Abstract
The advent of combination immunotherapy has revolutionized the treatment of metastatic renal cell carcinoma (mRCC), leveraging immune checkpoint inhibitors and tyrosine kinase inhibitors to improve survival outcomes. However, these advancements come with a significant challenge: the management of adverse effects, which can impair patients’ quality of life and lead to therapy discontinuation. This review explores the prevalence, mechanisms, and management of gastrointestinal (GI) toxicity associated with immunotherapy combinations for mRCC from the gastroenterologist’s perspective. Diarrhea, colitis, nausea, and vomiting are the most frequently reported adverse events, often necessitating a multidisciplinary approach for timely diagnosis and intervention to mitigate risks and ensure therapeutic continuity. We highlight the management strategies, ranging from symptomatic treatment and dietary modifications to advanced therapies for severe cases, even discussing the emerging approaches for refractory cases. By integrating oncological and gastroenterological expertise, clinicians can optimize outcomes for patients while minimizing GI-related complications.
Plain language summary
The introduction of combined immunotherapy and chemotherapy treatments has revolutionised the treatment of tumour diseases, such as advanced renal carcinoma. Immune checkpoint inhibitors enhance the immune system's ability to recognise and attack the tumour by ‘reactivating’ immune cells. Tyrosine kinase inhibitors acting by blocking signals that help the tumour grow, such as the formation of new blood vessels.
However, these therapies can cause significant gastrointestinal side effects, (i.e. diarrhea, colitis, nausea and vomiting), which negatively affect patients’ quality of life and may require discontinuation of treatment. This review highlights:
• the importance of multidisciplinary management of gastrointestinal side effects to ensure continuity of treatment.
• the spectrum of treatment options, ranging from symptomatic drugs to advanced therapies for the most severe cases.
• the new promising approaches emerging to address refractory cases.
Integration between oncologists and gastroenterologists is crucial to improve clinical outcomes and reduce gastrointestinal complications related to these innovative treatments.
Keywords
Introduction
The recent introduction of novel immune checkpoint inhibitors has led to a paradigm shift in the therapeutic landscape for advanced renal carcinoma. Currently, immunotherapy combinations represent the first-line standard of care for metastatic renal cell carcinoma (mRCC) and entail the use of two immunotherapy agents or two drugs with distinct mechanisms of action, immune checkpoint inhibitors (ICIs) and tyrosine kinase inhibitors (TKIs). 1 These therapeutic strategies are supported by a strong biological rationale and work synergistically to counteract two main mechanisms that promote tumor growth: angiogenesis and the immune evasion process. 2
Most solid tumors exhibit vascular abnormalities, which are attributed to the high concentrations of Vascular Endothelial Growth Factor (VEGF) and Angiopoietin 2 (ANG2) in the tumor microenvironment (TME). Drugs blocking these pathways promote vasculature normalization, thereby facilitating immune cells’ infiltration into the TME and enhancing their intrinsic antitumor action. 3
Immune cells play a crucial role in fighting cancer, but they undergo mechanisms of tolerance and loss of the ability to recognize and prevent tumor growth.
Programmed death 1 (PD-1), programmed death ligand 1 (PD-L1), and T lymphocyte-associated antigen 4 (CTLA-4) were identified as key players in the peripheral T-cell tolerance mechanism (PD1-PDL1 interaction) and the immune evasion process during the priming phase (CTLA4-CD80/86 interaction), respectively.4,5
These interactions, which normally prevent aberrant autoimmune responses under physiological conditions, in this instance, are used to turn off the immune cell response against tumor cells, resulting in premature immune cell inactivation, desensitization to immunological stimuli, and undisturbed tumor proliferation. PD-1, PD-L1, and CTLA-4 inhibitors stimulate the restoration of antitumor activity.
While ICIs attempt to reawaken exhausted immune cells in their innate fight against tumor cells, anti-angiogenics can restore the vasculature in the TME, enhancing the potential of infiltration of effector immune cells and thereby increasing therapy efficacy. 5
Several phase III studies have explored the efficacy of immunotherapy-based combinations in the mRCC first-line treatment and demonstrated unprecedented response rates and improved survival in the metastatic setting, compared with TKI monotherapy.6–15
As new drugs are introduced, patient risk stratification becomes crucial. This approach employs validated predictive factors to determine who may benefit from one treatment over another.
Two primary risk scores have been identified, incorporating known survival predictors: Memorial Sloan-Kettering Cancer Center and the International Metastatic RCC Database Consortium. 16 If none of the factors are present, the disease has a favorable prognosis; an intermediate prognosis is assigned if one to two are present, and a poor prognosis if more than three are present.
Despite significant progress made with the use of these drugs in combination, it is crucial to underline the need to successfully prevent and counteract side effects. This is the only way to ensure a good quality of life for patients along the course of treatment and, as a result, a successful outcome, keeping in mind that the occurrence of adverse events is the primary reason for drug discontinuation and dose reduction.
In this review, we analyze the evidence regarding gastroenterological adverse events of immunotherapy combinations in mRCC, focusing on their prevalence, prevention, and management.
