Abstract
Background:
Immune checkpoint inhibitors (ICIs), including Pembrolizumab, Nivolumab, Sintilimab, Tislelizumab, and Sugemalimab, have been approved in China as first-line treatments for advanced HER2-negative gastric cancer (GC) and gastroesophageal junction cancer (GEJC). However, the latest COMPASSION 15 study showed that Cadonilimab in combination with chemotherapy provided significant survival benefits.
Objective:
This study aims to evaluate the cost-effectiveness of Cadonilimab plus chemotherapy versus chemotherapy alone and the ICIs approved in China for first-line treatment of advanced HER2-negative gastric cancer or gastroesophageal junction carcinoma (GC/GEJC) from the perspective of Chinese payers.
Design:
The cost-effectiveness analysis.
Methods:
Based on the research data from COMPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303, we constructed a 15-year Markov model to evaluate the cost and health outcomes of Cadonilimab combined with chemotherapy versus chemotherapy alone and other ICIs in advanced HER2-negative GC and GEJC. This evaluation includes total cost, life years (LYs), quality-adjusted life years (QALYs), and incremental cost-effectiveness ratio (ICER).
Results:
Cadonilimab produced 0.73 QALYs (1.10 LYs) at a cost of $26,591. It required an additional investment of $17,826 to gain 0.25 QALYs (0.37 LYs), resulting in an ICER of $72,492.29 per QALY compared to chemotherapy alone. In comparison, other ICIs approved in China—Pembrolizumab, Nivolumab, Sintilimab, Tislelizumab, and Sugemalimab—incurred total costs of $11,735, $13,970, $16,346, $10,765, and $14,857, respectively, generating 0.68 QALYs (1.04 LYs), 0.69 QALYs (1.04 LYs), 0.73 QALYs (1.12 LYs), 0.82 QALYs (1.26 LYs), and 0.81 QALYs (1.25 LYs). Sensitivity analysis revealed that the cost of Cadonilimab, the utility value of progressive disease, and the risk of platelet decline in the Cadonilimab group were the most influential factors affecting the model’s stability. At a willingness-to-pay threshold of $37,386, Cadonilimab is not a cost-effective option for the first-line treatment of advanced GE/GEJC.
Conclusion:
Cadonilimab is not a cost-effective option for the first-line treatment of advanced HER2-negative GC/GEJC. In comparison to other ICIs approved in China, Tislelizumab appears to be a more favorable option.
Keywords
Introduction
Gastric cancer (GC) is the fifth most diagnosed cancer and the fifth leading cause of cancer-related deaths globally. According to the International Agency for Research on Cancer, there were an estimated 968,350 new cases and 659,853 deaths due to gastric cancer worldwide in 2022. 1 In China, gastric cancer ranks as the fifth most frequently diagnosed cancer and is the third leading cause of cancer-related deaths. In 2022, there were approximately 358,700 new cases of gastric cancer and about 260,400 associated deaths in China. 2 Although advances in endoscopic technology have improved the detection rate of early gastric cancer, 36.4% of patients are still diagnosed with advanced stages of the disease, resulting in a 5-year survival rate of only 7%. 3
Over the past few decades, the treatment of advanced gastric cancer has remained a significant challenge. The primary management approach has been chemotherapy, which offers limited improvements in overall survival (OS) and presents difficulties in managing chemotherapy-induced toxicity and side effects.4–7 Recently, targeted therapy has demonstrated efficacy in advanced gastric cancer. For instance, Trastuzumab, a monoclonal antibody targeting HER2, has been approved for first-line treatment of HER2-positive advanced gastric cancer,8–10 However, its indications still have limitations. With the emergence of immune checkpoint inhibitors (ICIs), the combination of programmed cell death protein 1 (PD-1) inhibitors and chemotherapy has become the standard first-line treatment for advanced gastric cancer (GC) and gastroesophageal junction cancer (GEJC). A phase III clinical trial, KEYNOTE-859 (NCT03675737), demonstrated a median OS of 12.9 months for patients receiving Pembrolizumab and 11.5 months for those receiving placebo in combination with the XELOX regimen (HR = 0.78, p < 0.0001). 