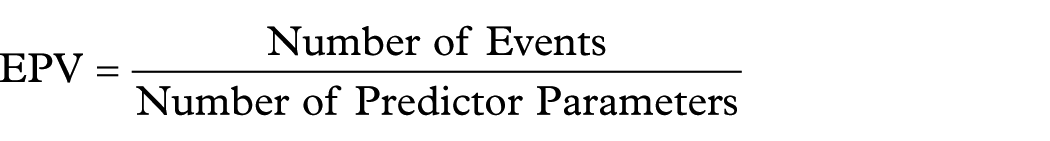Abstract
Background:
Gastric intraepithelial neoplasia (GIN) is a key precancerous lesion of gastric cancer, and discrepancies between preoperative endoscopic forceps biopsy (EFB) pathology and postoperative pathology after endoscopic submucosal dissection (ESD) pose significant challenges for accurate diagnosis and treatment planning.
Objective:
To analyze the risk factors for pathological upgrade in patients with GIN after an ESD procedure.
Design:
Retrospective study.
Methods:
A retrospective analysis was conducted on 682 patients diagnosed with GIN by gastric EFB pathology at the Digestive Endoscopy Center of Wuxi People’s Hospital from January 2018 to December 2024 (490 with low-grade intraepithelial neoplasia (LGIN) and 192 with high-grade intraepithelial neoplasia (HGIN)). Demographic characteristics (gender, age, BMI, chronic disease history, fecal occult blood), endoscopic features of lesions (location, morphology, size), and pathology data after the ESD procedure were collected. Univariate, multivariate, and stepwise logistic regression analyses were performed to identify independent risk factors for pathological upgradation.
Results:
The total pathological upgrade rate after ESD was 59.4% (51.2% in the LGIN group and 80.2% in the HGIN group). Stepwise regression analysis revealed that a maximum lesion diameter ⩾2 cm (odds ratio (OR) = 2.49) was the most significant risk factor for pathological upgrade. Meanwhile, age ⩾70 years, comorbid hypertension, Paris classification type IIc lesion, and a lesion location at the gastric angle or cardia were independent risk factors for pathological upgrade, whereas a type IIb lesion demonstrated a protective effect (OR = 0.61).
Conclusion:
Advanced age (⩾70 years), hypertension, lesion size ⩾2 cm, type IIc morphology, and lesions in the gastric angle/cardia significantly increase the risk of pathological upgrade after ESD. These findings provide a basis for preoperative risk assessment and individualized treatment strategies for GIN patients.
Plain language summary
A retrospective study were designed to analyze the risk factors for pathological upgrade in patients with gastric intraepithelial neoplasia (GIN) after endoscopic submucosal dissection (ESD). The study found that factors such as age, hypertension, lesion size, lesion location, and morphology were significantly associated with the risk of pathological upgrade. These findings emphasized the importance of preoperative risk assessment to guide individualized treatment strategies and improve patient outcomes.
Keywords
Background
Gastric cancer (GC) is a major global public health issue, ranking fifth in incidence and fourth in mortality among malignancies. 1 Although mortality rates have declined in recent years, prognosis varies significantly by stage: early gastric cancer (EGC) can be treated with endoscopic resection, with a 5-year survival rate exceeding 90%, while advanced GC has a survival rate below 30% even after surgical treatment. 2
According to the Correa cascade theory, GC develops progressively through “chronic gastritis → atrophy → intestinal metaplasia → low-grade intraepithelial neoplasia (LGIN) → high-grade intraepithelial neoplasia (HGIN) → adenocarcinoma.” 3 Gastric intraepithelial neoplasia (GIN) is a critical precancerous lesion. 4 While HGIN is widely recognized as requiring endoscopic resection, the management of LGIN remains controversial due to its heterogeneous malignant potential. 5 Endoscopic submucosal dissection (ESD) has become the preferred treatment for EGC and precancerous lesions due to its organ-preserving, minimally invasive, and high cure rate advantages.6,7 The decision to perform ESD in GIN patients depends on preoperative endoscopic forceps biopsy (EFB) results. The 2025 American College of Gastroenterology guidelines for the diagnosis and management of gastric precancerous lesions recommend repeat endoscopy at 12 months for low-risk LGIN patients, while HGIN patients, who likely already have active GC or will progress rapidly, should undergo repeat endoscopy and surgical evaluation within 3 months. 8 2025 ESGE/EHMSG/ESP recommends that patients with dysplasia (or indefinite for dysplasia) in whom no lesions are detected on gastroscopy should be referred for high-quality endoscopy. If no endoscopic lesions are identified again, follow-up high-quality endoscopy should then be performed, with intervals of 6 months for high-grade dysplasia and 12 months for low-grade dysplasia or indefinite for dysplasia. Patients with extensive endoscopic changes or advanced histological stages of atrophic gastritis are recommended to undergo high-quality endoscopic follow-up every 3 years. 9 The 2020 Chinese expert consensus on the management of gastric mucosal precancerous conditions and lesions suggests annual surveillance for LGIN with unclear borders on high-definition chromoendoscopy and semiannual surveillance for LGIN with clear borders not treated endoscopically. For LGIN with clear borders, endoscopic treatment may be considered, while HGIN and EGC should primarily undergo ESD. 10
