Abstract
Background:
A suitable disease classification is essential for individualized therapy in patients with Crohn’s disease (CD). Although a potential mechanistic classification of colon-involving and non-colon-involving disease was suggested by recent genetic and microbiota studies, the clinical implication has seldom been investigated. We aimed to explore the association of this colonic-based classification with clinical outcomes in patients with CD compared with the Montreal classification.
Methods:
This was a retrospective study of CD patients from a tertiary referral center. Patients were categorized into colon-involving and non-colon-involving disease, and according to the Montreal classification. Clinico-demographic data, medications, and surgeries were compared between the two classifications. The primary outcome was the need for major abdominal surgery.
Results:
Of 934 patients, those with colonic involvement had an earlier median (interquartile range) age of onset [23.0 (17.0–30.0) versus 26.0 (19.0–35.0) years, p = 0.001], higher frequency of perianal lesions (31.2% versus 14.5%, p < 0.001) and extraintestinal manifestations (21.8% versus 14.5%, p = 0.010), but lower frequency of stricture (B2) (16.3% versus 24.0%, p = 0.005), than those with non-colon-involving disease. Colon-involving disease was a protective factor against major abdominal surgery [hazard ratio, 0.689; 95% confidence interval (CI), 0.481–0.985; p = 0.041]. However, patients with colon-involving CD were more prone to steroids [odds ratio (OR), 1.793; 95% CI, 1.206–2.666; p = 0.004] and azathioprine/6-mercaptopurine (AZA/6-MP) treatment (OR, 1.732; 95% CI, 1.103–2.719; p = 0.017) than were patients with non-colon-involving disease. The Montreal classification was not predictive of surgery or steroids and AZA/6-MP treatment.
Conclusion:
This study supports the rationale for disease classification based on the involvement of colon. This new classification of CD is a better predictor of clinical outcomes than the Montreal classification.
Lay Summary
Colon-involving and non-colon-involving Crohn’s disease (CD) are different in gene expression patterns, suggesting a potential new classification according to disease location. We included 934 CD patients and classified them into colon-involving and non-colon-involving groups. We found that patients with colon-involving disease had an earlier median age of onset, higher frequency of perianal lesions and extraintestinal manifestations, but lower frequency of stricture. Also, colon-involving disease was a protective factor against major abdominal surgery. However, when the patients were stratified according to the Montreal classification (L1-L4), the association with disease prognosis was not significant. The results were consistent with the previous genetic study and supports a new classification that might provide better guidance for personalized therapy in CD.
Introduction
Crohn’s disease (CD) is a heterogeneous entity that primarily manifests as a chronic relapsing inflammation in the gastrointestinal tract with multiple other manifestations and complications. 1 A suitable classification is essential for individualized therapy in patients with CD. When it was first described, CD was considered to be restricted to the terminal ileum. However, it later became evident that the disease can affect any part of the gastrointestinal tract. 2 Although the Montreal classification is still the most advocated and widely used classification, 3 the significance of this conventional classification has been questioned recently and the data available on its reliability are limited. 4 Recent deeper phenotypic studies suggested a different stratification with respect to genetic and microbial phenotypes, based on absence or presence of colonic involvement. 5 Specifically, a landmark genetic study reported that the genetic profiles of patients with colon-involving (including colonic and ileocolonic) CD were distinct from the profiles of patients with ulcerative colitis or with ileal CD, which implied that colon-involving CD could potentially be a separate entity of inflammatory bowel disease (IBD). 6 Furthermore, a significant difference in the composition of gut bacteria was reported between colon-involving and ileum-restricted disease, 7 similarly suggesting a rationale of fundamentally two different phenotypes of CD according to colon-involving and non-colon-involving disease extent. However, only few studies have investigated the association of the clinical outcomes with disease stratified by the involvement of colon in CD. In this study, we aimed to explore the association of the clinical outcomes in patients with CD with either a colon-based classification or the conventional Montreal classification.
Materials and methods
Population and study design
A prospectively designed standardized follow-up schedule was applied to all patients in this observational cohort study. Patients with a diagnosis of CD who were followed up at a tertiary referral center (The First Affiliated Hospital, Sun Yat-sen University, China) between January 2008 and December 2017 were identified.
