Abstract
Background:
The current standard treatment paradigm for patients with unresectable and non-metastatic hepatocellular carcinoma (HCC) involves transarterial chemoembolization (TACE) as the primary local therapeutic modality, followed by systemic therapy upon disease progression. Emerging evidence suggests that the concurrent integration of immunotherapy and targeted therapy with TACE may yield synergistic therapeutic effects. Recent phase III trials—including EMERALD-1, LEAP-012, and TALENTACE—have provided important insights into the efficacy and safety of this multimodal approach.
Objectives:
This meta-analysis aimed to quantitatively synthesize the efficacy and safety of first-line TACE combined with immunotherapy and targeted therapy versus TACE alone in patients with unresectable, non-metastatic HCC.
Design:
Systematic review and meta-analysis of phase III randomized controlled trials (RCTs).
Data sources and methods:
A systematic search of PubMed and major conference proceedings was conducted up to July 6, 2025. Three phase III trials (EMERALD-1, LEAP-012, TALENTACE) were included. Pooled hazard ratios (HRs) for progression-free survival (PFS), odds ratios (ORs) for objective response rate (ORR), and risk ratios (RRs) for adverse events (AEs) were calculated using fixed-effect or random-effects models.
Results:
The combination of TACE with immunotherapy and targeted therapy demonstrated superior efficacy compared to TACE monotherapy. The pooled HR for PFS was 0.69 (95% confidence interval (CI): 0.60–0.79), indicating a significant delay in disease progression. The pooled OR for ORR was 1.83 (95% CI: 1.45–2.31), reflecting a markedly improved tumor response rate. However, overall survival (OS) data remain immature, and no statistically significant difference in OS was observed at this stage. In terms of safety, the combination therapy was associated with a higher incidence of AEs. Specifically, it significantly increased the risk of grade 3–5 AEs (RR: 1.88, 95% CI: 1.48–2.40) and serious AEs (RR: 1.65, 95% CI: 1.31–2.07).
Conclusion:
First-line treatment with TACE combined with immunotherapy and targeted therapy significantly improves tumor response and prolongs PFS in patients with unresectable and non-metastatic HCC, albeit at the cost of increased toxicity. This combination strategy represents a promising advancement in the therapeutic landscape for HCC, offering meaningful improvements in oncologic outcomes while highlighting the need for careful toxicity management.
Trial registration:
International Prospective Register of Systematic Reviews (PROSPERO; CRD420250652613).
Plain language summary
This study looks at the best first treatment for people with liver cancer that can’t be removed by surgery and hasn’t spread to other parts of the body. Researchers combined three treatments: transarterial chemoembolization (a procedure that delivers cancer-fighting drugs directly to the liver via blood vessels), immunotherapy (drugs that help the immune system attack cancer), and targeted therapy (drugs that target specific parts of cancer cells). They reviewed results from large, late-stage clinical trials to see how well this combination works. The goal was to find out if using all three together is more effective than single treatments. For patients with this type of liver cancer, finding the right first-line treatment is key to slowing the disease and improving quality of life. This analysis helps doctors and patients understand if combining these treatments could be a better option than using them alone.
Keywords
Introduction
Hepatocellular carcinoma (HCC) remains a major global health challenge and is among the leading causes of cancer-related mortality worldwide, with approximately 900,000 new cases diagnosed annually and a dismal 5-year survival rate of less than 20% in advanced stages.1,2 For patients with unresectable, non-metastatic HCC who are not candidates for curative treatments, transarterial chemoembolization (TACE) has served as the standard of care for over two decades.3,4 TACE achieves localized tumor control by delivering chemotherapeutic agents directly into the tumor vasculature while simultaneously inducing ischemia through embolization. However, its therapeutic benefits are frequently transient, with a median progression-free survival (PFS) of only 7–8 months and a high incidence of tumor recurrence.5,6 Moreover, repeated TACE procedures can progressively impair liver function, thereby limiting the feasibility of subsequent systemic therapy. 7 These limitations underscore the pressing need for novel strategies that can augment the therapeutic efficacy of TACE and prolong survival in this patient population.