However, EFB has limitations such as sampling error, insufficient tissue representation, and interobserver variability, often underestimating lesion severity.11,12 This leads to significant discrepancies between EFB and postoperative ESD pathology. Research shows that 12.1%–63% of cases may experience pathological upgrade, where preoperatively diagnosed LGIN lesions are reclassified postoperatively as HGIN or invasive carcinoma (IC).13–15 Such an upgrade has critical clinical implications, as it is associated with higher risks of lymphovascular invasion, submucosal infiltration, non-curative resection rates, and the need for additional surgery or chemoradiotherapy.16,17 Therefore, reliable predictive models integrating endoscopic and pathological features are urgently needed to identify high-risk GIN patients, particularly those with LGIN.
This study retrospectively analyzed 682 GIN patients who underwent an ESD procedure, using multivariate logistic regression to systematically evaluate demographic and endoscopic features, aiming to provide evidence-based support for preoperative risk assessment and individualized treatment strategies.
Materials and methods
Inclusion and exclusion criteria
This study was designed as a retrospective cohort study, consecutively enrolling 682 patients who were pathologically diagnosed with GIN by EFB and subsequently underwent ESD at the Endoscopy Center of Wuxi People’s Hospital between January 2018 and December 2024.
Inclusion criteria were as follows:
(1) As this study is a retrospective study, we obtained informed consent from participants through phone calls.
(2) A pathological diagnosis of LGIN or HGIN was confirmed by EFB within 3 months prior to ESD.
(3) Complete clinical, endoscopic, and postoperative pathological data were available.
Exclusion criteria were as follows:
(1) Those who are unwilling to participate in this study.
(2) Patients with a preoperative pathological diagnosis other than GIN.
(3) Previous history of gastric surgery, radiotherapy, or chemotherapy.
(4) Patients with immunodeficiency or concurrent malignancies in other organs.
(5) Patients with incomplete or missing data.
Data collection and grouping
The dataset was collected via the MedBit system developed by Unimed Scientific Inc (Wuxi, China).
Demographics (age, gender, tumor history, hypertension, diabetes), endoscopic features of lesions (location, morphology, size), and pathology data after the ESD procedure were collected. Lesion morphology was classified by the Paris classification 18 as protruding (0–I), superficial elevated (0–IIa), flat (0–IIb), superficial depressed (0–IIc), or excavated (0–III). Lesions were divided into <2 and ⩾2 cm groups. The study population was categorized into two groups based on pathological outcomes: (1) the non-upgrade group, comprising patients whose preoperative EFB results were either concordant with post-ESD pathology or demonstrated pathological downgrading and (2) the upgrade group, consisting of patients showing pathological progression to more advanced malignancies on post-ESD evaluation, specifically including progression from LGIN to HGIN or adenocarcinoma, as well as progression from HGIN to adenocarcinoma.
With regards to collinearity, we did an analysis to calculate variable inflation factors, and the values for each predictor are very close to 1, which means the predictor is independent (Supplemental Table 1).
Biopsy and ESD specimen processing
According to the Chinese Expert Consensus on Standardized Digestive Endoscopy Biopsy and Pathological Examination, for lesions <2 cm in diameter, generally 1–2 tissue specimens are obtained; for every additional 1 cm increase in lesion diameter, one additional biopsy specimen should be collected. In addition, biopsy sampling should avoid the central ulcerated area to prevent severe complications such as perforation or major bleeding.
