Abstract
Background:
Many women of childbearing age with inflammatory bowel disease (IBD) require advanced therapies. While biologics are largely low risk during pregnancy, the novel small molecules tofacitinib, filgotinib, upadacitinib and ozanimod (TFUO) have shown concerning teratogenic effects, and decreased fertility in animal studies. Therefore, their use in women of childbearing age needs careful consideration.
Design:
RAND/University of California Los Angeles (UCLA) Appropriateness Method (RAM).
Objective:
To evaluate the appropriateness of TFUO in women of childbearing age.
Methods:
We convened a panel of six gastroenterologists, two IBD nurses, one IBD pharmacist and three expert patients. Following a literature review, 13 statements were drafted and voted upon in 2 rounds.
Results:
All 13 statements were deemed appropriate. The panel concluded that women with IBD of childbearing age who wish to commence therapy with TFUO, need to use effective contraception and be counselled regarding the risk in unplanned pregnancies. For women using contraception while on Janus kinase inhibitor (JAKi) therapy, we suggest the preferred use of progesterone-only or non-hormonal long-acting contraception. TFUO are contraindicated during pregnancy and breast feeding. We recommend that women receiving TFUO cease therapy in time to establish clinical remission for at least 3 months prior to conception. Therapies other than TFUO should be considered as first-line therapy in women with IBD of childbearing age, except in select individual circumstances. TFUO may be appropriate for women of childbearing age after failure of, intolerance or contraindications to one biological agent.
Conclusion:
TFUO should be avoided during pregnancy and breastfeeding, and alternative therapies should be considered as first-line treatments.
Summary:
We provide clinical practice recommendations regarding the use of TFUO for IBD in women of childbearing age.
Plain language summary
The maintenance of remission is vital for good fetal and maternal outcomes in IBD pregnancies and biological therapies used in IBD have been associated with favourable pregnancy outcomes. The safety profile of novel small molecules in human pregnancy is however largely unknown.
We conducted a RAND appropriateness panel to agree recommendation son the use of Tofacitinib, Filgotinib, Upadacitinib and Ozanimod in women of childbearing age. Tofacitinib, Filgotinib, Upadacitinib and Ozanimod should be avoided during pregnancy due to serious concerns regarding birth defects. Small molecule therapies Tofacitinib, Filgotinib, Upadacitinib and Ozanimod should be given due consideration in the management of IBD in women of childbearing age, provided there are no immediate plans for conception. Counselling about potential risk of adverse effects during pregnancy, risk of venous thromboembolism and effective contraception are needed. Clinicians need to consider time to establish alternative therapies when switching away from Tofacitinib, Filgotinib, Upadacitinib and Ozanimod in the pre-conception period.
Future research should examine pregnancy outcomes in cases where Tofacitinib, Filgotinib, Upadacitinib and Ozanimod exposure occurred but data may be difficult to compare to biologics due to differences in exposure duration. Guidelines should be updated as further maternal and fetal safety data become available
Background
The peak incidence of inflammatory bowel disease (IBD) is between the second and fourth decades of life (age 15–40 years), which coincides with prime reproductive years, underpinning the importance of an astute understanding of a myriad of issues with reproductive planning by health care providers.1,2 Indeed, approximately 50% of females with IBD may be of childbearing. 3
Prospective registries and studies have provided considerable reassurance regarding the use of most biological therapies used to treat IBD in women during the preconception period, through pregnancy and postpartum.1,4–9 The recent (2023) ECCO consensus guidelines on sexuality, fertility, pregnancy and lactation continues to suggest that the risk of flare outweighs the risk of medication, with most drug treatments boosting clinician confidence with the use of most drug treatments preconception, during pregnancy and breastfeeding.2,10
Although the safety profile of anti-tumour necrosis factor (TNF) biologics4,11 and newer biologics including Vedolizumab 12 and Ustekinumab 13 during pregnancy has been largely reassuring, novel small molecule drugs must be treated with caution. In contrast to biologics, they cross the placenta passively during the first trimester leading to significant fetal exposure during this crucial period. Animal model data suggest adverse outcomes, 14 raising concerns regarding the theoretical risk of adverse human pregnancy outcomes. Likewise, these small molecule treatments have also been identified at various levels during breastmilk sampling in animal studies 14 and in one human case report, 15 suggesting the potential for babies to be exposed to the mother’s drug therapy.