Recent advancements in systemic therapies—particularly immune checkpoint inhibitors (ICIs) and agents targeting the vascular endothelial growth factor (VEGF) pathway, such as monoclonal antibodies and tyrosine kinase inhibitors (TKIs)—have transformed the treatment landscape for advanced HCC.8–12 Combinatorial regimens, such as atezolizumab (anti-PD-L1) plus bevacizumab (anti-VEGF), and dual immune checkpoint blockade (e.g., nivolumab plus ipilimumab), have demonstrated significant improvements in overall survival (OS), thereby establishing new benchmarks in first-line systemic therapy.13–15 Mechanistically, TACE-induced tumor necrosis and hypoxia may enhance the immunogenicity of tumors by promoting antigen release and upregulating PD-L1 expression, potentially sensitizing tumors to ICIs.16–18 Additionally, anti-angiogenic agents may counteract TACE-induced neovascularization and normalize tumor vasculature, thereby improving immune cell infiltration. This biological rationale has catalyzed growing clinical interest in integrating TACE with systemic therapies to enhance antitumor efficacy and address the unmet needs in patients with intermediate-stage HCC.
In this context, three pivotal phase III randomized controlled trials (RCTs)—EMERALD-1, LEAP-012, and TALENTACE—have recently investigated the clinical utility of combining TACE with immunotherapy and targeted therapy in patients with unresectable, non-metastatic HCC.19–21 The EMERALD-1 trial demonstrated that the addition of durvalumab (anti-PD-L1) and bevacizumab to TACE significantly prolonged median PFS compared to TACE monotherapy (15.0 vs 8.2 months; hazard ratio (HR) 0.77), with an acceptable safety profile. 19 Similarly, the LEAP-012 trial reported a notable PFS benefit when pembrolizumab (anti-PD-1) and lenvatinib (a multikinase inhibitor) were combined with TACE, compared to TACE plus placebo (14.6 vs 10.0 months; HR: 0.66), along with a non-significant trend toward improved OS. 20 The TALENTACE study found that TACE combined with atezolizumab and bevacizumab nearly doubled median PFS relative to TACE alone (10.32 vs 6.37 months; HR: 0.64). 21 Collectively, these trials underscore the potential of TACE-based combination regimens to redefine therapeutic algorithms in the first-line treatment of intermediate-stage HCC.
Despite the promising findings from these individual studies, no comprehensive synthesis has yet quantified the pooled efficacy and safety of this multimodal strategy. Furthermore, the limited statistical power of each individual trial constrains the ability to perform meaningful subgroup analyses for clinically relevant populations. Therefore, a meta-analysis of these phase III trials is warranted to provide a more robust estimate of treatment efficacy, assess the risk of high-grade and serious adverse events (AEs), and identify patient subgroups most likely to derive benefit from this approach. The present analysis seeks to consolidate the available evidence on TACE combined with immunotherapy and targeted therapy, with the goal of informing clinical practice and guiding future research in the management of intermediate-stage HCC.
Methods
Protocol and guideline
This study was registered in the International Prospective Register of Systematic Reviews (PROSPERO) and conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 22 The PRISMA checklist is provided as Supplemental Material.
Data source and search strategies
A comprehensive literature search on PubMed and major conference proceedings was conducted to identify RCTs evaluating the efficacy and safety of TACE in combination with immunotherapy and targeted therapy, compared to TACE alone, for the treatment of unresectable and non-metastatic HCC. The PubMed database was systematically searched for relevant studies published up to July 6, 2025. The following search strategy was applied: ((“Transarterial chemoembolisation”) AND ((“Immunotherapy” OR “Pembrolizumab” OR “Nivolumab” OR “Atezolizumab” OR “Durvalumab”)) AND (“Bevacizumab” OR “Sorafenib” OR “Lenvatinib”) AND (“Hepatocellular carcinoma” OR “Liver cancer”)).
Selection criteria
Studies were deemed eligible for inclusion in the meta-analysis if they met the following criteria:
(i) they were phase III RCTs comparing TACE combined with immunotherapy and targeted therapy versus TACE alone;
(ii) they enrolled previously untreated patients with unresectable and non-metastatic HCC; and
(iii) they reported survival outcomes, including HRs with corresponding confidence intervals (CIs).
