Abstract
Background:
Ulcerative colitis (UC) is associated with an increased risk of venous thromboembolism (VTE) and cardiovascular (CV) events, particularly during flares. While concerns have emerged regarding the intrinsic CV and thromboembolic risk of Janus kinase inhibitors, real-world data on baseline risk profiles in UC remain scarce.
Objectives:
This study aimed to assess the thromboembolic and cardiovascular risk profiles of UC outpatients initiating advanced therapies and to evaluate the incidence of related clinical events.
Design:
We conducted a cross-sectional study with prospective longitudinal follow-up at a single tertiary center.
Methods:
Consecutive UC outpatients who initiated an advanced therapy between June 2020 and December 2023 were enrolled. Baseline VTE and CV risk factors were assessed using medical records, a structured online questionnaire, and the International Physical Activity Questionnaire. CV risk was estimated using the Atherosclerotic Cardiovascular Disease and Systematic Coronary Risk Estimation 2 calculators. Patients were monitored for VTE and CV events until December 2024.
Results:
The study included 300 patients (median age 44 years; 45.3% female). Most had 0–1 VTE risk factor (61.0%) and elevated C-reactive protein was the most common (45.0%). CV risk stratification showed that most non-elderly patients had low or moderate risk, while elderly patients showed higher risk. During a median follow-up of 27 months (683 person-years), only four events (1.3%, incidence rate 0.59 per 100 P-Y) were recorded: two VTE (both in patients on ustekinumab, one with multiple risk factors and one with cirrhosis) and two CV events (angina and retinal ischemia in low-risk patients on vedolizumab and adalimumab).
Conclusion:
In our cohort, both thromboembolic and CV risks were overall low. CV risk was higher in elderly patients. The incidence of VTE and CV events during follow-up was low. These findings suggest that concerns regarding the intrinsic VTE and CV risks associated with Janus kinase inhibitors may not fully apply to UC patients.
Plain language summary
Ulcerative colitis, a chronic inflammatory bowel disease, is associated with an increased risk of blood clots and cardiovascular events, particularly during active disease phases. Certain UC treatments, such as JAK inhibitors, have raised concerns due data supporting an increased risk in patients with rheumatoid arthritis. In this study, we investigated 300 UC patients initiating advanced therapies, including JAK inhibitors, with a median follow-up of two years, and assessed baseline risk factors for thromboembolic and cardiovascular events and calculated long-term risk scores. Our findings indicate that most patients were young with few risk factors, aligning with the 4 events that occurred. These results suggest that UC patients generally have a low risk of thromboembolic and cardiovascular events, and that the risks associated with specific treatments may not be equally generalized to all immune-mediated inflammatory diseases. Effective inflammation control and promoting healthy lifestyle choices may be key strategies for mitigating future risk.