The inclusion criteria were as follows: (1) definite diagnosis of CD based on clinical manifestations, abdominal imaging, and intestinal pathology; (2) complete demographic and clinical data at the time of diagnosis with regular follow-up; (3) colonoscopy at first diagnosis; and (4) at least one of the following examinations for assessing the involvement of small bowel: computed tomography enterography, magnetic resonance enterography, small bowel enteroscopy, and capsule endoscopy.
The exclusion criteria were as follows: (1) history of malignancy and (2) previous abdominal surgery.
Data collection
Data, including age, sex, duration of disease, disease locations, behaviors, presence of perianal lesions, extraintestinal manifestations, medications, and surgeries, were collected from the prospectively maintained database.
Definition and outcomes
According to the Montreal classification, 8 disease locations were categorized as L1, L2, L3, and L4 for involvement of the terminal ileum, colon, ileocolon, and upper gastrointestinal tract, respectively. L4 can coexist with L1, L2, or L3. Mucosal erythema and edema alone are not sufficient to determine the disease involvement. 9 Colon-involving disease was defined as a disease with any involvement of the colon, while non-colon-involving disease was defined as a disease without any colonic involvement.
The primary outcome was the need for major abdominal surgery during follow-up, which was defined as bowel resection related to CD involvement (including small bowel resection, hemicolectomy, subtotal and total colectomy). To exclude some emergency cases that needed surgery within a short time, we only included patients with more than 3 months of follow-up. The secondary outcome was the requirement of steroids, azathioprine/6-mercaptopurine (AZA/6-MP), and infliximab within 1 year of follow-up. We used the Crohn’s Disease Activity Index (CDAI) to assess disease activity.
Statistical analyses
SPSS (version 22.0, IBM Corp., Armonk, NY, USA) was used for data analyses. Discrete data were reported as frequency and percentage with the use of chi-square test or Fisher’s exact test for comparisons. Continuous variables that were normally distributed were expressed as mean and standard deviation, while non-normally distributed ones were expressed as median and interquartile range (IQR) with the use of independent sample t-test or Mann–Whitney U tests for comparisons, as appropriate. Cox’s regression model was used to compare the risk of surgery between colon-involving and non-colon-involving disease. Kaplan–Meier curves were used to estimate the cumulative possibilities of surgery between the two groups. Logistic regression was applied to determine the independent factors associated with the use of medications. Variables with p < 0.1 in univariate analysis were included in the multivariate analysis. All statistical tests were performed two-tailed, with p < 0.05 denoting statistical significance.
Ethical considerations
The protocol was approved by the Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University (Approval Number: 2018224), which waived the requirement for informed consent.
Results
Patient characteristics
A total of 934 patients with CD who met the inclusion criteria were included in this study. As summarized in Table 1, 68.5% of patients were male and the median (IQR) follow-up was 15.5 (4.0–38.0) months. The median age at disease onset was 24.0 (18.0–32.0) years. The most common disease location was L3 (53.8%), followed by L1 (24.4%) and L2 (15.9%). Overall, 69.7% of patients had colon-involving disease, while 30.3% had non-colon-involving disease. B1 was predominant in disease behavior (72.5%), in contrast to 18.6% of B2 and only 8.9% of B3. Perianal lesions were present in 26.1% and extraintestinal manifestation in 19.6% of patients.
Baseline characteristics of the study population with Crohn’s disease.

Comparison between colon-involving and non-colon-involving disease
Demographic features and disease phenotype
Table 2 depicts the comparison between colon-involving and non-colonic patients. As shown, patients with colon-involving CD had an earlier disease onset than did those with non-colon-involving CD [median age 23.0 (17.0–30.0) versus 26.0 (19.0–35.0) years, respectively, p = 0.001]. Additionally, higher frequencies of perianal lesions (31.2% versus 14.5%, respectively, p < 0.001) and extraintestinal manifestations (21.8% versus 14.5%, respectively, p = 0.010) were observed in patients with colon-involving disease. However, B2 phenotype was less common when the colon was involved in comparison with when the colon was not involved (16.3% versus 24.0%, respectively, p = 0.005). In the Montreal classification, no differences were observed in the demographic and clinical features between the two types of colonic-involving disease, that is, L2 and L3, except that L2 had a higher chance of B2 phenotype (22.8% versus 14.3%, p = 0.005) (Supplemental Material Table 1 online).
Demographic and clinical features and serum biomarkers of colon-involving and non-colon-involving Crohn’s disease.

CD, Crohn’s disease; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; IQR, interquartile range; PLT, platelets; WBC, white blood cells.