Confidence and experience among clinicians in managing IBD in people of childbearing age may vary and a great deal of uncertainty currently exists around the appropriateness of novel small molecules in this cohort. These uncertainties include when to offer women with IBD small molecule therapy, appropriate contraception, counselling regarding pregnancy risks, wash out periods and optimising the timing of pregnancy. We convened a RAND/UCLA 16 expert panel from the United Kingdom with the aim of supporting informed and shared decision making among clinicians and women living with IBD. It should be noted that many pregnancies are unplanned and clinicians should initiate these discussions with women of childbearing age early and where appropriate include partners/significant others (such as parents during age appropriate paediatric/transition/adolescent clinics).
For uniformity, these consensus statements will refer to people of childbearing age as ‘women’ or ‘female’ although we recognise, respect and recommend sensitive conversations with people who may be able to conceive but identify with a different or no gender.
The purpose of the study was to develop best practice statements regarding the use of novel small molecules for women of childbearing age diagnosed with IBD.
Methods
The RAND/UCLA appropriateness method uses a modified Delphi panel approach and combines expert opinion with the best available evidence to determine the appropriateness of specific practices in certain clinical situations, with particular relevance in areas of uncertainty wherein evidence is insufficient to guide day-to-day clinical practice. 16 The aim of our RAND/UCLA panel was to provide clarity for informed and shared decision making around the use of novel small molecule (tofacitinib, filgotinib, upadacitinib and ozanimod (TFUO)) therapies among clinicians for women living with IBD.
A working group with expertise in IBD and pregnancy was established, with 12 delegates: 6 gastroenterologists, 2 IBD nurses, 1 IBD pharmacist and 3 expert patients. Participants were selected because of their relevant clinical experience, research expertise, involvement in previous national-level collaborative antenatal IBD work and/or involvement in national/international guidelines panels for IBD in pregnancy by C.P.S. and R.L. Patient participants were selected by clinicians based on their personal experience with IBD and family planning decisions. The initial consensus statements were created by a steering committee (R.L. and C.P.S.) based on the PICO format (population, intervention, comparison, outcomes).
A literature review was performed in Medline, Embase and PubMed to identify English language publications relevant to each statement, supplemented by data on animal and human pregnancy outcomes held by the relevant drug manufacturers with data published prior to the consensus meeting in early October 2023 included. In this manuscript all pregnancy exposure is only reported for maternal exposure unless explicitly stated as in statement. 12 The literature review was disseminated to all delegates. Delegates voted on the appropriateness of each statement through a personalised link to Google Forms. Voting results were assessed for appropriateness and agreement based on the RAND/UCLA appropriateness method. 16 Responses were recorded on a Likert scale ranging from 1 to 9 (completely inappropriate to completely appropriate). All delegates completed this step.
After the first round of remote voting, statements were refined based on the results and feedback. All delegates were invited to a meeting to discuss the statements without agreement. This allowed for interdisciplinary insight and ascertainment of patient views. A second round of voting then determined the agreement and appropriateness of the final consensus statements (Table 1). The median and IQR of the results were calculated. The AGREE and CREDES checklists were followed (Supplemental Material).17,18
Definitions of agreement and appropriateness.
