Abstract
Background:
Small intestinal-predominant Crohn’s disease (CD) exhibits distinct therapeutic patterns and is characterized by increased biological use and intestinal resection rates. However, systematic evaluations of the objective response of ustekinumab (UST), particularly related to disease location, are limited.
Objectives:
To compare the therapeutic efficacy of UST between small intestinal CD (SICD) and non-small intestinal CD (NSICD).
Design:
A prospective observational cohort study.
Methods:
Demographic and clinical characteristics were collected at baseline and 52-week follow-up, assessments including endoscopy, imaging, ultrasonography (US), histopathology, fecal calprotectin (FCP), C-reactive protein (CRP), and hemoglobin results. Patients with isolated ileal involvement (Montreal classification L1) were classified as having SICD, while NSICD was defined by L2 and L3. Primary outcome was objective remission, defined by meeting one of the following: (1) endoscopic remission (ER): Simple endoscopic score for CD (SES-CD) ⩽ 2. (2) US remission: normal bowel wall thickness and absence of Doppler signals or detectable complications, or (3) radiological remission: complete resolution of inflammatory features. Secondary outcomes included (1) mucosal healing (MH): absence of ulcerations. (2) transmural healing (TH): complete healing of all intestinal wall layers, and (3) biochemical remission: FCP/CRP normalization, (4) objective response: a clear improvement from baseline, and (5) endoscopic response: SES-CD ⩽ 4.
Results:
This study included 331 patients with CD (SICD 36.9%, NSICD 63.1%). Of these, 309 completed a 52-week follow-up. Objective remission, MH, and TH were achieved by 32.7%, 25.6%, and 13.6% of patients, respectively. Biochemical remission occurred in 41.5% (FCP) and 35.2% (CRP). Objective and endoscopic responses were observed in 70.2% and 42.1% of patients, respectively. Objective remission was achieved in 54/116 patients (46.6%) in the SICD group and 47/193 (24.4%) in the NSICD group. Regression analyses and propensity score matching showed significantly higher rates of objective remission, MH, and endoscopic response in the SICD group.
Conclusion:
Disease location influenced UST treatment outcomes, with SICD showing higher rates of objective remission, MH, and endoscopic response than NSICD.
Plain language summary
Crohn’s disease (CD) is a chronic condition that causes inflammation in the digestive tract. It can affect different parts of the gut, and where the disease occurs may influence how well treatments work. In particular, patients with CD affecting only the small intestine (called “small intestinal CD” or SICD) may respond differently to medication compared to those with inflammation in other areas, such as the colon.
This study looked at how effective a medication called ustekinumab (UST) is for treating CD in different parts of the gut. We followed 331 patients with CD for one year after they started UST treatment. About one-third of the patients had disease limited to the small intestine (SICD), while the others had disease involving the colon or both the small intestine and colon (NSICD).
We used several medical tests-including endoscopy, imaging, lab tests, and stool markers-to measure how well patients responded to treatment. We found that UST helped many patients, but those with SICD had better outcomes than those with NSICD. Specifically, patients with SICD were more likely to experience healing of the gut lining (mucosal healing), improvement seen on endoscopy, and overall objective signs of remission.
These results suggest that UST may be particularly effective for patients with CD limited to the small intestine. Understanding how disease location affects treatment response could help doctors make more personalized treatment decisions.