11 Four-year follow-up data from the phase III clinical trial CHECKMATE-649 12 (NCT02872116) demonstrated that patients with a programmed cell death-ligand 1 combined positive score (PD-L1 CPS) ⩾5 who received Nivolumab had a median OS of 15.5 months (95% CI: 11.9–21.1 months), compared to 9.6 months (95% CI: 8.0–12.1 months) in the control group. 13 Based on the efficacy data mentioned above, both the 2024 NCCN guidelines and the CSCO guidelines recommend Pembrolizumab and Nivolumab as the first-line treatment for advanced HER2-negative GC and GEJC.8,9 Furthermore, phase III clinical trials ORIENT-16 (NCT03745170), RATIONALE-305 (NCT03777657), and GEMSTONE-303 (NCT03802591), Sintilimab, Tislelizumab, and Sugemalimab have also demonstrated significant OS and progression-free survival (PFS) benefits.14–16 Therefore, these agents have been successively approved in China for first-line treatment of advanced HER2-negative gastric cancer or gastroesophageal junction adenocarcinoma (GC/GEJC). 9
In 2024, the American Association for Cancer Research (AACR) General Assembly announced the results of the phase III clinical study COMPASSION-15 (NCT05008783). This study examined the efficacy of Cadonilimab, a bispecific antibody targeting PD-1 and cytotoxic T lymphocyte antigen-4 (CTLA-4), in combination with a chemotherapy regimen for the first-line treatment of advanced gastric or gastroesophageal junction adenocarcinoma. The results indicated that the combination significantly prolonged OS and PFS in the overall population. OS was extended to 15.0 months compared to 10.8 months (HR = 0.62; 95% CI, 0.50–0.78; p < 0.001), and PFS was extended to 7.0 months compared to 5.3 months (HR = 0.53; 95% CI, 0.44–0.65; p < 0.001). The benefits were even more pronounced in patients with PD-L1 CPS ⩾5 scores, resulting in an OS of not estimable compared to 10.6 months (HR = 0.56; 95% CI, 0.39–0.80; p < 0.001) and a PFS of 6.9 months compared to 5.5 months (HR = 0.51; 95% CI, 0.37–0.70; p < 0.001). In the PD-L1 CPS <5 group, the combination of Cadonilimab significantly prolonged OS to 14.8 months compared to 11.1 months (HR = 0.70; 95% CI, 0.51–0.95; p = 0.011) and PFS to 6.9 months compared to 4.6 months (HR = 0.60; 95% CI, 0.45–0.79; p < 0.001). 17
Although Cadonilimab has demonstrated significant efficacy and safety in advanced GC/GEJC, its economic benefits have not been evaluated. This study aims to evaluate the cost-effectiveness of Cadonilimab combined with chemotherapy, compared to chemotherapy alone and other ICIs approved in China for first-line treatment of advanced HER2-negative GC/GEJC, from the perspective of Chinese payers.
Materials and methods
This study is a cost-effectiveness analysis based on a 15-year Markov model. The inclusion criteria are as follows: (1) phase III Randomized Controlled trials; (2) histologically confirmed advanced or metastatic GC/GEJC; (3) first-line treatment; (4) HER2-negative status; (5) ICIs; and (6) approval by the Chinese CSCO guidelines. Eligible studies include COPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303. In addition, this survey and report fully comply with the CHEERS guidelines 18 (Table S1).