Exclusion criteria included
(i) Non-randomized study designs, such as single-arm trials;
(ii) studies that did not use TACE as the control intervention;
(iii) studies in which TACE was not administered in the experimental arm; and
(iv) ongoing trials without publicly available results at the time of the literature search.
All eligible studies meeting these criteria were subsequently included in the analysis.
Data extraction and risk of bias assessment
The following information was extracted from each study, when available: trial name, publication year, study design, sample size, treatment regimen, patient characteristics, HRs with corresponding 95% CIs for PFS, number of patients achieving complete or partial responses based on RECIST v1.1 criteria, and the incidence of AEs. The risk of bias for each included study was systematically assessed using the Cochrane Collaboration’s Risk of Bias tool. 23
Statistical analysis
To evaluate efficacy, HRs with corresponding 95% CIs for PFS, as well as odds ratios (ORs) with 95% CIs for objective response rate (ORR), were extracted from individual studies and pooled to generate summary estimates. For the safety analysis, risk ratios (RRs) with 95% CIs for AEs were similarly calculated to provide an overall assessment of treatment-related toxicity.
Heterogeneity among studies was assessed using the I2 statistic and Cochrane’s Q test. Heterogeneity was considered significant if the I2 exceeded 50% and the p-value for the Q test was less than 0.1, in which case a random-effects model was applied. When heterogeneity was not significant, a fixed-effect model was used. Given the limited statistical power to detect true homogeneity in subgroup analyses, a random-effects model was preferentially employed to account for potential underlying heterogeneity that might not be statistically apparent.
All statistical analyses were conducted using R software (version 4.5.1), with statistical significance defined as a two-tailed p-value of <0.05.
Results
Study selection
The literature search initially identified six records. Of these, four were assessed for eligibility, and three studies met the inclusion criteria and were subsequently included in the final analysis.19–21 The study selection process is detailed in Supplemental Figure 1.
Characteristics of the included studies
Among the included trials, two were double-blind phase III RCTs, and one was an open-label phase III RCT. A total of 1231 patients were enrolled across the 3 studies, with 612 patients (49.7%) receiving TACE in combination with both immunotherapy and targeted therapy, and 619 patients (50.3%) receiving TACE monotherapy. Among the three trials, EMERALD-1 allowed patients with slightly impaired liver function (Child-Pugh B7), LEAP-012 restricted enrollment to Child-Pugh A and strictly non-metastatic disease, while TALENTACE included patients with a higher intrahepatic tumor burden (maximum tumor diameter + number of lesions ⩾6) and limited macrovascular invasion (Vp1-2). These population differences may partially explain the heterogeneity observed across trials. The systemic regimens evaluated included durvalumab plus bevacizumab (EMERALD-1), pembrolizumab plus Lenvatinib (LEAP-012), and atezolizumab plus bevacizumab (TALENTACE). The baseline characteristics of the included trials are summarized in Table 1.
Main characteristics of the included phase III trials.

BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; Ctrl, control; ECOG, Eastern Cooperative Oncology Group; Exp, experimental; HCC, hepatocellular carcinoma; HR, hazard ratio; mPFS, median progression-free survival; ORR, objective response rate; RCT, randomized controlled trial; TACE, transarterial chemoembolization.
Efficacy analysis
In the meta-analysis evaluating PFS (n = 1231), pooled results from the intention-to-treat (ITT) population demonstrated a significant benefit for the combination of TACE with immunotherapy and targeted therapy (HR: 0.69; 95% CI: 0.60–0.79; Figure 1(a)). Notably, no significant heterogeneity was observed among the included studies (I2 = 0%).