Gastrointestinal toxicity of TKIs treatment
Side effects associated with TKIs, including gastrointestinal (GI) effects, are a significant concern in cancer treatment. GI side effects are frequently reported in cancer patients treated with TKIs, indicating their high prevalence and can sometimes necessitate the premature discontinuation of life-saving cancer therapies.17,18 Toxicity is often nonspecific and associated with most antineoplastic drugs used to treat malignancies. 19
Mild diarrhea, graded 1–2 according to the Common Terminology Criteria for Adverse Events (CTCAE), is the most frequently reported manifestation. 19
Other toxicities include nausea, vomiting, constipation, stomatitis, dysgeusia, and dyspepsia. More severe manifestations, such as colon perforation and severe, life-threatening diarrhea and colitis, have rarely been described.20–23 From a pathogenetic perspective, potential mechanisms are widely debated, and many theories have been proposed. The role of several tyrosine kinase receptors in regulating chloride secretion is well known, and diarrhea appears to be mostly secretory. 24 Accordingly, due to altered regulation of the activation of chlorine channels, including the cystic fibrosis transmembrane conductance regulator and calcium-activated channels. 25 They increase fluid secretion in the intestinal lumen, while inhibition of intestinal sodium transport reduces fluid absorption. Epidermal Growth Factor Receptor (EGFR) has an inhibitory effect on chloride secretion, and therefore it has been hypothesized that its inhibition may result in excessive chloride secretion in the gut lumen. 25
However, diarrhea may also result from inhibition of epithelial growth and limited healing of the gastrointestinal mucosal lining. It must be considered that the EGFR and VEGF Receptor signaling pathways are highly expressed in the gut and have stimulatory effects on enterocyte proliferation and electrolyte transport, causing structural and functional changes when inhibited, reducing the gut absorptive capacity. 25
Other hypotheses include direct toxicity on mucosal cells and increased gastrointestinal inflammation.24,26 These theories are supported by the dose-dependent effects of toxicity. 26
In addition, increased levels of inflammatory mediators or toxic drug metabolites may also play a role in gastrointestinal adverse events (AEs). Interestingly, immunomodulatory effects of cabozantinib have been described, with a significant increase in CD81 T-lymphocytes and CD141 monocytes, which could also contribute to diarrhea. 27 CD81 is known to regulate the immune system, and probably contributes to colic inflammation due to its activity on cell-to-cell communication between antigen-presenting cells (APCs) and T-lymphocytes. 27
Focusing on TKIs used in first-line treatment of mRCC, diarrhea is frequently reported in patients treated with axitinib as monotherapy (incidence of 45% of all grades and 9% of grade 3). Other effects reported during axitinib are nausea, 20% of each grade, and 1% of grade 3.28,29
In case of cabozatinib therapy, the following data were reported: 75% incidence of diarrhea in all grades and 13% of grade 3, 53% and 34% incidence of nausea and vomiting across all grades, and 5% and 2% for grade 3, respectively. 30 The three main toxicities associated with lenvatinib are diarrhea, vomiting, and stomatitis. Multiple clinical studies have identified diarrhea (incidence rate of 45%–67%) as the most common gastrointestinal event associated with lenvatinib, with nausea, vomiting, and decreased appetite reported in 18%–75% of patients. 30
These side effects are generally characterized by rapid onset, mild and often self-limiting, and mostly related to the TKI dose. 20
Gastrointestinal AEs during ICIs
Of the adverse effects of immunotherapy, a notable role is reserved for gastrointestinal toxicity.
As previously discussed, these effects can often lead to therapy discontinuation and a worsening of patients’ quality of life, especially in those with chronic immune-related adverse events (IRAEs). 31
The severity of manifestations is graded from 1 to 5 on an increasing scale, according to the CTCAE. 28 Side effects may affect the upper and lower digestive tracts, the hepatobiliary system, and the pancreas. 32
The exact pathophysiology underlying these effects is not yet fully understood; however, they seem to be a consequence of the drug’s pharmacodynamics. Immunotherapy can interfere with immunotolerance mechanisms that prevent inappropriate reactions to self-antigens and commensal microorganisms, with hyperactivation of T-cell effectors, increased memory T-cells, lymphocyte infiltration, and cytokine activation.32,33 Specifically, some reports show that activated cytotoxic CD8+ T-cells appear to derive from tissue resident cells (Trm). 34 On the other hand, increased expression of genes coding for the integrin α4β7 receptor and integrin αEβ7 was also observed in the T-cells of these patients. These integrins, through their interaction with mucosal addressin cell adhesion molecule-1 (MadCAM) on the intestinal endothelium, participate in lymphocyte recruitment and retention of these cells in the gut. 35