Introduction
Stratifying Crohn’s disease (CD) by location provides valuable insights into its pathophysiology and therapeutic responsiveness, thereby facilitating more precise and individualized management strategies. Emerging evidence suggests that ileal-predominant CD is associated with a unique therapeutic profile, including higher reliance on biological agents and reduced treatment persistence. 1 Patients with isolated ileal involvement (Montreal classification L1) are classified as having small intestinal CD (SICD), a phenotype with distinct clinical and pathological features compared to non-small intestinal CD (NSICD), which encompasses colonic (L2) and ileocolonic (L3) involvement. Notably, SICD confers a seven-fold increased risk of disease progression and a nine-fold increased risk of intestinal complications compared with colonic CD. 2 Patients with SICD also exhibit a significantly higher likelihood of transitioning to stricturing or penetrating disease phenotype over a median follow-up of 10.5 years, with significantly greater cumulative risk of requiring surgery compared with patients with colonic CD. This underscores the need for early and effective therapeutic interventions.1,2 This aggressive course may be attributed to the ileum’s distinct anatomical, physiological, and immunological characteristics. The ileum exhibits unique pathological alterations, including reduced transmembrane mucin levels, decreased microbiota diversity, lower antimicrobial peptide levels, and compromised epithelial integrity. Immunologically, the ileal lamina propria is predominantly infiltrated by Th1 and Th17 cells, whereas the colonic mucosa is mainly Th1-driven. 3 These differences emphasize the need for targeted therapies. However, inconsistencies remain regarding the influence of disease location on the efficacy of biologic therapies.4–8
Ustekinumab (UST) has been widely used over the past decade and demonstrated efficacy in the clinical remission of patients with CD. Given the specific immunological and structural characteristics of the small intestine, therapeutic responses to UST may vary considerably between SICD and NSICD. However, real-world data on the effectiveness of UST across different intestinal segments are limited and controversial.9–11 Furthermore, most studies primarily relied on patient-reported outcomes, which may not accurately reflect underlying disease activity. Currently, treatment goals in CD extend beyond symptomatic relief to encompass deeper and more objective endpoints, including endoscopic remission (ER), mucosal healing (MH), and transmural healing (TH), to alter disease progression more effectively.12,13 Noninvasive methods such as intestinal ultrasonography (US) and magnetic resonance enteroclysis (MRE) have shown value in monitoring these treatment targets. 14 Despite the growing recognition of these objective endpoints, real-world systematic evaluations of UST efficacy in terms of objective remission, particularly with respect to disease location, are scarce. Specifically, the comparative effectiveness of UST in SICD and NSICD remains poorly defined. 15 Therefore, the aim of this prospective study was to evaluate the effectiveness of UST across different intestinal segments in CD, with a particular emphasis on comparing outcomes between patients with SICD and NSICD.
Methods
Study design and data collection
This prospective cohort study of patients with CD who underwent UST was initiated in October 2023 at the Second Affiliated Hospital, Zhejiang University School of Medicine. Enrollment, treatment, and follow-up were performed according to prespecified protocols. Patients were selected consecutively, and eligibility was determined by meeting the following criteria: (1) adults aged 18–80 years; (2) a confirmed diagnosis of CD established based on clinical, endoscopic, and/or histological findings; (3) presence of active disease or inadequate response to, or intolerance of, other medical therapies; and (4) willingness to receive long-term treatment with UST. Exclusion criteria were as follows: (1) known hypersensitivity or intolerance to UST; (2) pregnancy or breastfeeding; (3) prior exposure to UST for other indications; (4) concomitant malignancy requiring tumor-related treatments; (5) presence of acute infectious diseases; and (6) presence of severe organic diseases, unstable vital signs, rapidly progressive conditions, or end-stage disease.
Patients with isolated ileal involvement were classified as having SICD, while those with isolated colonic or ileocolonic involvement were categorized as having NSICD. Demographic and clinical characteristics at baseline, including disease location, disease behavior (according to the Montreal classification), the Harvey-Bradshaw index (HBI), history of surgery, prior medication exposure and modifications, and biomarkers (fecal calprotectin [FCP], C-reactive protein [CRP], and hemoglobin) were systematically collected. Endoscopic, imaging, ultrasonic, and pathological findings were also documented. The simple endoscopic score for CD (SES-CD) was used to evaluate endoscopic results. All patients were required to undergo endoscopy at baseline, and significant disease activity was defined as SES-CD > 6, consistent with an obviously thickened intestinal wall accompanied by enhancement in MRE/CT or stage III–IV blood supply by Doppler signals at baseline.
UST prescriptions (date of initiation, dosing, optimization, concomitant treatments, cessation of treatment, and reasons for cessation) were also specified. The entire cohort was treated uniformly during induction, with a baseline intravenous infusion based on weight ranges: <55 kg (260 mg), 55–85 kg (390 mg), and >85 kg (520 mg). During maintenance therapy, UST 90 mg was administered subcutaneously every 8 weeks. Treatment intensification, with a new IV infusion and/or interval shortening (injections every 4 or 6 weeks), could be performed from week 16 onwards in patients with an initial response and subsequent loss of clinical response at the discretion of the treating physician.
