Abstract
Background:
Esophageal distensibility, measured by endoluminal functional lumen imaging probe (EndoFLIP), identifies esophageal fibrostenotic changes and is impaired in patients with eosinophilic esophagitis (EoE). Early-phase clinical trials suggested dupilumab could increase esophageal distensibility. However, there are limited data on the long-term impact of dupilumab treatment on fibrostenosis.
Objectives:
To evaluate the long-term efficacy of dupilumab, including its impact on esophageal fibrostenotic progression, in adult patients with EoE.
Design:
The phase IV study is comprised of a randomized, double-blind, placebo-controlled trial period for 24 weeks, followed by an open-label extension for 104 weeks.
Methods and analysis:
In total, 69 adult patients with endoscopically and histologically active EoE have been recruited from 30 global sites and randomized 2:1 to receive dupilumab 300 mg once weekly (qw) or placebo during the double-blind treatment period. Eligible patients continuing into the open-label extension period will receive dupilumab 300 mg qw. The primary endpoint is absolute change from baseline in esophageal distensibility plateau at week (W)24 measured by EndoFLIP. Secondary endpoints include change in esophageal distensibility plateau at W76 and W128; histologic, endoscopic, and molecular features of EoE at W24, W76, and W128; and long-term safety. After the double-blind treatment period, endpoints will be summarized with descriptive statistics.
Ethics:
REMODEL will be conducted in accordance with the Declaration of Helsinki, the Council for International Organizations of Medical Sciences international ethical guidelines, and the International Council for Harmonisation Good Clinical Practice guidelines. The protocol was approved by an institutional review board before study initiation.
Discussion:
REMODEL will address whether long-term dupilumab treatment can mitigate fibrostenotic progression in patients with EoE and may provide new insights into the roles of interleukin-4 and -13 in the pathophysiology and progression of EoE.
Trial registration:
All patients will provide informed consent. REMODEL was registered on ClinicalTrials.gov (NCT06101095) on October 19, 2023.
Plain language summary

Introduction
Eosinophilic esophagitis (EoE) is a chronic, progressive disease characterized by esophageal eosinophilia and symptoms associated with esophageal dysfunction, and is rising in incidence and prevalence.1,2 EoE is driven by type 2 inflammation, a specific immune response involving pro-fibrotic and homeostatic signaling via the key type 2 cytokines interleukin (IL)-4, IL-5, IL-13, and IL-33, and the actions of activated mast cells, basophils, eosinophils, and pro-fibrotic macrophages.3,4 If left untreated, persistent uncontrolled type 2 inflammation contributes to pathologic fibrosis and epithelial remodeling,4 –6 leading to the development of a fibrostenotic EoE phenotype.7 –9 Fibrostenosis causes the clinical manifestations and complications of EoE, including dysphagia, strictures, and food impactions, 10 that can significantly impact patients’ quality of life (QoL). 11