Moreover, Interleukin (IL)-17, a pro-inflammatory cytokine produced by T helper (Th)17 cells, exerts a pivotal effect on immune activation, which is involved in the pathogenesis of autoimmune diseases and tumor progression. Serum levels of IL-17 are increased during ICI colitis, and elevated basal levels are associated with severe diarrhea. 35
The innate immune system also contributes significantly, particularly through IL-6: its blockade was associated with improved antitumor efficacy of αCTLA-4 and reduced gastrointestinal toxicity when combined with antibiotics. 36 Furthermore, the severity of ICI colitis correlates with the expansion of group 3 innate lymphoid cells (ILC3), which regulate bacterial immunity and microbiota balance. 35
The PD-1 axis is crucial in the maturation of ILC3, and the altered interaction between the immune system and the gut microbiota plays a central role. Some bacteria appear to trigger inflammation; Faecalibacterium prausnitzii is associated with increased proliferation of cytotoxic T-lymphocytes and recruitment of T-reg and α4β7 cells in the intestinal tract and tumor microenvironment, enhancing therapeutic efficacy but also promoting the occurrence of colitis. 37
Regarding the upper digestive tract, nausea, gastritis, duodenitis, and oesophagitis have been observed. Nausea is a nonspecific symptom, often associated with vomiting; it is reported in 12% of patients treated with PD-(L)1 inhibitors, in 19% of those treated with CTLA-4 inhibitors, and in 25% of subjects treated with immunotherapy combination. 38 Nausea and vomiting mainly occur as single manifestation but increase in severity when associated with other conditions such as infections, endocrinopathies or organ damage as reported below; however, only 2% of cases present with a grade >3. 38 Rarely, lymphocytic gastritis, cytomegalovirus-related gastritis, neutrophilic gastritis, gastric ulcerations hemorrhagic gastritis and ulcerative oesophagitis have been observed. 39
Rare cases of new onset of coeliac disease have also been described, the first characterized by enteropathy with villous atrophy (VA), and the second characterized by ulcerative enteritis, sometimes with massive gastrointestinal hemorrhage or perforation of the small intestine, but without VA.40,41
Diarrhea is a common side effect; however, it can be a warning sign of an underlying colitis. Colitis is an inflammatory condition of the colon, which represents a serious and potentially life-threatening adverse event, being considered the central gastrointestinal toxicity of ICIs.32,34
Focusing on drugs currently used in the treatment of mRCC, ipilimumab was associated with a 36% incidence of diarrhea of all grades and 5% of grade 3–4. For Nivolumab and Pembrolizumab, the incidence of this side effect is approximately 11%, with grades 3–4 less than 1%.42,43
The most significant toxicity is generally linked to anti-CTLA-4 therapy, probably due to a more generalized immune response producing massive T-cell proliferation, as compared to anti-PD1/PD-L1 drugs. 44 Notably, colitis resulting from PD-(L)1 inhibition is characterized by a variable onset throughout treatment and often by a more indolent presentation than that resulting from CTLA-4 inhibition. 44 Uncertain data concern the relationship between the incidence of colitis and therapy dosage: the use of high doses of ipilimumab, either as monotherapy or in combination with PD(L)1 inhibitors, results in an increased frequency of colitis compared to low doses.45,46 In general, the onset of colitis can occur both during treatment and after its end. CTLA-4 inhibitors cause a later onset of colitis than PD-1/PD-L1 inhibitors, while the combination of the two can lead to an earlier incidence. Specifically, the median onset of diarrhea is around 6–7 weeks for CTLA-4 inhibitors. In contrast, the time to onset with PD-1/PD-L1 inhibitors can be highly variable, ranging from as early as 1 week to as late as 2 years. For combination therapy (CTLA-4 inhibitors plus PD-1/PD-L1 inhibitors), the median onset of diarrhea is 7 weeks (range: 0–51 weeks). 47 Diarrhea is the main symptom of colitis, often associated with gastrointestinal bleeding and, in some cases, abdominal pain, fever, vomiting, and nausea. Colic perforation has rarely been described, especially in cases of late diagnosis. 48 Compared to chemotherapy, IRAEs generally last longer and are mild to moderate in severity. 49
Gastrointestinal AEs of mRCC immunotherapy combinations
Approved combinations as first-line therapy of mRCC are pembrolizumab + axitinib, nivolumab + cabozantinib, pembrolizumab + lenvatinib, and nivolumab + ipilimumab. 50 Gastrointestinal side effects identified in the most important clinical trials investigating the combination of TKIs and immunotherapy in advanced renal cell carcinoma are shown in Table 1.
Gastrointestinal side effects in the most important clinical trials investigating the combination of TKIs and immunotherapy in advanced renal cell carcinoma.