At follow-up (52 ± 10 weeks, with the exact time point determined by each patient’s condition), information including symptoms, surgeries, treatment regimen/alterations, and reexaminations was collected. Levels of inflammatory biomarkers were also evaluated. Data on adverse events through week 52 were recorded. Patients with missing data due to adverse events, bowel surgery, or loss to follow-up were excluded. All analyses were conducted using available data without imputation. Sample size was calculated based on a two-sided significance level (α) of 0.05 and a statistical power of 80%. According to published literature, the proportion of patients in the SICD group was estimated at 44%, with expected remission rates of P1 = 0.18 and P2 = 0.33 for the comparison groups.1,16 Using these assumptions, the required sample size was determined to be 296. With an anticipated 5% dropout rate, the final target enrollment was set at 311 participants. Sample size estimation was performed using Sealed Envelope (https://www.sealedenvelope.com/). The study is reported in accordance with the Strengthening and Reporting of Observational Studies in Epidemiology (STROBE) guidelines for cohort studies. 17
Definitions of outcomes
The primary outcome was objective remission, a composite endpoint defined by meeting one of the following three criteria at week 52: (1) ER: SES-CD ⩽ 2 according to STRIDE-II guideline. 18 (2) US remission: normal bowel wall thickness (⩽3 mm for the small bowel, ⩽4 mm for the colon) and the absence of Doppler signal or detectable complications, or (3) radiological remission: complete resolution of inflammatory features on MRE or CT.
Secondary outcomes included (1) MH: absence of ulcerations on endoscopy; (2) TH: complete healing of all intestinal wall layers (mucosa, submucosa, muscularis, and serosa), confirmed by a combination of endoscopic, ultrasonic, and radiological assessments; (3) biochemical remission: FCP/CRP normalization in patients with abnormal baseline values; (4) objective response: a clear improvement from baseline on any screening modality, with assessment based on physician judgment and the following criteria: a ⩾50% reduction in SES-CD; substantial improvement from baseline in bowel wall thickness, enhancement, ulceration, or inflammatory signals on MRE/CT; a ⩾25% reduction in bowel wall thickness compared with baseline; or a marked decrease in vascular signals as detected by US; and (5) endoscopic response: SES-CD ⩽ 4.
As individual patients might have undergone multiple examinations, a predefined hierarchical approach was applied: endoscopy was given the highest priority, with results considered definitive if available. In the absence of endoscopy, MRE or CT findings were used as the second level of evidence (MRE was considered superior to CT; moreover, patients did not undergo both MRE and CT simultaneously as a re-examination method to avoid unnecessary use of medical resources and costs), followed by US as the third. In cases where MH was achieved at follow-up, complementary assessment with MRE, CT, or US is required to ascertain whether TH has been achieved. UST discontinuation was defined as the absence of improvement in symptoms or examinations after 16 weeks of treatment, resulting in patients opting for therapy change. Safety outcomes included the incidence of drug-related adverse events and disease-related hospitalizations.
Statistical analysis
Descriptive statistics were used to analyze baseline characteristics. Quantitative variables were presented as mean (standard deviation (SD)) or median (interquartile range), depending on data distribution. Analyses were conducted using available data without imputation of missing data. Group comparisons were conducted with the χ² test for categorical variables and the Kruskal–Wallis test for continuous variables. Univariate and multivariable logistic regression analyses were performed to identify predictors of UST treatment success, and the need for intensification (post-hoc exploratory analysis), expressed as odds ratios (ORs) with 95% confidence intervals (CIs). Multivariate model 1 was adjusted to include known prognostic factors and variables influencing ER, MH, TH, and dose increase (model 2).
Baseline variables with p < 0.10 were used for multivariable analysis. p < 0.05 was considered to be significant. In addition, 1:1 propensity score matching was employed to minimize confounding factors and assess the independent association between SICD and outcomes. The following variables were included in the propensity score model: disease behavior, sex, age category, smoking status, baseline FCP level, perianal disease, concomitant medications, prior intestinal surgery, presence of granulomas on histology, and baseline CRP level. Matching quality was assessed using standardized mean differences (SMD). A threshold of SMD < 0.2 for all variables was considered to indicate adequate balance after matching. Analyses were performed using SPSS version 27.0 (SPSS Inc., Chicago, IL, USA).