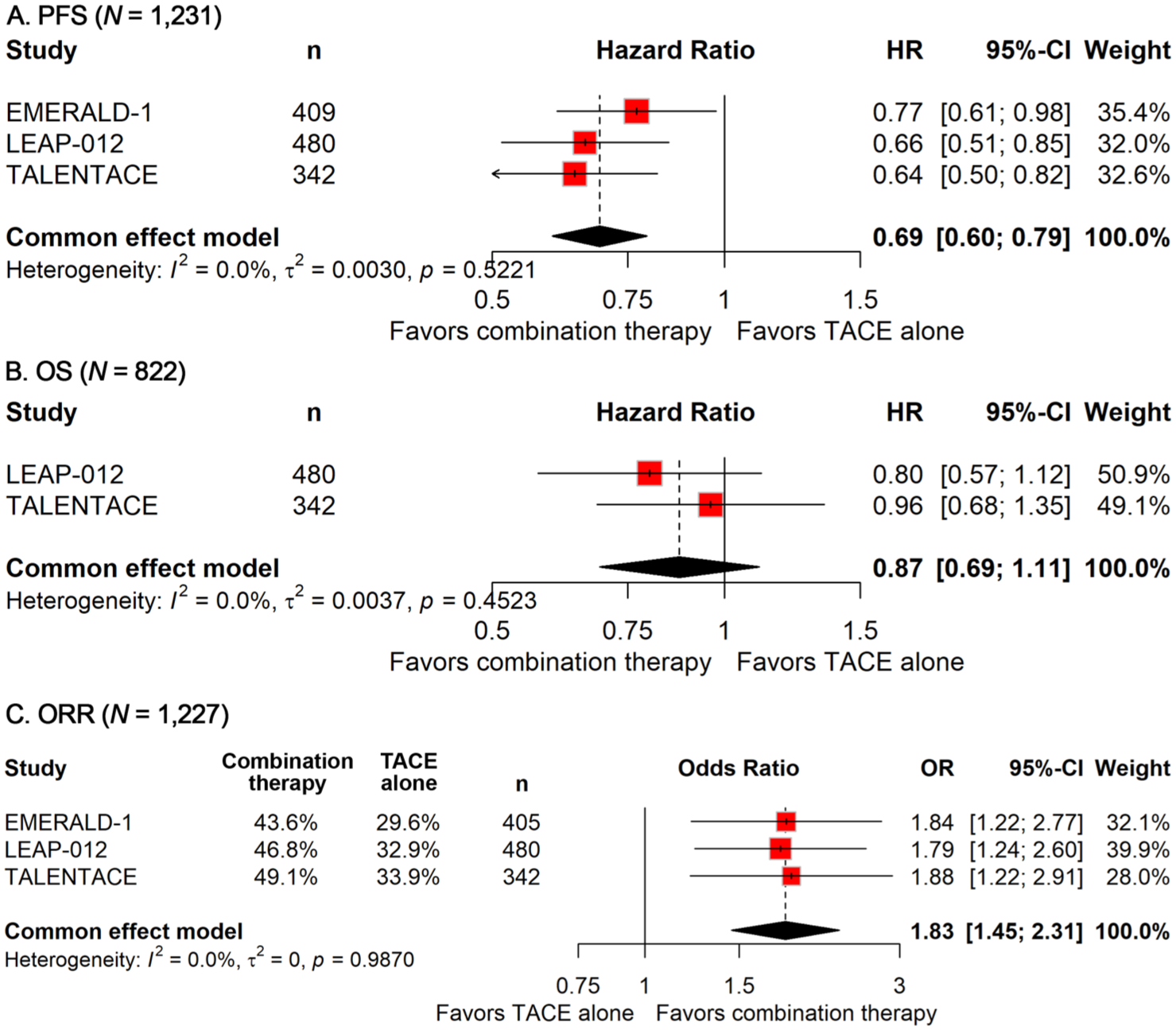
Forest plot of progression-free survival (a), overall survival (b), and objective response rate (c) comparing the combination therapy versus TACE alone in patients with hepatocellular carcinoma.
For OS (n = 822), the integration of immunotherapy and targeted agents with TACE did not result in a statistically significant reduction in mortality risk (HR: 0.87; 95% CI: 0.69–1.11; Figure 1(b)). However, it is important to note that the OS data remain immature. Similar to the PFS analysis, no heterogeneity was detected across the OS estimates (I2 = 0%).
