Abstract
Background:
Eosinophilic esophagitis (EoE) is a chronic, progressive, immune-mediated disease of the esophagus. Esophageal dysfunction in solid food dysphagia combined with eosinophil-dominant inflammation (⩾15 eos/high-power-field (hpf)) serves as diagnostic criteria. Treatment objectives for active EoE include induction and maintenance of clinical-histological remission. Topical budesonide therapy in the form of an orodispersible tablet (BUD-ODT) has shown effectiveness in both scenarios.
Objectives:
To examine the efficacy and safety of BUD-ODT therapy in adults with active EoE in a Real-World-Setting.
Design:
This retrospective study from two German EoE centers examined the efficacy and safety of BUD-ODT therapy in adults with active EoE between June 2018 and August 2022.
Methods:
Patient demographics, clinical characteristics, and EoE history were extracted from medical records. Evaluations included clinical (Straumann Dysphagia Index, range: 0–9, remission: ⩽3 points), histological (remission: ⩽15 eos/hpf), and endoscopic assessment (endoscopic reference scoring system (EREFS) score, range: 0–9, remission: ⩽2 points). Three periods were analyzed: Induction phase (induction therapy (IT): 1 mg BID, 6–12 weeks; n = 201), maintenance phase I (RM1: 0.5 mg BID or 1 mg QD, 14–24 weeks; n = 109), and maintenance phase II (RM2: same dosage as RM1, 52–104 weeks; n = 72).
Results:
A total of 221 adults with clinical-histologically proven active EoE were included. Clinical-histological remission was achieved in 75.1% (151/201) of patients after IT, maintained in 73.4% (80/109) after RM1, and in 76.4% (55/72) after RM2. The relapse rate was 26.6% (29/109) after RM1 and 23.6% (17/72) after RM2. Those patients underwent re-induction therapy for 6–8 weeks and were re-evaluated separately (remission: 36.0% after RM1 and 46.2% after RM2). Endoscopic remission was demonstrated in 88.1% (177/201) after IT. The most prevalent side effect was endoscopically detected combined oral-esophageal candidiasis.
Conclusion:
BUD-ODT demonstrated high effectiveness in inducing and maintaining remission in a real-world setting. The spectrum of side effects did not reveal any new aspects compared to known clinical data.
Keywords
Introduction
Eosinophilic esophagitis (EoE) is a chronic, progressive, immune-mediated disease of the esophagus characterized by the combination of dysphagia for solid foods and eosinophil (eos)-dominant inflammation of ⩾15 eosinophils per high-power field (eos/hpf).1 –3 Targeted medical and dietary therapies are employed to prevent progression of the disease, as continuous inflammation of the esophageal mucosa can ultimately lead to fibrotic remodeling, resulting in ring formation, esophageal narrowing and strictures that significantly impair solid food intake and quality of life (QoL).4,5 The treatment objectives for active EoE are thus not only to achieve clinical and histological remission but also to maintain it.5,6 For treatment of active EoE, various options are available. Pharmacological therapies include topical corticosteroids, proton pump inhibitor (PPIs), and the recently approved anti Interleukin-4 and -13 monoclonal antibody, Dupilumab.1 –3 Elimination diets represent a nonpharmacological treatment approach. Here, 2-, 4-, and 6-food elimination diets are utilized, systematically removing and reintroducing potentially allergenic foods to identify triggers. 7
In 2018, the first orodispersible tablet containing budesonide (BUD-ODT) was approved for the treatment of EoE in Europe, proving to be highly effective in addressing active EoE.2,8,9 Numerous studies have subsequently demonstrated the efficacy of BUD-ODT in controlled settings in recent years. The double-blind EOS-1 study published in 2020 revealed that 57.6% of patients achieved clinical-histological remission after 6 weeks, and 84.7% after 12 weeks of induction (1 mg BUD-ODT BID). 10 In another 48-week randomized controlled trial (RCT), 73.5% of patients (0.5 mg BUD-ODT BID) and 75.0% (1.0 mg BUD-ODT BID) maintained clinical-histological remission compared with 4.4% of the patients receiving placebo. 11
The aim of this study was to assess the effectiveness and safety of BUD-ODT in adult EoE patients within a real-world setting at two German EoE centers. The evaluation encompassed: (1) clinical-histological remission, (2) symptomatic remission, (3) endoscopic remission, (4) adverse events, and (5) an estimation of the number of topical steroid-refractory EoE patients, who require new advanced treatment options.