Patient population and intervention
In this study, we used patient cohorts and treatment data from the COPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303 trials. In the ORIENT-16, RATIONALE-305, and GEMSTONE-303 trials, the anti-PD-1 antibodies, Sintilimab (200 mg every 3 weeks), Tislelizumab (200 mg every 3 weeks), and Sugemalimab (1200 mg every 3 weeks), were combined with chemotherapy regimens: XELOX (capecitabine 1000 mg/m2 twice daily on days 1–14 and oxaliplatin 130 mg/m2 on day 1), FP (5-fluorouracil 800 mg/m2 on days 1–5 and cisplatin 80 mg/m2), or CAPEOX (oral capecitabine 1000 mg/m2 twice daily on days 1–14 and oxaliplatin 130 mg/m2 on day 1). For simplicity, the model uniformly adopted the XELOX regimen. After six cycles, maintenance therapy continued with Cadonilimab or placebo (in the Cadonilimab trial) or with Sintilimab, Tislelizumab, or Sugemalimab (in the ORIENT-16, RATIONALE-305, and GEMSTONE-303 trials) until disease progression or unacceptable toxicity, for a maximum of 2 years. In the KEYNOTE-859 and CheckMate-649 trials, patients received Pembrolizumab (200 mg every 3 weeks) or Nivolumab (360 mg every 3 weeks) plus the XELOX chemotherapy regimen, with treatment capped at 2 years. All patients were hospitalized every 3 weeks, and imaging assessments were performed every 6 weeks to monitor disease progression. Since the original article did not specify a second-line treatment regimen, we recommended one based on the 2024 NCCN and CSCO guidelines. Docetaxel (75–100 mg/m² intravenously every 3 weeks) was used as second-line treatment for patients with disease progression. The patient population was assumed to be male, 64 years old, weighing 65 kg, with a body surface area of 1.72 m2, to calculate the drug dose. 19
Model structure
In this study, we used TreeAgePro2022 software (TreeAge Software, Williamstown, MA, USA, https://www.treeage.com) to build a decision tree and a Markov model simulating patients with advanced gastric cancer treated with one of seven regimens (Cadonilimab + XELOX, placebo + XELOX, Pembrolizumab + XELOX, Nivolumab + XELOX, Sintilimab + XELOX, Tislelizumab + XELOX, or Sugemalimab + XELOX). Patients transitioned among three health states—PFS, progressive disease (PD), and death—with initial probabilities of 1, 0, and 0, respectively, to evaluate health and economic outcomes (Figure S1). All patients will receive 35 cycles of treatment and be followed regularly, unless disease progression or severe adverse reactions occur. If the disease progresses, second-line treatment will be administered, while the remaining patients will receive optimal supportive care. In conjunction with the patient medication regimen, we defined the model cycle as a 3-week cycle over a 15-year period (with more than 99% of patients dying) and applied a 5% discount rate20,21 to predict the total cost, life years (LYs), quality-adjusted life years (QALYs), and incremental cost-effectiveness ratio (ICER) of the treatment strategies. We also evaluated the cost-effectiveness based on the willingness-to-pay (WTP) value. Relevant literature indicates that the WTP value is typically calculated as three times per capita GDP for China,22,23 estimated at $37,386/QALY for 2024.
Survival estimates and model transitions
We used Getdata Graph Digitizer (version 2.20, available at: http://www.getdata-graph-digitizer.com/index.php) to extract survival data from the OS and PFS curves of the COMPASSION-15 KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303 trials. Patient data were then reconstructed using R Studio software (version 4.3.2, available at: https://www.rstudio.com). The log-normal, log-logistic, Weibull, exponential, and Gompertz distributions were selected to calculate the parameters described earlier. The best-fitting parameter distribution curves for the PFS and OS groups were selected based on the Akaike information criterion and Bayesian information criterion (Figure S2 and Table S2). The most suitable Weibull was chosen to fit the data, and the values of the two parameters, scale parameter (λ) and shape parameter (γ), were calculated using the R software. Assuming that the mortality rate during period t is denoted by M, and the mean OS time denoted as S(t), then the cause-specific mortality M at cycle t can be expressed as follows: M = [S(t) – S(t + 1)]/S(t). Based on the fitted parameter, S(t) can be further formulated as follows: exp
Cost input and utility estimate
The medical costs included in this model encompass drug costs, laboratory and imaging examination costs, hospitalization management costs, adverse reaction management costs, optimal supportive treatment costs, and palliative care costs (Table 1). The price—Cadonilimab, Pembrolizumab, Nivolumab, Sintilimab, Tislelizumab, Sugemalimab—is based on the official list price, while the prices of other drugs are derived from the official pricing at Xiangya Hospital of Central South University. Other costs were obtained from published literature21,23–26 and were included in the model calculation only when adverse reactions were ⩾grade 3 and their incidence was ⩾5% (all adverse reactions were assumed to occur once within the PFS and PD states). These prices have been adjusted for inflation to reflect current values using the Bureau of Labor Statistics Inflation Calculator (https://www.bls.gov/data/inflation_calculator.htm?ss=P&st_rid=null). All prices are expressed in US dollars (as of March 7, 2025, USD 1 = CNY 7.1705).
Model parameters: baseline values, ranges, and distributions for sensitivity analysis.

