications—such as infection, organ failure, and hepatic encephalopathy—can be reduced, thereby lowering mortality. However, because of persistent portal hypertension in liver cirrhosis, the risk of recurrent varices remains very high, necessitating further treatment.18,26 The standard of care for secondary prophylaxis of variceal bleeding involves a combination of nonselective beta-blockers and repeated EVL.19,20 The advantage of repeated endoscopic treatment is that, in addition to providing emergency hemostasis, it allows for the assessment of variceal severity through multiple follow-up endoscopic visits. For large and high-risk EGV, preventive treatment strategies can also be adopted, and individual treatment strategies can be formulated according to the specific conditions and characteristics of each patient. For different types of EGV, the most appropriate treatment methods can be selected to improve the treatment effect and reduce the mortality rate. The European Society for Gastrointestinal Endoscopy (ESGE) guidelines and the Baveno VII workshop recommended EVL for acute esophageal variceal bleeding, and endoscopic sclerotherapy injection for acute gastric variceal bleeding.20,22 However, some studies indicated that the combination of EVL with other treatments showed significant advantages in rapidly eradicating bleeding lesions, reducing the number of required treatments and lowering mortality and rebleeding rates. This made it an effective combined treatment strategy for gastrointestinal bleeding.27 –30
In this study, the mortality rate of the sequential endoscopy group was significantly lower than those of the remaining three groups. The highest mortality was observed in the emergency endoscopy plus TIPS and control groups. One patient died in the sequential endoscopy group. A detailed examination showed that the death of the patient was not a result of improper treatment strategies. Our analysis revealed that the patient had mixed-type allergic purpura, affecting the skin, joints, kidneys, and gastrointestinal tract. Gastrointestinal allergic purpura often results in gastrointestinal bleeding. In addition, the patient also had EGV and spontaneous peritonitis, which resulted in sepsis and ultimately led to death. The mortality rate in the emergency endoscopy group was relatively high, primarily owing to gastrointestinal rebleeding, followed by liver failure. As mentioned earlier, timely hemostasis helped reduce mortality, and preventing variceal rebleeding improved survival rates. 31 A similar RCT showed that sequential endoscopic variceal band ligation plus endoscopic variceal sclerotherapy exhibited fewer complications and lower recurrent bleeding rates than endoscopic variceal sclerotherapy alone (3% vs 20% and 3% vs 16%, respectively). 16 The above sequential endoscopic treatment method involved initially performing EVL, followed by repeated sclerotherapy every 1–2 weeks until the varices were eradicated. In this study, the sequential endoscopic treatment approach involved performing initial endoscopic hemostasis after acute EGVB, followed by endoscopic examinations every 1–3 months. In the absence of bleeding, treatment using band ligation, tissue glue, or sclerotherapy injections was performed to reduce variceal severity until the varices were eradicated. However, the time interval for the endoscopic follow-up was not unified. Some guidelines recommend repeating endoscopic treatment at intervals of 1–4 weeks or 4–8 weeks after controlling acute variceal bleeding until the varices are eradicated.20,23 In contrast, other studies have shown that performing EVL every 2 months is more effective than treatment every 2 weeks for esophageal varices, yielding a higher overall eradication rate, lower recurrence rate, and a reduced need for additional treatments. 26 Studies have shown that the interval for endoscopic treatments can be determined based on the judgment of the doctor and local logistical resources. 30 Because our hospital is situated in the economically less developed southwestern region of China, the intervals between endoscopic treatments were longer. Previous studies did not specify whether endoscopic treatment was administered without active bleeding to achieve variceal eradication during repeated endoscopic procedures.20,23,32 In contrast, our research was the first to clearly show that sequential endoscopic therapy started with endoscopic hemostasis, followed by additional endoscopic interventions in the absence of active bleeding, to effectively achieve variceal eradication.