Materials and methods
Patient cohort
This was a retrospective cohort study at two secondary care centers in Germany from 06/2018 to 08/2022. Patient demographics, clinical characteristics, EoE history, and procedural data were extracted from patients’ medical records. The study included all adult patients during this time period with a diagnosis of active EoE who were prescribed BUD-ODT only. Active EoE was defined as a prior diagnosis as per consensus guideline criteria: ongoing symptoms indicative of esophageal dysfunction and histologically confirmed eosinophil-predominant inflammation (⩾15 eos/hpf) of the esophagus, with exclusion of other systemic and/or local causes for esophageal eosinophilia. 1 The reporting of this study conforms to the HARmonized Protocol for rEal-world evidence Reporting (HARPER) guideline as stated in Table 3 from the cited publication. 12
Treatment regimen
In accordance with local protocols, patients were initially treated with induction therapy (IT) for 6–12 weeks at a dosage of 1 mg BUD-ODT BID and were subsequently evaluated. Patients who achieved remission proceeded to an initial maintenance therapy (RM1) with 0.5 mg BUD-ODT BID or 1 mg BUD-ODT QD and were subsequently assessed between week 14 and 24 (from the start of IT). For those who maintained remission, their dosage was continued in a second maintenance phase (RM2), and they were evaluated between week 52 and 104. Patients not achieving or maintaining remission in these phases were offered extended IT or re-induction therapy (re-IT), respectively, for 6–8 weeks at a dosage of 1 mg BUD-ODT BID and were evaluated separately (Figure 1). At each evaluation, patients’ clinical condition was assessed, EREFS score was evaluated, and a histological examination of the biopsies was conducted. Compliance was defined as the extent to which a patient’s behavior aligned with the prescribed treatment regimen, including adherence to monotherapy and overall adherence to the treatment plan. A possible use of PPIs or food elimination could not be fully excluded, despite patient instruction and repeated follow-up evaluations.
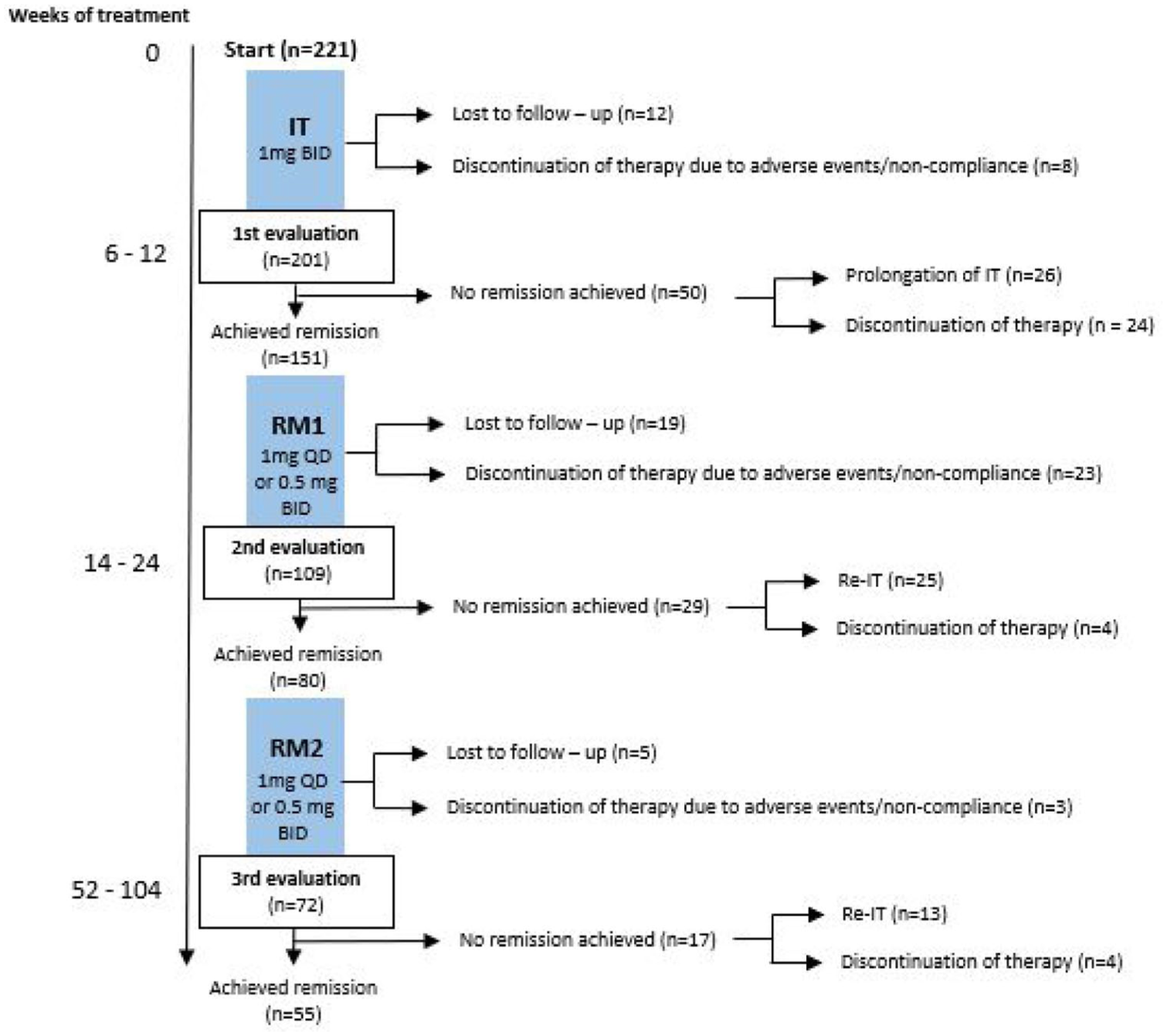
Patient flow chart.
Assessments and endpoints
Clinical assessment and clinical remission
The Straumann Dysphagia Index (SDI) was used for the measurement of dysphagia. It includes the frequency and severity of dysphagic events. The total possible score is 0–9,12,13 and clinical remission was defined as ⩽3 points. Complete absence of symptoms corresponded to an SDI score of 0. The SDI was calculated retrospectively during the data analysis based on information retrieved from patients’ medical reports.