AEs, adverse events; CX, Cadonilimab + XELOX; HR, hazard ratio; NC, Nivolumab plus chemotherapy; OS, overall survival; PC, Pembrolizumab plus chemotherapy; PFS, progression-free survival; PX, Placebo + XELOX; SC, Sugemalimab plus chemotherapy; SNC, Sintilimab plus chemotherapy; TC, Tislelizumab plus chemotherapy; —, unreported.
Utility values are used to assess a patient’s health-related quality of life (HRQOL), ranging from 0 (representing death) to 1 (indicating perfect health). Since the COPASSION15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303 studies did not provide measures like EQ-5D, SF-36, or other relevant HRQOL scales,27–29 we referred to existing literature to gather the required data for our calculations. 30 We also included disutility values related to adverse reactions in our analysis.21,31–33
Sensitivity analysis
Sensitivity analysis was conducted to assess model stability. In the one-way sensitivity analysis, a ±20% variation range was applied to this parameter, and the method described in the literature was used to assess its influence on the ICER. In the probabilistic sensitivity analysis, we conducted 10,00 Monte Carlo simulations on a population of 610 patients to assess the cost-effectiveness of Cadonilimab in combination with XELOX at the $37,386 WTP threshold. In addition, in subgroup analysis, we assessed the cost-effectiveness of different PD-L1 expression levels (PD-L1 CPS <5, PD-L1 CPS ⩾5) under the WTP threshold using survival curves. In other subgroup analyses, because complete Kaplan–Meier curves for PFS or OS were unavailable, we estimated the parameters using the following formulas:

Results
Base case results
The Cadonilimab + XELOX regimen yielded 0.73 QALYs (1.10 life years (LYs)) at a total cost of $26,591, compared with the placebo + XELOX regimen, which yielded 0.48 QALYs (0.73 life years) at $8765. Cadonilimab increased costs by $17,826 for a gain of 0.25 QALYs, resulting in an ICER of $72,492.29 per QALY. At a WTP threshold of $37,386 per QALY, the regimens produced the following outcomes: Pembrolizumab + XELOX: 0.68 QALYs (1.04 LYs) at $11,735; Nivolumab + XELOX: 0.69 QALYs (1.04 LYs) at $13,970; Sintilimab + XELOX: 0.73 QALYs (1.12 LYs) at $16,346; Tislelizumab + XELOX: 0.82 QALYs (1.26 LYs) at $10,765; and Sugemalimab + XELOX: 0.81 QALYs (1.25 LYs) at $14,857; Overall, the Cadonilimab + XELOX regimen did not meet the cost-effectiveness threshold for first-line treatment of advanced gastric cancer (Table 2).
