Abstract
Background:
Recurrent Clostridioides difficile infection (rCDI) disrupts health-related quality of life (HRQoL), often causing debilitating symptoms, emotional distress, and social withdrawal. Fecal microbiota, live-jslm (RBL; REBYOTA®) is approved by the FDA to reduce CDI recurrence. While the burden of rCDI is well established, less is known about the patient experience after RBL treatment and its impact on HRQoL.
Objectives:
To explore the burden of rCDI on HRQoL, and any improvements in symptoms and impacts after RBL administration by colonoscopy.
Design:
A qualitative sub-study embedded in the CDI-SCOPE phase IIIb clinical trial (NCT05831189).
Methods:
Qualitative interviews were conducted with 30 trial participants approximately 8 weeks after RBL treatment. Interviews were conducted using a semi-structured discussion guide; they were audio-recorded, transcribed verbatim, and thematically analyzed.
Results:
Thirty of 41 trial participants (73%) took part in the qualitative interviews (July 2023–September 2024). Participants (mean age 59 years; 87% female) had experienced multiple prior rCDI episodes. Before RBL treatment, participants reported severe symptoms, including diarrhea (30/30; 100%), abdominal pain (21/30; 70%), and fatigue (14/30; 47%), which negatively affected daily life, social interactions, and emotional health. Eight weeks after RBL treatment, no participants reported a recurrence. Diarrhea symptoms resolved (17/30; 57%) or improved (13/30; 43%) for all participants, typically within 1 month. Average severity scores for diarrhea declined markedly (from 8.9 to 1.3). Every participant (100%) reported improved ability to engage in daily activities; nearly all (29/30; 97%) described positive changes to social life/relationships, and 77% (23/30) reported improved emotional well-being.
Conclusion:
RBL administered via colonoscopy provided rapid, meaningful relief from rCDI symptoms, and due to symptom improvements, many participants reported that they were able to resume daily life. These findings highlight the broader value of RBL in restoring HRQoL and overall well-being.
Trial registration:
ClinicalTrials.gov: NCT05831189.
Introduction
Clostridioides difficile is a bacterium that causes colon infection with symptoms ranging from diarrhea to life-threatening colitis. 1 In 2017, C. difficile accounted for nearly half a million infections in the United States. 2 There has been a continued high overall incidence of C. difficile infection (CDI) and severity of disease.3–6
People diagnosed with CDI are at a high risk of recurrent CDI (rCDI), which most commonly presents as repeated diarrhea within 8 weeks of stopping antibiotic treatment against CDI. After an initial diagnosis of CDI, 10%–30% of individuals diagnosed with CDI experience at least one recurrence, and the risk of recurrence increases with each successive episode. 4 rCDI can persist for years, leading to antibiotic cycling, compromised health-related quality of life (HRQoL), hospitalizations, and increased mortality. 7
Fecal microbiota transplant (FMT) is a highly effective treatment for resolution of rCDI. 8 Ferring has developed fecal microbiota live-jslm (RBL; REBYOTA® (formerly RBX2660)) to prevent rCDI. The safety and efficacy of RBL has been investigated when delivered by rectal administration in five clinical trials.9–12 In the PUNCH CD3 trial, 71% of patients responded to RBL and did not experience a recurrence after 8 weeks; the results showed that treatment efficacy was 70% versus 58% with placebo. 13 RBL delivered by rectal administration was approved by the U.S. Food and Drug Administration on 30 November 2022 and by Health Canada on 06 March 2025 for the prevention of rCDI in adults.
More recently, Ferring conducted a multicenter, single-arm phase IIIb study (NCT05831189; CDI-SCOPE), evaluating the safety and clinical effectiveness of RBL when administered as a single dose via colonoscopy. Overall, 41 adults with rCDI received RBL treatment in the CDI-SCOPE trial; 39 study patients completed the 8-week follow-up period. The trial’s primary objective was to assess safety of RBL, and secondarily to explore effectiveness. Results from the CDI-SCOPE trial demonstrated that RBL was safe and effective against preventing rCDI with 95% of patients experiencing treatment success (no CDI diarrhea at 8-week follow-up visit). 14 Another secondary objective was to explore patient experience of RBL and its impact on HRQoL. Prior survey data captured in the PUNCH CD3 trial evidenced that RBL-treated patients reported more substantial and sustained disease-specific HRQoL improvements than placebo-treated patients. 13 However, in-depth qualitative data on the patient experience after RBL treatment and the impact on HRQoL are lacking and are explored further in this study.