Introduction
Ulcerative colitis (UC) is one of the main forms of inflammatory bowel disease (IBD), characterized by a twofold risk of venous thromboembolic (VTE) events compared to the general population. 1 Key risk factors include active disease, hospitalization, and surgery. Indeed, the European Crohn’s and Colitis Organisation guidelines recommend prophylactic doses of low molecular weight heparin or fondaparinux for IBD patients during hospitalization, major surgery, or severe flare-ups. 2 With regard to cardiovascular (CV) events, several meta-analyses have highlighted a slightly increased risk of ischemic heart disease, mesenteric ischemia, and cerebrovascular accidents,3–6 even though mortality does not differ from that of the general population. 4 This has led to the speculation that chronic systemic inflammation, rather than traditional CV risk factors, can play a crucial role. 7 The impact of advanced therapies on either VTE or CV events remains unclear. A French nationwide cohort study found that IBD patients receiving tumor necrosis factor alpha (TNF-α) antagonists had a reduced risk of acute arterial events, suggesting that uncontrolled inflammation is a modifiable risk factor for CV disease. 8
Following the results of the ORAL Surveillance study 8 on rheumatoid arthritis patients treated with Tofacitinib, concerns have emerged on the intrinsic VTE and CV risks associated with Janus kinase inhibitors (JAK-i). Consequently, regulatory agencies recommend using this class of drugs only when no suitable treatment alternatives are available for patients aged ⩾65 years, active or previous smokers, or those with risk factors for cancer or major adverse CV events. However, the ORAL Surveillance study included patients who were at least 50 years old, had at least one additional CV risk factor, and were mostly (>90%) active or former smokers. Therefore, extrapolating these findings to UC patients may be challenging due to differing baseline risk profiles.9–11 Long-term data on JAK-i in patients with UC have not raised significant safety concerns.12,13 In addition, a recent meta-analysis of 32 randomized control trials including 22,007 patients with UC found no increased risk of CV events during either the induction or maintenance phases of treatment with infliximab, ustekinumab, Tofacitinib, and Upadacitinib, compared to placebo. 14
Currently, real-life data on the prevalence of VTE and CV risk factors in patients with IBD remain limited. In this study, we examined these risk profiles in a large cohort of UC patients eligible for advanced therapies.
Methods
Study design
We conducted a cross-sectional study with a prospective longitudinal follow-up at the Fondazione Policlinico Universitario “A. Gemelli” IRCCS in Rome, Italy. The study included all consecutive adult outpatients with a confirmed diagnosis of UC for at least 3 months, who initiated an advanced therapy (as a first or second/third line) between June 2020 and December 2023 at our outpatient clinic. Patients with Crohn’s disease, indeterminate colitis (IBD, unclassified), or with an ileal pouch were excluded (Figure 1). Our hospital serves the general IBD population within the Italian National Health System, providing free access to care, diagnostics, and advanced therapies. Therefore, the study cohort represents an unselected, real-world population.

Study flow diagram.
At baseline, a cross-sectional evaluation was performed, collecting clinical data at the time of therapy initiation, using both medical records and an online questionnaire to ensure a comprehensive assessment of VTE and CV risk factors. In addition, the International Physical Activity Questionnaire (IPAQ) 15 was administered to evaluate patients’ physical activity levels. Afterward, a longitudinal evaluation was performed to investigate the occurrence of any VTE or CV events after therapy initiation until December 2024. Routine follow-up visits occurred every 2–4 months, and patients were instructed to notify the Center by email or telephone if they experienced any adverse events.
This study was conducted in accordance with the Declaration of Helsinki and was approved by the Local Ethics Committee (no. 2403) on March 28, 2024, with the need for written informed consent waived.
The STROBE (The Strengthening the Reporting of Observational Studies in Epidemiology) Statement guidelines were consulted during the manuscript preparation 16 (Supplemental Material).
Venous thromboembolism risk assessment
Currently, no validated thromboembolic risk scores exist for the IBD outpatient population. To address this limitation, we considered all variables recognized as VTE risk factors in both IBD17,18 and non-IBD patients,19,20 incorporating elements from risk assessment models validated in other clinical settings, such as the Caprini risk assessment score for surgical patients 21 and the IMPROVE Risk Score for hospitalized patients. 22 Most of these variables, when not available in clinical records, were collected through an online questionnaire as previously stated.
Baseline demographic and phenotyping data were collected, including sex, age, smoking status, ethnicity, disease duration, and body mass index (BMI). BMI was categorized according to the World Health Organization criteria: underweight (<18.5 kg/m²), normal weight (18.5–24.9 kg/m²), overweight (25–29.9 kg/m²), and obese (⩾30 kg/m²).
We explored the presence of varicose veins or prior surgical interventions for varicose veins, personal or family history of VTE, both deep venous thrombosis (DVT) and pulmonary embolism, known genetic thrombophilia, such as Factor V Leiden and prothrombin G20210A mutation, homozygous methylenetetrahydrofolate reductase gene mutation, deficiencies of antithrombin III, protein C and protein S; familial dysfibrinogenemia and antiphospholipid antibody syndrome, and other hematological and systemic conditions, such as polycythemia vera, essential thrombocythemia, idiopathic myelofibrosis, and sickle cell anemia.
