Abstract
Disorders of gut–brain interaction (DGBI) are among the commonest contributors to disease burden in children, yet remain difficult to diagnose and treat, in part because available tests inadequately capture underlying mechanisms or link physiology to symptoms. Body surface gastric mapping (BSGM) is a next-generation, noninvasive electrophysiological technology that records high-resolution gastric slow-wave activity with simultaneous symptom logging. This review summarizes emerging evidence that BSGM may fill a key diagnostic gap in pediatric DGBI. After outlining the burden and multifactorial pathophysiology of pediatric DGBI, we compare alternative investigations—including electrogastrography, gastric emptying scintigraphy, and antroduodenal manometry—and highlight their limitations in pediatrics, including regarding reproducibility, accessibility, invasiveness, and symptom correlation. We then describe BSGM methodology, validated spectral metrics, symptom integration, and standardized testing protocols. Early pediatric studies have demonstrated excellent feasibility, particularly in adolescents, established preliminary normative ranges, and identified phenotypes to distinguish neuromuscular disorders, delayed meal responses, and alternative symptom profiles to help inform targeted care. Concordance with antroduodenal manometry is also discussed. Finally, we outline research priorities, including the need for larger multicenter cohort studies, formalization of pediatric-specific reference intervals, and longitudinal studies assessing treatment responses. BSGM shows potential as a valuable noninvasive diagnostic tool for better characterizing pediatric DGBIs, though further validation is required.
Keywords
Disorders of gut–brain interaction
Disorders of gut–brain interaction (DGBI) represent one of the most common causes of chronic gastrointestinal (GI) symptoms in pediatrics, affecting 20%–25% of the pediatric population worldwide. 1 Symptoms such as abdominal pain, nausea, vomiting, bloating, early satiety, and heartburn are common gastroduodenal DGBI symptoms. Presentations of abdominal pain and nausea in children are some of the most common reasons for seeking medical care.2–5 Therefore, it is not surprising to find that abdominal pain is the most prevalent symptom in DGBI, 6 with functional dyspepsia (FD) the most prevalent diagnosis, with a reported rate range of 11%–15%. 1 The impact of DGBI on the well-being of children (and their families) is significant, affecting their physical health, emotional development, and social participation. 7 They often score significantly lower on quality-of-life outcomes compared to healthy children and to those with other GI disorders with similar or more severe symptoms. 8
Making a diagnosis
Children presenting with DGBI-like symptoms are challenging to diagnose due to the absence of reliable biomarkers and the lack of definitive confirmatory investigations. A positive diagnosis is primarily symptom-based and relies on the application of the Rome IV diagnostic criteria, 9 identifying and excluding any alarm features, and completing any targeted evaluation deemed necessary and appropriate.9,10 The range of investigations available has remained mostly unchanged over the last decade, and despite the application of Rome IV criteria and the absence of alarm features, diagnostic investigations are frequently pursued, often driven by a desire to minimize uncertainty.11,12
Common first-line assessments include comprehensive clinical assessment, blood tests, and stool studies, which are often used to exclude biochemical or infectious explanations. Investigations such as abdominal ultrasound, radiological imaging, and esophagogastroduodenoscopy are often performed to exclude structural, inflammatory, or obstructive causes of the gastrointestinal symptoms, although in the absence of alarm features, the outcome yields remain low.13–17
Children with symptoms suggestive of a delay in gastric emptying may have a gastric emptying scintigraphy (GES). While GES is considered the gold standard for diagnosing gastroparesis, studies have shown that GES weakly correlates symptoms with a gastroparesis outcome,18,19 has problematic reproducibility,20,21 and often fails to meaningfully differentiate children with similar presenting symptoms. 22 Other investigations, such as antroduodenal manometry (ADM), which is invasive, or autonomic testing, are considered for highly complex cases, often when symptoms are persistent and refractory to repeated treatment regimes. However, access to these specialized investigations is limited to certain jurisdictions and specialist tertiary centers, restricting their availability.
Negative testing does not always provide enough reassuring evidence to patients and families to accept a diagnosis of a DGBI. From a patient’s perspective, these negative tests can increase frustration as they continue to lack a definitive cause of their symptoms. 23 The absence of detectable abnormalities can lead to confusion or skepticism, and a perception that their child’s symptoms are being minimized or misunderstood, despite their ongoing distress and functional impairment. This can prompt parents to pursue additional consultations and ask for more investigations, in search of a more tangible explanation, leading to significant delays between the onset of symptoms and clinical management.24,25 Healthcare utilization often increases due to repeated and additional investigations and a cycle of trial-and-error treatment pathways.16,17,26
The low diagnostic yields and overall outcome ambiguity associated with many of the current investigative approaches used are partly attributable to the fact that these tests do not clearly distinguish between underlying pathophysiological mechanisms, objectively evaluate centrally mediated symptom relationships, and/or correlate symptoms to measurable gastric activity.