Methods
Clinical trial overview
Adults with a diagnosis of rCDI and a positive stool test for C. difficile toxin or toxigenic C. difficile were eligible for screening in the CDI-SCOPE trial. These individuals were either currently taking antibiotics or had just received a prescription for a course of antibiotics to control rCDI symptoms. To receive treatment with RBL, the stool diary must have demonstrated that CDI symptoms were under control, that is, no longer meeting the symptomatic criteria for CDI diarrhea. CDI diarrhea during the trial was defined as passage of ⩾3 unformed/loose stools (i.e., Bristol Stool Scale type 6–7) in 24 consecutive hours for ⩾2 consecutive days.
At baseline, after the antibiotic washout period and bowel preparation, subjects received treatment with RBL, administered by colonoscopy to the right colon. Follow-up visits occurred at 1, 2, 4, and 8 weeks, and 3 and 6 months after RBL treatment. Follow-up visit 4 was performed on-site 8 weeks ±3 days after RBL treatment and served as the timepoint for the primary endpoint. At this visit, the outcome was determined as one of the following: (1) treatment failure, that is, presence of CDI diarrhea, (2) treatment success, that is, absence of CDI diarrhea, or (3) indeterminate.
Interviews
Participation in the patient experience qualitative interview was optional; signed informed consent was obtained in-person at the clinical trial site. All 41 trial study patients who received the RBL treatment were invited to participate in one exit interview. Purposive sampling was utilized; rCDI patients who consented and completed the follow-up visit 4 at 8 weeks after RBL treatment, or who were documented as treatment failures (recurrence of CDI) at a CDI recurrence visit, were contacted via telephone or email to participate in the interview.
Interviews were conducted virtually (telephone or web-based conference) with the patient within 14 days of attending the respective visit (see the Study Flow Diagram in Figure 1). The 60-minute interviews were conducted by trained interviewers (M.M.-M. and C.U.-F.) using a semi-structured discussion guide. The first part of the discussion entailed open-ended questions designed to understand the patients’ prior experiences with rCDI, focusing on symptoms, symptom severity, and impacts. In the second part, patients were asked about their experiences after receiving treatment, including probes about symptom change and impact change. Individuals who completed the 8-week period were asked about the most meaningful or important benefit of not having another CDI episode. Those who experienced a recurrence (treatment failure) were asked what changes they would have liked to see. The final part of the interview explored how patients’ well-being might be impacted longer term, given the potential for recurrence of CDI.

Study flow diagram.
Analysis of interviews
Interviews were audio-recorded and transcribed verbatim by a third party, and patients’ responses were analyzed using thematic analysis. Thematic analysis is widely used in the health sciences and is a well-accepted, valid approach for interpreting qualitative data, thus was considered the most suitable for this study. 15 Two experienced coders (M.M.-M. and C.U.-F.) coded the transcripts using NVivo 14 software (Lumivero, Denver, CO, USA); coding was performed on a rolling basis, as interviews were completed. The first three transcripts were analyzed independently by each coder, and the results were compared by the study lead, who reviewed and discussed any areas of inconsistency with the team to reach consensus. Improvements were made to the codebook to ensure coder agreement. The reporting of this qualitative study adheres to the Consolidated Criteria for Reporting Qualitative Research (COREQ; Supplemental Material). 16
Ethics
All study procedures were in accordance with the ethical standards of the 1964 Helsinki Declaration and its later amendments, relevant laws, institutional guidelines, and approved by an institutional review board (Advarra IRB on November 9, 2022).
Results
Participant characteristics
Out of the 41 trial participants, 32 (78%) consented to participate in an interview and 9 (22%) did not. Eight of nine did not give a reason for declining interview participation. One participant said that at the time of consenting for the interview, they felt so ill, they did not wish to commit to an interview at a later date. The site followed up with this participant at a later timepoint in the 8-week period, but they still declined interview participation. Of the 32 patients who had consented to the patient impact interviews, two of these participants were lost to follow-up because the interviews could not be scheduled in a timely manner, despite efforts from the research team.