With respect to the ORR (n = 885), the addition of immunotherapy and targeted therapy to TACE in the ITT population was associated with a significantly improved response (OR: 1.83; 95% CI: 1.45–2.31; Figure 1(c)). Consistent with the PFS and OS results, no statistical heterogeneity was observed in the ORR estimates (I2 = 0%).
Safety analysis
A total of 361 out of 557 patients (64.8%) experienced grade 3–5 AEs in the group receiving TACE combined with immunotherapy and targeted therapy, compared to 206 out of 614 patients (33.6%) in the TACE monotherapy group. The meta-analysis demonstrated that the addition of immunotherapy and targeted therapy significantly increased the risk of grade 3–5 AEs (RR, 1.88; 95% CI: 1.48–2.40; Figure 2(a)). However, substantial heterogeneity was observed across the studies (I2 = 73.1%).

Forest plot of grade 3–5 AEs (a) and serious AEs (b) comparing the combination therapy versus TACE alone in patients with hepatocellular carcinoma.
Safety data on serious AEs were available from two trials. The incidence of serious AEs was 36.6% (117/320) in the combination therapy group versus 23.1% (86/373) in the TACE monotherapy group. The pooled analysis revealed a significantly higher risk of serious AEs in patients receiving combination therapy (RR, 1.65, 95% CI: 1.31–2.07; Figure 2(b)).
A detailed analysis was also conducted to examine the incidence of specific all-grade AEs as reported in the three phase III trials. The most frequently observed events included hypertension (37.7% in the experimental group vs 11.1% in the control group), proteinuria (36.4% vs 4.6%), and elevated aspartate aminotransferase levels (33.3% vs 25.6%). Other notable AEs included post-embolization syndrome, hypothyroidism, and hyperbilirubinemia, as illustrated in Supplemental Figure 2.
Subgroup analysis
Given the limited statistical power of individual studies to assess various clinically relevant subgroups, we conducted a comprehensive series of subgroup analyses to more precisely delineate the therapeutic efficacy of TACE combined with immunotherapy and targeted therapy across distinct patient populations. These analyses were based on demographic and pathological characteristics, with the aim of informing personalized treatment strategies.
Subgroup analysis stratified by demographic variables
To evaluate the influence of demographic factors on treatment response, subgroup analyses were performed according to age, sex, geographical region, and Eastern Cooperative Oncology Group (ECOG) performance status in patients with unresectable and non-metastatic HCC.
Among patients aged <65 years, the addition of immunotherapy and targeted therapy to TACE significantly prolonged PFS (HR: 0.67; 95% CI: 0.51–0.89; Figure 3(a)). A comparable PFS benefit was observed in patients aged ⩾65 years (HR: 0.71; 95% CI: 0.57–0.90; Figure 3(a)).

Forest plot of progression-free survival for the combination therapy versus TACE alone in hepatocellular carcinoma populations, stratified by age (a) and sex (b).
Sex-stratified analysis demonstrated that combination therapy significantly improved PFS in male patients (HR: 0.66; 95% CI: 0.54–0.80), whereas no statistically significant benefit was observed in female patients (Figure 3(b)).
Regarding ECOG performance status, patients with an ECOG performance status of 0 experienced a significant improvement in PFS (HR: 0.66; 95% CI: 0.55–0.79), while no significant benefit was observed in those with an ECOG performance status of 1 (Figure 4(a)).

Forest plot of progression-free survival for the combination therapy versus TACE alone in hepatocellular carcinoma populations, stratified by ECOG performance status (a) and region (b).
Region-specific analyses indicated that the addition of immunotherapy and targeted therapy to TACE significantly improved PFS in both the Asian (excluding Japan) subgroup (HR: 0.65; 95% CI: 0.49–0.85; Figure 4(b)) and the Japan and other regions subgroup (HR: 0.77; 95% CI: 0.59–1.00; Figure 4(b)).
Subgroup analysis stratified by disease pathology
To explore the impact of pathological characteristics on treatment outcomes, further subgroup analyses were conducted based on viral etiology, alpha-fetoprotein (AFP) levels, Barcelona Clinic Liver Cancer (BCLC) stage, and albumin-bilirubin (ALBI) grade.