It is evident that there is a need for diagnostic tools to better characterize DGBI, thus improve clinical decision-making, guide targeted interventions, and minimize the significant financial burden to the healthcare system,13,27 to improve health, economic, and psychosocial outcomes for children and their families. 28
Mechanisms of pediatric DGBIs
DGBI arises from complex multisystem mechanisms across neural, immune, endocrine, and GI systems. Central to DGBI is the dysregulation of bidirectional communication between the GI tract and the nervous system. Abnormalities in this communication pathway can result in visceral hypersensitivity, impaired central pain modulation, and altered motility patterns in the absence of visible structural disease. Other contributing factors, such as epithelial barrier dysfunction, the microbiome, endocrine activation via the hypothalamic–pituitary–adrenal axis, and immune activation also appear to play a key role in their development. 29 Importantly, gastric myoelectrical dysfunction has also increasingly been recognized as a contributing mechanism, particularly in conditions such as FD and gastroparesis in adults 30 and pediatrics. 22
The role of dysregulated central pain processing and its amplification affects both visceral and somatic hypersensitivity experienced by DGBI patients. This central altered pain processing phenomenon is both an amplifier and a cause of chronic GI pain. The added complexity of developmental factors such as stress, early life traumatic events, and personal and parental mental health may exacerbate or perpetuate neural and myoelectrical dysfunction, reinforcing symptom persistence. 31 Collectively, these pathophysiological processes contribute to the heterogeneity and chronicity of symptoms in DGBI, underscoring the diagnostic complexity and clinical management challenges.
The role of the gastric conduction system and DGBI
As current diagnostic tools fail to consistently capture and characterize some of the major physiological disturbances underlying DGBI, effective investigations to evaluate one of the key motility control systems—the gastric myoelectrical system—are required. Assessing gastric myoelectrical function requires a correct understanding of the conduction system.
The gastrointestinal tract is a continuous tube, with regions regulated by distinct mechanisms. 32 The esophagus is primarily controlled by its vagal innervation, which initiates and regulates peristaltic contractions. By contrast, the small and large intestines are governed by the enteric nervous system in concert with myenteric/myoelectrical mechanisms. 33
By contrast, the stomach’s motor function is predominantly regulated by myoelectrical control. 32 Gastric slow waves originate from a pacemaker zone in the greater curvature (Figure 1). These waves are generated by interstitial cells of Cajal (ICC), a heterogeneous network of mesenchymal cells classified by location, including intramuscular (ICC-IM) and myenteric (ICC-MP) subtypes.32,34–36

Overview of the human gastric conduction system (reproduced with permission).
ICC activity, driven by rhythmic ion channel oscillations, initiates depolarization, calcium influx, and smooth muscle contraction.34,37 Slow wave activity propagates circumferentially and longitudinally, with longitudinal (proximal-to-distal) propagation rapidly dominating. 38 The typical slow wave frequency is tightly controlled around 3 cycles per minute (cpm), 39 with conduction velocities around 3 mm/s, accelerating beyond 7 mm/s in the terminal antrum, creating approximately 6 cm spacing between successive wavefronts. 32
Propagation toward the fundus and cardia is limited due to functional specialization and relative hyperpolarization.34,40,41 Slow waves weaken and terminate proximally, with the fundus and cardia primarily involved with accommodation rather than peristalsis. 42 Fundic ICC-IM cells, in interaction with vagal fibers, promote neurotransmitter release and smooth muscle relaxation. 43
Thus, healthy gastric function relies on coordinated slow wave generation at about 3 cpm, effective propagation, and region-specific motor specialization, enabling efficient mixing, propulsion, and accommodation of gastric contents. Symptoms such as nausea, abdominal pain, or post-prandial distress may arise when any of the above-mentioned steps are dysfunctional. Gastric electrical activity may be measured clinically to recognize and evaluate these processes.
Measuring gastric myoelectrical activity
Legacy electrogastrography
Noninvasive measurement of gastric myoelectrical activity from the abdominal surface was first reported by Alverez in the 1920s, 44 who envisioned electrogastrography (EGG) as a diagnostic tool akin to electrocardiography. Subsequent decades saw many adult and pediatric studies,45–48 particularly attracted by EGG’s noninvasive nature in vulnerable populations. However, EGG failed to achieve widespread clinical adoption in adults and pediatrics due to multiple challenges.49–51
These challenges have been broadly categorized into protocol variability, signal acquisition and processing, metrics or biomarkers, and symptom correlation. 52 In legacy EGG, protocol variations in electrode placement, meal timing, meal composition, and recording duration significantly compromised study comparability. Most critically, signal acquisition compared to competing noise was often inadequately distinguished, and suboptimal electrode positioning and failure to adjust for anatomical differences frequently led to signal loss or contamination with non-gastric activity.
Analytically, artifact handling was predominantly a combination of manual and simple computational techniques, and frequently confused noise with gastric signals. 53 Reports of tachygastria >5 cpm, for example, which are absent in serosal recordings, suggest artifacts were often conflated with true gastric activity. 52 Due to the small number of electrodes and weak source signal, extracting reliable data from background noise was an insurmountable hurdle to widespread use. Finally, there was no standardized symptom reporting.