Results
Baseline characteristics
In total, 331 eligible patients with CD were enrolled, and a 52-week follow-up was initiated (Figure 1). Baseline demographic and clinical characteristics are summarized in Table 1. The cohort comprised 225 males (68.0%), with a mean age of 36.5 ± 13.4 years and a mean disease duration of 5.6 ± 5.5 years. Four patients had concomitant psoriasis. Intestinal stenosis was present in 140 patients (42.3%), and 52 (15.7%) had a penetrating disease phenotype at the most severe stage of the disease. A total of 158 patients (47.7%) had been previously treated with immunosuppressants (IMS), while 151 (45.6%) had prior exposure to other biologicals. The proportions of patients who had undergone prior bowel and perianal surgeries were 105 (31.7%) and 92 (27.8%), respectively. At the time of UST induction, 71 patients (21.5%) were receiving concomitant medications, including corticosteroids (oral controlled-release beclomethasone or budesonide) in 16 patients and other IMS agents (azathioprine, methotrexate, or thalidomide) in 55 patients.
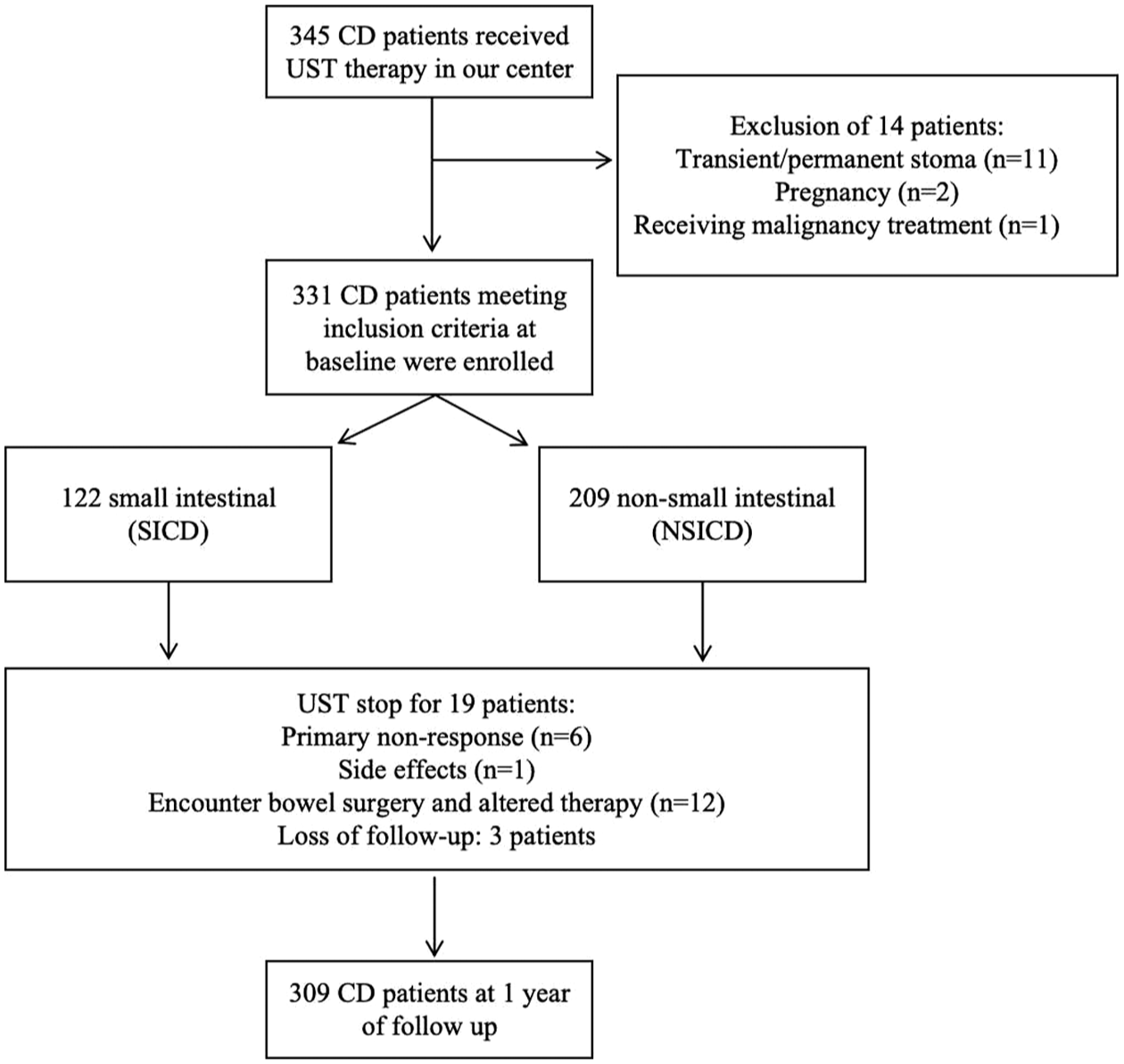
Flowchart of the overall population in the study.