Clinical guidelines for EoE recommend intervention with an empirical food elimination diet and/or pharmacologic therapies such as swallowed topical corticosteroids (STCs), proton-pump inhibitors (PPIs), and biologics. 12 While capable of inducing histologic remission in a proportion of patients with EoE, loss of response and relapse are common with PPIs and STC drugs.13,14 Dupilumab, a fully human monoclonal antibody that blocks the shared receptor component for IL-4 and IL-13, is approved in the USA and EU for the treatment of patients with EoE aged ⩾1 year, weighing ⩾15 kg. 15 Guidance on the use of dupilumab for EoE, particularly in patients with PPI-refractory disease, 12 is based on results from the pivotal phase III LIBERTY EoE TREET and EoE KIDS studies, which show improvements in histologic, symptomatic, and endoscopic features.16,17 Once EoE treatment has achieved control of underlying inflammation, maintenance therapy should be continued to prevent relapse. 18 Throughout treatment, in addition to determining patient symptom response, ongoing endoscopic and biopsy-based histologic monitoring is required to ensure effective management of eosinophilic mucosal inflammation. However, detection of esophageal strictures in EoE can be difficult via endoscopy. Although esophageal narrowing is obvious when the esophageal lumen is constricted and passage of a standard endoscope is more difficult, endoscopy is less sensitive than other tools for accurate assessment of esophageal diameter. 19 Endoluminal functional lumen imaging probe (EndoFLIP) is a sensitive method that can be used to assess fibrostenotic changes in EoE 12 and to quantify the esophageal distensibility plateau. Defined as the narrowest diameter along the esophagus that demonstrates resistance to distention, the esophageal distensibility plateau provides a quantitative assessment of esophageal remodeling 20 and has been shown to be significantly impaired in patients with EoE.21,22
There are limited data on how effective therapies may be at preventing fibrotic remodeling of the esophagus in EoE. 14 In a phase II study, compared with placebo, dupilumab was associated with increased esophageal distensibility at 12 weeks, measured by EndoFLIP in an exploratory endpoint. 23 However, there is a need for more data to determine the efficacy of dupilumab in reducing inflammation and preventing and/or reducing fibrostenosis over longer treatment periods. The REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term (REMODEL) trial aims to assess the long-term efficacy of dupilumab, including its impact on esophageal function and remodeling, in adult patients with EoE (ClinicalTrials.gov identifier: NCT06101095). The primary objective of REMODEL is to assess the efficacy of dupilumab in improving the esophageal distensibility plateau. The secondary objectives of the study include determining the long-term effect of dupilumab on esophageal function and histologic, endoscopic, and molecular features of EoE.
Methods
Study design
REMODEL is a multicenter, multinational, randomized, placebo-controlled, phase IV trial with a long-term open-label extension (Figure 1) that has recruited across 30 sites in Brazil, Canada, Israel, Switzerland, and the USA. Adult patients were eligible for enrollment if they had endoscopically and histologically active EoE with evidence of dysphagia symptoms at screening. Key eligibility criteria are detailed in Table 1.