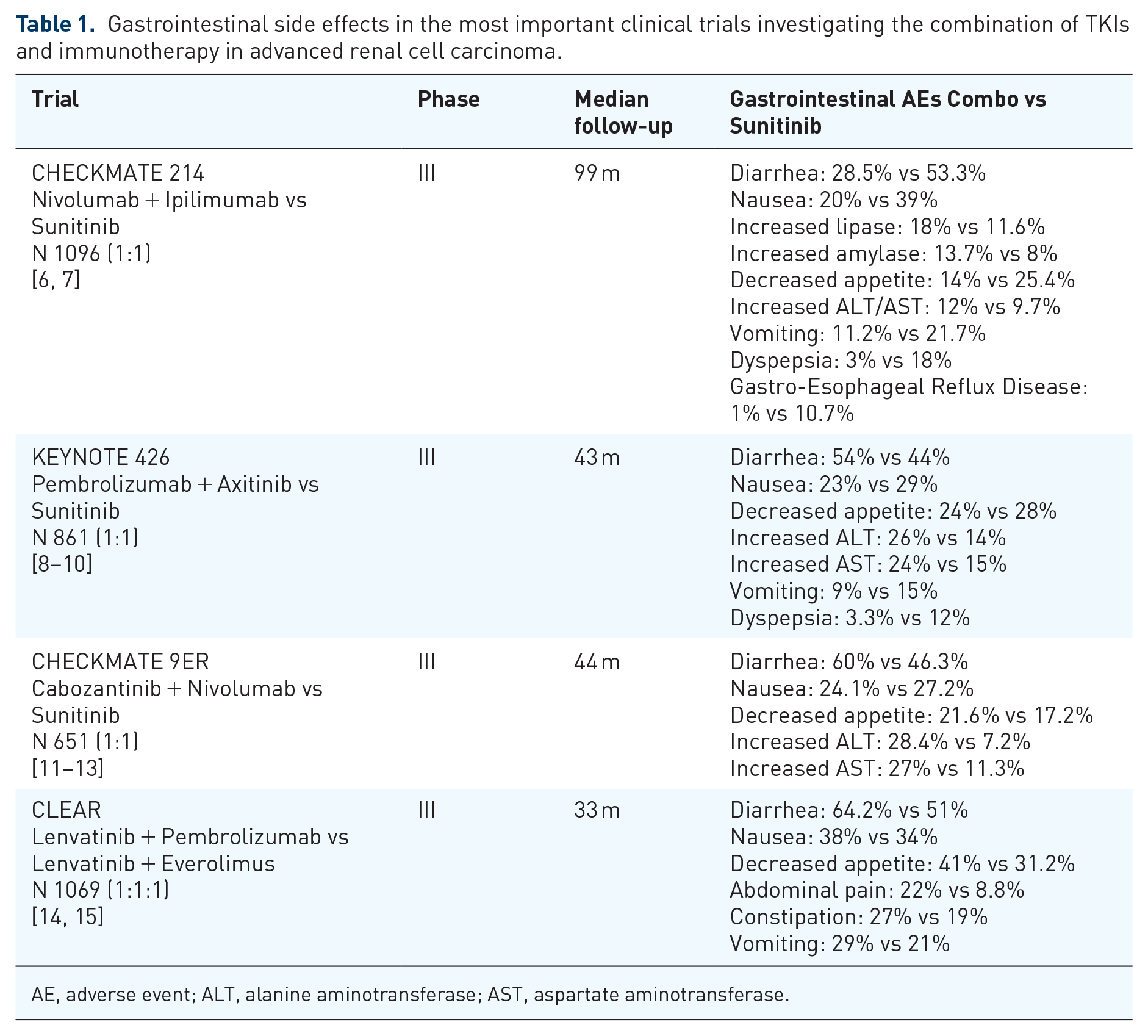
AE, adverse event; ALT, alanine aminotransferase; AST, aspartate aminotransferase.
In a recent meta-analysis, Rizzo et al suggested an increased risk of gastrointestinal toxicity for combination therapy, in particular, increased risk of diarrhea of all grades, grade 3–4 diarrhea, and grade 3–4 decreased appetite compared to sunitinib monotherapy. 45 Similar data also came from Kovalenko et al. 51
In case of pembrolizumab + axitinib, diarrhea was reported in 54.3% while immune-related colitis was reported in 3% of patients. 52
Focusing on the nivolumab + cabozantinib combination, in the study by Laccetti et al., the onset of diarrhea affected approximately 60% of patients, while nausea occurred in 31%–37.5%. 52
Furthermore, a systematic review and network meta-analysis showed that cabozantinib + nivolumab is the worst option in terms of grade ⩾3 vomiting, while the mean risk of vomiting of any grade is the highest for lenvatinib + pembrolizumab (27.9%). 53
Considering the combination of nivolumab + ipilimumab, diarrhea of any grade and grades 3–4 were reported in 13.5% and 8% of cases, respectively; no grade 5 GI IRAEs occurred after 24 months minimum of study follow-up. In general, the combination of the two immunotherapies leads to an earlier onset of GI toxicity than the single drug, with a median of 7 weeks and a range of 0–51 weeks. 54
In a recent meta-analysis, the incidence of diarrhea of all grades was analyzed in a phase III RCT testing immunotherapy combinations in mRCC. The results were as follows: avelumab + axitinib (86.4% of cases), pembrolizumab + lenvatinib (48.8%), pembrolizumab + axitinib (42.9%), nivolumab + ipilimumab (70.3%), nivolumab + cabozantinib (40.8%). 55 However, Nielsen et al., considering also other cancers, suggested that nivolumab + ipilimumab had a higher incidence of diarrhea occurring mainly in the phase of induction treatment. 56 Nowadays, we have no data on the toxicity and treatment efficacy of immunotherapy combinations toxicity.53–56
Gastrointestinal evaluation at baseline and monitoring during and after first-line mRCC therapy
From the gastroenterologist’s point of view, patients undergoing immunotherapy combinations should be educated at baseline about early recognition of symptoms, estimating bowel movements, and stool consistency to recognise any changes. 57 No specific gastroenterological examinations must be performed at baseline; however, general hematochemical evaluation, comprehensive of a metabolic panel, and thyroid balance is helpful. A crucial aspect is to thoroughly evaluate the patient’s medical history, paying attention to chronic diarrhea or occult symptoms.
Notably, a screening colonoscopy is not recommended for all patients before starting therapy, and fecal calprotectin assay at baseline is not indicated unless the patient has preexisting chronic inflammatory bowel disease (IBD) (Figure 1).