Clinical and demographic characteristics of the CD patients treated with UST at baseline.

Compare between the SICD group and NSICD group.
CD, Crohn’s disease; IMS, immunosuppressants; EEN, exclusive enteral nutrition; NSICD, non-small intestinal Crohn’s disease; SICD, small intestinal Crohn’s disease; UST, ustekinumab.
The bold font indicates statistical significance.
Regarding disease location, 122 patients (36.9%) had SICD, while 209 patients (63.2%) had NSICD (31 with isolated colonic involvement and 178 with ileocolonic disease). At baseline, the SICD group exhibited higher rates of strictures and prior bowel surgery. In contrast, the NSICD group had higher proportions of patients with perianal disease and a surgery history, concomitant medication use, and granulomas on histopathology. No significant differences were observed between the groups in baseline disease activity, HBI, CRP, or FCP levels.
Follow-up and UST drug survival
After the 52-week follow-up, 309/331 (93.4%) patients were available for objective outcomes assessment following continuous UST treatment, of whom 116 (37.5%) had SICD and 193 (62.4%) had NSICD. A total of 19/331 (5.7%) patients discontinued UST at a mean time of 28.6 weeks, and 3/331 (0.9%) patients were lost to follow-up. The reasons for treatment cessation were lack of response in 6/19 (31.6%) patients, inevitable bowel surgery followed by therapy alteration in 12/19 (63.2%), and 1/19 (5.3%) patient experienced a treatment-related adverse event that required drug discontinuation (Figure 1). Among patients who underwent surgery, four were in the SICD group (one small bowel resection with anastomosis and three small bowel strictures), and eight were in the NSICD group (five ileocolic resections, one small bowel strictureplasty, one incision and dilatation of rectal stricture, and one incision and drainage of perianal abscess). There were no discontinuations owing to stable remission.
Objective remission and response
A total of 309 patients were available for primary outcome assessment following the 52-week UST treatment, of whom 116 (37.5%) had SICD and 193 (62.4%) had NSICD. The proportions of patients evaluated by endoscopy, US, and imaging were 281 (90.9%), 116 (37.5%), and 152 (49.2%), respectively. Objective remission was achieved in 101 patients (32.7%) who received UST. In addition, MH and TH were observed in 79 (25.6%) and 42 (13.6%) patients, respectively. Objective response was achieved in 217 (70.2%) patients, and endoscopic response was observed in 130 patients (42.1%). Notably, the proportions of patients achieving objective remission, MH, and endoscopic response were significantly higher in the SICD group than in the NSICD group (Figure 2(a)).

Proportions of patients achieving different outcomes (if available) at 52 weeks. (a) Comparison of objective remission, mucosal healing, transmural healing, objective response, and endoscopic remission between patients with SICD and NSICD. (b) Comparison of laboratory indicators between SICD and NSICD groups among patients with abnormal baseline levels. (c) Proportions of patients who underwent treatment alterations.
Biochemical remission
Regarding laboratory parameters, FCP normalization (<250 µg/g) was achieved in 97 patients (41.5%), and CRP normalization (<5 mg/L) in 44 (35.2%) patients among those with abnormal baseline FCP/CRP levels. Anemia resolution was observed in 53 patients (50.5%) at week 52. Importantly, patients in the SICD group demonstrated higher rates of FCP normalization and anemia resolution than those in the NSICD group (Figure 2(b)), whereas CRP levels exhibited an opposite trend, although no significance was found.
Safety profile
Adverse events leading to therapeutic discontinuation occurred in one (0.3%) patient who reported chest tightness. Two additional patients developed transient fever and mild liver dysfunction following UST dose intensification. These symptoms resolved spontaneously with supportive care. After thorough evaluation and shared decision-making, UST treatment was continued in both cases, and no recurrence of adverse events was observed during the remainder of the follow-up period.
UST drug strengthens
UST dose was intensified in 161 patients (48.6%) during treatment. Specifically, 53 patients (43.4%) were in the SICD group and 108 patients (51.7%) in the NSICD group (Figure 2(c)). Factors associated with UST dose intensification were analyzed and are presented in Supplemental Table 1. Stricturing and penetrating disease phenotypes, elevated baseline CRP levels, and the presence of pathological granulomas were identified as risk factors for UST dose intensification. Intriguingly, a baseline HBI ⩾ 4 was inversely associated with the need for treatment escalation.