Thus, 30 of the 41 (73%) study patients ultimately participated in the patient experience interviews (Table 1). Average interview duration, excluding time used to resolve any technical issues and to introduce the study, was 38.3 minute (range: 11–57 minute). The interview sample was primarily female (26/30; 87%), non-Hispanic (29/30; 97%), and White (30/30; 100%); similarly, the trial sample was also primarily female (36/41; 88%), non-Hispanic (40/41; 98%), and White (39/41; 95%). Compared to the trial sample, which included participants aged 21 to 85 years, with a mean age of 61.2 years, the interview participants were slightly younger (ranging from 22 to 86 years, with a mean age of 59.1 years). On average, interview and trial participants had been diagnosed with a mean of 2.9 and 3.2 CDI episodes in their lifetimes, respectively. None of the 30 interview participants (0%) reported a CDI recurrence at the time of the interview. However, according to the clinical trial criteria for success, one participant had an indeterminate outcome, that is, neither the protocol-specified definition for treatment success nor treatment failure was met, as the participant did not complete the 8-week visit.
Demographic and clinical characteristics.
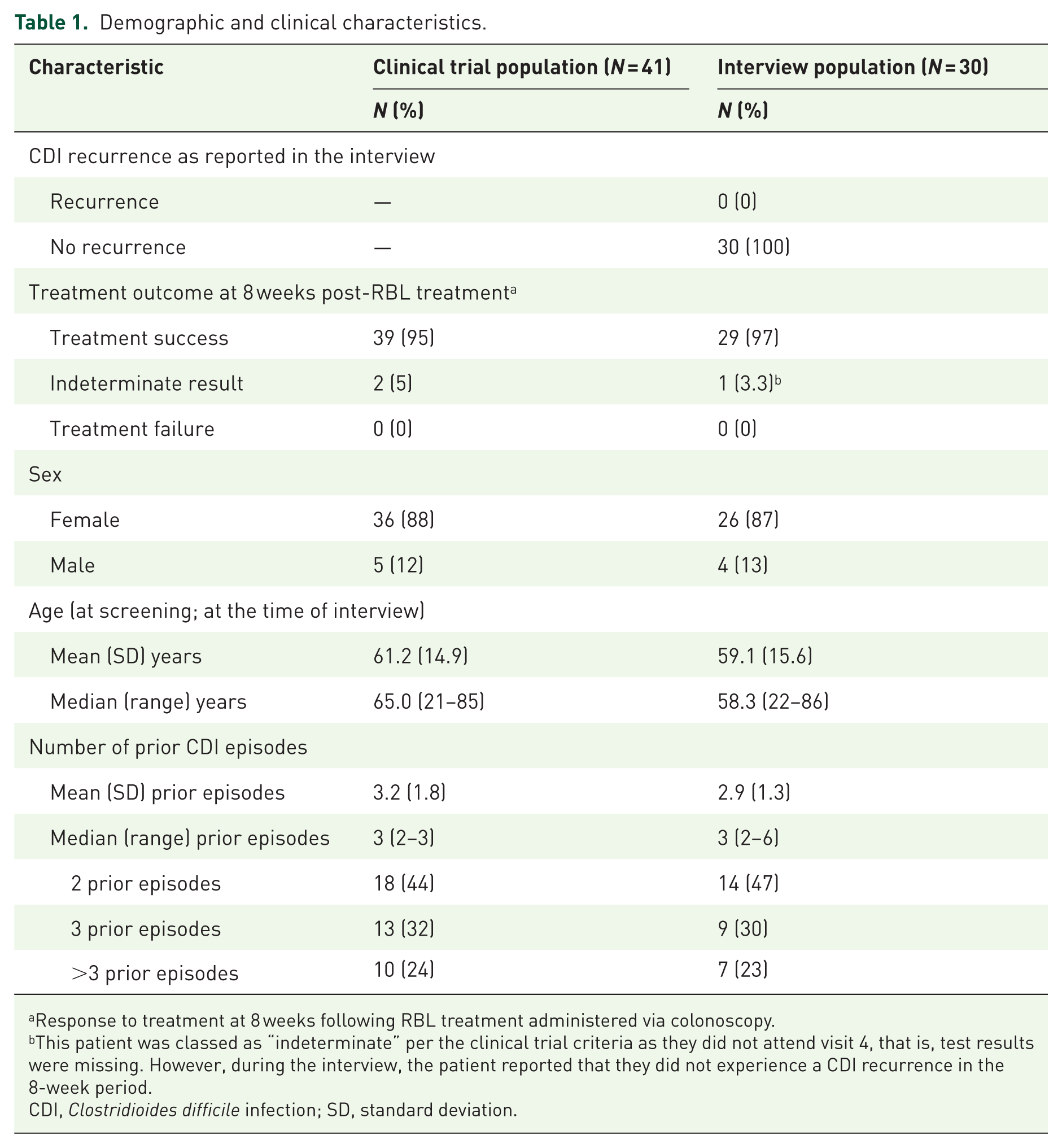
Response to treatment at 8 weeks following RBL treatment administered via colonoscopy.