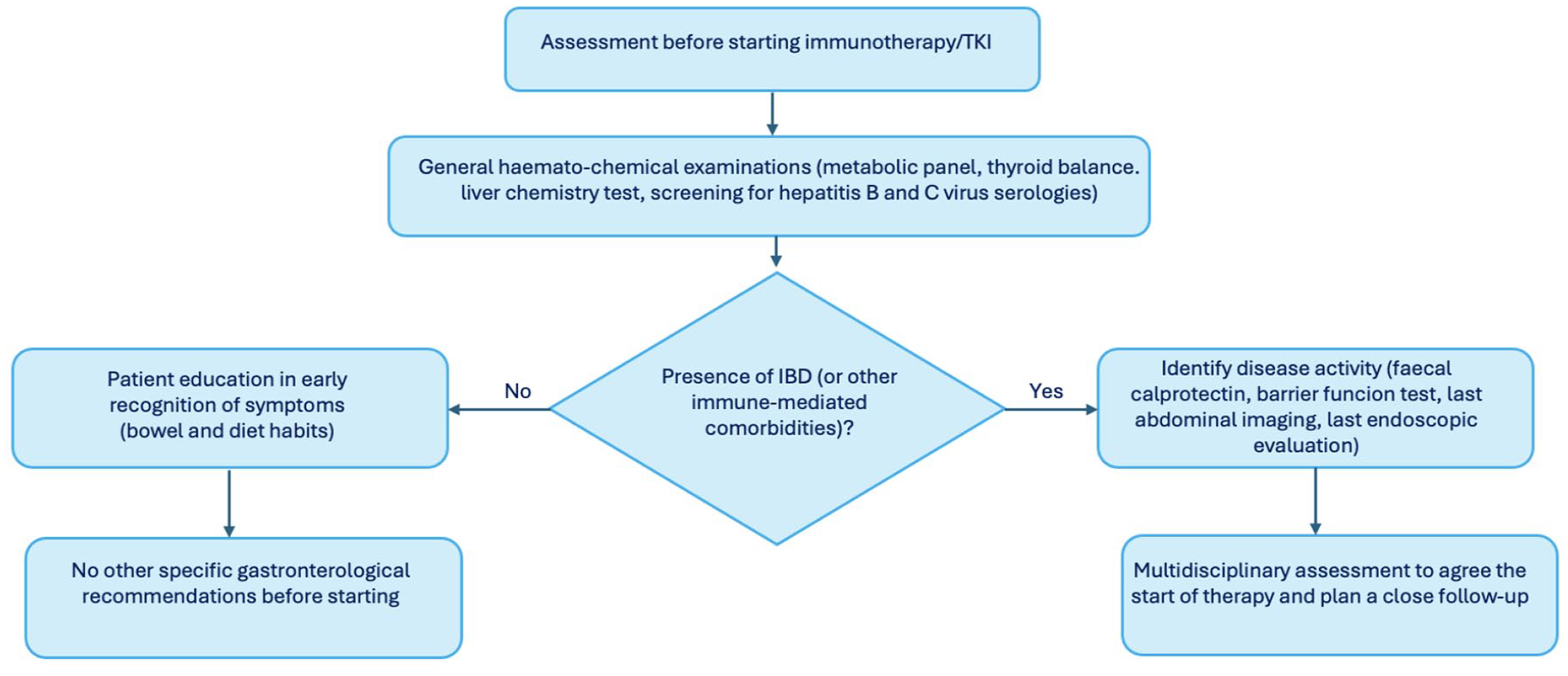
Practical considerations for assessment before starting immunotherapy/TKI.
The patient should undergo baseline assessment of liver chemistry tests (total bilirubin, alkaline phosphatase, aspartate aminotransferase (AST), and alanine aminotransferase (ALT)) and pre-treatment screening for hepatitis B and C virus serologies. Liver chemistry tests should be repeated before each course of ICI treatment, and management should be based on the CTCAE grade of toxicity. 58 Hepatitis is often asymptomatic and typically presents with elevated serum levels of ALT and AST. 59
It should be noted that other sources of ALT/AST increase include muscle. Hence, in case of AST/ALT elevation, it may be useful to evaluate creatine phosphokinase (CK) levels to ensure that myositis/myocarditis is not missed.
In addition, liver tests should be rechecked weekly for patients with grade 1–2 liver toxicity and every 1–2 days for patients with grade ⩾3 liver toxicity. 58
Notably, regarding the prevention of gastrointestinal toxicity, there are no specific recommendations. Budesonide has demonstrated a lack of efficacy as a prophylactic treatment in this setting, presumably attributable to its inability to fully suppress the various inflammatory pathways involved, as well as potential limitations in its absorption or specific site of action. 57 There are insufficient data for biomarkers predictive of toxicity, and those that have been described are not applicable in clinical practice, although IL-6 levels, IL-17, and some fecal microbial profiles at baseline were associated with the development of colitis in patients treated with ipilimumab.60–62
Interestingly, Lippenszky et al. have recently reported a novel approach for the prediction of IRAEs, including colitis, in patients treated with ICIs. 63 The colitis model, based on random forest and developed on real-world clinical data, showed good performance (AUC = 0.755). A strength of the model is its exclusive use of structured data already available in clinical practice, without the need for additional testing. However, the relatively high sensitivity (75%) is counterbalanced by a more modest specificity and positive predictive value, underlining the need for careful prospective validation before routine application.