Hospitalization and procedural risks were defined as recent hospital admissions within the past 6 months and major surgeries performed within the last 3 months, including fractures involving the hip, pelvis, or legs. Additional factors considered were the use of antipsychotic medications, ongoing treatments with estrogen-based therapies, hormone replacement therapy or tamoxifen, and elevated serum C-reactive protein (CRP) levels (cutoff >5 mg/dL).
Cardiovascular risk assessment
Cardiovascular risk was assessed using two validated tools: the ASCVD Risk Estimator Plus (atherosclerotic cardiovascular disease, ASCVD), 23 developed by the American College of Cardiology, and the systematic coronary risk estimation 2 (SCORE2), 24 recommended by the European Society of Cardiology. Both tools estimate the 10-year probability of cardiovascular events, including myocardial infarction and stroke, by integrating demographic and clinical variables. Risk categories were defined according to established ASCVD and SCORE2 thresholds, as detailed in Table S1.
For both scores, only patients with available lipid profiles were included in the analysis. After the assessment using the validated scores, we divided the population into “elderly” (⩾65 years old) and “non-elderly” (<65 years old) and compared the score results. Moreover, comparisons were made by grouping the lower and higher risk categories of the two scores.
Statistical analysis
Continuous variables were expressed as median with interquartile range (IQR), depending on distribution assessed by the Shapiro–Wilk test. Categorical variables were presented as absolute frequencies and percentages. Statistical comparisons between groups were performed using t test or Mann–Whitney U test for continuous variables, and chi-square test or Fisher’s exact test for categorical variables, as appropriate. A p-value <0.05 was considered statistically significant. We calculated person-years of follow-up and estimated incidence rates per 1000 person-years with exact Poisson 95% confidence intervals (for VTE, CV events, and their composite). We also estimated the number of expected CV events by applying each patient’s 10-year risk (ASCVD and SCORE2) to their observed follow-up, restricted to patients with available lipid profiles; expected VTE events were derived by applying available outpatients’ IBD incidence rates to the cohort’s accrued person-time.25,26 All analyses were conducted with STATA software version 18.5 (StataCorp LLC, located in College Station, Texas, USA).
Results
Study population
Out of 300 patients surveyed, all provided responses via the online questionnaire, achieving a 100% response rate (Figure 1). The median age of the cohort was 44 years (IQR 33–58), with females accounting for 45.3% of the population. The median BMI was 23.4 kg/m² (IQR 21.0–26.5), with 8.3% of patients classified as obese. Most patients were Caucasian (n = 293, 97.7%). Active smoking was reported by 60 patients (20.0%). The median disease duration was 7 years (IQR 2.9–16.0). Regarding advanced therapies, 122 patients (40.7%) received anti-TNF agents, 61 (20.3%) Ustekinumab, 51 (17.0%) Vedolizumab, and 66 (22.0%) JAK-i, most of whom were treated with Tofacitinib. Overall, 117 patients (39.0%) were naïve to advanced therapies.
Concerning physical activity, 139 patients (46.3%) were classified as inactive, 91 (30.3%) as sufficiently active, and 70 (23.3%) as active or very active.
Baseline patients’ characteristics are summarized in Table 1.
Baseline demographics and clinical characteristics of the study population.