This patient was classed as “indeterminate” per the clinical trial criteria as they did not attend visit 4, that is, test results were missing. However, during the interview, the patient reported that they did not experience a CDI recurrence in the 8-week period.
CDI, Clostridioides difficile infection; SD, standard deviation.
rCDI symptom and impacts prior to RBL treatment
All participants in the interview sample had experienced multiple episodes and/or bouts of C. difficile before entering the clinical trial. In the interview, participants were asked to describe their CDI symptoms and impacts in the episode prior to receiving RBL treatment; however, some participants spoke collectively about previous CDI episodes and bouts.
Pretreatment rCDI symptoms
The most commonly reported CDI symptom was diarrhea/loose stool (30/30; 100%), followed by urgency/accidents (21/30; 70%), and abdominal pain/cramping (21/30; 70%). An overview of the five most frequently reported CDI symptoms reported by participants is presented in Figure 2.

Top 5 rCDI symptoms reported by participants prior to RBL treatment (N = 30).a
Participants described the diarrhea as “violent,” “severe,” and that they could not “get off the toilet.” Participants were asked to rate the severity of the diarrhea/loose stool prior to RBL treatment. Of the n = 30 who experienced diarrhea/loose stool, n = 29 provided a rating (1 did not understand the question). On a severity scale of 0–10, where 0 indicates no symptoms and 10 indicates the worst possible symptoms, participants rated their diarrhea at 8.9 on average prior to treatment (range: 6–10).
In relation to feelings of urgency and experiencing accidents, participants described having to “run into the bathroom,” that they “couldn’t make it” to a toilet on time, and a lack of bowel “control.” “Urgency and accidents” emerged as a separate symptom concept during the coding process; thus, severity ratings were not captured during the interviews for this concept.
Participants described the abdominal pain as “constant,” “stabbing,” and “scary.” Of the n = 21 who experienced this symptom, n = 18 provided a severity rating (1 did not understand the question, 2 responses were missing). On a severity scale of 0–10 (none to worst), participants rated their abdominal pain and cramping at 7.5 on average (range: 5–10).
About half of the participants reported experiencing fatigue and/or weakness (14/30; 47%). They described feeling “weak,” “exhausted,” and “run down.” Of the n = 14 who experienced this symptom, n = 7 provided a rating (seven responses were missing). On a severity scale of 0–10 (none to worst), participants rated their fatigue and weakness at 8.1 on average (range: 6–10).
Participants (8/30; 27%) reported experiencing nausea and/or vomiting; these participants also reported experiencing dehydration, weight loss, and malnutrition due to nausea/vomiting. Of the n = 8 who experienced this symptom, n = 5 provided a rating (three responses were missing). On a severity scale of 0–10 (none to worst), participants rated their nausea and vomiting at 7.0 on average (range: 4–10).
Most bothersome symptoms
Participants were asked to identify the most bothersome symptom. Some participants could not specify only one symptom, thus more than one response per participant was possible. Diarrhea was considered the most bothersome of all symptoms (Table 2).
Most bothersome rCDI symptom experienced (N = 30).

More than one response could be given.
rCDI, recurrent Clostridioides difficile infection.
Pretreatment rCDI impacts
Participants were asked to describe the impacts of rCDI on their lives prior to receiving treatment. Impact concepts which were probed included: physical, emotional, cognitive, daily activities, social activities, relationships, work/finances, and sleep. All participants described only negative impacts of rCDI; no positive impacts were mentioned. Participants described their experience with rCDI as “a literal living nightmare,” “all-consuming,” and “pure hell.” rCDI ruined their lives for months at a time, “derailing any normal existence,” and making it “impossible to function or do anything.” Table 3 provides an overview of rCDI impact concepts and subcodes.
Impacts of rCDI reported by participants prior to treatment (N = 30).

More than one response could be given.
rCDI, recurrent Clostridioides difficile infection; UTI, urinary tract infection.