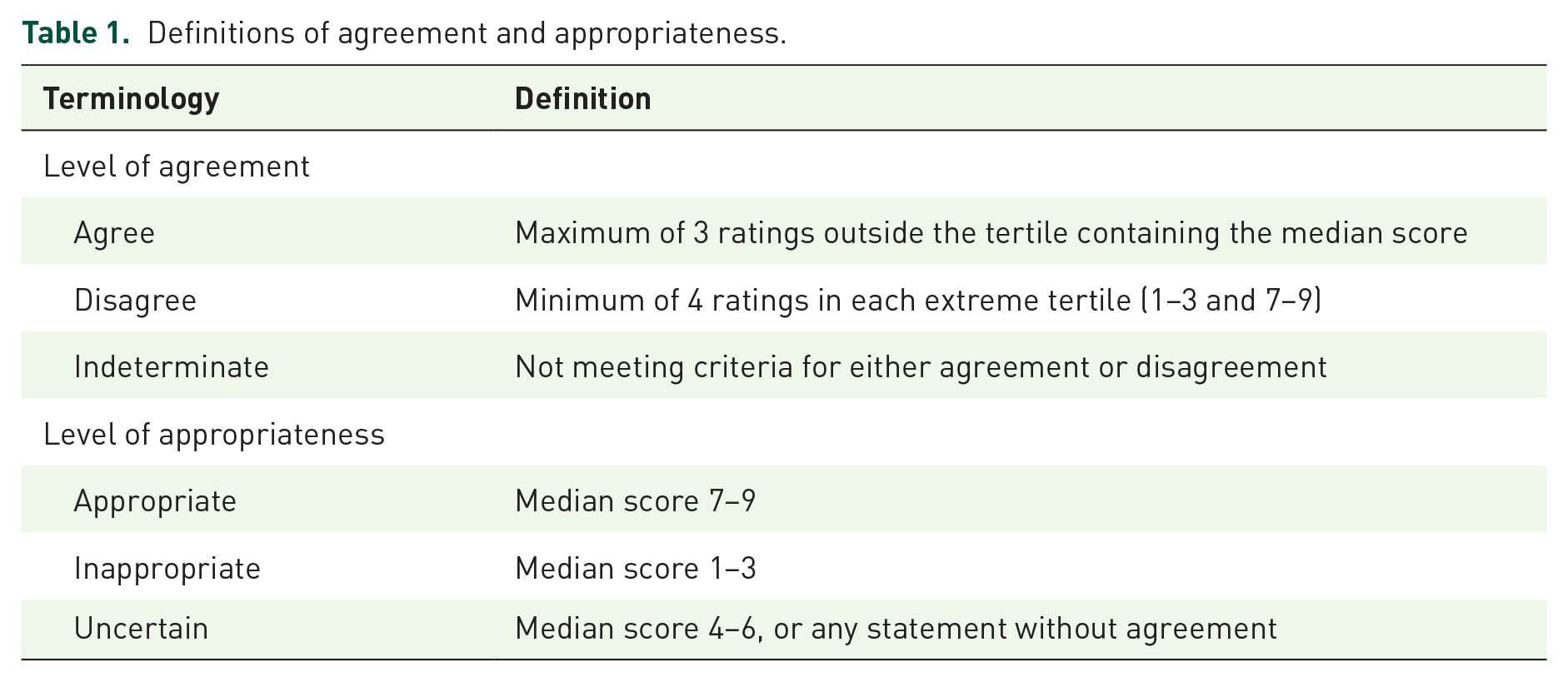
Given the current lack of conclusive evidence, all statements are intended as an aid to joint patient-clinician decision making and in individual circumstances, it may be appropriate to consider alternative courses of action.
Results
All 13 consensus statements met criteria for inclusion based on voting (Table 2) and are listed and described below.
1. Women with IBD, who are of childbearing age and wish to commence therapy with TFUO, need to use effective contraception. Counselling regarding the risk of TFUO in unplanned pregnancies should be provided prior to commencing therapy.
There are significant concerns over the reproductive safety of TFUO based on animal studies. 14 Tofacitinib has been reported in pre-clinical animal data (in rats and rabbits) as being teratogenic and negatively affects female fertility (decreased pregnancy rate; decreases in the numbers of corpora lutea, implantation sites, and viable foetuses and an increase in early miscarriages) at doses 6.3 times higher than human equivalent dose of 10 mg BD in rabbit models, with a higher No-Adverse-Effect Level in murine studies. Data on exposure of doses comparable to human exposure have not been reported. 19 For filgotinib, limited human data have shown 2 serious congenital malformations in 55 live births. 14 Studies on rats and rabbits revealed that dose equivalent to 200 mg of filgotinib for humans were associated with internal hydrocephaly, dilated ureters, multiple vertebral anomalies and visceral malformations of the lungs and cardiovascular system). 20 One serious congenital malformation was reported in 21 live births. For upadacitinib, musculoskeletal and cardiovascular malformations were observed in pregnant rats and rabbits exposed to the equivalent of human level dosing (1.5 times the equivalent dose of 15 mg) with upadactinib. No congenital malformations were reported in 30 live births. 21 Animal studies for ozanimod revealed generalised oedema/anasarca and malpositioned testes in rats, malpositioned caudal vertebrae and malformations of the great vessels in rabbits. 22 One congenital malformation was observed in 31 live births.