Ultimately, as a consequence of its poor sensitivity, specificity, and lack of methodological consensus, legacy EGG did not find a routine role in clinical diagnostic pathway algorithms. 54 Consequently, EGG remained largely a research tool in both pediatric and adult gastroenterology, rather than becoming a routine clinical diagnostic procedure.49,55,56
Body surface gastric mapping
Advances in bioinstrumentation, computational modeling, and flexible biocompatible electrode materials have modernized legacy EGG technology, facilitating a new generation of approaches.41,57 This has resulted in the development of “Body Surface Gastric Mapping (BSGM),” a new, noninvasive technology that integrates physiological and subjective data collection to disentangle the complex interconnections between gastroduodenal dysfunction, gut–brain interactions, and symptoms and presents a positive diagnostic aid. 58 The first commercial system has been developed (Gastric Alimetry, Alimetry Ltd; Auckland, New Zealand), and is now available in the United States, the United Kingdom, and New Zealand.
As of 2024, BSGM became FDA-cleared for individuals aged 12 years and older, paving the way for clinical usage in pediatrics. The test employs a large, flexible 64-electrode array to capture a wide field of gastric myoelectrical activity, generated by extracellular ion flows during slow wave depolarization and repolarization. 59 Rigorous validation studies confirm that the recorded signals are gastric in origin and not artifacts of contractile movement.60,61
As gastric myoelectrical signals are in the order of 100 times weaker than cardiac signals, the dense array greatly enhances the sensitivity of signal capture through summation of many individual sources. Figure 2 illustrates the differences between legacy EGG electrodes and the new BSGM multi-channel electrode array.
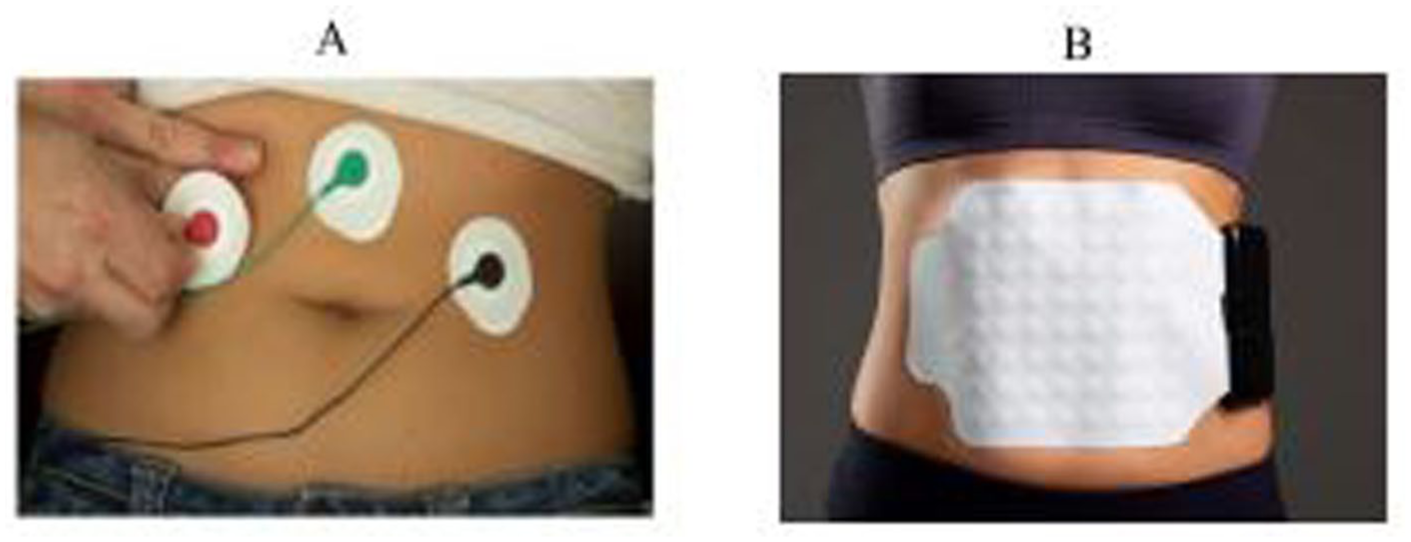
(a) Example of legacy EGG electrodes and their placement compared to (b). The placement of BSGM, which is a flexible array embedded with 64 electrodes designed to accommodate anatomical variations.
Given the large volume of data generated, sophisticated automated algorithms have also been developed and validated to accurately detect and exclude artifacts, supporting reliable data interpretation.53,62
To optimize gastric specificity and directly address the reliability issues of earlier single-channel EGG systems, BSGM also incorporates person-specific anatomical measurements for precise array placement.63,64 The array design also accommodates anatomical variability across ages and body mass index (BMI) and can be modified (e.g., cut along preset lines) to fit patients with feeding tubes without compromising signal quality. In addition, the ability to modify the array has enabled BSGM to be used effectively and safely in children as young as 5 years old in research contexts. 65 Furthermore, the generous size and adaptability of the array are also well-suited to accommodate chronological anatomical variations of the shape and position of the stomach observed in children. 66
A standardized protocol
BSGM follows a standardized protocol, including 6 h of fasting, specific preparation before placing the array, a 30-min pre-prandial recording, consumption of a standardized meal over 10 min, and a 4-h post-prandial recording. 67 This protocol has been demonstrated to be feasible and safe in children (Figure 3). 65

Standard BSGM protocol.