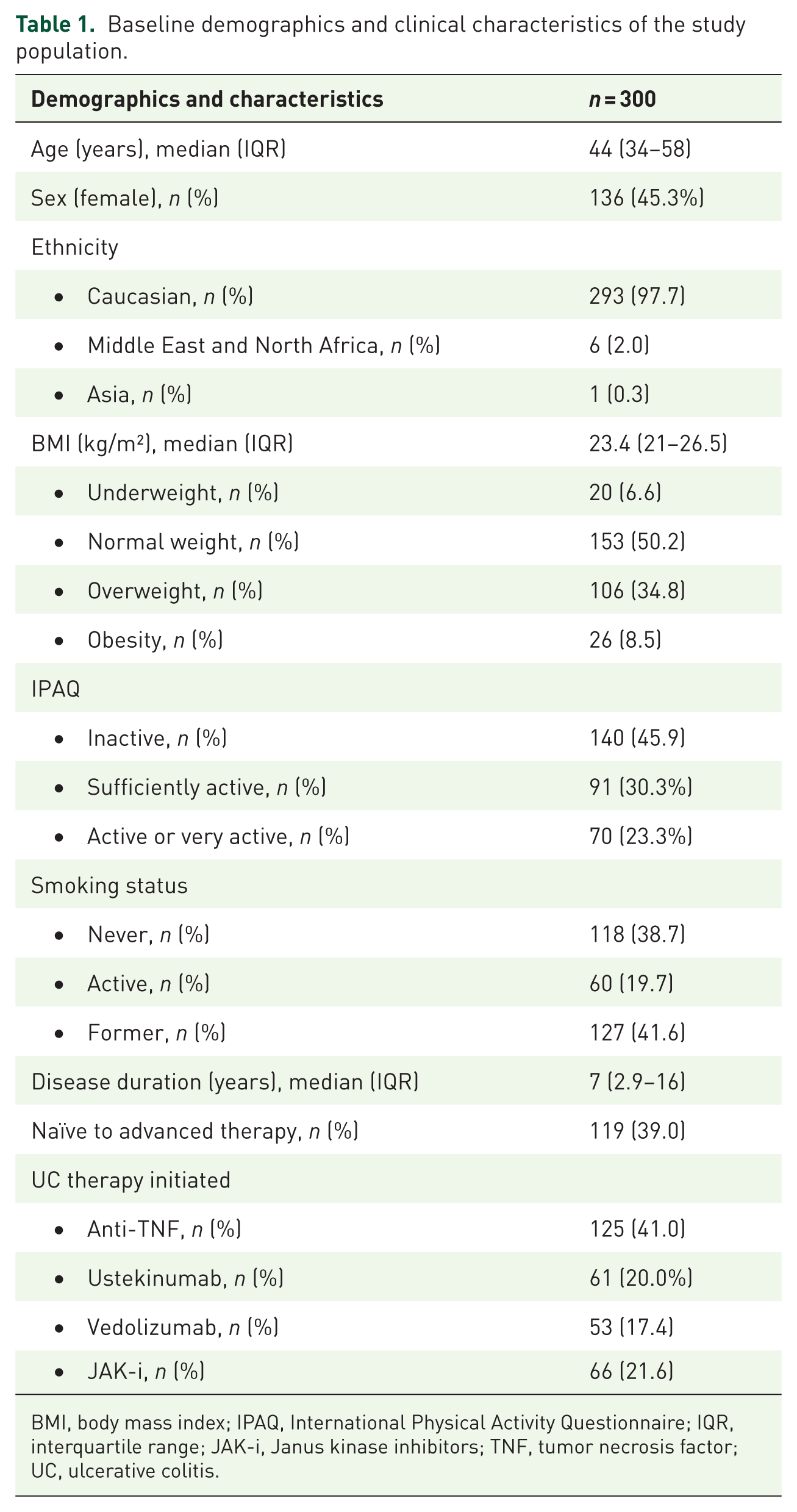
BMI, body mass index; IPAQ, International Physical Activity Questionnaire; IQR, interquartile range; JAK-i, Janus kinase inhibitors; TNF, tumor necrosis factor; UC, ulcerative colitis.