Final voting results.

The IQR indicates the range within which the central 50% of ratings fell, highlighting the consensus among panel members.
IBD, inflammatory bowel disease.
As the human data stem largely from clinical trials with very limited duration of pregnancy exposure, no reassurance can be drawn from these. S1P receptors are involved in regulating events during embryogenesis, such as angiogenesis, cardiogenesis, limb development and neurogenesis. 23 While some post-marketing reports have included patients with longer first trimester exposure with reasonable outcomes,14,24,25 the numbers are very small at present and patients must be advised of the risk of potentially serious complications in unplanned pregnancies exposed to these four agents. Taken together, these data underpin the importance of effective contraception for all women of childbearing age starting novel small molecule therapy for IBD. Clinicians need to inform women of the potential risk in unplanned pregnancies by individualised counselling.
2. For women using contraception while on tofacitinib, filgotinib or upadacitinib, we suggest the preferred use of progesterone-only or non-hormonal long-acting contraception formulations to combined oestrogen-progesterone formulations to reduce the risk of venous thromboembolism.
Based on an observed increased risk of venous thromboembolism (VTE) among older patients with rheumatoid arthritis exposed to Tofacitinib, 26 the Medicinces and Healthcare products Regulatory Agency (MHRA) and European Medicines Agency (EMA) have issued a warning for increased VTE risk with all JAKi. Oral contraception with the combined oral contraceptive pill that contains oestrogen is associated with an increased risk of VTE. 27 In contrast, other studies reporting on the risk of VTE with combined oral contraceptives containing progesterone (drospirenone (DSRP)) concluded that DSRP-containing pills were associated with a slight to no increase in risk as compared with other pills.28–30
It is therefore inadvisable to augment the risk of VTE (already heightened by active IBD) with additional medication-related risk factors. IBD clinicians should encourage patients to discuss alternative effective contraception for example the use of progesterone-only or non-hormonal long-acting contraception formulations to combined oestrogen-progesterone formulations with their primary care practitioner.
3. When offering TFUO therapies to women of childbearing age, clinicians need to consider that nearly half of pregnancies in the United Kingdom are not planned.
Although the global unintended pregnancy rate appears to be declining, it is estimated that there are >90,000 unplanned pregnancies per year. 31 Globally, 44% (90% UI 42–48) of pregnancies and 23% (22–26) of births were unintended; 56% (90% UI 53–60) of all unintended pregnancies were terminated in 2010–2014. 31 The largest UK dataset regarding pregnancy planning from the third National Survey of Sexual Attitudes and Lifestyles (Natsal-3) surveyed 15,162 men and women. 32 Women who had been pregnant within the last year provided data regarding their attitude to and planning of their pregnancy: 16.2% of pregnancies scored as ‘unplanned’, 29.0% as ‘ambivalent’ and 54.8% as ‘planned’. The proportion of unplanned pregnancies was highest among 16–19 year olds, although the highest number of overall pregnancies occurred in the women aged 20–34 years, who consequently accounted for two thirds of unplanned pregnancies. Only 55% pregnancies were ‘planned’ in terms of contraception, timing, partner/family discussion and pre-conception health preparations (e.g. folic acid). 32 This is important, given the association of unplanned pregnancies with adverse health outcomes for mother, baby and children long term. 33 Factors associated with unplanned pregnancy include smoking, sexual intercourse before 16 years of age, drug use other than cannabis and depression. 32
There are few data on unplanned pregnancies in IBD. In a Dutch study, 17% of pregnancies in a tertiary IBD antenatal clinic were unplanned, in contrast to 29% in the general Dutch population.34,35 Worryingly, 23% of women with IBD do not use contraception and up to 83% are not on highly effective methods of contraception, increasing the risk of mistimed or unplanned pregnancies. 36 A high unplanned pregnancy rate should be considered when treating a patient with a potentially teratogenic medication, and effective contraception should form part of routine conversations with adults of childbearing age considered for TFUO.
