Abstract
Background:
Colorectal cancer (CRC) remains a multifaceted disease with variations in aetiology, clinical presentation and prognostic factors.
Objectives:
This study explores the features and outcomes of sporadic (S-CRC), inflammatory bowel disease-associated CRC (IBD-CRC), early-onset CRC (EO-CRC) and late-onset CRC (LO-CRC).
Design:
This is a systematic review and meta-analysis performed following the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) Statement, comparing S-CRC versus IBD-CRC and EO-CRC versus LO-CRC.
Data sources and methods:
The literature search was conducted on PubMed and Embase databases. The primary endpoint was the overall 5-year survival rate of CRC. Secondary aims included the features of CRC at diagnosis.
Results:
Fifty studies and 6,148,851 patients with CRC were included in the analysis. Comparing S-CRC and IBD-CRC, the overall survival was higher in S-CRC (61.88 (range 41.3–78.7) vs 55.54 (51.9–80.9) months). IBD-CRC showed a minor mean age of diagnosis (63.5 (45–78) vs 69.1 ((40–78) years), a minor risk of stage IV (odd ratio (OR) 1.091; 95%CI 1.031–1.155,
Conclusion:
IBD-CRC was associated with a younger mean age at diagnosis, higher risk of mucinous cancers, emergency presentation, and synchronous neoplasia compared to S-CRC. EO-CRC had a higher risk of being diagnosed at stage IV and of mucinous tumours versus LO-CRC. IBD-CRC seemed to have an overall shorter survival rate and a higher prevalence of mucinous cancers, suggesting different pathways of progression and more aggressive features.
Trial registration:
Prospero Registration ID1021182.
Plain language summary
Colorectal cancer (CRC) remains a multifaceted disease with variations in etiology, clinical presentation, and prognostic factors. This study explores the characteristics of sporadic CRC (S-CRC), inflammatory bowel disease-associated CRC (IBD-CRC), early-onset CRC (EO-CRC), and late-onset CRC (LO-CRC). The review was conducted through a systematic review and meta-analysis comparing sporadic CRC with IBD-CRC and EO-CRC with LO-CRC. The literature search was conducted using PubMed and Embase databases. The primary endpoint was 5-year overall survival rate for CRC. Upon completion of the review, 50 studies and 6,148,851 CRC patients were included in the analysis. When comparing S-CRC and IBD-CRC, overall survival was higher in S-CRC, with IBD-CRC showing a lower mean age at diagnosis (63.5 [45-78] vs 69.1 [40-78] years), a lower risk of stage IV, higher risk of mucinous tumor, emergency diagnosis, and synchronous neoplasia. When comparing EO-CRC and LO-CRC, overall survival was higher in EO-CRC, although it had a higher risk of being diagnosed at stage IV and of presenting mucinous tumors compared to LO-CRC. When comparing IBD-CRC, EO-CRC, and LO-CRC, the first presented the shortest OS (61.88 months), the highest rates of mucinous cancer (13%) and emergency diagnosis (24%), while metastatic disease at diagnosis was more frequent in EO-CRC (22.6%). In conclusion, different analyzed groups of CRC presented differential features, suggesting different pathways of progression, as well as more aggressive features, particularly in IBD.
Keywords
Introduction
Colorectal cancer (CRC) is among the leading causes of cancer-related mortality worldwide. 1 Despite significant advancements in early detection and treatment, CRC remains a multifaceted disease with variations in aetiology, clinical presentation, and prognostic factors. 2 This diversity is particularly evident among its subtypes, each characterised by unique features and behaviours. For instance, inflammatory bowel disease-associated CRC (IBD-CRC) differs significantly from sporadic CRC (S-CRC), 3 as well as early-onset CRC (EO-CRC) presents distinct characteristics compared to late-onset CRC (LO-CRC). 4
S-CRC represents the majority of CRC cases showing no predisposing conditions in the affected subjects. 5 Its pathogenesis is primarily driven by genetic and epigenetic changes contributing to the adenoma-carcinoma sequence. Key molecular alterations include APC, KRAS and TP53 gene mutations. Moreover, lifestyle factors such as diet, obesity and smoking also contribute to its development.6,7 On the contrary, IBD-CRC is believed to arise in the setting of chronic inflammation primarily due to long-standing IBD, which contributes to the development of a carcinogenic microenvironment. Unlike the adenoma-carcinoma sequence seen in S-CRC, IBD-CRC often follows an inflammation-dysplasia-carcinoma pathway. Key molecular alterations in this setting also include early TP53 mutations and a higher prevalence of CpG island methylator phenotype alterations.8,9 The latter alterations may also contribute to differences in histological features compared to S-CRC. According to available literature, IBD-CRC more frequently shows mucinous and signet-ring cell subtypes, which may contribute to its more aggressive behaviour and poorer prognosis compared to S-CRC. 10