After the ESD procedure, the specimen should be flattened and fixed on a dedicated foam board, with clear labeling of the oral and anal sides. It should then be placed in 10% neutral buffered formalin for 24–48 h before being sent for pathological examination. The pathological sectioning process generally involves the following steps: Pre-sampling documentation: Photograph the entire specimen to record its size, color, texture, shape, and distance to resection margins. Use different-colored dyes to mark horizontal and vertical margins. Sampling: Begin sectioning from the shortest distance between the lesion edge and resection margin. Subsequently, parallel tissue strips are cut every 2–3 mm and sequentially placed into embedding cassettes while recording their order. Embedding: Place a layer of sponge in the cassette, followed by the tissue strips, and cover with another sponge layer. Mark the same side to ensure consistent embedding orientation. Processing: After dehydration and paraffin infiltration, rotate the tissue strips 90° for sectioning, with the first and last slices embedded in reverse to comprehensively assess mucosal margins. Sectioning and staining: The paraffin-embedded tissue is cut into 2 μm-thick sections, stained with hematoxylin and eosin, and analyzed under a microscope.
Pathological diagnosis
The pathological diagnostic criteria are based on Robbins & Kumar Basic Pathology. The diagnostic criteria for LGIN primarily involve mild abnormalities in the gastric mucosal tissue structure, including elongated and hyperplastic gastric pits; the glands are composed of enlarged columnar cells with blue-stained cytoplasm, slender cigar-shaped pseudostratified nuclei exhibiting polarity, and are located at the base. The diagnostic criteria for HGIN mainly include significant alterations in both mucosal tissue structure and cellular morphology; the glands are disorganized, densely crowded, and irregular in shape, with branching, twisting, or folding, showing marked cellular atypia, pseudostratified arrangement, loss of polarity, hyperchromatic nuclei, increased mitotic figures, and involvement of the entire epithelial layer without stromal invasion. In this study, both preoperative biopsy pathology and post-ESD pathology were independently diagnosed by two senior pathologists. If their diagnoses differed, a third, more senior pathologist was consulted for the final diagnosis.
Pathological upgrade definition
Pathological upgrade was defined as cases where the preoperative biopsy pathology diagnosed LGIN but was upgraded to HGIN or GC after ESD, or where the preoperative EFB pathology diagnosed HGIN but was upgraded to GC after ESD.
Calculation and justification of the sample size
Since real-world data are used in this study, we applied “⩾10 events per predictor variable (EPV)” rule of thumb to estimate the sample size. It recommends at least 10 outcome EPV to maintain reliable estimates in logistic regression. The formulation is as below.
In our study, the number of events (pathological upgrade) is 405, and the number of predictor parameters (the total number of degrees of freedom) is 18, so 405/18 ≈ 22, this value satisfies the 10 EPV rule, which indicates that our sample size is enough to maintain reliable estimates in a logistic regression model.
Statistical analysis
Categorical variables were shown as N (%), and the chi-square test was employed to assess the difference. A logistic regression model was employed to identify associated risk factors for the pathological upgrade from EFB to ESD. All statistical analyses were performed using R 4.4.0 software (https://www.r-project.org/), with a significance threshold established at p < 0.05.
Reporting guidelines
This study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 19
Results
General clinical characteristics of patients with GIN
Of the 868 patients who underwent ESD at Wuxi People’s Hospital between 2018 and 2024, 829 had pathology reports from EFB performed within 3 months prior to ESD. In total, 147 patients whose EFB pathology indicated GC or was non-GIN were excluded; finally, a total of 682 patients were included in the study (277 in the non-upgraded group and 405 in the upgrade group; Figure 1).

Flowchart of the study.