4. TFUO are contraindicated during pregnancy due to serious concerns over teratogenicity seen in animal studies.
Due to their low molecular weight, the small molecules JAKi and S1P inhibitors like ozanimod are able to cross the placenta passively throughout pregnancy, with consequent fetal exposure. However, there are limited data regarding outcomes of human pregnancies exposed to small molecules. Therefore, current concerns regarding the teratogenicity of JAKi and S1P inhibitors are based on data from pre-clinical animal studies.
As outlined in the supporting text for statement 1TFUO showed concerning malformations in animal experiments. A recent meta-analysis by Monfared et al. published the human data from clinical trials and real-world observations. 14 Data were collated from six publications reporting pregnancies in patients prescribed small molecules, alongside data supplied by the four manufacturers. Three hundred fifteen pregnancies were reported, resulting in 130 healthy live births (41%), 51 medical terminations (16%), 49 miscarriages (16%), 4 congenital malformations and 3 other complications (2%). Seventy-eight (25%) were lost to follow-up. 14 These data provide some reassurance but are insufficient to outweigh concerns regarding teratogenicity of these small molecules. Recent reports of tofacitinib exposure throughout pregnancy in less than five patients have been reassuring.24,25 Large registries, such as the PIANO (Pregnancy Inflammatory bowel disease And Neonatal Outcomes) study, are likely to collate prospective data of IBD patients becoming pregnant on TFUO. Until more data are available, we advise avoiding the use of these agents during pregnancy.
5. Regardless of serum clearance periods, we recommend that women currently receiving tofacitinib, filgotinib, upadacitinib or ozanimod cease therapy with enough time to establish clinical remission ideally for at least 3 months prior to conception. This window allows to safely establish whether the alternative (or no) therapy can maintain remission. Remission should be achieved and maintained prior to conception.
IBD activity at the time of conception is a key determinant of the course of IBD during pregnancy.35,37–39 Unplanned or mistimed pregnancies are associated with a higher risk of delayed pre-conceptual care, increased risk of preterm birth, low birth weight and adverse maternal and neonatal outcomes.37–39,40,41 Furthermore, IBD activity during pregnancy is associated with adverse pregnancy-related outcomes, such as miscarriage, intrauterine growth retardation and preterm birth.42–45
A systematic review and meta-analysis including 28 studies reported an association of flares of IBD during the periconception and pregnancy period and a higher risk of pregnancy complications in comparison with patients who had quiescent IBD. 41 The pooled odds ratios (ORs) for various adverse pregnancy outcomes compared to those in women with inactive IBD were: low birth weight (3.8 (95% CI 1.8–8.0)), small for gestational age (i.e. a birth weight less than the 10th percentile for gestational age (OR 1.5 (95% CI 1.2–1.9)), preterm birth (OR 2.4 (95% CI 1.7–3.4)), preeclampsia (OR 2.8 (95% CI 0.7–11.6), early pregnancy loss (OR 1.9 (95% CI 1.2–3.0)) and stillbirth (OR 2.3 (95% CI 1.0–5.0). 41
Although patients with IBD who are in remission at conception have up to a 38% risk of increased activity in pregnancy,46,47 this is similar to flare rates reported in non-pregnant women.37,38 Conversely, the risk of continued disease activity throughout pregnancy is nearly doubled among patients with flaring IBD at conception.37,38,41,48 Moreover, in patients who experienced an IBD flare during pregnancy, the risk of active IBD during subsequent pregnancies may be increased.39,49 Taken together, this emphasises the importance of achieving clinical remission for a minimum period of 3 months prior to conception, though some increased risk of flares exists even for patients with disease activity prior to 3 months before conception. Sufficient time is required to enable a suitable therapy to be selected and used effectively to achieve and maintain remission.2,50–52
6. When clinicians plan to initiate TFUO in women of childbearing age with IBD, the patient’s current family plans, potential length of time before the patient may contemplate potential pregnancies and willingness to use effective contraception need to be discussed.
As outlined earlier, animal data for small molecules have demonstrated significant fetal anomalies,53,54 and current human data cannot provide reassurance. 14
In light of these concerns, clinicians need to discuss family planning with women when considering TFUO as therapeutic options, to establish whether these are suitable options. Clinicians should not assume a woman’s family planning wishes. An open conversation will help clinicians and patients in making appropriate choices on an individualised basis.