Baseline results.
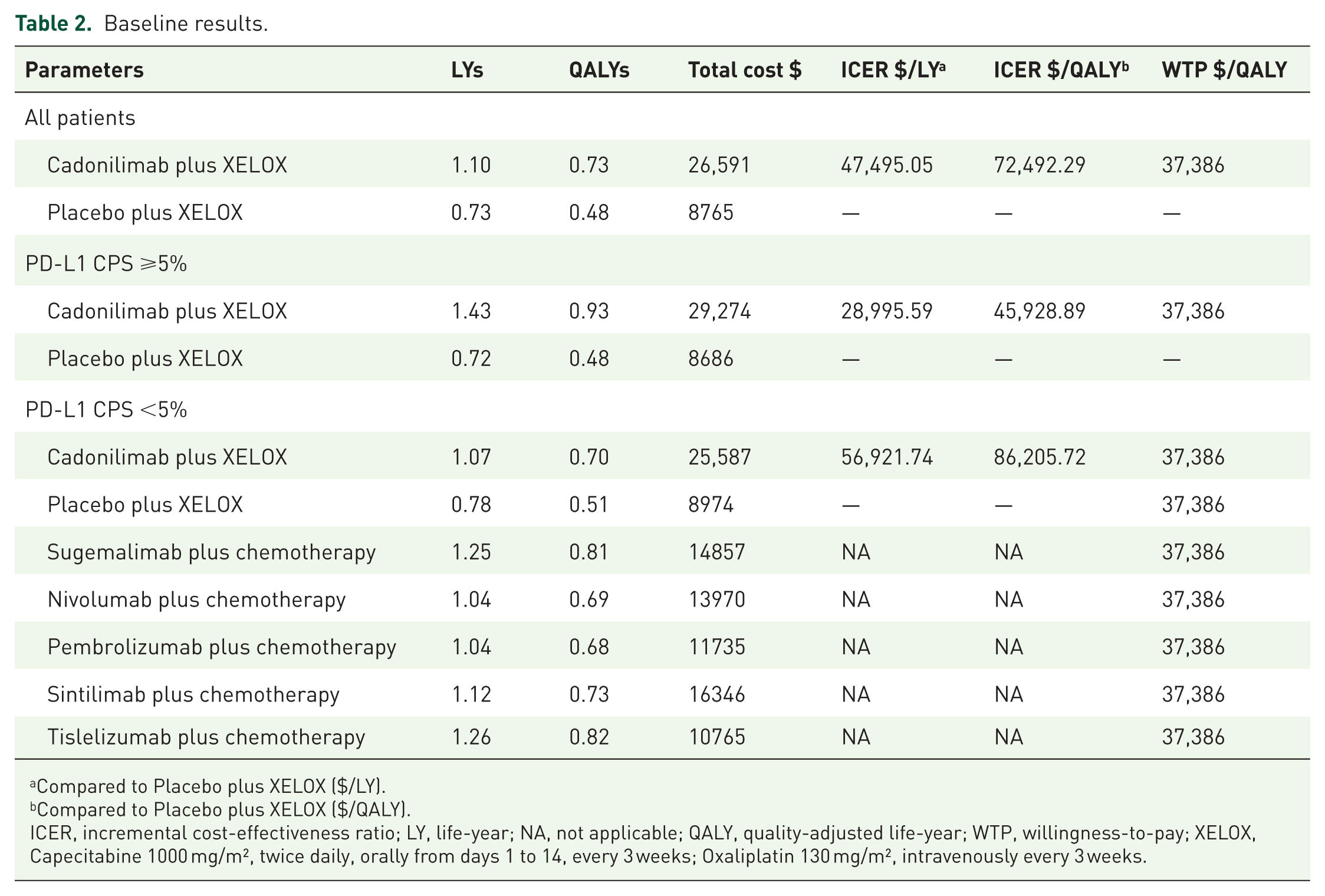
Compared to Placebo plus XELOX ($/LY).
Compared to Placebo plus XELOX ($/QALY).
ICER, incremental cost-effectiveness ratio; LY, life-year; NA, not applicable; QALY, quality-adjusted life-year; WTP, willingness-to-pay; XELOX, Capecitabine 1000 mg/m², twice daily, orally from days 1 to 14, every 3 weeks; Oxaliplatin 130 mg/m², intravenously every 3 weeks.
Sensitivity analysis results
One-way sensitivity analysis revealed that the cost of Cadonilimab, the utility value associated with disease progression, and the risk of Cadonilimab-induced thrombocytopenia were key factors affecting model stability (Figure 1). Among these variables, the cost of Cadonilimab was the most influential. As the per-cycle cost of Cadonilimab ranged from $1245 to $1867, the ICER varied from $62,219.72/QALY to $82,764.37/QALY. The per-cycle costs of oxaliplatin, docetaxel, and hypokalemia management had minimal impact on model outcomes. Furthermore, variations in ICER values within their respective ranges had negligible effects on the overall model outcomes. In the PD-L1 CPS ⩾5% subgroup, the most influential factors affecting model stability were the utility value of PD, the cost of Cadonilimab, and the risk of platelet decline in the Cadonilimab group (Figure 2). In the PD-L1 CPS <5% subgroup, the cost of Cadonilimab, the risk of platelet declines in the Cadonilimab group, and the utility value of PFS were the most influential factors affecting model stability (Figure 3). During a comprehensive comparison of multiple clinical trials, such as COMPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303, we conducted a sensitivity analysis to assess the net monetary benefits (NMB). The results reveal that the utility value of PD, the utility value of PFS, and the cost associated with decreased platelet count are the three most influential factors affecting model stability. Conversely, the costs of Cadonilimab, Sugemalimab, Nivolumab, and Pembrolizumab have minimal impact on the robustness of the model (Figure 4).

The one-way sensitivity analyses for Cadonilimab plus chemotherapy (all patients).

The one-way sensitivity analyses for Cadonilimab plus chemotherapy (PD-L1 CPS ⩾5%).

The one-way sensitivity analyses for Cadonilimab plus chemotherapy (PD-L1 CPS <5%).

The one-way sensitivity analyses for all groups (COPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303).