REMODEL study design.
REMODEL key inclusion and exclusion criteria.
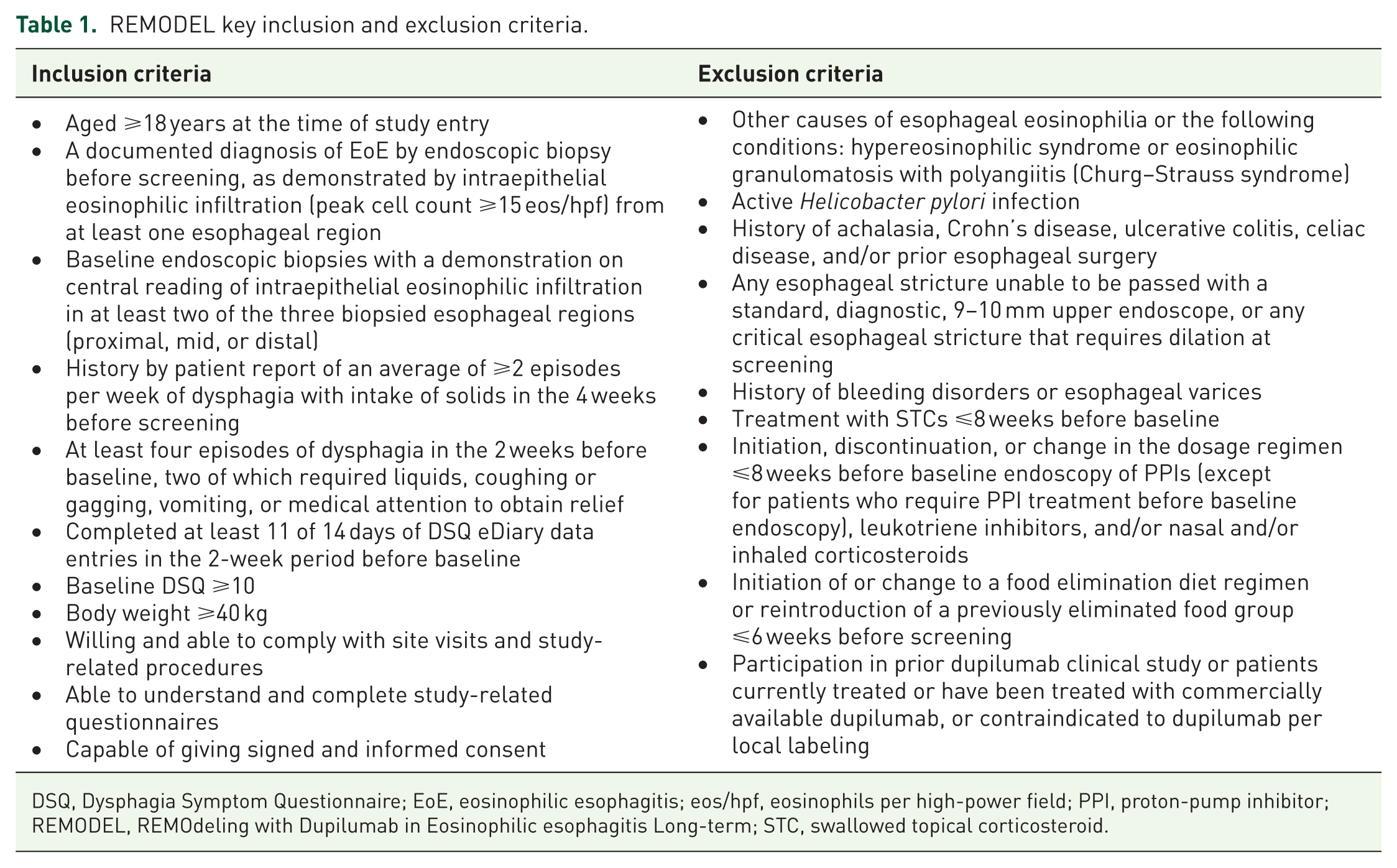
DSQ, Dysphagia Symptom Questionnaire; EoE, eosinophilic esophagitis; eos/hpf, eosinophils per high-power field; PPI, proton-pump inhibitor; REMODEL, REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term; STC, swallowed topical corticosteroid.
The study was preceded by an up to 12-week screening period during which baseline characteristics were recorded, such as patient demographics, medical history including current and/or history of treatment and existing comorbidities, and molecular biomarkers. For further information regarding prohibited concomitant and historic therapies, see the Supplemental Appendix. Following screening, eligible patients were randomized 2:1 to receive dupilumab or a matching placebo for 24 weeks. Concealed treatment randomization was stratified by dilation history and performed centrally using an interactive response technology. The interactive response technology telephone number and/or log-in information were provided along with directions for use to each site prior to activation.
During the 24-week double-blind treatment period, all patients, care providers, and investigators will be blinded to the study intervention. To achieve blinding, study patients, care providers, and investigators will not have access to the randomization list, and study drugs will be visually indistinguishable. If rescue treatment is medically necessary, and at the discretion of the investigator, patients can switch to open-label treatment or receive emergency dilation, and the patient’s intervention will be unblinded before rescue. The investigator will have sole responsibility for determining if unblinding of a patient’s intervention assignment is warranted, with patient safety being the first consideration in making the decision. Sponsor staff may unblind the intervention assignments for any patient with a serious adverse event. The study will be unblinded at the first early analyses, when all patients complete the 24-week treatment period, or upon early discontinuation from the study (see Supplemental Methods for further information). The 24-week, randomized, double-blind, placebo-controlled period will be followed by an open-label period of 104 weeks in which all patients will receive dupilumab. Recruitment for REMODEL ended in May 2025, and the primary data readout of the double-blind, placebo-controlled treatment period is anticipated in December 2025.
Study interventions
For the first 24 weeks, patients will be randomized to receive either dupilumab 300 mg once weekly (qw), corresponding with the US Food and Drug Administration-approved dupilumab dosing in adults with EoE, or placebo. 16 During the open-label period, all patients will receive dupilumab 300 mg qw. Dupilumab 300 mg and placebo will be provided in identically matched, 2 mL prefilled syringes and will be administered by subcutaneous injection, which, after appropriate training, can be administered by the patient at home. Patients’ adherence to treatment will be monitored during the study by site investigators (see Supplemental Methods).