Validated measurement outcomes
BSGM myoelectrical outcome measures
BSGM myoelectrical outcome measures were substantially revised to improve on legacy EGG approaches, including separating frequency and rhythm, identifying dominant frequency bands from noise, and adjusting metrics for BMI. The four key metrics currently included in FDA-cleared devices are as follows:
Principal Gastric Frequency: Sustained stable frequency.
Gastric Alimetry Rhythm Index (GA-RI): Measure of rhythmic stability.
BMI-Adjusted Amplitude: Adjusts for signal attenuation by adipose tissue.
Fed-to-Fasted Amplitude Ratio: Quantifies the post-prandial gastric response.
Normative reference intervals for adults have been established,58,68 with adolescent intervals currently under development. 69 In the absence of current adolescent reference ranges, we advise applying adult reference ranges, however, with caution. In particular, early pediatric data suggest that the rhythm index ranges may be slightly lower in adolescents, particularly in the later post-prandial test period, meaning that borderline-low results should be assessed cautiously.
BSGM with Gastric Alimetry has also been found to be reproducible in the short and long term, with no significant differences observed in the BSGM metrics following re-testing at 1 week and 6 months (all p-values >0.05). Reproducibility appears superior to comparable gastric investigations, such as gastric emptying testing. 20
Symptom and well-being integration
In the Gastric Alimetry system, BSGM integrates real-time symptom logging using pictograms validated in children and young people to capture within-test symptoms such as nausea, bloating, and abdominal pain,25,70,71 which allows temporal correlation of symptoms with gastric myoelectrical patterns (Figure 4). The use of pictograms has been shown to improve patient symptom reporting through enhanced recognition and differentiation of GI symptoms, which can otherwise cause confusion or overlap. 72

Gastric Alimetry app for logging symptoms and severity showing (a) the symptom screen with all symptoms with their reported severity scores and the “tap to record” events. (b) An individual symptom with a sliding severity scale.
In adults, mental well-being, a key modulator of gut–brain interaction, is also assessed concurrently using the validated Alimetry Gut Wellbeing Survey (AGBW). 73 An adolescent version has recently been co-developed with young people and validated (AGBW-Y), 74 with clinical implementation pending. Incorporating psychological dimensions with physiological symptoms helps to differentiate physiological from psychogenic contributions to symptoms.70,75–77
Presenting the outcomes of BSGM
BSGM metrics are visually presented through intuitive spectral plots that display changes over time, allowing for easy interpretation of gastric electrophysiological patterns in relation to meals and symptoms, and a table with the averaged metrics over time. Logged symptoms are presented individually over time, as a total score out of 70 to illustrate severity, and correlations with gastric activity are visually presented (Figure 5).
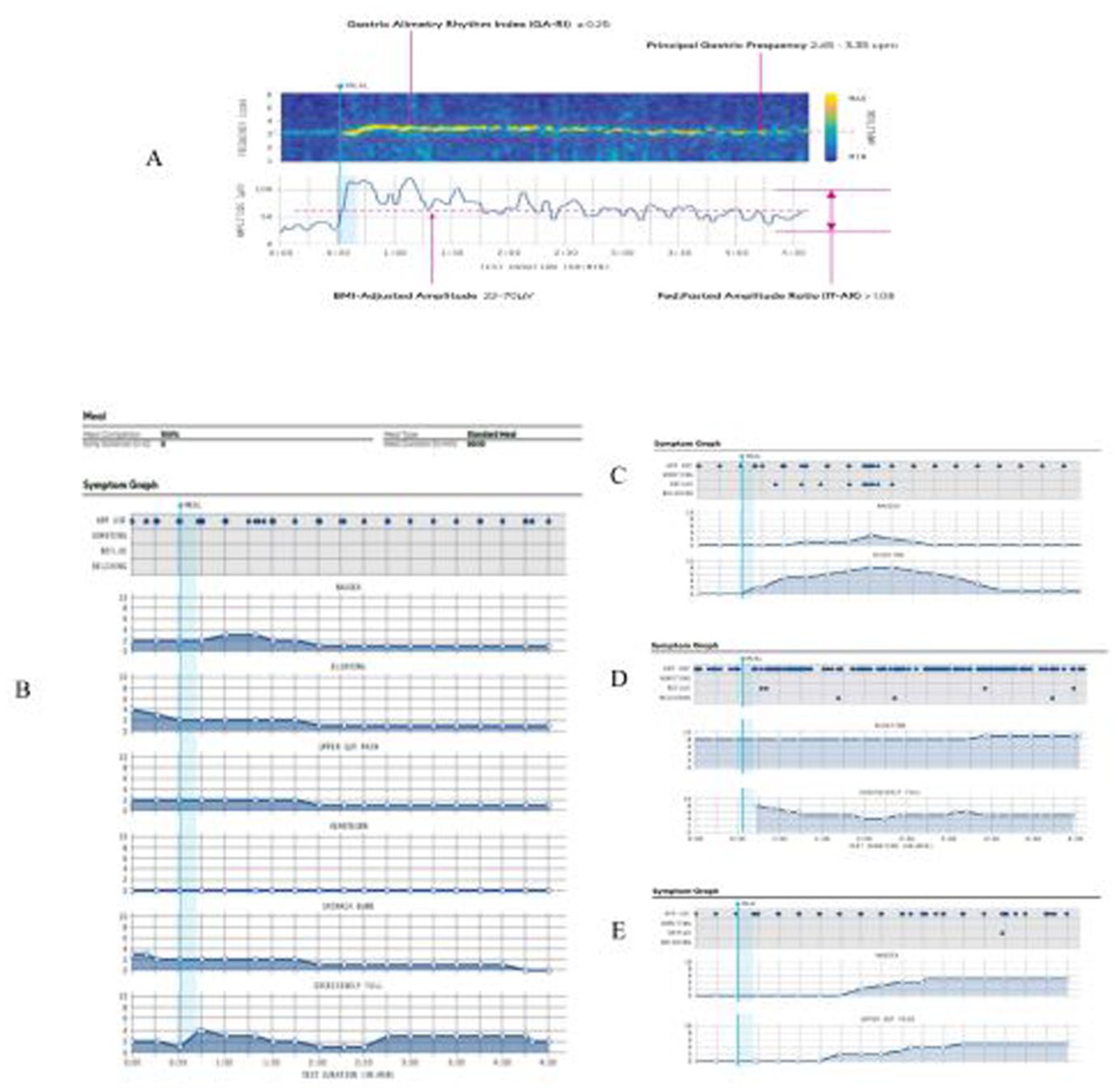