As illustrated in Figure 1, the proportion of patients with remission at the time of diagnosis was significantly lower for patients with colon-involving disease than for those with non-colon-involving disease (24.2% versus 35.3%, p = 0.003), while the proportions of moderate (43.1% versus 27.9%, p < 0.001) and severe disease (4.2% versus 1.0%, p = 0.028) were higher in the colon-involving group compared with the non-colon-involving group. In contrast, a lower proportion of mild activity was observed in L2 compared with L3 (20.4% versus 30.7%, p = 0.036) but not in other grades of disease activity (Supplemental Figure 1).
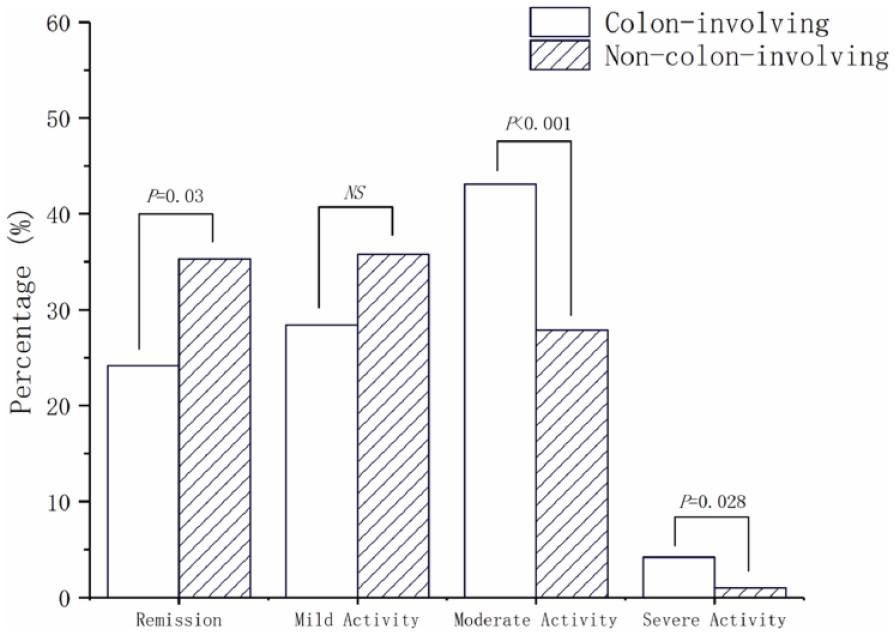
Disease activity between patients with colon-involving and non-colon-involving Crohn’s disease at the time of diagnosis.
The levels of serum inflammatory biomarkers at the time of diagnosis summarized in Table 2 indicate higher inflammatory activity in colon-involving than in non-colon-involving disease. Compared with patients with non-colon-involving CD, those with colon-involving CD had higher levels of white blood cells [7.9 (6.4–9.9) versus 6.7 (5.3–8.2) × 109/L, p < 0.001], platelets [349.0 (284.0–428.0) versus 295.0 (243.0–356.0) × 109/L, p < 0.001], erythrocyte sedimentation rate [43.1 (26.0–66.0) versus 26.0 (12.0–43.1) mm/h, p < 0.001], and C-reactive protein (CRP) [14.6 (9.3–28.3) versus 9.1 (2.6–15.0) mg/L, p < 0.001] but a lower level of hemoglobin [114.0 (97.0–128.0) versus 123.0 (105.0–137.0) g/L, p < 0.001]. In the Montreal classification, no differences were observed between L2 and L3 in serum inflammatory biomarkers (Supplemental Table 1).
Surgery
A total of 741 patients had a follow-up period of more than 3 months, including 523 patients with colon-involving and 218 with non-colon-involving disease. The median follow-up period was not significantly different between the two groups [22.0 (9.0–45.0) versus 24.0 (11.0–47.0) months, p = 0.653].
Patients with colon-involving disease had a lower risk of major abdominal surgery than did those with non-colon-involving disease during the follow-up (15.9% versus 23.4%, p = 0.015). The cumulative probability of first surgery in those with colon-involving disease was lower than that in those with non-colon-involving disease (p = 0.014) (Figure 2). In the multivariate Cox’s regression analysis, colon-involving disease was still an independent protective factor for surgery [hazard ratio, 0.689; 95% confidence interval (CI), 0.481–0.985; p = 0.041] (Table 3).

Kaplan–Meier curves for surgery-free survival between patients with colon-involving and non-colon-involving Crohn’s disease during follow-up.