Histological assessment and histological remission
For a standardized histological assessment, a minimum of six graded tissue samples were collected and analyzed during each endoscopic examination, sourced from at least two segments of the esophagus including the proximal region and areas with suspected pathological patterns/findings. 14 The European guideline stipulates that to histologically confirm eosinophilic esophagitis, a threshold of ⩾15 eos/hpf (equivalent to 0.3 mm2), must be demonstrated in at least one of the esophageal biopsies.1,2 Histological remission was accordingly defined as <15 eos/hpf. Deep histological remission according to Greuter et al. was defined as 0 eos/hpf. 13
Endoscopic assessment and endoscopic remission
Results were categorized based on the validated modified endoscopic reference score (EREFS) criteria in the esophagogastroduodenoscopy (EGD) report, calculated by the physician conducting the examination. The total possible score ranges from 0 to 9, 15 and remission was defined as a total EREFS score ⩽2. 16 Deep endoscopic remission was defined as the complete absence of typical EoE findings (EREFS = 0).
Safety
Side effects were monitored and analyzed at all evaluations starting at the beginning of the study.
Statistics
Categorical variables were presented as counts (n) and percentages (%). Descriptive statistics included mean with standard deviation (SD) or standard error of the mean (SEM), as well as median and range. To compare subgroups, a one-sided t-test was applied, while a chi-square test (χ²) was used to examine factors associated with remission rates. Statistical significance was defined as a p-value < 0.05 in both analyses. Pooled remission rates including missing data were calculated using multiple imputation analyses (MI) in SPSS Statistics 31.0 (IBM Corp., Armonk, NY, USA). All remaining analyses used ‘observed cases’ and dropouts/lost to follow-ups were not included in calculations of remission.
Results
Patient disposition and characteristics
We identified 221 patients who underwent treatment with BUD-ODT and were included in this study. Of these patients, 201 completed IT, 151 of whom proceeded to maintenance therapy. Among these patients, 109 completed the RM1 phase, 80 entered the RM2 phase, and 72 completed the RM2 phase. The full patient disposition is shown in Figure 1.