All participants reported that they experienced impacts to activities of daily living (ADL) (30/30; 100%). Eighty percent of participants (24/30) said that their sleep was impacted, and they described “waking up to run to the restroom” and difficulty going back to sleep. Many participants (19/30; 63%) described how rCDI symptoms impacted their eating since they described not having much of an appetite for food and/or avoiding food entirely. Participants said rCDI impacted their lives in that they constantly needed to be in close proximity to toilets and/or be able to map the distance to a toilet at any given time (21/30; 70%), to the point that several participants said that they could not leave their house (14/30; 47%). Three out of five participants (18/30; 60%) said that rCDI symptoms impacted their work and/or finances. They spoke about missing workdays, being unable to complete tasks such as meetings, client visits and burdening other coworkers, and a few mentioned the additional cost of medical care related to rCDI: laboratory tests, emergency room visits, antibiotics and being prescribed unaffordable treatment as shared by one participant, “a new medication they were trying to get me on and it was like $6,000, my insurance wouldn’t cover it.” Participants described how rCDI limited their ability to travel and to participate in hobbies and outdoor activities, due to lack of energy, weakness, or feeling poorly.
Social life was negatively impacted by rCDI. Many (19/29; 66%) stopped or reduced socializing with others when they had rCDI. Others (9/29; 31%) missed important family events (weddings, funerals, vacations) due to rCDI. Almost half of the participants expressed that their relationships with partners and family members were impacted due to this infection (13/29; 45%). The social/relational impacts and the emotional impacts were frequently mentioned together (n = 11).
Nearly all participants (28/30; 93%) reported emotional/psychological impacts from their experience with rCDI. They experienced anxiety, worry, and stress (14/28; 50%); for some (8/28; 29%), the negative emotions reached a level of depression. They felt embarrassed (7/28; 25%) about having the illness and did not want others to know about it or know the extent of it.
Eighty percent of participants (24/30) reported physical or bodily impacts due to rCDI, including low energy, exhaustion, and feeling miserable. Others described bodily impacts in relation to their diet/eating, such as malnutrition, dehydration, unwanted weight loss, hair loss, bruising, and brittle nails.
Finally, rCDI resulted in cognitive impacts for some participants (14/30; 47%). They described the impact of being unable to plan their day due to the unpredictability of the infection/symptoms and being unable to think about future plans (10/14; 71%).
Most bothersome impact
Participants were asked to identify the most bothersome impact. Some participants could not specify a single impact, as some impacts intersected (e.g., physical and psychological). Thus, more than one response per participant was possible. The “inability to function” was considered the most bothersome impact, followed by the psychological impact or burden of rCDI, and the social and relational impacts (Table 4).
Most bothersome rCDI impact experienced (N = 30).

More than one response could be given.
rCDI, recurrent Clostridioides difficile infection.
Improvements in function and HRQoL after RBL treatment
Experiences with colonoscopy procedure
Participants were asked to describe their experience after receiving RBL treatment via colonoscopy. More than half of participants (19/30; 63%) reported they “felt better,” 9 (30%) participants reported they were still experiencing symptoms, and 2 (1%) could not recall the experience shortly after the procedure. Nine of 30 (30%) participants spontaneously shared their experiences with the RBL treatment administration via colonoscopy. Eight of 9 (89%) participants reported a positive experience, with one participant noting that “having this done by a colonoscopy, to me was really important, because after having C. difficile for a while, I wanted to make sure everything was okay [with their large intestine and rectum].” However, 2 of the 8 reported experiencing fatigue due to preparations required for the colonoscopy, though one of these participants stated: “I feel very strongly about the procedure and, um, I feel very positive about the results.” One participant reported that they had experienced two prior colonoscopies and that the administration for the RBL treatment was more “intense”; further descriptions were not provided.
Post-treatment symptom change
Participants were asked to describe how their CDI symptoms had changed since receiving RBL treatment. Interviewers probed on symptoms previously mentioned by the participant but also allowed for spontaneous reports of changes in any other symptoms. Overall, participants described improvements in diarrhea, urgency, abdominal pain, fatigue/weakness, and nausea/vomiting (Figure 3).

Changes after RBL treatment for the most commonly reported rCDI symptoms (N = 30).
All participants (30/30; 100%) reported that their diarrhea symptoms had resolved completely (17/30; 57%) or were less severe (13/30; 43%). None reported that symptoms had worsened or remained the same as before. Participant-reported symptom severity ratings decreased sharply from 8.9 (SD ±1.3) on average (29/30 reporting) to 1.3 (SD ±1.8) on average (23/30 reporting). The median for posttreatment severity ratings was 0 (no symptoms); prior to treatment, the median was 10 (worst possible symptoms).