Assessments
Baseline assessments are taken on day 1 of the study or, in the case of measures obtained via endoscopy, taken during screening and up to 21 days before study initiation. All primary, secondary, and exploratory endpoints assessed throughout the study are listed in Table 2. The primary objective and endpoint of the study will be to assess absolute change from baseline in esophageal distensibility plateau as measured by EndoFLIP at week 24. The secondary objective will be to assess long-term effects of dupilumab on esophageal function by measuring the percent change from baseline in esophageal distensibility plateau as measured by EndoFLIP at week 24 (the key secondary endpoint) and absolute and relative change from baseline in esophageal distensibility plateau at weeks 76 and 128. Additional secondary objectives will be to assess the long-term effects of dupilumab on histologic, endoscopic, and molecular features of EoE, measuring endpoints at weeks 24, 76, and 128. The long-term safety of dupilumab for the treatment of EoE will also be assessed as a secondary objective. Incidents of adverse events of special interest, serious adverse events, and treatment-emergent adverse events will be monitored throughout the study. Exploratory objectives will be to assess the long-term effect of dupilumab on disease modification, the need for rescue treatments, peak eosinophil count, gene and protein expression, and symptoms and QoL at weeks 24, 76, and 128.
Primary and secondary endpoints assessed throughout REMODEL.

AESI, adverse event of special interest; EDP, EoE Diagnostic Panel; EndoFLIP, endoluminal functional lumen imaging probe; EoE, eosinophilic esophagitis; EoE-EREFS, EoE‑Endoscopic Reference Score; EoE-HSS, EoE histology scoring system; EoE-QoL-A, adult EoE QoL questionnaire; eos/hpf, eosinophils per high-power field; HADS, Hospital Anxiety and Depression Scale; HCRU, healthcare resource utilization; NES, normalized enrichment score; QoL, quality of life; REMODEL, REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term; SAE, serious adverse event; TEAE, treatment-emergent adverse event.
Endpoint measures
During endoscopy, esophageal distensibility will be assessed using EndoFLIP 3.0 (Medtronic, Minneapolis, MN, USA) by both local and central reading; the central read will be used for the endpoint data. The EndoFLIP device is a catheter-based procedure that measures the cross-sectional area at multiple sites along the esophagus with simultaneous intraluminal pressure recordings during volumetric distension of the esophagus. The analyses of cross-sectional area versus pressure relationships of the esophagus allow for the determination of esophageal compliance as well as the distensibility plateau, defined here as the narrowest esophageal lumen diameter that fails to expand despite increasing intra-balloon measures. The device consists of a 240 mm-long, 3 mm outer diameter catheter, with a 16 cm balloon mounted on the distal end (EF-322N; Medtronic). The catheter within the balloon contains 16 paired impedance planimetry electrodes spaced at intervals of 1 cm. The balloon is filled with a conductive fluid via an 80 mL syringe, and serial measurements are taken, starting from 30 mL, with 10 mL step-ups to 40 mL, 50 mL, 60 mL, and finally to a max fill of 70 mL. Esophageal distensibility is calculated based on simultaneous measurement of pressure within the balloon by the solid-state transducer.24,25 EndoFLIP measurements for the calculation of the esophageal distensibility plateau will be taken at the distal region of the esophagus only.