Patients were stratified by viral etiology into hepatitis B virus (HBV), hepatitis C virus (HCV), and non-viral groups. The addition of immunotherapy and targeted therapy to TACE significantly improved PFS in patients with HBV (HR: 0.68; 95% CI: 0.49–0.94) and in those with non-viral etiologies (HR: 0.63; 95% CI: 0.44–0.92), while no significant benefit was observed in patients with HCV infection (Figure 5(a)).

Forest plot of progression-free survival for the combination therapy versus TACE alone in hepatocellular carcinoma populations, stratified by viral etiology (a) and alpha-fetoprotein (b).
AFP-based stratification revealed that patients with AFP levels <400 ng/mL experienced a significant PFS benefit from combination therapy (HR: 0.69; 95% CI: 0.56–0.85), whereas no significant improvement was noted among those with AFP levels ⩾400 ng/mL (Figure 5(b)).
When stratified by BCLC stage, patients with stage B disease demonstrated a significant improvement in PFS with combination therapy (HR: 0.64; 95% CI: 0.50–0.82; Figure 6(a)). However, no significant differences were observed in patients with stage A or stage C disease (Figure 6(a)).

Forest plot of progression-free survival for the combination therapy versus TACE alone in hepatocellular carcinoma populations, stratified by BCLC stage (a) and ALBI grade (b).
Lastly, ALBI grade-based analysis showed that combination therapy significantly improved PFS in patients with ALBI grade 1 (HR: 0.65; 95% CI: 0.50–0.84), whereas no significant benefit was detected in patients with ALBI grade ⩾2 (Figure 6(b)).
However, it is important to note that the above subgroup analyses for PFS were exploratory and were based on data from two trials (EMERALD-1 and LEAP-012), as such detailed data were not available from the TALENTACE trial.
Risk of bias
The risk of bias across the included trials is graphically summarized in Supplemental Figures 3 and 4. Both the EMERALD-1 and LEAP-012 trials employed double-blind designs, effectively minimizing the risk of performance and detection biases. In contrast, the TALENTACE trial adopted an open-label design, which inherently introduced a high risk of performance bias due to the absence of blinding. Nevertheless, TALENTACE exhibited low risks of selection bias, attrition bias, and reporting bias, as judged by its study design and execution. All three trials were assessed as having a low risk of selection and attrition biases. Owing to their double-blind methodology, EMERALD-1 and LEAP-012 were further determined to have low risk across all evaluated domains, including performance, detection, and reporting biases. Additionally, neither the funnel plot nor Egger’s test indicated the presence of publication bias (Supplemental Figure 5).
Discussion
This meta-analysis presents the first quantitative synthesis of data from three pivotal phase III RCTs—EMERALD-1, LEAP-012, and TALENTACE—evaluating the efficacy and safety of combining TACE with immunotherapy and targeted therapy in patients with unresectable and non-metastatic HCC. Our findings offer compelling evidence that this triplet combination regimen significantly improves PFS and ORR compared to TACE alone, albeit at the cost of increased grade 3–5 and serious AEs. These results underscore the evolving treatment landscape of intermediate-stage HCC and raise important clinical, mechanistic, and practical considerations that merit further discussion.
The principal finding of this meta-analysis is the significant improvement in PFS with the addition of ICIs and anti-angiogenic agents to TACE, indicating a 31% reduction in the risk of disease progression or death compared to TACE monotherapy. Notably, the absence of statistical heterogeneity (I2 = 0%) across the included trials reinforces the robustness and consistency of this benefit, regardless of drug combinations, patient demographics, or trial design.