(a) Example spectral plot for a single participant with annotations showing how the BSGM metrics (Principal Gastric Frequency, Gastric Alimetry Rhythm Index, BMI-Adjusted Amplitude, and Fed to Fasted Amplitude Ratio) and their reference intervals are visualized. (b) Presents the visualization of all the individual gastric symptoms and which symptoms have been logged during BSGM over time. (c, d) Illustrate specific symptom profiles that have emerged, for example, (c) presents a Sensorimotor profile, where the symptoms are meal-responsive and decline as gastric amplitude reduces following a post-prandial decay curve pattern. (d) Is the Continuous profile, where reported symptoms are continuous throughout the test, with little if any variation. They also do not correlate with either the meal or the gastric activity. (e) Presents a Post Gastric profile, where symptoms are not meal-responsive but uptrend late in the test, often as gastric amplitude declines.
Advances in diagnostic understanding in adults through BSGM
A growing body of research in adults is demonstrating the effectiveness of BSGM as a diagnostic aid. In a seminal study, patients with chronic nausea and vomiting syndromes and those diagnosed with gastroparesis presented with clinically indistinguishable symptom profiles. 78 Evaluating these patients with BSGM identified two distinct subgroups. The majority (62%) had normal BSGM spectral results but higher rates of psychological comorbidities and anxiety, whereas a smaller group had markedly abnormal BSGM spectral metrics—considered to reflect underlying neuromuscular dysfunction, with these abnormalities correlating with the severity of gastrointestinal symptoms such as nausea, pain, fullness, early satiety, and bloating. This finding revealed distinct underlying phenotypes despite initial overlapping symptom presentations. The authors argue that this finding may inform more targeted clinical management by identifying true gastric motility disorders from competing causes and thus enabling a reduction in trial-and-error cycles of care. 78
Subsequently, the ability to identify distinct BSGM “phenotypes” based on objective physiological measures and symptom correlations has also led to several studies incorporating BSGM into the clinical evaluation of patients with complex and often refractory chronic gastroduodenal symptoms to assess its utility for guiding clinical management. For example, a cohort of 50 patients with chronic gastroduodenal symptoms, BSGM outcomes aided changes in management decisions in 84% of cases, medication de-escalation occurred in 20% of patients, and overall changes in management decisions resulted in a significant decrease in healthcare resource utilization. 30 Similarly, in a series of 10 patients on parenteral nutrition and assumed to have intestinal failure secondary to gut dysmotility, BSGM outcomes facilitated successful weaning of parenteral nutrition in six patients at a median of 5 months of follow-up, with substantial quality of life and health economic implications. 79
The growing understanding of the underlying mechanisms and symptom correlations is providing new insights and avenues for the stratification and classification of current conditions beyond traditional diagnostic labels like FD and gastroparesis.77,80 This emerging phenotype-based classification schema enables patients to be phenotyped into cohorts comprising those with spectral abnormalities (indicating an objective motility abnormality) (Figure 6(a) and (b)), those with symptoms correlated with gastric activity, and those with symptoms independent of gastric activity (Figure 6(c) and (d)).