In terms of gender distribution, the non-upgrade group comprised 62.5% males and 37.5% females, while the upgrade group showed an increased male proportion (71.6%) and a decreased female proportion (28.4%). For age distribution, the non-upgrade group had 56.3% of patients aged <70 years and 43.7% aged ⩾70 years, whereas the upgrade group exhibited a lower proportion of <70-year-olds (42.2%) and a higher proportion of ⩾70-year-olds (57.8%). Regarding BMI categories, the non-upgrade group consisted of 4.3% underweight (<18.5 kg/m2), 52.0% normal weight (18.5–24 kg/m2), 37.2% overweight (24–28 kg/m2), and 6.5% obese (⩾28 kg/m2). The upgrade group showed similar distributions: 2.0%, 54.8%, 37.5%, and 5.7%, respectively. Fecal occult blood test results were negative in 94.9% of the non-upgrade group and 93.3% of the upgrade group, while positive rates were 5.1% and 6.7%, respectively. In comorbidities/history, diabetes prevalence was 17.3% in the non-upgrade group and 21.0% in the upgrade group. Tumor history was present in 5.4% of the non-upgrade group and 6.4% of the upgrade group. Hypertension prevalence increased from 39.7% in the non-upgrade group to 50.9% in the upgrade group. For pathological features, tumor location distribution in the non-upgrade group was: antrum (58.8%), angle (17.0%), cardia (10.5%), and fundus/corpus (13.7%). The upgrade group showed the following proportions: antrum (44.0%), angle (21.7%), cardia (16.3%), and fundus/corpus (18.0%). Lesion size analysis revealed that in the non-upgrade group, 65.0% were <2 cm, 13.7% were ⩾2 cm, and 21.3% had missing data. In the upgrade group, the proportions were 62.7%, 30.9%, and 6.4%, respectively. Macroscopic type distribution in the non-upgrade group was as follows: IIa (43.0%), IIb (36.1%), IIc (15.2%), I (3.6%), and missing (2.2%). The upgrade group showed the following: IIa (38.8%), IIb (24.0%), IIc (34.1%), I (1.7%), and missing (1.5%). The indicators among the above that showed statistically significant differences in proportions between the two groups were gender, age, hypertension, pathological features location, lesion size, and macroscopic type (Table 1).
General clinical characteristics of patients with GIN.

Black bold italic indicates statistical significance.
GIN, gastric intraepithelial neoplasia.
Comparison of pathological diagnosis between EFB and ESD specimens
By comparing EFB with ESD pathology results, it was shown that among the 490 patients preoperatively diagnosed with LGIN by EFB, 239 (48.8%) remained LGIN post-ESD, 71 (14.5%) were upgraded to HGIN, and 180 (36.7%) were upgraded to GC, yielding an overall upgrade rate of 51.2%. Among the 192 patients preoperatively diagnosed with HGIN by EFB, 19 (9.9%) remained HGIN post-ESD, 154 (80.2%) were upgraded to GC, and 19 (9.9%) were downgraded to LGIN, resulting in an overall upgrade rate of 80.2%. The overall concordance rate between LGIN and HGIN was only 37.8% (258/682), while the overall upgrade rate was as high as 59.4% (405/682), indicating a significant discrepancy between EFB and post-ESD pathology, with EFB demonstrating a notable tendency for underestimation (Table 2).
Comparison of pathological diagnosis between EFB and ESD specimens.

EFB, endoscopic forceps biopsy; ESD, endoscopic submucosal dissection; GC, gastric cancer; HGIN, high-grade intraepithelial neoplasia; LGIN, low-grade intraepithelial neoplasia.
Univariate logistic regression analysis for pathological upgrade in patients with GIN
Univariate logistic regression analysis was performed on the clinical characteristics of the included subjects, including gender, age, BMI, history of chronic diseases (cancer, hypertension, diabetes), and fecal occult blood. In terms of demographic features, female patients had significantly lower upgrade rates than males (52.5% vs 62.6%, OR = 0.66), while elderly patients ⩾70 years showed significantly increased risk (65.9% vs 52.3%, OR = 1.76). Regarding pathological features, lesions located in the cardia (69.5%, OR = 2.08), gastric angle (65.2%, OR = 1.71), and fundus/corpus (65.8%, OR = 1.76) all demonstrated significantly higher upgrade risks compared to the antrum. Patients with type IIc macroscopic appearance (76.7%, OR = 2.49) and lesions ⩾2 cm (76.7%, OR = 2.33) exhibited the highest upgrade risks. In addition, patients with hypertension also showed significantly increased upgrade risk (65.2% vs 54.4%, OR = 1.57). Factors such as gender, BMI, tumor history, and fecal occult blood showed no significant associations in this study (Table 3).