In the COMPASSION-15 trial, probability sensitivity analysis showed that, at a WTP threshold of $37,386 per QALY, the probability of Cadonilimab in combination with XELOX being cost-effective for the Chinese population was 27% (Figure S3). The cost-effectiveness acceptability curve further suggested that chemotherapy alone might be a more cost-effective treatment option (Figure 5). In comparison with other ICIs approved in China, the acceptability curve indicates that Tislelizumab is a relatively better option for first-line treatment of advanced gastric cancer (Figure 6).

Acceptable curve plots (COMPASSION-15).
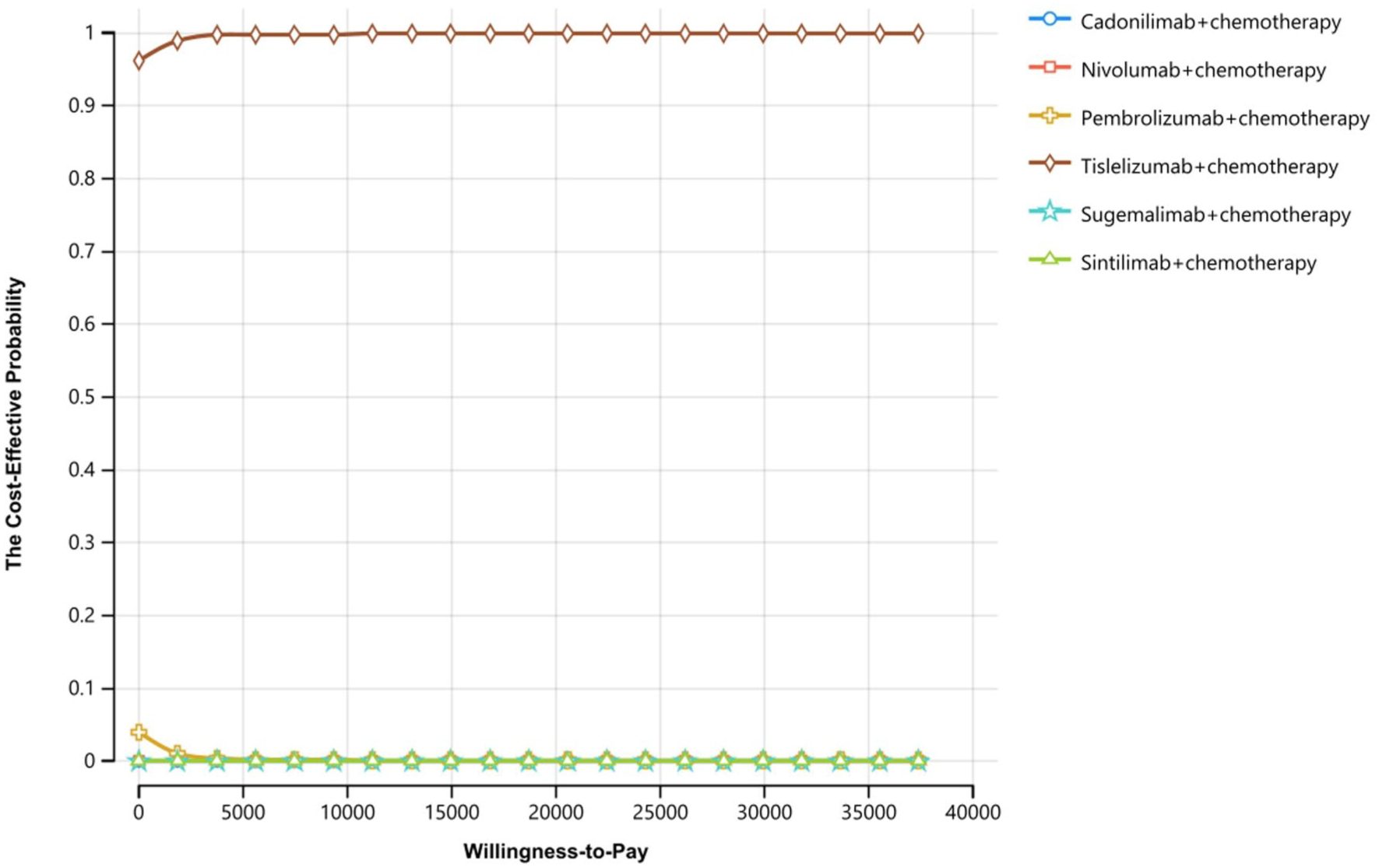
Acceptable curve plots (COMPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303).