In addition, a real-life, multicentre, retrospective Italian observational study found that a higher BMI was significantly correlated with a higher occurrence of IRAEs of any degree, including those regarding the gastrointestinal and hepatic tract, compared to normal-weight patients. 64
It is crucial to note that the risk of gastrointestinal AEs differs between anti-CTLA-4 and anti-PD-1 therapies. The distinct mechanisms of anti-CTLA-4 and anti-PD-1 therapies, central versus peripheral immune checkpoint modulation, result in their different safety profiles, with anti-CTLA-4 inducing broader immune dysregulation during early thymic selection of T-cells. Consequently, AEs such as nausea, diarrhea, colitis, and hepatitis are more frequently observed with CTLA-4 inhibitors compared to anti-PD-1 agents. Furthermore, combination therapy with CTLA-4 and PD-1 inhibitors is associated with a significantly higher incidence of IRAEs than monotherapy. This increased GI toxicity risk with anti-CTLA-4 inhibitors may influence treatment decisions, particularly in patients with preexisting autoimmune gastrointestinal conditions.4,5
The lack of reliable methods able to identify patients at increased risk of developing gastrointestinal toxicities represents an unmet need in this context, and in particular in patients with preexisting GI disorders. For example, in patients with autoimmune diseases, especially IBD, the safety of such therapies is debated. In those patients, a fourfold higher risk of developing adverse gastrointestinal events compared to patients without IBD has been described, with increased risk of colitis primarily related to anti-CTLA-4 administration. 65 A recent meta-analysis reported an overall incidence of IBD exacerbations after cancer treatment of 30%, with a significantly increased risk of gastrointestinal toxicity from ICI. 66 Current expert recommendations suggest starting therapy only during clinical remission, with the precaution to avoid combination therapy (anti-CTLA plus PD-(L)1 inhibitors). In these patients, it is usually recommended to perform a complete endoscopic and biomarker assessment of disease activity before treatment. Close follow-up is critical to provide timely intervention and ensure positive outcomes. 67 While maintenance treatment management for IBD (immunosuppressants or biological drugs) should be coordinated with the oncologist, considering the risk of oncological progression.
Management of gastrointestinal side effects
Nausea and vomiting treatment
Nausea and vomiting are frequent symptoms in patients receiving those therapies. Notably, they may be an expression of conditions affecting the upper gastrointestinal tract, including infections, endocrinopathy, gastrointestinal metastases, or IRAEs. 58
There are several effective options for treating nausea, and the most suitable agent often depends on the individual patient’s assessment. 68 Generally, corticosteroids are not recommended for therapy-related nausea and vomiting. Patients who develop grade ⩾2 ICIs-related nausea and vomiting should receive antiemetics (centrally or peripherally acting as ondansetron, metoclopramide, or domperidone). 69 If no benefit is observed within 5–7 days, esophagogastroduodenoscopy should be considered to rule out the adverse effects on the upper digestive tract. 69
Diarrhea management (grade 1–2 CTCAE)
Management of diarrhea is usually guided by the degree of toxicity according to the CTCAE grading system. Although different conditions, diarrhea may be an expression of an underlying colitis. The grading system and its corresponding management strategies outlined in the guidelines are applicable to both conditions.
Regardless of the grade, a diagnostic work-up should be performed, including complete blood tests: blood count, electrolytes, C-reactive protein, thyroid function, copro-culture, parasitological examination, Clostridium difficile test (toxin and glutamate dehydrogenase antigen), and quantitative fecal calprotectin assay. 57 It is crucial to document the absence of infection as a priority before starting any corticosteroid therapy 57 (Figure 2). In addition, rarer etiologies, such as new-onset coeliac disease and immune-mediated pancreatic insufficiency, should also be investigated in patients who do not respond to conventional therapy or who report steatorrhea.41,42

Management algorithm for diarrhea/colitis.
For patients receiving TKI/immunotherapy combination, the AGA (2021) and ASCO (2021) guidelines recommend against discontinuation of ICI in case of grade 1 diarrhea/colitis, while discontinuation of both drugs is considered from grade 2 onwards.57,69
Initially, it may be helpful to only consider the TKI discontinuation and reassessment after 2 or 3 days. The pharmacokinetic characteristics of the different molecules are useful in identifying the drug involved. Considering the relatively short half-life of TKIs and their rapid onset of action, a prompt improvement of symptoms (48–72 h) without corticosteroid administration suggests that toxicity is probably TKI related.70,71 In grade 1 or 2 cases, symptomatic treatment is based on a fiber-free diet, adequate hydration, and antidiarrheal treatment with antisecretory agents or intestinal absorbents (es diosmectite) rather than transit-slowing treatments (es loperamide).57,69 Although the latter drugs are suggested in some guidelines, they should be avoided in the first instance, especially in cases of fever, as microbial overgrowth must always be suspected and ruled out. Hospitalization and endoscopic evaluation are generally unnecessary at this stage.72,73
If gastrointestinal symptoms are TKI related, these drugs can be reintroduced with enhanced preventive measures as discussed above. 72
Although not mentioned in the guidelines due to a lack of solid efficacy data, at this stage it might be useful to administer probiotics, containing Lactobacillus rhamnosus, Bacteroides fragilis, or Lactobacillus reuteri, due to the immunomodulatory effects shown in mice; only a few data are available on humans in this context.73,74
Notably, the oral intake of the probiotic VSL#3® 450B for reducing the incidence of ICI-induced colitis in human patients (NCT06508034) is currently under investigation.