“Urgency and accidents” improved for many participants. Change direction and severity score were not routinely captured for this symptom concept in the interviews, as it emerged as a new concept during the interview process. Still, of those who experienced urgency/accidents prior to treatment, 4/21 (19%) spontaneously stated that it was resolved, and 5/21 (24%) said that it was less severe. The remaining 12/21 (57%) had all experienced an improvement in diarrhea symptoms and did not spontaneously mention feelings of urgency or accidents during the discussion about improvements. However, improvement in this area was alluded to in various ways in the interviews, for example, “I haven’t needed to go,” “regular bowel movements on a normal basis,” and “I don’t have to sleep on waterproof sheets anymore.”
Overall, 21 participants reported improvements in abdominal pain and cramping either as being less severe (13/21; 62%) or completely resolved (8/21; 38%). None reported that symptoms had worsened or remained the same as before. As with diarrhea symptoms, abdominal pain symptom severity ratings decreased sharply from 7.5 (SD ±1.5) on average (17/21 reporting) to 1.6 (SD ±1.6) on average (14/21 reporting).
Similar trends were reported for fatigue/weakness and nausea/vomiting. For fatigue/weakness, 4/14 (29%) stated that it was resolved, and 9/14 (64%) said that it was less severe. The remaining 1/14 (7%) did not spontaneously mention any feelings of fatigue and weakness after treatment. Severity scores dropped from 8.1 (SD ±1.3) on average before (7/14 reporting) to 1.7 (SD ±1.2) on average after (6/14 reporting). For nausea/vomiting, 4/8 (50%) stated that it was resolved. The remaining 4/8 (50%) did not spontaneously mention any feelings of nausea or vomiting after treatment; however, all of these (4/4; 100%) had reported overall improvement in symptoms. Participant-reported symptom severity ratings decreased from 7.0 (SD ±2.0) on average (6/8 reporting) to 0 (SD ±0) on average (4/8 reporting).
Time till symptom improvement
Participants were asked how soon after RBL administration they experienced improvement in some or all of their symptoms. They generally reported a rapid improvement in symptoms, with 77% (23/30) seeing an improvement within 1 month of treatment; 14/30 (47%) reported symptom improvement of some or all of their rCDI symptoms within 1 week of treatment, 3/30 (10%) within 1–2 weeks, and 6/30 (20%) within 3–4 weeks.
Of the remaining seven participants, 4 (57%) did not report a specific time frame for symptom improvement but indicated general resolution of rCDI symptoms by the time of the interview; two reported initial improvement within 1 week followed by fluctuating symptoms, and one reported persistent diarrhea for 3 weeks posttreatment with subsequent symptom attenuation.
Post-treatment impact change
Participants described how the treatment had impacted their lives. The majority (25/30; 83%) spontaneously described an improvement in their overall quality of life. They said that they finally had their normal lives back: “It just, it gives me a new life (laughs) . . . a brand new life. That’s what it gives me, the freedom and choice to do things and travel again and go places and. . . it’s marvelous!” Some called the treatment “life changing” and “a miracle”: “I feel really good. It has not come back. I’m, it literally, I think it, I mean, I, I, I know this sounds super dramatic and I never thought I’d say this ever, but I really think it saved my life.” Five participants (5/30; 17%) had experienced some improvement in a few impact areas, but, at the time of the interview, they were still hoping for more change to occur: “Every week it’s a little better, so hopefully I’m . . . I still have hopes that this is gonna get back to normal.” Figure 4 illustrates the changes to HRQoL impacts.

Changes to HRQoL impacts after RBL treatment (N = 30).a
All 30 participants (100%) described improvements in their daily activities (on average: 3.6 improved ADLs per participant). They could resume their leisure activities, travel, and hobbies (19/30; 63%). They described improvements in sleep (15/30; 50%), work/finances (15/30; 50%), household activities and cleaning (13/30; 43%), as well as diet/eating (9/30; 30%). Because the feelings of urgency had resolved, they were able to leave the house without needing to remain constantly near a toilet (15/30; 50%). A small number of participants noted that some impact areas were not yet improved or back to normal: Four of 30 participants (13%) noted some impact areas that had not improved yet or were not back to normal. These were related to impacts on leisure time, cleaning/sanitizing, work/finances, diet/eating, and sleeping. One participant reported poor diet/eating, and one reported obsessive cleaning.