Subgroup analysis
In the subgroup with PD-L1 CPS levels, the ICER exceeds the WTP threshold. When PD-L1 CPS ⩾5%, Cadonilimab + XELOX and placebo + XELOX produced 0.93 QALYs (1.43 LYs) and 0.48 QALYs (0.72 LYs), respectively. The ICER was $45,928.89 per QALY ($28,995.59 per LY). When PD-L1 CPS <5%, Cadonilimab + XELOX and placebo + XELOX produced 0.70 QALYs (1.07 LYs) and 0.51 QALYs (0.78 LYs), respectively, with ICERs of $86,205.12 per QALY ($56,921.74 per LY) (Table 2). This indicates that Cadonilimab was not a cost-effective option regardless of PD-L1 CPS levels (Figure 5), a conclusion also supported in other subgroups (Table S3).
Discussion
Gastric cancer is one of the most common malignancies in China. Due to the large population, the number of gastric cancer cases in China accounts for nearly half of the global total. 35 Statistical data indicate that approximately 35.1% of patients survive more than 5 years after diagnosis, 36 showing that gastric cancer remains a major health threat in China. Furthermore, the economic burden of gastric cancer treatment is significant. In 2017, total cancer treatment expenditure in China was 304.8 billion yuan, with cancer-related hospitalization costs reaching 241.9 billion yuan. Gastric cancer ranked among the top three cancers for hospitalization costs from 2005 to 2017. 37 In tertiary hospitals, gastric cancer had the highest inpatient costs compared to other cancers. 35 Therefore, when a treatment program provides significant benefits, evaluating its economic impact is crucial to help patients and clinicians make informed clinical decisions.
The COMPASSION-15 study showed that the combination of Cadonilimab and XELOX chemotherapy improved OS in the entire population, with more pronounced OS benefits when PD-L1 CPS ⩾5%. However, this study used a Markov model to analyze cost-effectiveness and concluded that Cadonilimab was not cost-effective. Subgroup analysis further showed that for patients with PD-L1 CPS ⩾5%, the treatment regimen was also not cost-effective. Furthermore, the probability of Cadonilimab being cost-effective at a WTP threshold of $37,386 was 43.7%. When PD-L1 CPS <5%, the probability of Cadonilimab being cost-effective at the WTP threshold of $37,386 dropped to 22.6%. Sensitivity analysis showed that the cost of Cadonilimab per cycle was the most influential factor in the entire population affecting the model. When the cost of Cadonilimab was reduced by 70%, the total cost and QALYs generated by the Cadonilimab + XELOX chemotherapy regimen were $17,750 and 0.73 QALYs, respectively. Compared to the placebo + XELOX chemotherapy regimen, the total cost increased by $8985, while generating an additional 0.25 QALYs. The resulting ICER was $36,538.97 per QALY. At a WTP threshold of $37,386, Cadonilimab is a cost-effective option for first-line treatment of advanced gastric cancer. In patients with PD-L1 CPS ⩾5%, when the cost of Cadonilimab per cycle is reduced by 30%, the Cadonilimab + XELOX regimen results in a total cost of $25,196 and generates 0.93 QALYs. Compared to the alternative treatment, this regimen incurs an additional cost of $16,510 and provides an incremental gain of 0.45 QALYs. The resulting ICER is $36,831.90 per QALY, indicating that Cadonilimab is cost-effective at this threshold.