The BSGM spectral phenotypes: (a) Dysrhythmic: The spectrogram shows a high degree of yellow scatter with no clear signal band, signaling poor Gastric Alimetry rhythm stability (low GA-RI). Symptom profile is variable. (b) High frequency: Defined by a sustained high frequency >3.35 cpm as illustrated in the spectrogram. Symptom profile is variable but is often continuous. The symptom profiling phenotypes: (c) Sensorimotor profile: Defined by normal spectral plot with meal-responsive symptoms and post-prandial decay curves, all correlating with gastric activity. (d) Continuous profile: Defined by normal spectral analyses, however, the reported symptoms are continuous, often presenting during the fasting baseline and with little variation throughout the test. The symptoms do not correlate with either the meal stimulus or in response to gastric activity. The specific symptoms reported may be mixed, but the continuous pattern is unchanged.
The important clinical implication is that it appears plausible to define significant subsets of patients with chronic gastroduodenal symptoms into primarily gastric pathophysiology versus gut–brain-axis mediated (or alternative non-gastric disorder) subgroups. This allows for improved patient selection for principally psychological or central nervous system targeted therapies versus gastric targeted therapies such as prokinetics or endoscopic interventions.70,80
Adult comparative and other clinical studies
In a study of 75 adult patients with chronic gastrointestinal disorders where BSGM and GES were simultaneously undertaken, BSGM identified motility abnormalities in a higher proportion of patients (33.3%) compared to GES (22.7%), identified superior correlations with patient-reported symptoms and anxiety levels, and improved patient differentiation compared to GES. These results indicate BSGM’s potential for enhanced diagnostic profiling and personalized management of these conditions, and potential complementarity with gastric emptying testing. 19
In addition, BSGM has been able to characterize and define different types of gastric myoelectrical abnormalities in individuals with long-term type 1 diabetes (T1D), both with and without gastrointestinal symptoms. Symptomatic T1D patients exhibited more unstable gastric myoelectrical activity and identified specific phenotypes, including those with markedly unstable gastric rhythms (12.5%—thought to be a biomarker of ICC damage) and those with abnormally high gastric frequencies (28%—thought to be a biomarker of autonomic neuropathy). Deviations in gastric frequency were positively correlated with worse symptoms, including bloating, upper gut pain, nausea, vomiting, and fullness. The authors conclude that these new insights offer more specificity in diagnosis, monitoring, and therapy in diabetic gastroenteropathy. 81
BSGM is also being successfully used in research to evaluate the effectiveness of procedures, such as fundoplication, 82 esophagectomy, 83 pancreaticoduodenectomy, 84 and transcutaneous auricular vagus nerve stimulation. 85 Currently, clinical trials are also evaluating the clinical success of Gastric PerOral Endoscopic Myotomy (G-POEM), to determine whether BSGM can help identify who would best benefit from receiving the G-POEM procedure. 86
In addition, a separate clinical trial has recently shown potential to identify prokinetic responders, with patients showing low post-prandial amplitudes appearing to experience symptom relief from prokinetics, compared to an apparent exacerbation of symptoms in patients with dysrhythmic gastric activity. 87 These should be considered preliminary findings, with larger multicenter studies awaited.
Gaining new understandings of the underlying pathophysiology and influencers
A recent study used BSGM to investigate the effect of liraglutide injection, a GLP-1 analog, on gastric electrophysiology in healthy participants. 88 GLP-1 analogs have been found to cause gastric symptoms, similar to those in gastroparesis, and often are the reason for discontinuing the drug. The results showed that the GLP-1 independently decreased both BMI-adjusted gastric amplitude and gastric emptying, with both effects fully reversing on drug cessation. Another study evaluated the effects of corticotropin-releasing hormone (CRH) on meal-related gastrointestinal symptoms, gastric electrical activity, and gastric sensorimotor function. After CRH exposure, the gastric emptying time was decreased, with significantly increased post-prandial gastric amplitude captured by BSGM, accompanied by a rise in reported post-prandial symptoms. The authors conclude that CRH modifies gastric physiology and emptying and produces symptoms, thus providing a further co-regulatory mechanism of the gut–brain axis. 89
It is well reported that females are over-represented in DGBI. A recent study explored the effect of the menstrual cycle and menopause on gastric electrophysiology. 90 Lim et al. report higher gastric frequencies and faster gastric emptying (via a computational model) in females during the luteal versus follicular phases, measured by BSGM. Borderline increases in BSGM frequency during the luteal phase in females can therefore be considered normal.
Body surface gastric mapping and pediatric DGBI
While much of the current evidence supporting the utility of BSGM as a diagnostic aid comes from studies in adult populations, a body of evidence is beginning to emerge for the pediatric population. The noninvasive nature of BSGM, and the use of validated pediatric symptom pictograms to aid symptom recognition and reporting25 makes it particularly well-suited for use in children, offering a patient (and family) friendly alternative to more invasive diagnostic techniques or those involving radiation.