The mean age at the time of study inclusion was 43.5 (standard deviation (SD) ± 13.8) years and 69.0% of the patients were male. Patients had symptoms for an average of 2.5 years (range: 0–7) before diagnosis. Concomitant atopic conditions were reported: in total, 52.0% (115/221) of patients presented atopic comorbidities, with allergic asthma (36.2%) and food allergy (13.6%) being the most prevalent ones. At initial diagnosis, 23.6% (52/221) of patients had a history of endoscopically confirmed food bolus obstruction (FBO) and 29.0% (64/221) with reported bolus obstruction without intervention according to medical records, resulting in a total of 52.5% (116/221)) with a history of bolus obstruction. Additionally, 16.7% (37/221) of patients had endoscopically detected strictures, of which 27.0% (10/37) were classified as fibrostenotic and required endoscopic dilatation. The remaining 73.0% (27/37) were of inflammatory origin; among these, three strictures required dilatation due to their severity. The decision to perform dilatation was made on a case-by-case basis by the treating center and physician. In all instances, balloon dilatation was utilized, as it was the standard approach across all centers. In total, 5.9% (13/221) of patients underwent dilatation therapy for the treatment of strictures (Table 1).
Patient characteristics.
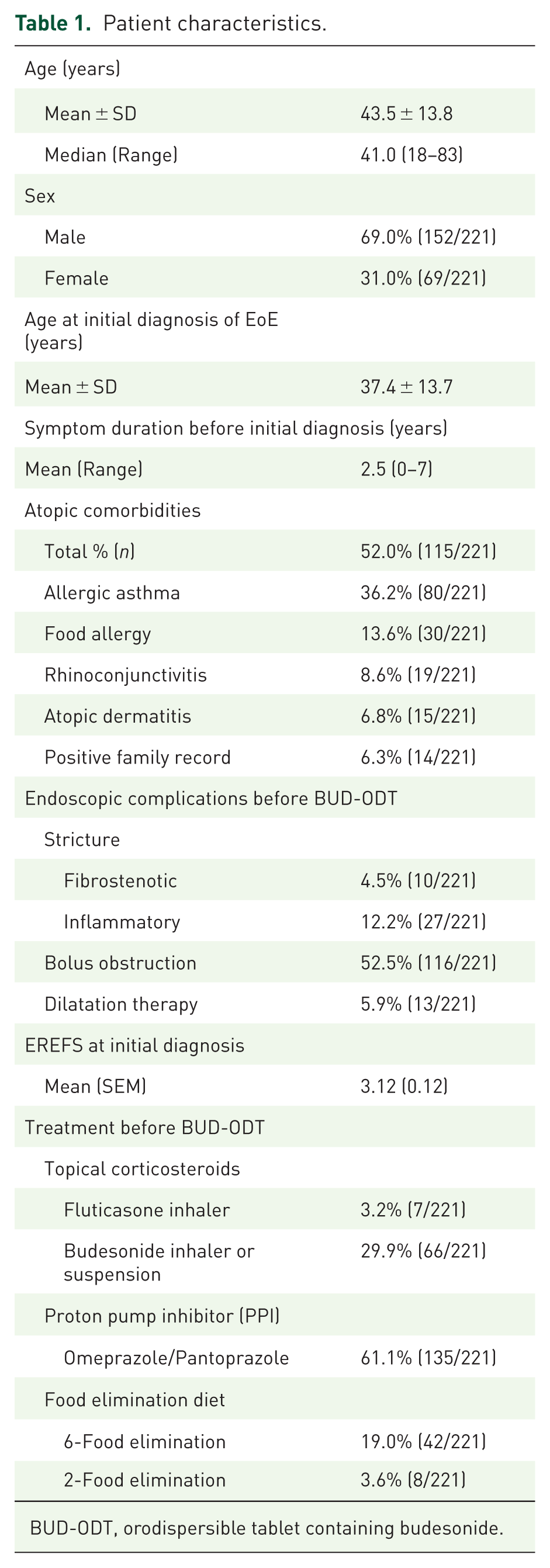
BUD-ODT, orodispersible tablet containing budesonide.
Treatment response to BUD-ODT
Clinical-histological remission
After 6–12 weeks of induction therapy with BUD-ODT, 75.1% (151/201) of the total patient population achieved combined clinical and histological remission, with clinical remission observed in 89.1% (179/201) and histological remission in 75.1% (151/201). Remission rates varied by treatment duration, with the following breakdown: 72.6% (69/95) of patients achieved remission at 6 weeks, 75.0% (30/40) at 8 weeks, 66.7% (2/3) at 10 weeks, and 79.4% (50/63) at 12 weeks. At the end of RM1, clinical-histological remission was maintained in 73.4% (80/109) of patients, 83.4% (91/109) demonstrated symptom remission, while 73.4% (80/109) achieved remission of histological findings. A final follow-up after 1–2 years (RM2) revealed maintenance of clinical-histological remission in 76.4% (55/72) of the patients, with 83.3% (60/72) in histological remission and 76.4% (55/72) in clinical remission (Table 2).