Additional endoscopic features of EoE will be assessed using the EoE-Endoscopic Reference Score (EoE-EREFS), a validated scoring system that assesses the inflammatory and remodeling features of EoE using an overall score and scores for each individual characteristic (edema, rings, exudates, furrows, and strictures). 26 The EoE-EREFS will be performed by both the investigators performing the endoscopies and a centralized reading center using endoscopy imaging. Proximal and distal esophageal regions will be scored separately (range 0–9) using EoE-EREFS, and the overall score calculated from the sum of both regions (range 0–18). During endoscopy, esophageal epithelial impedance will be measured using mucosal impedance analysis (MiVu; Diversatek Healthcare, Milwaukee, WI, USA), which provides real-time, quantitative assessment of epithelial integrity and esophageal barrier function. 27 A total of 12 mucosal pinch biopsies will be collected during endoscopy, from three esophageal regions: four proximal, four mid, and four distal. Three samples from each region will be used for histology and other tissue analyses, including but not limited to immunohistochemistry, RNA in situ hybridization, RNA sequencing, and spatial transcriptomic/proteomic analysis. Any remaining tissue samples will be banked. Biopsy samples will be processed locally and sent to a central laboratory (Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA) to be assessed for peak eosinophil counts per high-power field, spatial biomarker analyses (transcriptomic and proteomic) bulk RNA sequencing analysis, and histologic severity using the EoE histology scoring system, a validated scoring system used to measure the severity (grade) and extent (stage) of histopathologic features in EoE. 28
The impact of disease on patient health-related QoL will be assessed using the adult EoE QoL questionnaire, a validated and EoE-specific patient-reported outcome (PRO) tool11,29 that evaluates the impact of disease on several domains, including social and emotional functioning and daily life experiences. 30 Changes in anxiety and depression experienced by patients will be assessed using the Hospital Anxiety and Depression Scale, a validated PRO tool used extensively across multiple diseases to screen for symptoms specific to anxiety and depression regardless of disease state. 31
Ethical considerations and data collection
REMODEL will be conducted in accordance with (i) consensus ethical principles derived from international guidelines, including the Declaration of Helsinki and the applicable amendments 32 and Council for International Organizations of Medical Sciences international ethical guidelines, 33 and (ii) applicable regulatory requirements in accordance with the International Council for Harmonisation Good Clinical Practice E6 (R2) guidelines. 34 The protocol and other relevant documents were submitted for institutional review board (IRB) review and were approved before the study was initiated. Any substantial amendments to the protocol will require health authority and IRB/independent ethics committee (IEC) approval before implementation, except for changes necessary to eliminate an immediate hazard to study patients. The reporting of this study conforms to the SPIRIT statement (Supplemental Appendix). 35
During enrollment, patients were informed that their participation was voluntary and were required to sign a statement of informed consent that meets the requirements of 21 Code of Federal Regulations 50, local regulations, International Council for Harmonisation guidelines, privacy and data protection regulations, and the requirements of the IRB/IEC and/or study center. The informed consent form contains two separate sections, each subject to independent consent, that address the use of patients’ data and/or samples for future research. Written consent was obtained by the investigator or an authorized representative. Data will be collected and stored in accordance with the General Data Protection Regulation and any other applicable data protection laws (details related to data access and dissemination are found in the Supplemental Methods).
A placebo arm consisting of patients who may be exposed to background therapies, including PPIs and/or dietary therapy, was deemed necessary to assess natural disease progression and robustly measure dupilumab treatment effects while controlling for other factors, for example, background treatment adherence. A randomization ratio of 2:1 for the active treatment versus placebo was used to limit the number of patients exposed to placebo and avoid excessive and unjustified burden on patients. All patients were appropriately informed and provided written consent for the potential eventuality of receiving a placebo and may withdraw from the study intervention or from the study at any time. Furthermore, if medically necessary and at the discretion of their physician, patients may receive other treatments, switch to open-label treatment, and/or have emergency dilation at any time.
Statistical analysis
The study has recruited 69 adult male and female patients with active EoE. Assuming a 10% dropout rate by week 24, this sample size will provide 90% power with a 0.05 two-sided significance level to detect a treatment difference (standard deviation) of 2.3 (2.42) mm in esophageal distensibility between dupilumab and placebo groups. This design is based on a conservative prediction of potential treatment effect, powered to detect a value that is less than that seen in the phase II trial. 23 Baseline values are defined as the last available value before the first dose of the double-blind study drug. The primary endpoint will be compared between the dupilumab and placebo groups using an analysis of covariance model, with dilation treatment history and baseline EndoFLIP measurements as covariates. For patients who undergo rescue treatment for EoE, data collected after rescue treatment will be set to missing and imputed by multiple imputation. For patients who discontinue the study drug without having rescue treatment, the data collected after treatment discontinuation will be included in the analysis. If a patient still has missing data after all efforts, the missing data will be imputed by multiple imputation. Statistical inference obtained from all imputed data will be combined using Rubin’s rule. Binary secondary endpoints at week 24 will be analyzed using the Cochran–Mantel–Haenszel test to assess the difference in the proportion of responders, adjusting for dilation history as the randomization stratification factor. Continuous secondary endpoints at week 24 will be analyzed using the analysis of covariance model in the same fashion as for the primary endpoint. After the double-blind treatment period of the study, all efficacy endpoints will be summarized with descriptive statistics. Early analysis is planned at week 24.