The first study investigating BSGM in children and young people aged 5–18 years demonstrated that the BSGM protocol was feasible, well-tolerated, and safe for children as young as 5 years. It also showed that BSGM metrics could distinguish between 32 healthy controls and 12 patients meeting the Rome IV criteria for FD and gastroparesis. 65
In a separate study, 32 healthy pediatric controls (mean age 13.9 years, range 6–18), the authors found that the BSGM spectral metric outcomes in children were broadly comparable to those observed in healthy adults. The key difference was a reduced gastric rhythm stability in the latter stages of the post-prandial period. 69 As more healthy controls are included in the development of normative reference intervals for adolescence, the initially observed reduction in gastric rhythm stability during the later stages of the post-prandial period can be better evaluated to determine whether it represents a normal feature of pediatric gastric physiology.
A recent important study evaluated differences between children diagnosed with FD and those with gastroparesis confirmed by delayed gastric emptying scintigraphy (GES)—two conditions with overlapping symptoms that may exist on a disease spectrum. 21 To explore whether BSGM could improve patient differentiation beyond these standard diagnostic labels, 35 healthy controls, and 25 patients with FD or gastroparesis, aged 12–20 years, from 2022 to 2024 were evaluated using BSGM. 22 The results confirmed that BSGM could distinguish patients from healthy controls, as was reported in the earlier feasibility study. 65 No significant differences in symptoms, quality of life, functional disability, or BSGM metrics were identified between the two standard diagnostic groups of FD and gastroparesis. Although study numbers are small, when patients were stratified directly according to their unique BSGM metrics and symptom profiles, clear differences emerged. BSGM outcomes categorized adolescent patients into three distinct phenotype profiles. BSGM normal: No measurable differences in BSGM metrics were identified between this patient cohort and healthy controls. The primary distinction lay in symptom reporting, with this cohort experiencing continuous symptoms unrelated to gastric activity. The second phenotype category was BSGM delay (slow onset of meal response). This was illustrated by a visible lag in gastric activation (rise in amplitude) which occurred ⩾2.5 h after the meal. The symptom pattern for this cohort showed an increase immediately post-prandial and declined with the onset of gastric activity. The final phenotype that was identified was the low stability/low amplitude (dysrhythmic) phenotype. This phenotype had a distinct pattern of yellow scatter in the spectrogram, signaling minimal to no cohesive synchronous band of gastric activity. The distinct spectrograms of these three phenotypes are presented in Figure 7.

BSGM spectrograms showing the difference between the pediatric phenotypes: (a) BSGM normal: No measurable differences in BSGM metrics were identified between this patient cohort and healthy controls. The primary distinction lay in symptom reporting, with this cohort experiencing continuous symptoms unrelated to gastric activity. (b) BSGM delay (slow onset of meal response) is illustrated by the lag in gastric activation (rise in amplitude), which occurs ⩾2.5 h after the meal. The symptom pattern increases immediately post-prandial and declines with the onset of gastric activity, and finally, (c) low stability/low amplitude (dysrhythmic) phenotype. This phenotype is illustrated by the yellow scatter in the spectrogram, signaling minimal to no cohesive synchronous band of gastric activity.
Using validated questionnaires, these BSGM phenotypes also correlated with gastroduodenal symptoms, quality of life, and psychometric measures. Notably, the low stability/low amplitude (dysrhythmic) phenotype was associated with higher levels of nausea, anxiety, and functional disability scores compared to both other groups. 22 This low gastric stability (dysrhythmic) group has been considered indicative of a true neuromuscular dysfunction, 80 and points to a more severe disease subgroup requiring more intensive management.
In a pilot study exploring the impact of intrapyloric Botox injections, BSGM was used to monitor and evaluate changes in gastric myoelectrical activity and symptoms before and after injections for eight pediatric patients with FD or gastroparesis (7 females, age 9–20). 91 Initial data indicated decreases in gastric amplitude and gastric rhythm index, suggesting a physiological modulation of gastric function following pyloric Botox treatment in children. Notably, these objective changes were accompanied by self-reported improvements in real-time symptoms at a median of 16 days post-injection, also suggesting a potential therapeutic benefit. These results, while preliminary, highlight the utility of BSGM in capturing both physiological and symptomatic responses to targeted interventions, offering a promising approach for evaluating and guiding treatment strategies in children with gastric dysfunction. 91
Irritable bowel syndrome (IBS) was reported as the most common functional gastrointestinal disorder in pediatrics for two consecutive decades.92,93 Upper GI symptoms are common for the IBS cohort, and Benitez et al. utilized BSGM to evaluate gastric activity for IBS patients. Ten IBS patients with 10 sex- and age-matched controls (aged 9–22) were included in their study. An abnormal GA-RI (low rhythm stability) was detected in 70% of IBS cases, and GA-RI was significantly lower versus controls (0.24 vs 0.55, p < 0.001). Total symptom burden was significantly different between cases and controls (9.80 vs 0.85, p = 0.006), and worse symptoms were moderately correlated to the IBS cases with lower GA-RI (R = −0.44). 94 These data highlight the fact that DGBI often presents with pan-GI involvement.