Thromboembolic risk factors
The most common VTE risk factor was elevated CRP (45.0%), followed by recent hospitalization (18.7%) and family history of VTE (14.7%). The frequencies of individual VTE risk factors are detailed in Table 2.
Thromboembolic and cardiovascular risk factors in the study cohort.

VTE, venous thromboembolism; TIA, Transient Ischemic Attack.
A total of 183 patients (61.0%) had 0–1 risk factor, while 71 (23.7%) had 2, and 33 (11.0%) had 3. Only a small proportion of patients (8%) presented with 4 or more risk factors, particularly 8 patients (2.7%) with 4 and 5 patients (1.7%) with 5 or 6. The overall distribution of considered VTE risk factors among patients is shown in Figure 2.

Distribution of VTE risk factors per patient in the study cohort.
Cardiovascular risk scores
The frequencies of individual CV risk factors are shown in Table 2. The majority of patients (63.3%) had 0 or 1 cardiovascular risk factor, 20.0% had 2, and 11.0% had 3. Only a small proportion had 4 (3.3%) or 5 or more (2.3%) risk factors. The distribution of cardiovascular risk factors per patient is illustrated in Figure 3. Cholesterol levels were available for 170 patients, but 29 had LDL values below 70 mg/dL, precluding the calculation of the ASCVD score. Patients with available lipid data had a significantly higher median age (49 years, IQR 40–60) compared to the overall study population (44 years, IQR 33–58, p < 0.001) and even more so compared to those without lipid data (37 years, IQR 27–51, p < 0.001). Accordingly, the ASCVD risk score was calculated only for 151 patients, with a median age of 49 years (IQR 40–60). Of these patients, 103 (67.3%) were classified as low risk, 10 (6.5%) as borderline risk, 32 (20.9%) as intermediate risk, and 8 (5.2%) as high risk. Among non-elderly patients, 102/129 patients (79.1%) were categorized as low risk, 8 (6.2%) as borderline risk, 16/129 (12.4%) as intermediate risk, and 3/129 (2.3%) as high risk. Conversely, in the elderly group, only 1/24 patient (4.2%) was classified as low risk, while 16/24 (66.7%) were at intermediate risk and 5/24 (20.8%) were at high risk. These results are summarized in Table 3.

Distribution of the number of known CV risk factors per patient in the study cohort.
ASCVD risk stratification by the elderly (⩾65 years) and non-elderly (<65 years).
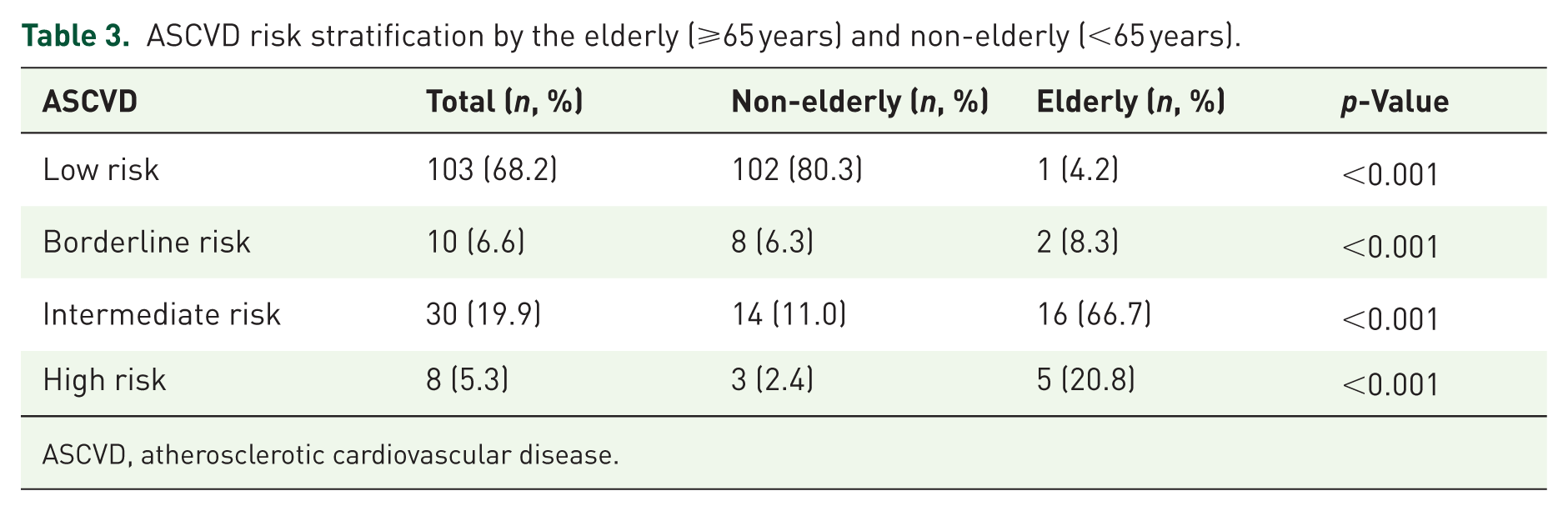
ASCVD, atherosclerotic cardiovascular disease.