Univariate and multivariate logistic regression analyses for pathological upgrade in patients with GIN.

Black bold italic indicates statistical significance.
GIN, gastric intraepithelial neoplasia; OR, odds ratio.
Multivariate logistic regression analysis for pathological upgrade in patients with GIN
We further included the eight high-risk factors obtained above in the multivariate regression analysis. The results showed that lesions in patients aged ⩾70 years had a significantly increased risk of upgrade (OR = 1.50, p = 0.020). In addition, a history of hypertension (OR = 1.45, p = 0.036) was also identified as a risk factor for pathological upgrade. Among the anatomical locations, lesions in the gastric angle (OR = 1.90, p = 0.006) and cardia (OR = 2.02, p = 0.011) exhibited higher upgrade risks compared to those in the gastric antrum. Morphological analysis revealed that type IIc lesions had a significantly higher upgrade risk than type IIa lesions (OR = 2.39, p < 0.001), while type IIb lesions showed a protective effect (OR = 0.61, p = 0.019). Lesions with a maximum size ⩾2 cm also had an increased upgrade risk (OR = 2.49, p < 0.001; Table 3; Figure 2).

Forest plot of multivariate logistic regression analysis for risk factors associated with pathological upgrade. The plot displays the ORs and their corresponding 95% CIs for various clinicopathological features. The analysis identifies several independent risk factors for pathological upgrade: age ⩾70 years (OR = 1.50), hypertension (OR = 1.45), lesion location at the gastric angle (angulus; OR = 1.90) or cardia (OR = 2.02), macroscopic type IIc (OR = 2.39), and a lesion size ⩾2 cm (OR = 2.49). Conversely, macroscopic type IIb was found to be a protective factor (OR = 0.61).
Discussion
This study identifies that age (⩾70 years), the presence of hypertension, lesion size (⩾2 cm), a type IIc morphology, and a location in the gastric angle or cardia are significant risk factors for pathological upgrades in patients with GIN. Conversely, a type IIb morphology was found to be a protective factor. This evidence may be used to optimize preoperative risk assessment and guide personalized treatment strategies. Therefore, it is recommended that clinicians prioritize these risk factors to enhance diagnostic accuracy, treatment efficacy, and patient outcomes.
GIN, as a precancerous lesion of GC, is classified into LGIN and HGIN, both carrying a risk of progression to cancer. Asian studies have shown that 18%–50% of LGIN cases persist, with 23% progressing to malignancy within 10–48 months. 20 European research has reached similar conclusions. A Dutch nationwide cohort study found that the cumulative incidence of GC at 1, 5, and 10 years of follow-up exhibited a significant gradient: 0.3%, 0.6%, and 0.8% in the atrophic gastritis group; 0.7%, 1.2%, and 1.8% in the intestinal metaplasia group; 2.1%, 3.1%, and 3.9% in the mild-to-moderate dysplasia group; and as high as 24.9%, 29.5%, and 32.7% in the severe dysplasia group. 21 A domestic prospective study revealed that, within a 5-year follow-up period, 0.6% of LGIN patients progressed to cancer annually, while the annual progression rate for HGIN patients was 1.4%. 22 These findings suggest heterogeneity among GIN patients, highlighting the importance of early identification of high-risk individuals for timely intervention and improved survival rates.
Conventional white-light endoscopy combined with EFB for pathological diagnosis remains the primary method for screening and diagnosing GIN and EGC, as well as guiding treatment decisions. ESD is recommended as the preferred endoscopic treatment for most superficial gastric lesions, 9 with advantages such as high safety, minimal invasiveness, rapid postoperative recovery, and short hospital stay. The main complications include bleeding, perforation, etc. There are several methods to be used to reduce the risk of bleeding, and intraoperative spraying of hemostatic powder can reduce the risk of bleeding and perforation. 23 In case of delayed bleeding, endoscopic electrocoagulation can be performed for hemostasis. If intraoperative perforation or delayed perforation is detected, endoscopic titanium clip closure can be performed.