In line with the PFS benefit, the combination regimen also yielded a markedly higher ORR, reflecting an 83% increase in the likelihood of achieving radiological response. This finding is particularly relevant because ORR has been shown to correlate with longer survival in HCC and may serve as a surrogate endpoint in future trials.24,25 The improved tumor response with combination therapy likely stems from the complementary mechanisms of action: TACE induces immunogenic cell death and releases tumor neoantigens, priming the immune response, while ICIs sustain T-cell activation and anti-VEGF/VEGFR therapies remodel the tumor microenvironment to facilitate immune infiltration and improve drug delivery.16–18
Importantly, these benefits were consistently observed across various clinically relevant subgroups. Subgroup analyses stratified by age, geography, ECOG performance status, viral etiology, BCLC stage, and AFP levels reaffirmed the generalizability of the results. Patients under and over 65 years of age both derived significant PFS benefit, as did patients with HBV-related or non-viral HCC, BCLC stage B disease, and ALBI grade 1 liver function. These observations align with prior findings that intermediate-stage patients with preserved liver function and higher tumor burden may derive the most benefit from combination approaches.26–28
Despite these encouraging efficacy signals, the impact of the combination strategy on OS remains uncertain. Our analysis did not reveal a statistically significant OS benefit at the time of data cutoff (HR: 0.87; 95% CI: 0.69–1.11), a finding that likely reflects the immaturity of survival data, the influence of subsequent therapies, and the potential for crossover in clinical trials. Given these nuances, the observed OS trend—though not statistically significant—should not be interpreted as a lack of survival benefit. In the LEAP-012 trial, for instance, the median OS in the combination arm had not yet been reached at the time of interim analysis, suggesting potential for delayed separation of survival curves. 20 Future pooled analyses with longer follow-up and individual patient data will be critical to elucidate the OS impact of this strategy.
The integration of immunotherapy and targeted therapy with TACE, while efficacious, is not without cost. Our safety analysis revealed a marked increase in both grade 3–5 and serious AEs with the combination regimen. Nearly two-thirds of patients receiving combination therapy experienced grade 3–5 AEs, compared to one-third in the TACE monotherapy group, and the risk of serious AEs was also significantly elevated. The most frequently observed toxicities—hypertension, proteinuria, and elevated liver enzymes—are consistent with the known safety profiles of anti-VEGF agents and TKIs, and their incidence was substantially higher in the experimental arms. These findings underscore the necessity of vigilant monitoring, proactive management of AEs, and judicious patient selection to balance the benefits of enhanced disease control against the risks of increased toxicity. Multidisciplinary care, involving hepatologists, oncologists, and interventional radiologists, is essential to optimize outcomes and mitigate harm, particularly in patients with compromised hepatic reserve or comorbidities that may predispose to treatment-related complications.
The compelling clinical efficacy observed with TACE plus immunotherapy and targeted therapy is underpinned by a strong biological rationale. TACE induces tumor ischemia and necrosis, which in turn triggers local inflammation and antigen release, thereby transforming a “cold” tumor into a more “immunogenic” phenotype. 17 This process may increase PD-L1 expression and enhance T-cell priming, rendering the tumor more susceptible to ICIs. Simultaneously, VEGF blockade serves multiple functions: it normalizes the tumor vasculature, facilitates T-cell infiltration, and reduces immunosuppressive cell populations such as regulatory T cells and myeloid-derived suppressor cells. 16 By disrupting the immunosuppressive tumor microenvironment, VEGF/VEGFR inhibition synergizes with ICIs to potentiate durable immune responses. These mechanistic insights lend strong support to the observed clinical outcomes and justify ongoing trials evaluating similar combinations across HCC stages.
Historically, intermediate-stage HCC has been managed primarily with TACE, often repeated until disease progression or hepatic decompensation. However, accumulating evidence—including the present meta-analysis—suggests that this strategy may be suboptimal for patients with high tumor burden or aggressive biology, who are unlikely to benefit from repeated locoregional therapy alone. In this context, the integration of systemic therapy into the first-line treatment of intermediate-stage HCC represents a paradigm shift. TACE-based combination regimens could become the new standard of care for selected patients, especially those classified as “TACE-unsuitable,” as defined by the Kinki criteria or the “up-to-7” rule.29–31 In fact, expert consensus from recent international liver meetings increasingly supports a personalized, biology-driven treatment algorithm rather than a strict stage-based approach.