Grouping lowest (low and borderline) versus highest risk categories (intermediate and high), 87.5% of elderly patients were classified in the high groups (p < 0.001), as opposed to 86.7% of non-elderly patients in the lowest groups (p < 0.001).
Compared to the lowest groups, patients in the intermediate/high-risk group had a significantly higher BMI (median 25.2 kg/m2, IQR 23.8–27.6 vs 23 kg/m2, IQR 20.6–25.8, p < 0.001) and were more likely to be men (81.6% vs 18.4%, p < 0.001).
With regard to SCORE2 analysis, 92/170 patients (54.1%) were classified as low risk, 50/170 (29.4%) as moderate risk, 18/170 (10.6%) as high risk, and 10/170 (5.9%) as very high risk. Stratification by age showed that in the non-elderly group (n = 143), most patients were categorized as low (92/143, 64.3%) or moderate risk (44/143, 30.8%), with smaller proportions classified as high (6/143, 4.2%) or very high risk (1/143, 0.7%). By contrast, in the elderly group (n = 27), the majority were classified as high (12/27, 44.4%) or very high risk (9/27, 33.3%), with no patients in the low-risk category. These results are summarized in Table 4.
SCORE2 risk stratification by elderly (⩾65 years) and non-elderly (<65 years).

SCORE2, systematic coronary risk estimation 2.
Grouping lowest (low and moderate) versus highest risk categories (high and very high), 77.7% of elderly patients were classified in the high groups (p < 0.001), compared to 95.1% of non-elderly patients in the low groups (p < 0.001). Furthermore, patients in the high/very high group were more likely to be male (82.1% vs 17.9%, p = 0.02) and active/former smokers (92.9% vs 7.1%, p < 0.001).
Follow-up
All patients underwent at least three reassessments over a minimum of 1 year, with a median follow-up of 27 months (IQR 19–35), and none were lost to follow-up. Over 683 person-years of follow-up, 2 VTE and 2 CV events occurred, corresponding to incidence rates of 0.29 per 100 person-years (95% CI, 0.04–1.06) for both VTE and CV events; the combined endpoint (VTE or CV events) comprised 4 events, 0.59 per 100 person-years (95% CI, 0.16–1.50). Applying previously reported outpatients’ VTE IBD incidence rates (overall 1.8 per 1000 person-years; 6.4 per 1000 during active disease) to our person-time yielded 1.2 expected events.25,26 For CV events, among patients with available lipid profiles (n = 151 for ASCVD; n = 170 for SCORE2), we calculated the expected events by scaling each individual’s risk to their observed follow-up duration. This yielded 1.78 expected events using the ASCVD score and 1.38 expected events using the SCORE2.