Discrepancies between preoperative EFB pathology and post-ESD pathology are common, including both pathological upgrades and downgrades, with pathological upgrades significantly impacting treatment decisions. Japanese studies report a pathological discrepancy rate of 33.6% in the LGIN group and 42.7% in the HGIN group. 24 Korean research indicates an overall pathological upgrade rate of 25.7%, including 19% of LGD cases upgraded to HGD and 6.7% HGD to carcinoma. 25 A study from China shows a diagnostic concordance rate of 66.0% between pre- and post-ESD pathology, with an upgrade rate of 29.8%, including 4.2% upgraded to LGIN, 26.9% to HGIN, and 68.9% to IC. 26 The primary reasons for these discrepancies include biopsy quantity and strategy, which affect diagnostic accuracy. Graham et al. 27 demonstrated that obtaining >3 biopsy specimens increases diagnostic accuracy from 70% to 95% compared to a single biopsy. 27 Guideline form China recommends taking at least one mucosal biopsy from the antrum, body, and gastric angle during endoscopy, labeling them separately, and performing additional biopsies for suspicious lesions. 10 Although multiple biopsies improve diagnostic accuracy, 11 they may lead to submucosal fibrosis, increasing the difficulty of subsequent ESD, reducing complete resection rates, and elevating the risks of bleeding and perforation. Thus, the necessity of multiple biopsies for patients scheduled for ESD requires careful consideration. Variations in sampling depth and quality are critical factors influencing preoperative EFB pathology. Gastric wall thickness varies by location and is affected by insufflation and peristalsis. If biopsies fail to include the muscularis mucosae or are of poor quality, accurate assessment of lesion nature and invasion depth becomes challenging. Heterogeneous distribution of cancerous tissue, where focal HGIN or cancer may be hidden within LGIN background mucosa, making targeted biopsies difficult and contributing to discrepancies between biopsy and resection specimens. 28 Endoscopic adjunct techniques, such as chromoendoscopy, image-enhanced endoscopy, and artificial intelligence-assisted endoscopy, can improve lesion localization and diagnostic accuracy. 29 In this study, the overall concordance rate between preoperative and post-ESD pathology was only 37.8%, with an overall upgrade rate of 59.4%. Among preoperative LGIN cases, 51.2% were upgraded postoperatively, while the upgrade rate for preoperative HGIN cases reached 80.2%, consistent with previous reports. Although existing studies have explored risk factors for post-ESD pathological upgrades, conclusions remain controversial.14,30,31 For instance, the significance of age, male sex, and lesion size varies across studies, and the role of comorbidities like hypertension remains unclear.
This study identified hypertension as a significant risk factor for pathological upgrades. The association between hypertension and gastric precancerous lesions or cancer remains unclear. Some studies suggest shared biochemical pathways, such as endogenous mitogens and oncogenes activating inositol triphosphate, elevating cytoplasmic calcium levels, which may contribute to both hypertension and early gastric mucosal lesions. 32 A Korean study found that 55.4% of elderly patients with gastric dysplasia undergoing endoscopic resection had hypertension, making it one of the most common comorbidities. 33 This study is the first to propose hypertension as a potential risk factor for post-ESD pathological upgrades, offering a new perspective for managing patients with chronic conditions. However, there is the possibility of confounding; this finding requires the larger cohorts and a prospective study in a certain age group to validate the causality.
Age was another significant risk factor in this study. Patients aged ⩾70 years had a significantly higher risk of pathological upgrade (OR = 1.50), consistent with prior research. 26 This may be attributed to higher rates of gastric mucosal atrophy, intestinal metaplasia, and immune decline in elderly populations. In addition, comorbidities in older patients may impair mucosal repair, increasing cancer risk.
Lesion size was strongly associated with postoperative pathology. A Korean study reported a 2–3 times higher upgrade risk for lesions>2 cm. 34 Lee et al. found diagnostic concordance rates of 39% for lesions >2 cm versus 62% for those <2 cm. 35 This study similarly observed a significantly higher upgrade risk for lesions ⩾2 cm, likely due to greater heterogeneity and sampling errors in larger lesions. Moreover, larger lesions may exhibit deeper invasion, further increasing upgrade risks.