Discussion
REMODEL is a parallel-group, phase IV study that consists of a 24-week, randomized, double-blind, placebo-controlled treatment period followed by an open-label period during which all patients receive dupilumab for 104 weeks. Previous phase III dupilumab clinical studies established the link between treating type 2 inflammation and achieving durable (up to 52 weeks) improvements in histologic, symptomatic, endoscopic, and molecular (transcriptomic) features of EoE. 16 REMODEL aims to address an important data gap—that is, to explore whether long-term treatment of type 2 inflammation potentially modifies fibrostenotic progression of EoE. Addressing this data gap could have implications for whether treatment could prevent or modify overall EoE disease progression.
Esophageal eosinophilia often does not correlate with symptoms or endoscopic features of EoE, including esophageal distensibility, 36 nor is it a predictor for the development of clinical complications. 37 A more comprehensive evaluation of EoE beyond eosinophil counts to determine disease extent and response to treatment is therefore required. 38 In addition to its application in research, EndoFLIP has the potential to provide valuable clinical information in EoE. 25 Decreased esophageal distensibility, as measured by EndoFLIP, has been shown to be associated with an increased risk of clinical complications, including food impaction and the requirement for dilation in patients with EoE. 25 As a measure of fibrostenotic disease, EndoFLIP may provide information relevant to risks for clinical complications that have been shown to directly impact patient QoL. 11 Demonstrating prevention of fibrostenotic progression and reversal of existing fibrosis in EoE is therefore a key target for long-term management of the disease. Dupilumab has previously been shown to have the potential to reverse fibrosis in EoE, increasing esophageal distensibility in a phase II study. 23 In REMODEL, EndoFLIP will be used to provide a quantitative assessment of esophageal fibrosis and rigidity in response to dupilumab treatment compared with placebo, and may therefore provide novel evidence of an EoE therapy that can reverse and modify disease progression.
In addition to the primary aims of the study, REMODEL will assess several secondary endpoints to build upon findings from previous studies, including the effect of dupilumab on epithelial and transcriptomic endpoints associated with EoE disease activity. Growing evidence suggests that type 2 inflammation may contribute to impaired barrier function of the esophageal epithelium, ultimately contributing to esophageal remodeling.10,39 Several genes in the epidermal differentiation complex, of which down-regulation by type 2 cytokines results in barrier dysfunction of the skin, are also down-regulated in primary esophageal epithelial cells in vitro. 39 Furthermore, a reduced expression of the epidermal differentiation complex gene FLG and the esophagus-specific esophagin (small proline-rich protein 3) was observed in esophageal biopsies from patients with EoE compared with controls. 39 Expression of these genes was normalized in the phases II and III trials of dupilumab for the treatment of EoE. 40 This suggests that, in addition to driving an inflammatory response, type 2 cytokines may directly contribute to epithelial barrier dysfunction in the esophagus, indicating that treatment targeting type 2 inflammation has the potential to address esophageal epithelial remodeling and improve barrier function and consequently esophageal fibrosis. Data from REMODEL may help to further elucidate the molecular mechanisms linking type 2 inflammation with esophageal remodeling and fibrostenotic development in EoE.41,42 Furthermore, REMODEL will provide novel data through its application of MiVu to measure mucosal impedance, which could be used to assess the correlation of mucosal integrity with endoscopy, EndoFLIP, and spatial biomarkers—analyses that have not previously been explored in EoE.