Clinical-histological, clinical, histological, and endoscopic remission rates, including deep remission.

IT, induction therapy; re-IT, reinduction therapy; SDI, Straumann Dysphagia Index.
Complete symptom resolution (SDI = 0) was reported by 39.8% (80/201) of patients after IT, 66.1% (72/109) after ET1, and 52.8% (38/72) after completion of ET2. Deep histological remission (0 eos/hpf) was observed in 70.0% (141/201) of patients after IT, 48.6% (53/109) after ET1, and 69.4% (50/72) after ET2 (Table 2).
The pooled mean remission rate after multiple imputation (n = 221) was 68.1% (95% confidence interval (CI): 61.9–74.2) at IT, 62.7% (95% CI: 56.2–69.2) at RM1 and 69.9% (95% CI: −2.0 to 141.8) at RM2. This wide confidence interval indicates a high degree of uncertainty due to the large number of missing data (n = 149).
Endoscopic remission
Mean (standard error) EREFS score at week 0 was at 3.12 ± 0.12, whereas after 6–12 weeks (IT) it declined to 0.75 ± 0.08. It continued to slightly increase to 0.97 ± 0.13 (RM1) but remained at 0.64 ± 0.13 after 52–104 weeks (RM2). The reduction in EREFS at each evaluation, compared to the baseline EREFS at week 0 was shown to be highly significant (p < 0.01) (Figure 2). Deep endoscopic remission with an EREFS score of 0 was identified in 60.2% (121/201) after IT, 51.4% (56/109) after ET1, and 62.5% (45/72) after ET2 (Table 2). The total reduction over the entire observation period was 2.5 points. 88.1% (177/201) of the patients were in remission (EREFS ⩽ 2) after IT.
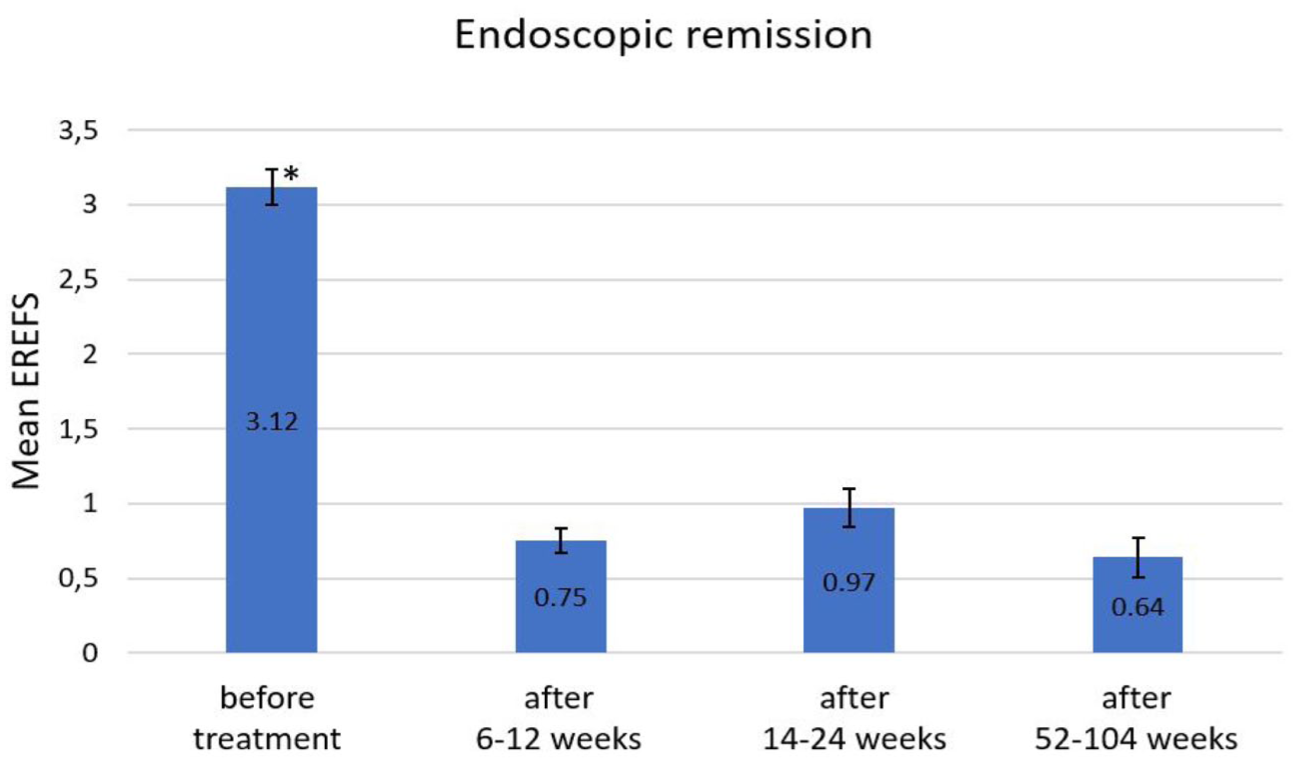
Endoscopic remission.