During the sensitivity analyses of the clinical trials COMPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and GEMSTONE-303, a more appropriate approach involved introducing NMB as a key metric. Traditionally, cost-effectiveness ratio sensitivity analyses involve pairwise comparisons. When multiple intervention regimens are considered, selecting a common reference group, typically the standard treatment, is necessary. Subsequently, the ICERs of other regimens relative to this reference are calculated individually, followed by the sensitivity analysis. This method restricts the clear illustration of the comparative performance of all regimens across various WTP thresholds in a single graphical representation. Transforming the analysis through the formula: NMB = Effectiveness * λ – Cost (where λ represents WTP) shifts it from a ratio-based to a linear framework. For any given WTP threshold, the NMB of each treatment strategy can be directly calculated, identifying the strategy with the highest NMB as the most cost-effective at that threshold. Among the parameters influencing NMB, the utility values for PD and PFS, along with the costs associated with decreased platelet count, are the three most influential factors affecting model stability. Specifically, higher utility values for both PD and PFS enhance cost-effectiveness. When the utility value of PD ranges from 0.4616 to 0.6924, the corresponding NMB varies from $16,511 to $23,364. Similarly, when the utility value of PFS ranges from 0.6376 to 0.9564, the corresponding NMB varies from $17,014 to $22,861. In addition, lower costs associated with decreased platelet count enhance cost-effectiveness. When the costs associated with a decline in platelet count range from $2988 to $4482, the corresponding NMB varies from $19,252 to $20,623. By contrast, the costs of Cadonilimab, Sugemalimab, Nivolumab, Pembrolizumab, and Sintilimab had minimal impact on the results of the sensitivity analysis. This is because these treatments were not identified as cost-effective options compared to Tislelizumab in the model.
PD-1/PD-L1 antibodies are widely used in the first-line treatment of gastric cancer, supported by data from phase III clinical trials such as KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and Gemstone-303. Consequently, Pembrolizumab, Nivolumab, Sintilimab, Tislelizumab, and Sugemalimab have been successively approved by the Chinese CSCO guidelines 9 for the first-line treatment of HER2-negative advanced GC/GEJC. In this study, we evaluated the cost-effectiveness of Cadonilimab compared to other ICIs. Our results indicate that Cadonilimab is not cost-effective compared with other ICIs. Tislelizumab emerges as the most cost-effective option. We also performed a linear analysis of incremental NMB across various WTP thresholds, which further confirmed this conclusion (Figure 7). Pembrolizumab is more cost-effective than Nivolumab, consistent with the findings of Zhu et al. 23 However, Chinese studies30,38,39 have found neither Nivolumab nor Pembrolizumab to be cost-effective, which does not contradict our results. Our analysis compares Pembrolizumab primarily with Cadonilimab, not with chemotherapy alone as in the KEYNOTE-859 and CheckMate-649 trials. Likewise, the reported cost-effectiveness of Sintilimab in China 40 aligns with our study’s conclusions.

Linear analysis chart of NMB.
This study has several limitations: First, the data are derived from the COMPASSION-15, KEYNOTE-859, CheckMate-649, ORIENT-16, RATIONALE-305, and Gemstone-303 trials, and any biases in those trials may influence our findings. Second, because these trials did not report health utility values by treatment group, we used utility estimates from external studies, which may introduce bias. Third, for adverse reaction cost estimation, we excluded grade 1–2 events with an incidence below 5%, affecting direct medical costs. However, one-way sensitivity analyses showed that this exclusion did not compromise model stability. Fourth, in the absence of head-to-head trials, we relied on indirect comparisons to evaluate the drugs’ cost-effectiveness. This approach may introduce heterogeneity due to differences in trial cohorts. However, the enrolled populations share similar baseline characteristics, which mitigates this heterogeneity. Sixth, this study is based on data from China’s healthcare system and evaluates the cost-effectiveness of Cadonilimab as a first-line immunotherapy for advanced GC and GEJC from the perspective of Chinese healthcare payers. Consequently, the findings and implications of this study are specifically applicable within the context of China’s healthcare system. Finally, clinical guidelines recommend multiple second-line therapy options after disease progression. In our analysis, we assumed all patients received docetaxel, which may not reflect real-world practice where clinicians individualize treatment based on each patient’s condition. However, one-way sensitivity analysis showed that variations in second-line treatment costs had a negligible effect on the ICER.
Conclusion
In conclusion, from the perspective of Chinese payers, Cadonilimab is not cost-effective compared to chemotherapy alone or to other ICIs approved in China, whereas Tislelizumab represents the most cost-effective option among all treatments evaluated.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251381143 – Supplemental material for Cost-effectiveness analysis of Cadonilimab in first-line treatment of advanced HER2-negative gastric cancer or gastroesophageal junction cancer
Supplemental material, sj-docx-1-tag-10.1177_17562848251381143 for Cost-effectiveness analysis of Cadonilimab in first-line treatment of advanced HER2-negative gastric cancer or gastroesophageal junction cancer by Kaiqi Zhu, Mengyao Qin, Zhaoyi Pan and Jin Huang in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