If toxicity is immunotherapy-related, corticosteroid (CS) treatment should be considered. They can be administered systemically by the oral route (prednisolone 1 mg/kg) or by the digestive route (budesonide 9 mg/day). However, locally acting corticosteroids are not recommended in major guidelines, despite the positive results reported with budesonide in ICI-induced microscopic colitis and in colitis recurrences.74–76
Grade 2 diarrhea/colitis requires the use of CS until symptoms regress to grade 1 or less. Once it improves, CS tapering can be initiated over 4–6 weeks until termination. In addition, ICI should be temporarily discontinued until improvement or, preferably, resolution of symptoms.57,70 Generally, grade 2 diarrhea/colitis requires outpatient management; however, hospitalization may be considered in case of systemic symptoms (e.g, tachycardia, dizziness, and high fever). 73
Early endoscopic evaluation should be strongly considered, as this allows physicians to obtain diagnostic biopsies and to recognise high-risk group patients (presence of deep ulcers and/or extensive colonic involvement), who are more frequently steroid-refractory colitis. 77 In this setting, the optimal extent of endoscopic examination has not been established. Usually, if the patient’s condition permits, a complete colonoscopy is preferred, as there may be regional variability in the inflammation severity. Otherwise, a flexible sigmoidoscopy is often sufficient to make a diagnosis. 57
In addition, shorter steroid treatment and fewer recurrences have been described in patients who underwent endoscopic evaluation within 1 month, particularly within 1 week of symptom onset. 77
Furthermore, approximately one-third of patients with ICI enterocolitis respond inadequately to first-line glucocorticoid treatment and may require second-line immunosuppression. Patients who do not respond to high-dose glucocorticoids within 72 h of therapy start or who do not have a complete response within 1 week should be considered for second-line immunosuppression. 78
Patients developing recurrent symptoms during steroid tapering or after completing a steroid course should be considered for second-line immunosuppression. 79 Interestingly, colonic ulceration is the only currently identified predictive factor associated with the need for secondary immunosuppression, whereas CTCAE grading was not predictive; consequently, identifying patients with colonic ulceration is one of the crucial roles of endoscopic evaluation of patients with suspected enterocolitis. 79
Moreover, the impact of CS on tumor growth is still unclear. As a result, exposure to CS should be minimized, and the dosage of CS therapy should be reduced as soon as possible. 80
Diarrhea management (grade 3–4 CTCAE)
In the case of grade 3–4 or no improvement in grade 2 diarrhea, hospitalization should be considered. Endoscopy with biopsies and abdominal imaging should be performed to exclude the presence of perforations or mechanical complications. In case of severe involvement, a surgical consultation should be sought.57,58 Treatment starts with high-dose intravenous (IV) corticosteroids (methylprednisolone 1 mg/kg/d) in addition to the recommendations for the previous grades. If there is no clinical improvement after 4–5 days of corticosteroid therapy, initiation of biological therapy is required.
Notably, in case of refractoriness to CS or persistent/recurrent symptoms, if not previously performed, a second-level endoscopic examination with biopsy should be considered to exclude Cytomegalovirus infection. 81
Recent data have shown that in high-risk patients, early introduction of infliximab (IFX) or vedolizumab (VDZ) was strongly associated with reduced hospitalizations and duration of steroid treatment. The selection of patients was mainly based on the severity of colitis and response to CS therapy, high calprotectin levels, the presence of extensive and deep mucosal ulceration, and inflammation extending beyond the left colon. 82 Overall, the response to biological therapy is around 96%. 83 To date, the choice of biological treatment and its dosage is based on experience and knowledge gained with IBD. 84 Therefore, current guidelines recommend IFX as first-line therapy for high-risk patients and CS refractory colitis. VDZ could be a viable alternative.78–80
Biological drugs usually act rapidly, with visible responses in less than a week. Complete resolution of symptoms may occur after a single infusion of IFX or VDZ, although the relapse rate may be lower in patients receiving three infusions compared to 1 or 2.84,85 The recurrence incidence was estimated to be around 44% for CTLA-4 inhibitors and 34% for PD-1/PD-L1 inhibitors.
Of note, in approximately 1%–1.5% of cases, ICI colitis may be complicated by colic perforation, requiring prompt surgical intervention. In these cases, emergency colectomy with subtotal colectomy is the treatment of choice due to the extensive nature of the inflammation.69,86
Resumption of therapy
Factors advising against the resumption of anti-cancer therapy include severe or life-threatening IRAE and the need for prolonged or multiple immunosuppressants.