The Japanese guidelines highlight type IIc (superficially depressed) as a typical morphology of EGC. 36 This study found that type IIc lesions carried a higher upgrade risk (OR = 2.39) compared to type IIa (superficially elevated), while type IIb (superficially flat) showed a protective effect (OR = 0.61). This may reflect differences in histological features and invasion depth. Type IIc lesions often involve deeper mucosal infiltration, making biopsy sampling challenging, 37 while type IIb lesions are more superficial, improving diagnostic accuracy. 12
Lesion location was another critical risk factor. Notably, lesions in the gastric angle and cardia exhibited higher upgrade risks than those in the antrum. This may stem from anatomical and physiological differences. The cardia, adjacent to the lower esophagus, is frequently exposed to acid and bile reflux, promoting intestinal metaplasia and dysplasia. The proximal stomach is prone to postoperative pathological upgrading due to numerous mucosal folds, limited visualization, and high lymphatic density. The gastric angle, a transitional zone between the body and antrum, experiences higher mechanical friction, potentially accelerating lesion progression.38,39 These findings suggest endoscopists should remain highly alert to these regions, increase biopsy times, follow hierarchical management strategies as per guidelines, 9 and use high-quality endoscopy (including virtual chromoendoscopy (VCE)) with VCE-guided biopsy to assist diagnosis and staging, thereby determining subsequent endoscopic treatment and follow-up strategies.
In summary, discrepancies between EFB and post-ESD pathology impact therapeutic efficacy and patient prognosis. Endoscopists must recognize the limitations of EFB, particularly in high-risk GIN patients (males, elderly, hypertensive patients); precise preoperative pathological evaluation is necessary to develop ESD procedure or follow-up plans. For high-risk lesions (⩾2 cm, type IIc, cardia/gastric angle), standardized biopsies, adjunct endoscopic techniques, and close collaboration with pathologists are essential to improve preoperative accuracy. In the future, consideration may also be given to verify the universality of these high-risk predictors in a multicenter cohort, and these indicators can be potentially integrated into pre-ESD risk stratification protocols.
This study has some limitations. First, its retrospective design and exclusion of surgically treated cases may introduce selection and information bias. Some of the results we obtained, such as hypertension as a risk factor for pathological upgrading after ESD procedure, still need further prospective research to verify. Second, pathological upgrades were assessed solely based on histologic grade (inflammation → LGIN → HGIN → cancer), omitting molecular markers (e.g., p53, Ki-67) that could reveal early malignant features. 40 Third, single-center data may lack generalizability; future multicenter prospective studies integrating clinical, morphological, and molecular data are needed to refine predictive models in the future.
Conclusion
In this study, we identified that age ⩾70 years, with hypertension, lesions ⩾2 cm, type IIc lesions, and lesions located at the gastric angle or cardia are independent risk factors for pathological upgrading in patients with GIN after ESD, whereas type IIb lesions serve as protective factors. These findings suggest the potential clinical value of enhanced preoperative monitoring in helping reduce postoperative pathological upgrading risk in GIN patients, offering a preliminary basis for refining preoperative risk assessment and individualizing management.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251397847 – Supplemental material for Risk factors analysis for pathological upgrade after endoscopic submucosal dissection in patients with gastric intraepithelial neoplasia: a single-center retrospective study
Supplemental material, sj-docx-1-tag-10.1177_17562848251397847 for Risk factors analysis for pathological upgrade after endoscopic submucosal dissection in patients with gastric intraepithelial neoplasia: a single-center retrospective study by Lijia Ding, Wenying Tian, Jiale Lv, Jinglue Han, Rong Liu, Yunan Zhang, Xiaoxue Zhang, Zhifa Lv, Ke Chen, Qinglin Zhang, Juntao Li, He Nie, Fengjun Sun, Qiang Zhan, Fangmei An, Shuping Si and Lin Ji in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-2-tag-10.1177_17562848251397847 – Supplemental material for Risk factors analysis for pathological upgrade after endoscopic submucosal dissection in patients with gastric intraepithelial neoplasia: a single-center retrospective study
Supplemental material, sj-pdf-2-tag-10.1177_17562848251397847 for Risk factors analysis for pathological upgrade after endoscopic submucosal dissection in patients with gastric intraepithelial neoplasia: a single-center retrospective study by Lijia Ding, Wenying Tian, Jiale Lv, Jinglue Han, Rong Liu, Yunan Zhang, Xiaoxue Zhang, Zhifa Lv, Ke Chen, Qinglin Zhang, Juntao Li, He Nie, Fengjun Sun, Qiang Zhan, Fangmei An, Shuping Si and Lin Ji in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.