Overall, 29/30 (97%) reported improvements in their social life. They were able to spend time with their friends and families once again (23/30; 77%). They no longer needed to isolate themselves in their homes or stay away from others. Participants shared that they had resumed socializing and going out with others (19/30; 63%). One participant mentioned that they had not yet resumed spending time with family fully, as their family members were still concerned about contagion. Another noted that they did not feel comfortable yet in big crowds, and one had difficulty answering the question.
Mental health and well-being improved for 23/30 participants (77%) after RBL treatment. Participants described themselves as happier, free, relieved, and having a more positive outlook. Despite this, anxiety and stress were still present for some individuals (10/30; 33%), while others reported improvement in this impact area (4/30; 13%). Anxiety and stress were most often related to concerns of recurrence of symptoms or rCDI. Other changes to emotional impacts, which were less frequently mentioned, included: fear of contagion (n = 4), posttraumatic stress syndrome or PTSD (n = 3), isolation (n = 2), and discouragement (n = 1).
One-third of participants (10/30; 33%) noted an improvement in cognitive impacts. They were able to plan again (n = 10). They no longer found themselves needing to cancel meetings or trips, now that their symptoms had resolved. They felt more reliable when it came to planning—both in their work life and social life. One participant mentioned having greater mental capacity (fewer consuming thoughts about rCDI), and while another stated that they felt able to focus again. One person still felt that they were “unreliable” when it came to planning meetings or outings, as they did not yet feel fully recovered.
Most important (meaningful) change in functional ability and HRQoL
Of all the changes that participants experienced, they were asked to name one that was the most meaningful to them. Generally, participants found that the functional improvements (ability to participate in life again and feel well again) were the most important to them (23/30; 77%). Table 5 provides an overview of the most important (meaningful) changes after RBL treatment reported by participants. Of note, one participant had experienced some improvements in symptoms; however, throughout the interview, they described some impacts that had not improved (no change), some that had worsened, and some new (negative) impacts.
Most important (meaningful) change after RBL treatment (N = 30).

N/A, not applicable; RBL, fecal microbiota, live-jslm.
Long-term impact of CDI on well-being
Participants primarily shared positive thoughts about how an rCDI-free life would positively impact their emotional/psychological well-being (87%). Others, however, shared their thoughts and concerns about the future, focusing primarily on a fear of recurrence. More than half of the participants expressed an ongoing fear of recurrence (18/30; 60%). They also emphasized the role of episode-free time (i.e., in weeks, months, etc.) in building their confidence in their complete healing.
Discussion
These 30 patient experience interviews were embedded in Ferring’s CDI-SCOPE phase IIIb clinical trial of RBL, a microbiota-based therapy. The interviews highlighted the cumulative burden of rCDI, as participants described persistent symptoms that disrupted their ability to maintain work, relationships, and routines. Diarrhea was the most commonly experienced and the most bothersome symptom. However, there were other key symptoms such as abdominal pain, cramping, fatigue, weakness, urgency, nausea, and vomiting, which align with symptoms reported in other CDI research.17,18 Together, these symptoms contributed to diminished quality of life.
The interviews were conducted approximately 8 weeks after administration of RBL via colonoscopy. At this timepoint, none of these 30 interviewed participants experienced a recurrence. All participants reported either a resolution or reduction in diarrhea severity, with a few describing some other less severe but lingering symptoms. This corresponds with other patient experience data showing that while FMT immediately resolves diarrhea, other symptoms may take several months and up to 1 year to fully resolve. 19
Nonetheless, RBL treatment represented an important turning point for many participants. Relief from diarrhea allowed patients to reclaim basic activities and social participation. They were no longer bound to bathrooms and their homes, avoiding other people and their daily life. They could participate once again, which was the most important benefit for the majority of the participants interviewed. Further, these improvements had a profoundly positive impact on the emotional well-being of participants.
This is consistent with other research that has demonstrated significant improvements in the severity of depression and anxiety symptoms with FMT. 20 However, the experience of having had rCDI has residual emotional effects on individuals, such as fears of recurrence,19,21 as illustrated by the experiences shared in these interviews. While rCDI causes multiple physical ailments, participants clearly described that it also causes far-reaching emotional impacts, as one person reflected: “C. diff doesn’t affect your brain, but (laughs) that’s the biggest [impact].”