Factors influencing remission rate
Prior dietary intervention showed to have a statistically significant impact on clinical-histological remission rates. After induction therapy, a higher clinical-histological remission rate was observed in patients with prior dietary measures (χ²(1) = 4.89, p = 0.027; Cramér’s V = 0.16), indicating a mild association. This effect remained significant during the first maintenance phase (RM1) (χ²(1) = 4.21, p = 0.04; Cramér’s V = 0.20), suggesting a mild-to-moderate association. These findings support the positive influence of dietary intervention on treatment outcomes, possibly reinforced by continued adherence during therapy. No other factors showed a statistically significant association with clinical-histological remission rates.
Further procedure
Patients who did not attain clinical-histological remission after the induction phase (24.9%, 50/201) were either offered an extension of therapy for an additional 6–8 weeks (52.0%, 26/50) or patients chose to discontinue treatment (30.0%, 15/50), due to side effects. After the extension of induction therapy, another follow-up appointment was conducted, and clinical-histological remission was observed in 57.7% (15/26) of these patients. Patients achieving remission after extension of IT or re-IT were separately followed and excluded of the original cohort (Figure 1).
At the end of RM1, a clinical-histological relapse was documented in 26.6% (29/109) of the patients. All relapses were confirmed histologically, with nine patients also exhibiting clinical symptoms. Among those experiencing relapse, 86.2% (25/29) opted to start re-IT for an additional 6–8 weeks, with 36.0% (9/25) re-achieving clinical-histological remission within that period. The remaining patients experiencing relapse, 13.8% (4/29), decided to discontinue treatment due to side effects, such as digestive issues (n = 3) and numbness of the mouth (n = 1).
During weeks 52–104 of treatment (RM2), the relapse rate was at 23.6% (17/72), with all relapses showing both histological and clinical disease activity. The procedure was the same as after RM1: 76.5% (13/17) of patients underwent re-induction therapy for an additional 6–8 weeks, of whom 46.2% (6/13) re-achieved clinical-histological remission (Table 3).
Treatment failure and further treatment at different time points.

IT, induction therapy; re-IT, reinduction therapy.
For those who did not achieve remission even after extension or restart of induction therapy (16.9%, 34/201), an alternative treatment, such as an elimination diet and/or biologic agents, was proposed. 14 These patients were then categorized as steroid-refractory non-responders (Table 3).
Compliance
Noncompliance was documented in 10.4% (23/221) of cases. A total of 19 of these patients discontinued treatment before remission could be assessed after IT, RM1, or RM2. The remaining four patients continued therapy but did not achieve clinical-histological remission (three after IT and one after RM1). They subsequently started either a prolonged IT or a re-IT but did not complete the treatment. Accordingly, none of these patients were classified as steroid nonresponders based on our criteria.
Safety
Adverse events
Adverse events encompassed weight gain, reported by 2.7% (6/221) of participants, and esophageal complications, including retrosternal pain and reflux, observed in 9.1% (20/221) of the patients (Table 4). Problems associated with the lower gastrointestinal tract affected 6.8% (15/221) of the patients, while 8.2% (18/221) experienced neurological symptoms such as numbness/tingling in the mouth, headaches, or dizziness. Other side effects were reported in 6.3% (14/221) of patients.
Adverse events.