While our meta-analysis provides a comprehensive synthesis of the available evidence, several limitations warrant consideration. The primary limitation is the immaturity of OS data, which precludes definitive conclusions regarding the long-term survival benefit of combination therapy. The heterogeneity of systemic regimens employed across the included trials introduces variability in efficacy and safety profiles, although the consistency of PFS and ORR benefit across studies lends confidence to our findings. The relatively small sample sizes in certain subgroups limit the statistical power to detect differences and render these analyses exploratory rather than confirmatory. Furthermore, the subgroup analyses were based on data from two trials (EMERALD-1 and LEAP-012), as the TALENTACE trial did not report detailed subgroup data, and therefore should be interpreted with caution. Additionally, the open-label design of one included trial introduces potential for performance and detection bias, although the overall risk of bias was deemed low. Lastly, the generalizability of our findings to real-world populations, who may have greater comorbidity burden and more advanced liver dysfunction, remains to be established.
The promising results of this meta-analysis open several avenues for future research. First, the ongoing maturation of OS data from existing and future trials will be essential to clarify the long-term impact of combination therapy on survival. Besides, ongoing trials such as IMPACT and EMERALD-3 will provide further data on the role of systemic therapies in intermediate-stage HCC, potentially enabling more refined patient stratification and biomarker-driven treatment approaches. Furthermore, the identification and validation of predictive biomarkers—such as PD-L1 expression, immune gene signatures, or molecular subtypes—hold promise for refining patient selection and personalizing therapy, thereby maximizing benefit while minimizing harm. Moreover, optimal sequencing and duration of TACE and systemic therapy remain unclear. Trials exploring induction systemic therapy followed by TACE (or vice versa), maintenance ICIs after TACE, or de-escalation strategies after deep response are warranted. Additionally, future analyses should consider quality-of-life outcomes, patient-reported symptoms, and hepatic function preservation as key endpoints in addition to traditional survival metrics. Finally, the accumulation of real-world evidence will be critical to validate the findings of randomized trials in broader, more heterogeneous patient populations and to monitor long-term safety in routine clinical practice.
Looking beyond TACE, the therapeutic landscape for intermediate-stage HCC is expanding to include other locoregional modalities. Ongoing trials are actively investigating combinations of systemic therapy with transarterial radioembolization, which may offer a distinct efficacy and safety profile.32,33 The results of these studies will further expand the therapeutic arsenal and help define the optimal integration of locoregional and systemic approaches.
Conclusion
This meta-analysis substantiates the therapeutic value of combining TACE with immunotherapy and targeted therapy in patients with unresectable and non-metastatic HCC. The triplet regimen significantly improves tumor response and delays progression, with manageable but increased toxicity. While OS data remain immature, the consistent benefit in PFS and ORR across multiple subgroups supports this strategy as a rational and promising first-line option for appropriately selected patients. These findings represent a substantial advance in the treatment of HCC, providing a strong foundation for the integration of combination therapy into clinical guidelines and informing the design of future studies aimed at further improving outcomes for this challenging patient population. As the therapeutic landscape continues to evolve, the ongoing refinement of combination strategies, informed by biomarker-driven patient selection and real-world experience, holds the promise of transforming the prognosis of unresectable HCC and delivering durable benefits to patients worldwide.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251396424 – Supplemental material for Transarterial chemoembolization combined with immunotherapy and targeted therapy as first-line treatment for unresectable and non-metastatic hepatocellular carcinoma: a meta-analysis of phase III trials
Supplemental material, sj-docx-1-tag-10.1177_17562848251396424 for Transarterial chemoembolization combined with immunotherapy and targeted therapy as first-line treatment for unresectable and non-metastatic hepatocellular carcinoma: a meta-analysis of phase III trials by Lichao Wang, Bo Luo, Mingrui Zhao and Jinhai Shen in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251396424 – Supplemental material for Transarterial chemoembolization combined with immunotherapy and targeted therapy as first-line treatment for unresectable and non-metastatic hepatocellular carcinoma: a meta-analysis of phase III trials
Supplemental material, sj-docx-2-tag-10.1177_17562848251396424 for Transarterial chemoembolization combined with immunotherapy and targeted therapy as first-line treatment for unresectable and non-metastatic hepatocellular carcinoma: a meta-analysis of phase III trials by Lichao Wang, Bo Luo, Mingrui Zhao and Jinhai Shen in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