Taken together, these qualitative findings suggest that an effective treatment for rCDI would not only lead to improved symptoms and better functioning and HRQoL in the short term, but it could also have a positive and lasting impact on emotional health and well-being for those recovering from rCDI.
Strengths and limitations
First, the trial in which these interviews were embedded was a single-arm trial, and therefore there was no comparator group, for example, placebo, standard-of-care antibiotics only, or administration by enema. These exploratory findings on the HRQoL benefits of colonoscopic administration of RBL provide useful insights for future research but would be strengthened by additional qualitative studies including a comparator group.
Although the experience of colonoscopy preparation or patient preferences for alternative administration methods were not a primary focus of this qualitative research, nine patients spontaneously shared their experiences regarding the procedure with most (~90%) describing it as a positive experience. Also, all physicians who administered RBL by colonoscopy in the trial assessed participant benefit as “much” or “very much” improved. 14 Past research related to patient perception of FMT in ulcerative colitis found that 77% of patients interviewed reported FMT delivery by single sedated colonoscopy as their preferred method of administration. 22 This preference may be due to the potential benefit from the efficiency of having only one procedure for administration rather than requiring multiple visits for management. 14
Additionally, people who have been diagnosed with rCDI report significant barriers to FMT access and provider unawareness as a treatment option. 19
Administration of RBL via rectal and colonoscopy expands options for approved modes of FMT administration. Still, colonoscopies are associated with pain, discomfort, and embarrassment; they require bowel preparation, and they may be costly. 23 Future qualitative research should include direct questions about procedural burden and patient preference regarding FMT administration methods (e.g., via colonoscopy, enema, or other methods) to better understand the overall patient experience and impact.
The research team aimed to recruit 100% of trial participants for the optional interviews. The majority of trial participants consented to the interviews (32/41; 78%), and nearly all of those who consented participated in the interview (30/32; 83%). Compared to other published in-trial interview studies, the current study captured a high proportion of the clinical trial population. 24 Although the qualitative interview sample was small, the characteristics of this sample (female: 87%, mean age: 59.1 (15.6)) and average number of prior CDI episodes 2.9 (1.3) mirrored the full trial population (N = 41), which was primarily female (88%), had a mean (SD) age of 61.2 (14.9) years and the mean (SD) of 3.2 (1.8) previous CDI episodes. The population characteristics also resemble populations from rCDI systematic reviews, which show predominantly female patients and an average of 3.7 prior CDI episodes. 21
The interview sample did not include any patients who experienced a recurrence during the 8-week follow-up period, which is a limitation; it would be important to capture information from these individuals to understand all perspectives after treatment with RBL.
Finally, participants expressed the importance of time, particularly with respect to the emotional impacts of rCDI. As these interviews were conducted within 8–10 weeks of RBL treatment, reflecting a key timepoint for follow-up within the CDI-SCOPE trial, any long-term benefits of treatment would not yet be captured. It could be insightful to collect additional qualitative data at a later timepoint to capture any potential improvement in emotional impacts.
Conclusion
Qualitative interviews with individuals diagnosed with rCDI suggest that RBL administered via colonoscopy can provide rapid, meaningful improvements in symptoms and daily functioning. Participants reported resolution or significant reduction of diarrhea, improved physical health, renewed ability to engage in social and professional activities, and enhanced emotional well-being. Importantly, participants described not only symptomatic relief but a restoration of everyday life—a return to routines, relationships, and opportunities that had been disrupted by rCDI. These findings underscore that the impact of RBL extends beyond clinical endpoints, supporting functional and emotional recovery critical to long-term well-being.
By capturing the patient voice, this study highlights the broader value of microbiota restoration and supports the use of patient-centered outcomes in evaluating treatments for rCDI.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848251392102 – Supplemental material for Patient-reported benefits of colonoscopicallyadministered microbiota restoration therapy: a qualitative study of adult patients with recurrent Clostridioides difficile infection
Supplemental material, sj-pdf-1-tag-10.1177_17562848251392102 for Patient-reported benefits of colonoscopicallyadministered microbiota restoration therapy: a qualitative study of adult patients with recurrent Clostridioides difficile infection by Megan McCool-Myers, Cindy Umanzor-Figueroa, Tara Symonds, Margarita Landeira, Tahany Awad, Daniel Armandi, Beth Guthmueller and Sahil Khanna in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