Women of childbearing potential should be counselled on the possible risk to the foetus in unplanned pregnancies. There needs to be agreement on the use of effective contraception during treatment or clear documentation of reasons where this may not be appropriate for medical or social reasons. Women should agree not to get pregnant for at least 3 months after stopping small molecule therapies, while other suitable treatments are considered.2,51,55 A shared decision-making approach in clinical conversations with patients is encouraged, in selecting a highly effective contraceptive method. This should take into consideration the values and preferences of the patient in the context of efficacy and risks of contraceptive method.55,56
7. The minimum suggested period between ceasing therapy and safely conceiving for TFUO are 4 weeks, 1 week, 4 weeks and 3 months respectively, according to manufacturers’ information.
Manufacturers of TFUO have provided suggested periods for continuing effective contraception after ceasing therapy in women of childbearing potential, based on the calculated wash out period for each drug.19–22 We have summarised these in Table 3 and we recommend that these time frames are explained to the patient before trying to conceive, after ceasing these small molecule therapies. Of note, this period only allows for the medication to be washed out but not for any alternative therapy to be established prior to conception, as is often desirable.
Manufacturer’s recommendations for minimum period between ceasing therapy and conception.

It must be emphasised that while the serum half-life of TFUO is short, tissue half-life and biological effects may be much longer. For example, the effect of ozanimod on absolute lymphocyte counts may take up to 8 weeks to resolve. 57 We therefore recommend following the manufacturer recommendations, over using half-lives as to ascertain residual biological effects when determining when to stop therapy prior to planned conception.
8. Therapies other than TFUO should be considered as
All patients with IBD should be offered the most appropriate therapy with the key considerations being efficacy and safety of proposed therapies. As discussed earlier, teratogenicity of TFUO in animal models and scarcity of pregnancy outcomes in patients using these medications implies that they are contraindicated in pregnancy 14 and agents other than TFUO should be considered as first-line advanced therapy for IBD for most women of childbearing age. There may be specific individual circumstances, however, where TFUO might represent an appropriate first-line option. These may include women not wanting to biologically carry a foetus and currently using effective contraception, or women where other available therapeutic options are medically not appropriate and they are aware and accepting of the potential known and unknown risks of these medications during pregnancy. Additionally, for women who have completed their family, TFUO may also be considered. In the event of initiating TFUO in women of childbearing age, it is strongly advised that an effective and safe contraception method is used.
When deciding on treatment, it is important that we utilise shared decision making with the patient. Clinicians should ask women about their reproductive plans and preferences. Benefits and consequences of all available treatment options must be discussed including which agents are not considered to be compatible with pregnancy. When TFUO are being used, discussion about their safety should be continually revisited as the patient may change their reproductive plans and desires.
9. TFUO may be appropriate choices for women of childbearing age after failure of, intolerance of or contraindications to one biological agent licensed for IBD.
Although contraindicated in pregnancy and in women actively considering becoming pregnant, TFUO, they deserve appropriate consideration in women of childbearing age on an individualised basis. .
Each time a new treatment is needed in women of childbearing age, however, it is important to discuss the safety profile of these agents with the patient as they may change their reproductive plans and desires. Upon starting TFUO the importance of effective contraception should always be emphasised.
10. In women who wish to have children in the future a minimum treatment period for TFUO should be considered. Where they wish to start a family in less than 1 year in the future it is recommended to consider agents other than TFUO.
For women who have plans for future pregnancy, it is important to consider the probable duration of treatment. Stopping TFUO therapies, to prepare for pregnancy, would require a wash out period, for the drug and the biologic effects of the drug on the immune system to wear off. As active IBD is associated with adverse pregnancy outcomes, 2 additional time must be factored in to establish an alternative effective therapy. This is important to ensure that cessation of TFUO does not trigger a disease flare-up with the associated risk of adverse pregnancy outcomes.
Consequently, for women actively considering pregnancy within a relatively short timeframe, typically less than 1 year, alternative agents are likely more appropriate. An exception to this might be a scenario where these agents are used as induction agent only, as a bridging therapy to a slower acting agent that would be considered safe in pregnancy. Where women have already been exposed to all other available therapies, advice from an expert in IBD antenatal care should be sought.