Risk factors for major abdominal surgery (colon-involving versus non-colon-involving Crohn’s disease).

CI, confidence interval; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; Hb, hemoglobin; PLT, platelets; WBC, white blood cells.
In the Montreal classification, disease location was not a predictor of major abdominal surgery in Cox’s regression analysis (Supplemental Table 2). Kaplan–Meier curves did not demonstrate differences between the different disease locations (Supplemental Figure 2).
Medications
Overall, 529 patients who had more than 1 year of follow-up were analyzed for the use of steroids, AZA/6-MP, and infliximab, the only biologics available in China during the period of data collection (Supplemental Table 3). Of them, 368 patients had colon-involving disease and 161 had non-colon-involving disease, with an overall mean follow-up period of 34.0 (21.0–57.0) months versus 35.0 (21.0–52.0) months, respectively (p = 0.677).
During the follow-up, the percentage of patients with colon-involving CD who required steroids was significantly higher than that of those with non-colon-involving disease (44.8% versus 30.4%, p = 0.002). Additionally, multivariate analysis demonstrated that colon-involving disease was an independent risk factor for steroid use [odds ratio (OR), 1.793; 95% CI, 1.206–2.666; p = 0.004] (Table 4). In the Montreal classification, L3 was an independent risk factor for steroid use when L1 served as the reference in the multivariate analysis (OR, 1.711; 95% CI, 1.105–2.650; p = 0.016) (Supplemental Table 4); however, no significance could be observed when L2 was used as the reference (OR, 1.145; 95% CI, 0.691–1.898; p = 0.599).
Risk factors for steroid use during follow-up (colon-involving versus non-colon-involving Crohn’s disease).

CI, confidence interval.
The proportion of patients with colon-involving CD who required AZA/6-MP treatment was higher than the proportion among patients with non-colon-involving disease (81.8% versus 70.8%, p = 0.005). Colon-involving disease was an independent risk factor for AZA/6-MP use in the multivariate analysis (OR, 1.732; 95% CI, 1.103–2.719; p = 0.017) (Table 5). Compared with L1, L3 was an independent risk factor for AZA/6-MP use (OR, 1.689; 95% CI, 1.104–2.814; p = 0.044) (Supplemental Table 5) but not when compared with L2 (OR, 1.489, 95% CI, 0.797–2.782, p = 0.212).
Risk factors for azathioprine/6-mercaptopurine use during follow-up (colon-involving versus non-colon-involving Crohn’s disease).