Comparison studies in pediatrics
While considered a gold standard investigation for dysmotility, ADM is highly invasive and not widely accessible or available across jurisdictions. A recent study by Sadaka et al. investigated the potential of BSGM as a noninvasive supplement to ADM to support the diagnosis of neuropathic gastroduodenal disorders in pediatric patients. 95 The research involved 15 children aged 10–19 who underwent simultaneous BSGM and ADM assessments. The findings revealed a correlation between neuropathic patterns identified by ADM and a low GA-RI reported by BSGM, which indicates dysrhythmic gastric activity. Patients exhibiting this low stability phenotype on BSGM also reported significantly higher levels of nausea and bloating, similar to that reported in the study by Humphrey et al. 22 . Conversely, normal ADM results corresponded with normal BSGM readings in the majority of cases. These results suggest that BSGM could be a reliable diagnostic tool for detecting gastric neuropathy and associated symptoms noninvasively, offering a promising supplement to ADM, especially in settings or jurisdictions where ADM is not feasible. Further data on these relationships are awaited.
Considerations and limitations
Much of the current BSGM research is in adults; therefore, it is important to avoid conflating the adult findings to pediatrics without adequate evidence. It is likely that the current synergy of early findings in adolescent studies with those of adult research is reflective of close similarity in the underlying physiology, given that core mechanical and secretory functions of the stomach are mature and operate at adult levels by early adolescence. 96
Nonetheless, adolescence is a time of biological, hormonal, and physical changes. While it is not a period of organogenesis, it is characterized by scaling—including in stomach capacity—as well as maturation of cellular functions and hormonal control systems. The role of gastric hormones such as Ghrelin (hunger regulation) and gonadal hormones (such as estrogen and testosterone) is highly growth-centric during early adolescence.97,98 These physical changes may impact normal gastric function, hence, establishing normative references for BSGM metrics in an adolescent population represents a key step toward establishing clinical applications for this population.
Psychosocial development during adolescence and the interplay between psychosocial stress, the developing adolescent brain and gastric function is also well reported.99,100 However, there are few validated mental wellbeing assessment tools specifically for adolescents with chronic gastroduodenal symptoms. 101 As noted earlier, pediatric BSGM research shows associations between phenotypes, quality of life, and mental well-being. These may support evidence that suggests that during this rapid biological developmental phase, adolescents appear to be more susceptible to experiencing some gastric symptoms, such as abdominal pain, in the presence of physiologically normal gastrointestinal motility. 102 Therefore, implementing the new validated youth gut well-being survey 74 into the BSGM protocol, and evaluating the outcomes alongside the physiological assessments in large-scale studies will help provide clearer evidence into these connections.
Overall, there is now also a clear need for large-scale research using BSGM in young people with chronic gastroduodenal symptoms to validate early phenotyping observations. This should be complemented by prospective interventional expert groups to inform and support evidence-based clinical management.
Conclusion
DGBI in children remains a challenging field defined by overlapping symptom disorders with multiple contributing factors. After the clinical failure of legacy EGG, BSGM is rapidly emerging as a valuable next-generation tool for assessing gastric myoelectrical activity, with a robust body of evidence now supporting its utility in adult populations. Clinical studies in adults have demonstrated that BSGM can reliably identify dysrhythmic gastric activity (indicating underlying neuromuscular dysfunction), correlate with symptom severity, and assist in phenotyping patients with disorders such as gastroparesis and functional dyspepsia. Its noninvasive nature positions BSGM as a positive (and possibly superior) alternative to traditional tests like gastric emptying studies, which often weakly correlate with symptom burden or underlying ability to distinguish pathophysiology in pediatric populations.
Encouragingly, a commercial BSGM device has recently been cleared by the FDA in pediatrics (ages 12+), and with a growing number of studies is now exploring the application of BSGM in pediatric populations. Early research, including recent findings by Humphrey et al. showing three distinct and actionable phenotypes and Sadaka et al., highlight that BSGM can detect neuropathic gastric patterns in children with gastrointestinal symptoms, with initial correlations to invasive ADM results for both normal and neuropathic patterns.22,95 These findings underscore the potential of BSGM as a noninvasive diagnostic alternative in a clinical space where diagnostic procedures are often limited by reproducibility, tolerability, and accessibility. Moreover, the technology’s ability to provide real-time, pediatric validated patient-reported symptom correlations adds an important dimension to pediatric care, where distinguishing and communicating symptoms may be more challenging.
However, BSGM in pediatrics remains in its early stages, and several important developments are required. This includes the validation of pediatric-specific normative reference intervals. The integration of BSGM into routine clinical pathways and practice, particularly in pediatric gastroenterology, will also require continued validation through larger, multicenter studies. New longitudinal research to better understand the predictive value of BSGM in treatment response, such as Botox injection 91 and prokinetic therapies, 87 and disease progression, as well as its role in guiding individualized care between gut–brain and intensive gastric therapies. Future directions may also include refining specific pediatric diagnostic criteria based on BSGM phenotypes and exploring its utility across a broader range of gastrointestinal conditions. As the evidence base expands, BSGM is well-positioned to transform the diagnostic landscape for gastric disorders, offering new objective, noninvasive, and physiologically meaningful insights for pediatric patients.