Combined oral-esophageal candidiasis required endoscopic confirmation for diagnosis. Therefore, the total number of patients examined is the same as the number of patients assessed after each treatment period. Candidiasis presented as follows: After 6–8 weeks, Candidiasis was found in 18.9% (38/201) of the patients, which decreased to 8.3% (9/109) after 14–24 weeks. Subsequently, the prevalence increased to 16.7% (12/72) after 52–104 weeks. The total number of patients exhibiting candidiasis was 29.4% (59/201; Table 4). Only 30.5% (18/59) of these cases exhibited symptomatic candidiasis, of which 27.8% (5/18) discontinued their treatment, making candidiasis one of the less common reasons for treatment discontinuation, as it was effectively managed with antifungal agents such as amphotericin B or nystatin during the therapy.
Worsening of preexisting symptoms and esophageal complications
No food bolus obstruction was documented during the course of therapy. Worsening of dysphagia was reported by 10.9 % (22/201), 14.7 % (16/109), and 20.8 % (15/72) of the patients during the respective study period. Those patients were all counted as “no remission achieved.”
Discontinuation of treatment
The overall treatment discontinuation rate for the study cohort was 15.4% (34/221). Primary reasons included adverse effects, accounting for 6.9% of cases (15/221), and noncompliance with 8.6% of the patients (19/221).
Discussion
In this real-world study of BUD-ODT for EoE, 75.1% (151/201) of patients achieved clinical-histological remission after induction therapy (IT), with 72.6% (69/95) after 6 weeks. In comparison, a 2019 conducted RCT study reported a 57.6% clinical-histological remission rate after 6 weeks of induction therapy (1 mg BID) and histological remission was shown in 93.2%. 10 In an open-label study involving 181 patients, clinical-histological remission was observed in 69.6% of patients after 6 weeks. 15
After successful induction therapy, 73.4% (80/109) of our patients were able to maintain their remission for a total of 14–24 weeks. A 2020 study on the long-term efficacy of BUD-ODT demonstrated that, in a RCT setting, 73.5% (0.5 mg BUD-ODT BID) and 75% (1 mg Bud-ODT BID) of patients maintained clinical and histological remission after 48 weeks of treatment, deep histological remission (0 eos/hpf) was achieved in 79.4% (1 mg) and 76.5% (0.5 mg) of the cases. 11
Following RM1, 76.4% (55/72) of our patients maintained remission after a total of 52–104 weeks of treatment. A study published in 2022, evaluating the long-term efficacy of BUD-ODT treatment in an open-label setting, reported comparable results: 84.0% of patients receiving a daily dosage of 1× or 2 × 0.5 mg budesonide BID maintained clinical remission after a total of 96 weeks, with histological remission demonstrated in 80.1% of patients. Remarkably, 62.2% (57/91) patients were able to sustain this remission even over a period of 3 years. 16 Regrettably, this study is the only investigation that encompasses treatment with BUD-ODT of this duration, highlighting the need for further research to assess its long-term effectiveness.
The multiple imputation analysis showed that clinical-histological remission rates for IT and RM1, respectively, were consistent with the observed-case analysis, indicating minimal impact of missing data. For RM2, however, the imputed estimate showed a very wide confidence interval, reflecting considerable uncertainty. This demonstrates that while multiple imputation analysis helps as a useful method to handle missing data, the validity of the findings is still constrained by the inherent limitations of real-world evidence. In particular, a limited number of patients can substantially reduce the precision of the estimates, leading to a wide confidence interval even after imputation.
Endoscopic remission was determined using the EREFS—score: after IT (6–12 weeks), mean EREFS decreased from 3.12 ± 0.12 at initial diagnosis to 0. 75 ± 0.08. This average remained consistent throughout the treatment period, with values of 0.97 after 14 weeks and 0.64 at the end of treatment. Endoscopic remission with ⩽2 points was shown in 88.1% (177/201) of the patients after induction therapy, in 75.2% (82/109) after RM1 and in 84.7% (61/72) after RM2. Several controlled studies indicate a similar endoscopic remission rate as secondary endpoints.11,15,16
A prior food elimination diet (FED) was conducted by 22.6% of patients (50/221) before starting BUD-ODT therapy and showed a statistically significant, though moderate, positive effect on clinical-histological remission. This suggests that FED may induce lasting immunological changes affecting EoE activity beyond the diet period. 17 Microbiome studies have also linked FED to specific mucosal alterations potentially contributing to remission. 18 These findings highlight the potential synergistic effects of dietary interventions alongside topical corticosteroids. However, the choice of diet is crucial, as long-term adherence to extensive elimination diets like the 6-FED remains limited.7,17
The relapse rate averaged 25.0% over the entire duration of the study. This appears to be more significant in a real-life setting than previously documented in RCT studies: a 48-week BUD-ODT study reports a histological relapse rate of 13.2% (0.5 mg BUD-ODT BID) and 10.3% (1 mg BID) as well as a clinical relapse rate of 10.3% (0.5 mg BUD-ODT BID) and 7.4% (1 mg BUD-ODT BID).11,15 Compared to the tightly controlled environment of RCTs, real-world settings often involve less-intensive monitoring and patient support, which may negatively affect adherence and, consequently, therapeutic outcomes. The study by Breedenord et al. 19 underscores the low adherence to medication therapy among adults with EoE, which is primarily attributed to long disease duration, severe symptoms, and diminished beliefs in necessity of treatment.