11. TFUO are contraindicated during breastfeeding.
There are limited data regarding breastfeeding and, with the exception of a single case report on Tofacitinib, all data are supplied by the manufacturers. 15 Tofacitinib is found in breast milk in animal studies (rats) and humans (single case report).15,19,22 Filgotinib was present in nursing rat pups likely due to exposure from milk in rats given filgotinib with an exposure equivalent to 6% of maternal exposure. 20 Upadacitinib is found in the milk of lactating rates in concentrations leading to 30-fold the exposure of maternal serum. 21 Ozanimod has been found in the milk of lactating rats. 22
Tofacitinib can be found in human breast milk in amounts that may be sufficiently large to exert negative effects on the infant. A case report of a woman who expressed milk for research purposes after commencing Tofacitinib 10 mg BD (and did no longer breastfeed her infant) showed detectable levels in all milk samples (median 9.9 ng/mL, range 2.0–54.5). 15 Exclusive breastfeeding could lead to a daily exposure of 0.0082 mg tofacitinib per kg bodyweight to the foetus, which may be sufficient to cause potential harm. 15 A second case of breast feeding an infant while exposed to 5 mg Tofacitinib showed similar levels of breast milk exposure with good infant outcomes. 24
Breastfeeding woman needing to commence TFUO should be recommended to switch to formula feeding rather than exposing the infant to a potentially serious risk. However, it is currently difficult to quantify risk through breast milk exposure. Alternatively, women should be offered therapies other than TFUO with a more acceptable risk versus benefit profile for breastfeeding.
12. If a woman conceives while taking tofacitinib, filgotinib, upadacitinib or ozanimod, the medication should be ceased, she should receive counselling about potential known and unknown maternofetal complications, and alternative therapy may then be commenced depending on individual circumstances.
If a woman conceives while taking TFUO, several aspects of her care need to be addressed promptly. We recommend that TFUO therapy ceases immediately to avoid any potential additional harm to the foetus. Even if a woman plans a medical termination of pregnancy, it is the opinion of the RAND/UCLA panel that therapy should cease regardless, as the short-term flare risk for up to 4 weeks will be low. It is recognised that it can be very emotive for a woman to have an unplanned pregnancy while undergoing these therapies. She may change her mind following consultation with her specialists, about the known and unknown effects. Therapy should therefore be stopped immediately to allow the woman time to come to a decision. For those opting for a termination, therapy may be restarted after termination of pregnancy.
All women who have an unplanned pregnancy while on TFUO should be counselled by an expert in obstetric or fetal-maternal medicine with input from gastroenterology where required. It should be highlighted that while animal data are concerning, human data (with drug exposure limited to the first 5–8 weeks of pregnancy like in this scenario) have been relatively reassuring and there is a reasonable chance of a healthy baby. Counselling should include advice on how much reassurance can be drawn from routine ultrasound scanning of the foetus and how much uncertainty remains despite normal scans. The individual choice for a pregnant woman on whether to continue a pregnancy or not will largely depend on the degree of willingness to accept the uncertainty over potential adverse fetal outcomes and her desire to have a biological child.
All patients continuing the pregnancy should receive advice on alternative therapies with suitable biologic agents to maintain remission throughout pregnancy. Woman should be advised on the importance of maintaining remission for optimal maternal and infant outcomes 2 and reassured regarding the safety profile of suitable biological agents. The choice of therapy depends on individual circumstances such as duration and level of remission, previous therapy and current symptom burden.
Clinicians should be aware of the psychological impact of unplanned pregnancies, which is associated with postpartum depression, among other complex social issues relating to negative impact on relationships and financial pressures. 58 The impact of an unplanned pregnancy with the added stress of possible adverse pregnancy outcomes cannot be underestimated. Counselling women in such situations must respect relevant sensitivities around the issue but also provide best available evidence, in a nuanced manner ensuring that women have ongoing support.