Factors associated with objective remission, MH, TH, and endoscopic response
Factors associated with objective remission at 52 weeks are summarized in Table 2. In model 1, SICD was positively associated with objective remission, while stricturing and penetrating disease phenotypes (B2 and B3) showed negative associations. After adjusting for variables in model 2, baseline FCP > 1000 µg/g was associated with objective remission. Age, sex, smoking, perianal disease, body mass index, previous and concomitant treatment, disease activity, HBI, CRP levels at baseline, surgical history, and presence of pathological granuloma were not found to predict objective remission.
Univariate and multivariate logistic regression analysis of predictive factors associated with objective remission with UST at week 52 (n = 309).

Multivariate model 1 was adjusted to model 2 to include known prognostic factors (perianal disease, prior biologic, surgical history, as well as baseline FCP and CRP).
CRP, C-reactive protein; FCP, fecal calprotectin; HBI, Harvey-Bradshaw index; IBD, inflammatory bowel disease; IMS, immunosuppressants; SICD, small intestinal Crohn’s disease; UST, ustekinumab.
The bold font indicates statistical significance.
Similarly, SICD, stricturing and penetrating disease phenotypes, along with baseline FCP levels, were found to predict MH, TH, and endoscopic response (Tables 3–5). Male sex, presence of pathological granuloma, and nonsmoker were associated with a higher likelihood of achieving endoscopic response.
Univariate and multivariate logistic regression analysis of predictive factors associated with mucosal healing with UST at week 52 (n = 309).

Multivariate model 1 was adjusted to model 2 to include known prognostic factors (smoking, perianal disease, and baseline CRP).
CRP, C-reactive protein; FCP, fecal calprotectin; HBI, Harvey-Bradshaw index; IBD, inflammatory bowel disease; IMS, immunosuppressants; SICD, small intestinal Crohn’s disease UST, ustekinumab.
The bold font indicates statistical significance.
Univariate and multivariate logistic regression analysis of predictive factors associated with transmural healing with UST at week 52 (n = 309).

Multivariate model 1 was adjusted to model 2 to include known prognostic factors (smoking, perianal disease, prior biologic, significant disease activity at baseline, surgical history, as well as baseline CRP).
CRP, C-reactive protein; FCP, fecal calprotectin; HBI, Harvey-Bradshaw index; IBD, inflammatory bowel disease; IMS, immunosuppressants; SICD, small intestinal Crohn’s disease; UST, ustekinumab.
The bold font indicates statistical significance.
Univariate and multivariate logistic regression analysis of predictive factors associated with endoscopic response with UST at week 52 (n = 309).

Multivariate model 1 was adjusted to model 2 to include known prognostic factors (perianal disease, prior biologic, significant disease activity at baseline, surgical history, and CRP).
CRP, C-reactive protein; FCP, fecal calprotectin; HBI, Harvey-Bradshaw index; IBD, inflammatory bowel disease; IMS, immunosuppressants; SICD, small intestinal Crohn’s disease; UST, ustekinumab.
The bold font indicates statistical significance.
To further clarify the impact of disease location on treatment outcomes, propensity score matching was used to evaluate the independent association between SICD and outcomes. The analysis revealed that patients with SICD had significantly higher rates of objective remission, MH, and endoscopic response compared with those with NSICD, regardless of other potential confounding factors (Table 6).
Propensity score matching and chi-square analysis of SICD and treatment outcomes at week 52 (N = 162).

CRP, C-reactive protein; FCP, fecal calprotectin; MH, mucosal healing; NSICD, non-small intestinal Crohn’s disease; SICD, small intestinal Crohn’s disease; TH, transmural healing.
The bold font indicates statistical significance.