Several other factors may also contribute to the higher relapse rate observed in this real-world cohort. First, the use of reduced maintenance doses, although consistent with current guideline recommendations, may be insufficient for certain patients, depending on disease severity or individual response. 2 Additionally, disease progression over time could explain relapses occurring later in the treatment course, particularly during the second maintenance phase (RM2). Furthermore, noncompliance is likely underestimated in this setting, as only documented instances could be recorded.
In contrast, in our study, 16.9% of patients were classified as steroid-refractory, which is lower than previous reports on corticosteroid treatment of EoE. 20 This result likely reflects both the specific formulation of BUD-ODT versus off-label steroids for EoE as well as our treatment regimen, in which patients lacking or losing response to BUD-ODT were offered re-induction therapy.
Furthermore, symptomatic response did not always correlate with histological remission. Following induction therapy, 89.1% of the patients achieved clinical remission, while only 75.1% exhibited histological remission. A similar pattern was observed after RM1 with 83.4% in clinical and 73.4% in histological remission (Table 2). These findings are consistent with the 2022 guideline and align with the results of several RCTs2,6,11,15,21,22 as well as retrospective studies investigating this phenomenon. 23 This effect is thought to result from coping mechanisms developed by patients over time. Given the delay between initial diagnosis and the initiation of topical steroid therapy (mean: 2.5 years in our study), patients often adapt strategies to maintain their quality of life despite impaired swallowing capacity. These learned techniques include chewing thoroughly, taking smaller bites per swallow, and increasing water intake during meals. 24 As a result, perceived clinical remission is more favorable than the corresponding histological or endoscopic outcome.
Adverse events and noncompliance were the primary reasons for discontinuing therapy. Surprisingly, while oral-esophageal candidiasis was the most common side effect, it was not the leading cause of discontinuation, as it was effectively managed with antifungal treatment. Overall, 29.4% (59/201) of patients developed a combined oral-esophageal candidiasis, for which physicians employed various antifungal agents. Similar findings were reported in other BUD-ODT studies.11,15,16 Extra-esophageal adverse effects, such as neurological symptoms, dermatological manifestations, and lower gastrointestinal complications, were the most common reasons for discontinuing therapy. This may be attributed to the observation that only 30.5% (18/59) of the patients diagnosed with candidiasis by endoscopy also experienced symptomatic candidiasis.
However, this study also has significant limitations. First, the retrospective design of the study increases susceptibility to errors, as reliance on patient records may lead to inaccuracies in documentation. Nonetheless, it can be assumed that the physicians conducting the interviews and examinations followed established guidelines, ensuring a consistent treatment approach. Despite facing certain challenges, this real-life approach provides a much more accurate representation of current treatment practices. Furthermore, the inclusion of patients from two major EoE centers introduces a diverse array of cases, capturing the variability inherent in EoE. Although the study initially included a sufficiently large number of patients, the decrease in this number over time due to therapy discontinuation may compromise the reliability of conclusions regarding the long-term effectiveness of the treatment. Nevertheless, this study is one of the few analyzing a long-term treatment with BUD-ODT of EoE for over 1–2 years.
In conclusion, our study provides real-life results of BUD-ODT treatment from two German EoE-centers. Treatment with BUD-ODT demonstrated high effectiveness in both inducing and maintaining remission of active EoE in adults. Despite three-fourth of the patients achieving clinical-histological and endoscopic remission in every phase, the overall relapse rate was 24.1%, with several patients even classified as steroid-nonresponders. It is essential for these patients to have access to additional treatment options, as untreated or inadequately treated EoE can progress and lead to complications such as stricture formation and / or food bolus obstructions. However, regarding the safety of treatment, it was demonstrated that although candidiasis was the most common adverse event, it did not lead to treatment discontinuation as often as noncompliance and other (far less prevalent) side effects.