The first DVTs occurred in a 76-year-old female with a total of 6 VTE risk factors and treated with ustekinumab. The second case was a portal vein thrombosis in a cirrhotic 43-year-old male, previously undergone a liver transplant for primary sclerosing cholangitis, also treated with ustekinumab.
Regarding CV events, one was exertional angina in a vedolizumab-treated 54-year-old man with a history of dyslipidemia on alirocumab therapy (a PCSK9 inhibitor), at low CV risk according to the SCORE2. The second event was a retinal ischemia in a 51-year-old female smoker, receiving adalimumab, who was also classified as low risk according to both ASCVD and SCORE2.
Discussion
To the best of our knowledge, our study is the first to evaluate the thromboembolic and CV risk profiles, applying the ASCVD and SCORE2 in a UC outpatients’ population at the time of initiation of an advanced therapy.
Previous studies reported an increased risk of VTE in IBD patients,27–31 particularly during disease flares.31–33 However, quantifying this risk remains challenging due to the heterogeneity of available studies in terms of design, sample size, and settings.
In our study, the prevalence of thromboembolic risk factors was relatively low, with most patients presenting 0 or 1 VTE risk factor.
Due to the lack of validated thromboembolic risk scores specifically designed for IBD outpatients, we could not assign a precise score to everyone. However, to avoid underestimating or overestimating risk, we systematically included all known VTE risk factors, incorporating more variables than conventional risk models such as the Caprini Score 34 or the IMPROVE Risk Score, 22 both validated in different settings. This low prevalence gives good reason for the low incidence of VTE events in our cohort, with only 2 records among 300 patients followed up for a median of 27 months.
We also compared our VTE crude rate with ambulatory IBD cohorts. Over 683.1 person-years, we observed 2 VTE (2.93 per 1000 person-years; 95% CI, 0.40–10.6). Applying ambulatory IBD incidence (1.8 per 1000 person-years; 6.4 per 1000 during active disease) to our person-time suggests 1.2 expected events. Accordingly, our count is slightly above the ambulatory baseline but below active-disease estimates, falling within the IBD reported range. However, these cross-cohort comparisons should be interpreted cautiously due to differences in disease activity, case definitions, prophylaxis practices, and therapy distributions, as well as the imprecision resulting from small event numbers and lack of full age-sex standardization.25,26
With regard to CV risk, our findings confirm the low prevalence of traditional CV risks among UC patients. When stratified by age, very few non-elderly patients were classified as high risk (ASCVD) or high/very high risk (SCORE2). By contrast, the proportion of patients at higher risk increased significantly in the elderly group, reaching 20.8% and 33.3% according to ASCVD and SCORE2, respectively.
When considering the highest risk categories grouped—intermediate/high for ASCVD and high/very high for SCORE2—these percentages rose to 87.5% and 77.7% in elderly patients, whereas most non-elderly patients remained in the low/borderline risk range for ASCVD (86.7%) and low/moderate range for SCORE2 (95.1%). It is important to note that CV risk scores were calculated only for patients with available serum lipid values and in our cohort patients with available lipid data had a significantly higher median age (49 years, IQR 40–60) compared to the overall study population (44 years, IQR 33–58) and to patients without lipid data (37 years, IQR 27–51). This represents an important selection bias. It is therefore likely that, if cholesterol data had been available for the entire cohort, the observed age-related differences in risk scores would have been even more pronounced, as cholesterol levels are known to rise with age, a finding consistently reported in large epidemiological studies.35,36
In our cohort, we found a high proportion of patients classified as inactive based on the IPAQ questionnaire, as well as a significant number of overweight and obese individuals. Although these factors are not included in ASCVD and SCORE2 risk calculations, they are well-recognized contributors to CV risk. Encouraging patients to engage in regular physical activity and adopt a healthy diet should be a key component of CV risk management in UC.