Discussion
In the treatment of CD, the pursuit of objective and reliable measures of effectiveness has become increasingly important, with the aim of providing accurate prognostication and comprehensive feedback on strategies. Clarifying how disease location influences therapeutic response is essential for the optimization of treatment strategies in CD. Although disease location is believed to affect disease course and therapeutic response, current evidence on its prognostic value remains limited, indirect, and inconclusive. Some studies suggest that SICD is associated with a more aggressive or disabling disease course, 1 while others indicate that both ileal and colonic locations may predict disease severity.19,20 Our findings revealed an association between SICD and higher UST response rate, irrespective of behavior, offering hope to patients requiring surgical intervention. This aligns with research indicating that colonic and ileocolonic involvement are inversely associated with clinical remission after 1 year of UST treatment. 21
This study identified additional potential clinical predictors of comprehensive remission under UST treatment in a real-world population, with findings largely consistent with existing literature. Male patients were more likely to reach endoscopic response, supporting prior evidence that female patients have a higher risk of biologic discontinuation, potentially reflecting greater disease activity.22,23 Penetrating and stricturing disease behaviors clearly emerged as significant predictors of poorer objective remission and healing outcomes. While smoking has been implicated in worsening the clinical course of CD, 24 other studies suggest that it does not affect the biologic use, intestinal resection, or maintenance of treatment. 25 The present study showed a significant association between smoking and endoscopic response. However, a key limitation was the inability to accurately assess irregular smoking patterns over the follow-up, which may have affected the analysis of the temporal association between smoking and treatment outcomes.
Although biologic-naive patients have been reported to show better effectiveness outcomes, this study showed no association between biologic history and UST response. Notably, variations in dosage, treatment regimen (including intensification), and administration route across and within studies may hinder the comparison of UST efficacy. Our single-center study was conducted using a standardized treatment protocol, ensuring consistency. The safety profile of UST is well established and has already largely been studied in psoriasis, with long-term evidence from the PSOLAR registry providing reassurance. 26 Consistent with findings from several real-world CD cohorts,27–29 no safety concerns have been reported.
A major advantage of this study is its comprehensive assessment of disease activity, providing powerful data to explore how location impacts UST efficacy while accounting for potential confounders. Clinical manifestations of SICD are often ambiguous and indolent, thereby hindering effective monitoring. 30 Conventional symptom scoring, such as HBI, may underestimate the manifestations and severity of ileal disease, which could result in under-treatment. 31 To overcome these constraints, an innovative multidimensional assessment framework was used to ensure accurate evaluation of treatment response in this study.
This study has several limitations. First, the subjectivity of individual providers performing endoscopic, ultrasonic, or radiologic reassessments, as well as variability in re-examination timing (52 ± 10 weeks), may have introduced bias into the outcome data. Second, some patients underwent different assessment methods at baseline and follow-up, potentially introducing variations in evaluation standards. Third, the classification of ileocolonic CD remains debated. While some studies suggest that ileocolonic CD resembles isolated ileal CD more closely than colonic CD, others indicate that isolated ileal and colon-involving CD maintain distinct microbiotas, suggesting different underlying mechanisms. Given the heterogeneity of evidence and regional particularity, we classified ileocolonic CD within the colonic group to ensure adequate sample size for statistical comparison and to adhere to our original protocol. Future studies with larger cohorts are required to better clarify the optimal grouping of ileocolonic disease.
Conclusion
Disease location had a significant impact on UST effectiveness, with patients with SICD showing higher rates of objective remission, MH, and endoscopic response than NSICD, independent of other common clinical predictors. Patients with SICD may be more likely to derive benefit from UST compared to those with NSICD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251403009 – Supplemental material for Effectiveness of ustekinumab in Crohn’s disease across intestinal locations: spotlight on isolated small intestinal involvement
Supplemental material, sj-docx-1-tag-10.1177_17562848251403009 for Effectiveness of ustekinumab in Crohn’s disease across intestinal locations: spotlight on isolated small intestinal involvement by Yue Yu, Wen Hu, Shuyan Li, Hanwen Chen and Yan Chen in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251403009 – Supplemental material for Effectiveness of ustekinumab in Crohn’s disease across intestinal locations: spotlight on isolated small intestinal involvement
Supplemental material, sj-docx-2-tag-10.1177_17562848251403009 for Effectiveness of ustekinumab in Crohn’s disease across intestinal locations: spotlight on isolated small intestinal involvement by Yue Yu, Wen Hu, Shuyan Li, Hanwen Chen and Yan Chen in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to all the patients and clinical staff involved in this study. Special thanks to Dr. Niveditha Devasenapathy and Dr. Thomas Bradbury from The George Institute for Global Health for their valuable advice on study design and data interpretation. We thank Dr Zexin Chen, from the Second Affiliated Hospital, who assisted in the statistical procedure.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