We recognise that in some circumstances women may have already had exposure to all other alternative therapies and only a TFUO therapy has provided sufficient control of their IBD. In that scenario, some patients may opt to continue TFUO during a pregnancy. The risk of this is currently difficult to quantify; there have been three patients reported in the published literature who continued Tofacitnib through all three trimesters of pregnancy with favourable pregnancy outcomes.24,25 If continuation of TFUO is considered, this should be done after counselling by an expert in IBD and antenatal care and close follow-up by gastroenterologists and experts in fetal-maternal medicine. The risk of continuation on fetal outcomes is currently difficult to quantify.
13. Male patients taking tofacitinib, filgotinib, upadacitinib, or ozanimod do not have to cease these agents while trying to conceive.
The data regarding male fertility, sperm motility and quality mainly stem from unpublished data available from the manufacturers. Tofacitinib had no effects on male fertility, sperm motility or sperm concentration in animal studies. 19 Upadacitinib had no effect on fertility in male rats, 21 and ozanimod had no effect on male fertility in animal studies. 22
In animal studies, filgotinib exposure was associated with decreased fertility, impaired spermatogenesis and histopathological effects on male reproductive organs were observed. 20 Two dedicated phase II clinical studies (MANTA and MANTA RAy, n = 240) evaluated human testicular safety in men with inflammatory arthritis diseases and IBD. 59 The studies did not reveal a difference between treatment groups in the proportion of patients who had a 50% or more decrease from baseline in semen parameters at weeks 13 and 26 (pooled primary endpoint: filgotinib 6.7%, placebo 8.3%). 59 In addition, no relevant changes were observed in sex hormone levels or any change from baseline in semen parameters across treatment groups. 59 The dedicated human studies, therefore, suggest no significant effects on male fertility, sperm motility and quantity.
In 29 pregnancies with 30 outcomes (1 twin pregnancy) where the male partner was exposed to ozanimod, there were 21 live births (1 spontaneous abortion, 8 outcomes unknown) and 3 congenital abnormalities (Hirschsprung’s disease, congenital hydrocele, and partial atrioventricular septal defect) were observed. 22
Based on reassuring animal data for tofacitinib and upadacitinib, there were no significant concerns over fathering children during exposure. Human data for filgotinib and ozanimod are also reassuring. TFUO therapy, if effective, should not be discontinued in men planning to father a child.
Discussion
These consensus statements combine input from a multidisciplinary group of experts to provide up-to-date guidance on the use of small molecule agents in people with IBD of childbearing age. Although guidance may change in the future as more data accumulates, caution is currently advised for this patient group due to concerning findings in animal studies and the limited human data available.
The main strength of our work relates to the thorough systematic review of the literature 14 prior to the RAND/UCLA panel convening, significant patient involvement and the adherence to the RAND/UCLA process. We applied a multi-disciplinary team approach to our work involving clinicians of all relevant backgrounds (gastroenterologists, IBD specialist nurses and pharmacists). Patients were involved in all aspects of the work and their testimony (Box 1) highlights the importance of joint decision making. We acknowledge that the evidence on which our statements are based is often preliminary or limited but there is an urgent need for clinical guidance for TFUO therapy for women of childbearing age. Most statements were informed by animal studies, which showed significant concerns. Humans studies were more reassuring but severely limited by short exposure time to the medications, insufficient follow-up and low numbers of pregnancies. Etrasimod has not been included in the statements as the licence for use was not yet granted at the time of the consensus meeting. It is, however, likely that the same considerations would apply.
Patients statements.

Conclusion
In conclusion, it is appropriate for women of childbearing age to be offered TFUO at least as second line advance therapy but due to the risk of adverse pregnancy outcomes, counselling for effective contraception and use of alternative therapies for those considering pregnancy is vital. Future studies reporting pregnancy outcomes in women (largely accidentally) exposed to TFUO are needed to provide a more informed evidence base.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241299737 – Supplemental material for Appropriateness of small molecule agents for patients with IBD of childbearing age – a RAND/UCLA appropriateness panel
Supplemental material, sj-docx-1-tag-10.1177_17562848241299737 for Appropriateness of small molecule agents for patients with IBD of childbearing age – a RAND/UCLA appropriateness panel by Christian Selinger, Robyn Laube, Jimmy K. Limdi, Kate Headley, Alexandra Kent, Klaartje Kok, Aileen Fraser, Victoria Newman, Helen Ludlow, Fiona Rees, Nidhi Sagar and Erin Walker in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