The main indications for TIPS are acute variceal bleeding and refractory ascites.23,33,34 TIPS can rapidly decrease portal vein pressure, promptly halting bleeding and lowering the risk of severe blood loss. Multiple meta-analyses have shown that, compared with endoscopic therapy, TIPS can reduce the incidence of variceal rebleeding; however, it does not improve survival, and it is associated with a higher incidence of HE.35 –38 Similarly, an RCT showed that compared with EVL plus propranolol, TIPS effectively reduced the risk of variceal rebleeding in advanced liver cirrhosis but had no significant impact on mortality. 39 These findings were consistent with the results of this study, wherein the mortality rate was higher in the emergency endoscopy plus TIPS group than in the sequential endoscopy and emergency endoscopy groups. In addition, a multivariate analysis showed that the postoperative Child–Pugh score was an independent risk factor for increased postoperative mortality (OR: 2.59, 95% CI: 1.46–4.59), in accordance with previous research findings. 40
In this study, the rebleeding rate of the sequential endoscopic group was lower than that of the other three groups. The rebleeding rate was higher in the emergency endoscopy and control groups. Sequential endoscopic therapy employs various effective hemostatic measures, including endoscopic ligation, sclerosing injections, and tissue glue injections. These measures directly act on the bleeding site of EGV, effectively stopping bleeding and reducing the incidence of rebleeding. In addition to direct hemostasis, sequential endoscopic therapy can adopt preventive treatment strategies to reduce the risk of rebleeding. For example, for large and high-risk EGV, preventive endoscopic ligation or injection of sclerosing agents can be performed to reduce the risk of rebleeding. Through regular follow-up and re-examination of patients, risk factors for rebleeding—such as the re-dilation of varicose veins or the emergence of new bleeding points—can be identified and promptly addressed, allowing for timely interventions to prevent rebleeding and reduce its incidence. Therefore, sequential endoscopic therapy may reduce the rate of rebleeding in patients with esophageal and gastric variceal bleeding. The rebleeding rate was higher in the emergency endoscopy group, primarily because patients did not receive regular follow-ups. Notably, for patients with significant and high-risk EGV, the lack of timely intervention resulted in repeated episodes of acute EGVB, necessitating repeated endoscopic treatments each time, which in turn contributed to an elevated rebleeding rate. However, the rebleeding rate in the emergency endoscopy plus TIPS group was lower than that in the emergency endoscopy group. TIPS can treat acute EGVB and reduce portal pressure, thereby achieving the goal of preventing rebleeding. In concordance with the findings of Holster et al., among patients with cirrhosis who underwent successful endoscopic hemostasis for variceal bleeding, covered TIPS was more effective than EVL combined with beta-blockers in reducing variceal rebleeding; however, it did not improve survival rates. 13 Consistent with the results of previous studies, TIPS reduced the variceal rebleeding rate compared with endoscopic therapy.35 –39
Currently, no studies have compared the effects of sequential endoscopic treatment with those of other treatment strategies on mortality and rebleeding rates in EGVB. We found that sequential endoscopic treatment may significantly reduce mortality in patients with EGVB and also show some effectiveness in lowering the rebleeding rate. However, despite the promising clinical results of sequential endoscopic therapy observed in this study, some limitations must be noted. First, this was a retrospective study with a temporal recall bias. Second, the baseline patient age varied significantly among the four groups, with the emergency endoscopy plus TIPS group having a lower mean age than that of the remaining groups. Owing to the young ages of the patients in this group, better physical functioning and a relatively lower mortality rate are expected. However, the high mortality rate (33%) for our primary outcome aligns with existing literature stating that TIPS does not reduce mortality in cases of variceal hemorrhage.13,35 –39 In addition, sequential endoscopic treatment was defined as the patient receiving endoscopic hemostatic therapy after the first episode of acute EGVB, followed by endoscopic examinations every 1–3 months. Endoscopic treatment was performed in the absence of bleeding until the varices were eradicated. However, cases of variceal bleeding recurrence within 1 month after successful endoscopic hemostasis, as well as cases identified as not requiring treatment upon follow-up, were not included in the sequential endoscopic treatment group, resulting in selection bias. Finally, the limited number of cases and potential follow-up bias reduce the objectivity and reliability of the conclusions. Further research with larger sample sizes involving multiple centers is needed to validate the clinical efficacy of sequential endoscopic treatment.
In summary, sequential endoscopic therapy has significant advantages in treating EGVB and may effectively reduce mortality and rebleeding rates. This technique is worth considering for broader clinical applications. The findings of this study suggest the need for increased research investment in this field. Collaborative efforts to promote the development of sequential endoscopic therapy could enhance treatment outcomes improve the and quality of life for patients in the future.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to all those who contributed to the research and writing of this article, and all authors for their dedication to the project. Furthermore, we would like to acknowledge the invaluable support provided by the Project of the Bureau of Science & Technology Nanchong City and Sichuan Province Science and Technology Department Key Research and Development Project throughout the duration of this study. We are also grateful to the participants of this study, whose cooperation and involvement were essential to its success. Their commitment to advancing medical knowledge is truly commendable.