A retrospective analysis of patients who exhibited grade ⩾ 2 toxicity with immunotherapy evaluated rechallenge. 87 Half of the patients underwent rechallenge with immunotherapy (n = 40). Toxicity was grade 2 in 46% and grade 3 or 4 in 54% of patients. Of the entire cohort, 43% experienced a recurrence of the initial toxicity, and 13% experienced a different toxicity. Overall, the new AEs were no more severe than the first toxicities. Progression-free survival and overall survival were no different between the two cohorts. Patients who have experienced grade 2 IRAE may be retreated with ICI therapy if toxicity was resolved or is controlled with ⩽10 mg prednisone (or equivalent) per day. The decision to restart treatment in a patient who has experienced grade 3 or 4 IRAE must be risk-adjusted according to the expected benefit of therapy versus potential toxicity.58,88
Future promising therapies
In analogy to the IBD model, the literature also reports cases of use of other therapies, such as IL-12-23 blockers and Janus kinase (JAK) inhibitors.32,89
Ustekinumab, a monoclonal antibody inhibiting interleukin-12 and interleukin-23, has shown promising results in patients refractory to steroids or other biological therapy. However, only rare case reports of very few patients are available in the literature.90,91
Similarly, Jak inhibitors, blocking JAK-TYK2-STAT pathway, have the potential to regulate downstream an endless array of key molecular mediators in the regulation of inflammatory processes, and have shown interesting results in this field. 91
Bishu et al. described Tofacitinib efficacy in 3 out of 4 patients (75%), with clinical remission and rapid endoscopic improvements. 92 In general, the cases available concern patients who experienced treatment failures across various conventional and advanced therapies. Although it should be noted that in a few cases, Tofacitinib was applied as the first advanced therapy after steroids, with positive efficacy.92–95 Notably, for both drugs, no new safety alerts emerged in these exploratory studies, but like efficacy, safety is an objective that should be investigated in larger studies.
Considering future innovative approaches, a recent prospective phase Ib/2 study evaluated the effect of local adiponectin induced by extracorporeal photophoresis (ECP) as a treatment for IRAEs. 96 The study was conducted on 14 patients presenting with colitis, hepatitis, dermatitis, or combinations of these conditions and reported high efficacy and safety. In particular, the colitis-specific complete clinical remission rate was 100% (95% CI: 63.06%–100%). The ECP-adiponectin axis appears to reduce the activation of intestinal mucosal resident T-cells and CD4+IFN-γ+ cells in patients with ICI-induced colitis, without evidence of loss of antitumor immunity. However, these are only a few cases, which, although promising, deserve further investigation.
Another promising future treatment, given the role of the gut microbiota in the pathogenesis of ICI colitis, is fecal microbiota transplantation (FMT). 97 The presence of dysbiosis has been proposed as one of the triggering mechanisms of colitis, and for these reasons, FMT has been proposed as a therapeutic approach for patients with refractory ICI-induced colitis. There are already case series in the literature demonstrating a clinical and endoscopic response to this treatment.97–99
Specifically, in the cases described by Halsey et al., 10 of 12 patients (83%) achieved symptomatic improvement after FMT, with 7 patients (58%) having a complete clinical response. 98 Wang et al. reported that both of their patients treated with FMT experienced complete resolution of clinical symptoms, with significant improvement in mucosal inflammation and ulceration after FMT. 97 In addition, Elkrief et al., in a small number of patients, described the efficacy of repeat transplantation after the initial loss of response. 99
In contrast, Davar et al. and Baruch et al. reported more modest efficacy rates, with 6 out of 15 patients and 3 out of 10 showing clinical benefit after FMT. Notably, these exploratory studies showed no alarming safety issues.100,101
Interestingly, analysis of the gut microbiome revealed that complete responders to FMT showed greater dysbiosis than nonresponders, with depletion of the bacterial genera Bifidobacterium and Collinsella. After FMT, a reduction in CD8+ T-cell density in the colic mucosa has been observed, with a concomitant increase in CD4+ FoxP3+ T-cells. 97 In addition, increased intestinal microbial diversity was noted with beneficial enrichment of Bifidobacterium and Akkermansia, Collinsella, and a decrease in potentially pathogenic species of the genus Escherichia.97,99,101
Although there are data on microbial profiles associated with an increased risk of developing colitis, there are currently no accepted criteria for selecting patients deserving of FMT. Such treatment is usually reserved for patients refractory to other therapies.
Regarding safety, cases of bacteremia from Escherichia coli producing extended-spectrum beta-lactamase after FMT have been described. 102 However, these cases have been linked to the same feces donor. Notably, these risks are very rare, while most of the AEs reported in the literature concern self-limiting gastrointestinal symptoms. 103 Nevertheless, it is necessary to emphasize the increasingly stringent and innovative donor screening processes and new techniques such as “washed” microbiota preparation, to limit the transmission of microorganisms that could lead to adverse infectious events. 104
Finally, these data suggest that modulation of the gut microbiome through FMT may be associated with a significant and rapid improvement of refractory ICI-associated colitis, with initial insights into potential mechanisms; however, larger and longer-term prospective studies are needed to further characterize the efficacy and safety of FMT in these patients and to identify specific microbial signatures that can guide patient selection and predict treatment response.
Conclusion
The management of gastrointestinal side effects associated with first-line immunotherapy combinations for advanced RCC represents a significant challenge for the clinician. Expertise in managing toxicity during therapy enables oncologists to prevent drug dosage reductions or interruptions that may negatively impact oncological outcomes. In cases of severe or rare toxicity, gastroenterological consultation is mandatory to ensure the efficiency of the patient care process.