Additional secondary endpoints of REMODEL will provide data for the long-term efficacy of dupilumab in improving histologic, symptomatic, endoscopic, and molecular (transcriptomic) features of EoE by assessing its efficacy over 128 weeks (~2.5 years). This study will also provide long-term safety data in EoE, adding to the years of follow-up data for dupilumab in other type 2 inflammatory diseases.43,44 In addition, REMODEL will assess disease-related anxiety and depression, endpoints that are not often assessed in EoE clinical trials, despite patients being at an increased risk of decreased mental health. 45
Limitations
EndoFLIP is not often used in clinical practice, 12 and therefore, standardization of the procedure in patients with EoE is not well established. REMODEL may provide evidence to help standardize this process and support the use of EndoFLIP for patient monitoring, particularly in patients with fibrostenotic disease. Fibrotic remodeling in EoE likely progresses over substantial periods of time,7,8,10 with observational data indicating that increased EoE duration is directly correlated with a fibrostenotic phenotype. 9 Clinically significant fibrostenosis may therefore take longer to develop than the study duration, limiting potential conclusions regarding disease modification. In addition, stipulating evidence of dysphagia as an eligibility criterion limits the study population to patients who likely have established fibrostenotic disease. Although this population is likely to derive treatment benefit, this may preclude assessment of dupilumab in preventing the initial stages of fibrostenotic progression. Data collected during the extended treatment period may be influenced by open-label bias, particularly in PROs. However, evidence suggests that the potential for bias should not prevent the interpretation of potentially meaningful treatment effects. 46
Conclusion
REMODEL aims to build upon findings from previous dupilumab EoE studies, providing long-term data for the efficacy and safety of dupilumab to improve histologic, endoscopic, and molecular features of EoE. In addition, the primary endpoint of REMODEL is novel and will assess the impact of dupilumab on fibrostenotic progression and esophageal remodeling in patients with EoE, exploring the potential for long-term EoE disease modification with dupilumab. The study may also increase our understanding of the roles of IL-4 and IL-13 in fibrostenotic progression of EoE and provide valuable insight into anxiety and depression experienced by patients with EoE. Recruitment for REMODEL ended in May 2025, and the estimated primary completion date is December 2025.
Supplemental Material
sj-doc-3-tag-10.1177_17562848251383782 – Supplemental material for Study design of the phase IV, randomized, placebo-controlled REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term (REMODEL) trial
Supplemental material, sj-doc-3-tag-10.1177_17562848251383782 for Study design of the phase IV, randomized, placebo-controlled REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term (REMODEL) trial by Evan S. Dellon, Edoardo V. Savarino, Sherif Zaghloul, James T. Angello, Mei Zhang, Bram P. Raphael, Amr Radwan and Albert J. Bredenoord in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251383782 – Supplemental material for Study design of the phase IV, randomized, placebo-controlled REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term (REMODEL) trial
Supplemental material, sj-docx-2-tag-10.1177_17562848251383782 for Study design of the phase IV, randomized, placebo-controlled REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term (REMODEL) trial by Evan S. Dellon, Edoardo V. Savarino, Sherif Zaghloul, James T. Angello, Mei Zhang, Bram P. Raphael, Amr Radwan and Albert J. Bredenoord in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-1-tag-10.1177_17562848251383782 – Supplemental material for Study design of the phase IV, randomized, placebo-controlled REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term (REMODEL) trial
Supplemental material, sj-pdf-1-tag-10.1177_17562848251383782 for Study design of the phase IV, randomized, placebo-controlled REMOdeling with Dupilumab in Eosinophilic esophagitis Long-term (REMODEL) trial by Evan S. Dellon, Edoardo V. Savarino, Sherif Zaghloul, James T. Angello, Mei Zhang, Bram P. Raphael, Amr Radwan and Albert J. Bredenoord in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We would like to acknowledge the sad passing of Dr. Ikuo Hirano, who contributed significantly to the gastroenterology community and to the concept of this study and manuscript. Medical writing/editorial support was provided by Emily Prior, MSc, of Adelphi Group, Macclesfield, UK, funded by Sanofi and Regeneron Pharmaceuticals, Inc., according to the Good Publication Practice guidelines.
Declarations
Trial status
Protocol version number 1 (electronic 2.0). Recruitment began on November 29, 2023 and ended in May 2025. The estimated primary completion date is December 2025.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