Data on CV risk profile among patients with IBD remain very limited and inconsistent.37–39 Evidence increasingly supports that inflammation,40,41 rather than traditional risk factors, plays a pivotal role in CV risk for patients with IBD, as suggested by the observation that disease flares and persistent disease activity are associated with increased risks of myocardial infarction, stroke, and CV mortality. Furthermore, anti-TNF therapies have also been associated with reduced CV risk42,43; while CRP levels are associated with an increased risk.44,45 Given the low incidence of both VTE and CV events in our study, a direct causal relationship with disease activity can be determined.
Our study has several strengths. First, it is based on a large cohort, providing robust real-world data on thromboembolic and CV risk factors in UC patients initiating or swapping advanced therapies. In addition, we applied two validated cardiovascular risk scores (ASCVD and SCORE2), allowing for a structured and comprehensive assessment of long-term CV risk. Another strength is the detailed collection of thromboembolic risk factors using a structured questionnaire, which supplements retrospective chart reviews and enhances data reliability. Lastly, the inclusion of physical activity assessment via the IPAQ questionnaire highlights a modifiable lifestyle factor in this population.
Our study has some limitations. First, it was primarily cross-sectional, although a longitudinal prospective follow-up with a median duration of 27 months was incorporated to capture potential CV and VTE events. However, the low number of observed events did not allow for statistical analyses to detect significant differences between treatment groups. Moreover, our cohort was generally younger and had fewer CV risk factors compared with the population enrolled in the ORAL Surveillance study. 8 Another limitation stems from the real-world clinical setting, where lipid profiles were not available for the entire cohort. Specifically, the missing lipid data predominantly involved younger patients, likely resulting in an overrepresentation of older, higher-risk individuals in the cardiovascular risk analysis. Consequently, our risk estimates may slightly overestimate the overall cardiovascular risk of the cohort. If complete lipid data were available, the observed age-related differences in risk scores would likely be even more pronounced. Third, as a single-center study, the findings may have limited external validity. Furthermore, the thromboembolic risk assessment was not based on a validated VTE risk score specifically designed for UC outpatients, given the lack of such a tool, even though we incorporated multiple known risk factors from existing models, which could have led to some degree of over- or underestimation of the risk.
Conclusion
In conclusion, our study confirms that the prevalence of thrombotic and CV risk factors among UC patients is quite low and mostly related to older age. Accordingly, concerns regarding the intrinsic VTE and CV risks of JAK-i, as observed in rheumatological cohorts, may not be entirely applicable to UC patients, given their different baseline risk profiles.
The prevalence of modifiable lifestyle factors, such as physical inactivity and obesity, highlights the need for targeted interventions to optimize long-term outcomes. Further prospective studies are needed to refine risk stratification and assess the long-term safety of advanced therapies in UC.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251388355 – Supplemental material for Thromboembolic and cardiovascular risk profiles in patients with ulcerative colitis initiating advanced therapies
Supplemental material, sj-docx-1-tag-10.1177_17562848251388355 for Thromboembolic and cardiovascular risk profiles in patients with ulcerative colitis initiating advanced therapies by Valentin Calvez, Giuseppe Cuccia, Angelo Del Gaudio, Ivan Capobianco, Laura Parisio, Giuseppe Privitera, Antonio Gasbarrini, Franco Scaldaferri, Luigi Carbone and Daniela Pugliese in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251388355 – Supplemental material for Thromboembolic and cardiovascular risk profiles in patients with ulcerative colitis initiating advanced therapies
Supplemental material, sj-docx-2-tag-10.1177_17562848251388355 for Thromboembolic and cardiovascular risk profiles in patients with ulcerative colitis initiating advanced therapies by Valentin Calvez, Giuseppe Cuccia, Angelo Del Gaudio, Ivan Capobianco, Laura Parisio, Giuseppe Privitera, Antonio Gasbarrini, Franco Scaldaferri, Luigi Carbone and Daniela Pugliese in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