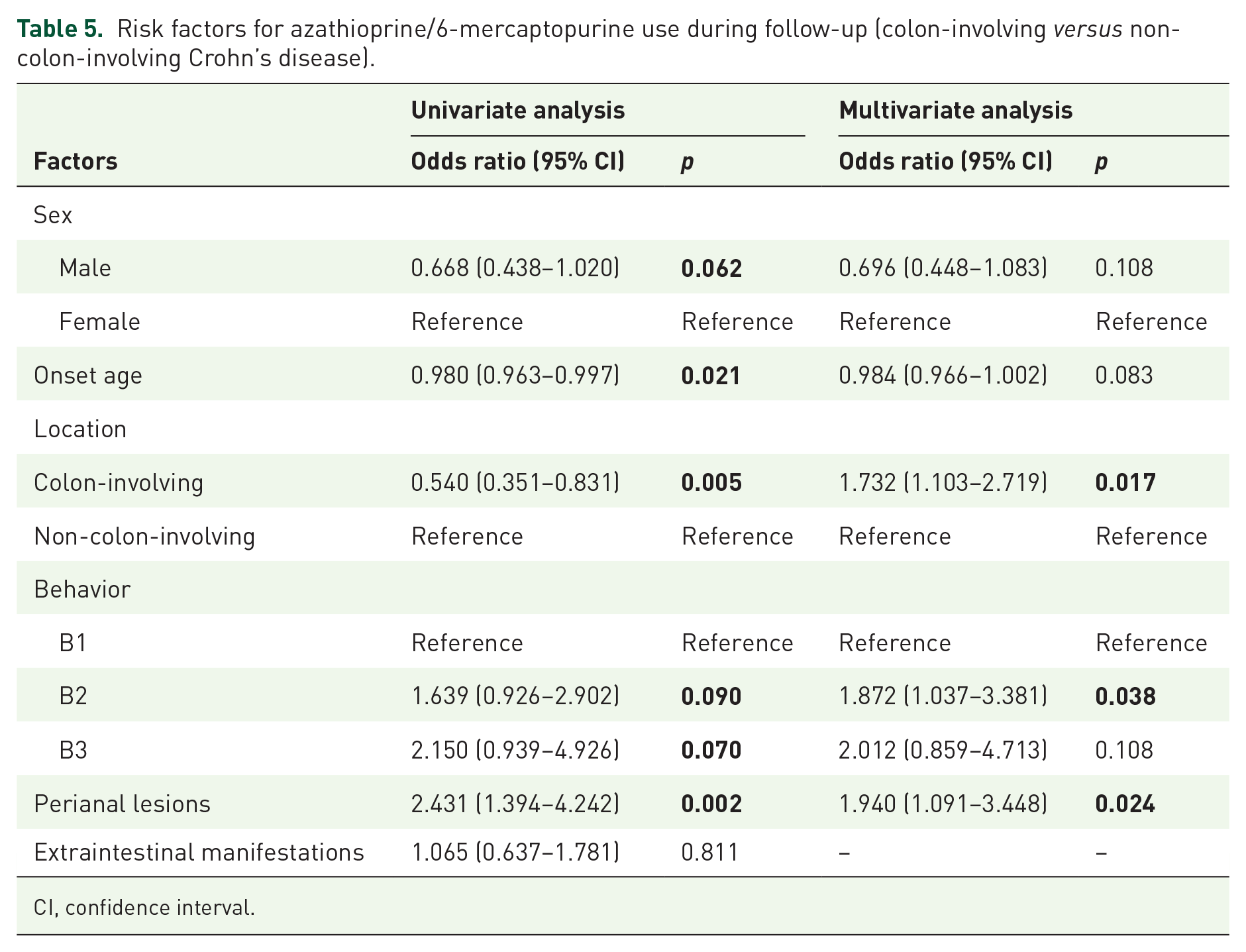
CI, confidence interval.
Although the proportion of patients who required infliximab was higher in those with colon-involving CD than in those with non-colon-involving CD (26.6% versus 17.4%, p = 0.022), colon involvement was not a predictor for the use of infliximab (OR, 1.418; 95% CI, 0.870–2.311; p = 0.161) (Supplemental Table 6). Similar results were observed in the Montreal classification; disease location was not an independent predictor of infliximab use (Supplemental Table 7).
Discussion
Practical classification of CD remains a challenge despite the use of the conventional Montreal classification. The differences in disease locations have been demonstrated in earlier studies. 10 Gene expression profiles were different between colon-involving disease and non-colon-involving disease, such as the association of JAK2 variations with colon disease and NOD2 mutations with ileal disease.11–14
Extensive research has been carried out on the differences in the genetic phenotypes between colon-involving and non-colon-involving CD. Nonetheless, very little is available in the literature with respect to comparisons of disease behavior and activity based on colonic involvement, which was the focus of the present study. For disease behavior, stricture is considered one of the most common complications of CD, which influences the quality of life and may ultimately lead to bowel resection. The results in this study indicated a lower rate of stricture phenotype (B2) in those with colon-involving CD than in those with non-colon-involving CD. This finding was consistent with the study by Louis et al. which reported an association between the development of stricture and disease locations. 15 In accordance with previous reports,16–18 a higher percentage of perianal lesions was observed in patients with colon-involving disease in this study. Two previous studies that investigated the inflammation burden of CD in patients with different disease locations demonstrated significantly higher CRP levels in those with colon-involving disease; however, they did not report on the disease activity.19,20 We found higher levels of serum inflammatory markers in patients with colon-involving disease, which is in agreement with results in these studies.19,20 It could, therefore, be suggested that inflammation burden is higher and more symptoms develop when the colon is involved.
A 10-year prospective study previously reported that patients with colon-involving disease had a lower risk for surgery in comparison with those with non-colon-involving disease (terminal ileum only). 21 Similarly, the results of this study demonstrated that colon-involving disease was a protective factor for surgery, while male sex, B2 and B3 locations were risk factors. They also supported the results of other studies that reported that colon involvement was a protective factor against bowel resection.22,23 It is interesting to note that there is a contrast between a relatively higher level of disease activity and lower surgery risk in colon-involving disease. One possible explanation for this is the changing pattern of disease behaviors. Although disease locations tend to be stable in the course of CD, disease behaviors change, especially in non-colon-involving disease, which is more likely to develop strictures over time,24,25 which may also be related to a possible delay in diagnosis of small bowel disease, less often witnessed with colon-involving CD. 26 Diagnostic delay could in turn potentially lead to an increased risk for surgery in non-colon-involving disease. 27
Other than surgery, the need for steroids, immunosuppressants, and biologicals is another indicator of the prognosis in CD. Consistent with prior findings, we found that the likelihood of AZA/6-MP use in colon-involving disease was higher than that in non-colon-involving disease. However, the same previous study suggested that colon-involving disease had a lower probability of steroid use than non-colon-involving disease, 28 which was contrary to the results of the present study. This discrepancy may be due to the relatively short duration of follow-up in this study. In the present study, there was no significant difference in the use of infliximab between colon-involving and non-colon-involving disease, which may be attributable to the relatively small sample size of patients using infliximab.
We also compared the demographic and clinical features between the different types of CD according to the conventional Montreal classification. 8 Specifically, we could not find significant differences between L2 and L3 phenotypes. Moreover, long-term prognosis, assessed by the risk of surgery and the use of steroids or immunosuppressants, was not different between L2 and L3, thereby questioning the relevance of these two sub-classifications within the Montreal methodology for determining clinically important outcomes. These observations suggest the possible need to consider further modifications of the conventional Montreal classification.
This study has certain limitations. First, although the data were collected with a standardized follow-up schedule, it was a single-center retrospective study from a tertiary hospital; therefore, selection bias might be inherent in the study design. A multi-center study with a prospective design in the future is needed. Second, we used CDAI to assess the disease activity at the time of diagnosis, which might also have resulted in some bias. Third, the serum biomarkers used in the assessment of the inflammatory burden mostly reflect the systematic inflammatory conditions. It is beyond the scope of this study to determine the exact inflammatory burden of the involved intestine. Specific markers of intestinal inflammation, such as calprotectin, should be included in future studies. 29
Conclusion
This study demonstrates that classifying CD into colon-involving and non-colon-involving disease is more relevant to the disease activity, disease behavior, risk of surgery, and the use of medication than is the Montreal classification. This supports the findings of previous studies on the differences in the genetic expression and microbiota between colon-involving and non-colon-involving disease. Further multi-center and large-scale investigations are needed to verify these findings.
Supplemental Material
sj-pdf-1-tag-10.1177_1756284820968732 – Supplemental material for Classifying Crohn’s disease into colon-involving versus non-colon-involving groups is a better predictor of clinical outcomes than the Montreal classification
Supplemental material, sj-pdf-1-tag-10.1177_1756284820968732 for Classifying Crohn’s disease into colon-involving versus non-colon-involving groups is a better predictor of clinical outcomes than the Montreal classification by Si-Nan Lin, Dan-Ping Zheng, Yun Qiu, Sheng-Hong Zhang, Yao He, Bai-Li Chen, Zhi-Rong Zeng, Ren Mao and Min-Hu Chen in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-1-tag-10.1177_1756284820968732 – Supplemental material for Classifying Crohn’s disease into colon-involving versus non-colon-involving groups is a better predictor of clinical outcomes than the Montreal classification
Supplemental material, sj-tif-1-tag-10.1177_1756284820968732 for Classifying Crohn’s disease into colon-involving versus non-colon-involving groups is a better predictor of clinical outcomes than the Montreal classification by Si-Nan Lin, Dan-Ping Zheng, Yun Qiu, Sheng-Hong Zhang, Yao He, Bai-Li Chen, Zhi-Rong Zeng, Ren Mao and Min-Hu Chen in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-tif-2-tag-10.1177_1756284820968732 – Supplemental material for Classifying Crohn’s disease into colon-involving versus non-colon-involving groups is a better predictor of clinical outcomes than the Montreal classification
Supplemental material, sj-tif-2-tag-10.1177_1756284820968732 for Classifying Crohn’s disease into colon-involving versus non-colon-involving groups is a better predictor of clinical outcomes than the Montreal classification by Si-Nan Lin, Dan-Ping Zheng, Yun Qiu, Sheng-Hong Zhang, Yao He, Bai-Li Chen, Zhi-Rong Zeng, Ren Mao and Min-Hu Chen in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We thank all the members in IBD-MDT group of The First Affiliated Hospital, Sun Yat-sen University for their help in collecting data and clinical care of patients. We also thank all the patients for their kind participation.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the National Natural Science Foundation of China (grant number 81970483).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