Historically, CRC has been considered a disease affecting individuals of later ages. However, over the past decades, there has been a notable increase in the incidence of CRC among patients younger than 50, particularly in Western regions. 11 The mechanisms of cancer onset and progression in this subset of CRC (EO-CRC) are yet to be elucidated. While hereditary syndromes partially contribute to its pathogenesis, they account for only a small proportion of EO-CRC. As for IBD-CRC, EO-CRC is often associated with more aggressive histological subtypes, including mucinous and signet-ring cell carcinoma.12,13 Most of the studies also show a wide variety of differences between both age-of-onset CRCs.14,15
Despite these differences, survival outcomes between EO-CRC and LO-CRC remain debated. Some studies suggest that younger patients may have longer survival due to greater physiological resilience and increased likelihood of receiving aggressive treatment.16 –18
While numerous studies have investigated the characteristics of S-CRC versus IBD-CRC and EO-CRC versus LO-CRC, no comprehensive meta-analysis has systematically and comprehensively synthesized these findings. A detailed comparison is necessary to clarify discrepancies in survival outcomes, histopathological differences and tumour features across these CRC subtypes, which could eventually impact the current screening programmes.
The current study explores the multiple facets of EO-CRC, LO-CRC and IBD-CRC, aiming to provide a refined characterization of subtypes that could aid in optimising tailored strategies for prevention, early detection and management.
Methods
The current systematic review and meta-analysis were performed following the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) statement. 19
Search strategy and data sources
The literature search was conducted on PubMed and the Embase database. Two screeners independently performed the literature screening (G.F. and O.N.). Data were extracted independently from included studies by the reviewers; any discrepancy between the two reviewers would be resolved by a third reviewer. This meta-analysis was prospectively registered (PROSPERO ID 1021182). The detailed search strategy is shown in Supplemental Table 1.
Inclusion and exclusion criteria
Only studies that included patients with CRC and compared sporadic versus IBD-associated cancers or EO-CRC versus LO-CRC were included in the analysis. Studies with non-extractable data, with no full-text available, written in languages other than English, and with a number of patients included lower than 1000 were excluded. In case of more than one study conducted at the same centre, only the most recent one was included.
Study aims and endpoint measures
The primary aim of the study was to assess the survival in each group of CRC, using as endpoint the 5-year overall survival (OS).
Secondary endpoints included the mean age of cancer diagnosis, tumour location, presence of metastatic disease at the diagnosis, histological features, synchronous cancer/s and emergency diagnosis.
Direct comparisons were made between S-CRC versus IBD-CRC, and EO-CRC versus LO-CRC using a meta-analytic approach. Features of IBD-CRC, EO-CRC and LO-CRC were also compared.
Statistical analysis
All collected data regarding the endpoints were reported in an Excel table showing the number of patients involved in the studies and the extracted data associated with the endpoints. A meta-analysis was realized using MedCalc® (MedCalc Software Ltd, Acacialaan, Ostend, Belgium) statistical software. The meta-analysis was performed according to the Cochrane Collaboration the Quality of Reporting of Meta-analyses (QUORUM) guidelines. 20 The estimated effect measures are reported as odds ratios (OR) with 95% confidence intervals (CI). The ratio represented the probability of occurrence of an adverse event in the group of patients and the point estimate of OR was considered statistically significant if the 95% CI did not include the value ‘1’. The meta-analysis regarding differences in colon versus rectal localization was expressed in relative risk (RR) with 95% CI.
Risk of bias assessment and quality of the studies
The Joanna Briggs Institute’s (JBI) critical appraisal checklist for studies reporting prevalence data was utilized to evaluate the quality of the studies. 21 This tool assessed studies according to 10 questions. If the answer was yes, the question was assigned a score of 1. If the answer was no, unclear, or not applicable, it was assigned a score of 0.
Results
The search yielded 10,136 studies and 4524 duplicates were excluded. After removing abstracts, reviews, meta-analyses, and case reports, the titles of 5612 articles were screened; all their full-text articles were assessed for eligibility. The flowchart for study inclusion is reported in Figure 1. There was 100% agreement among reviewers in the extraction of data.

Flow chart of study selection according to PRISMA statement.
In total, 50 studies,12,22 –70 including 6,148,851 patients with CRC, were included in the analysis. The characteristics of the included studies are summarized in Table 1.
Characteristics of included studies.
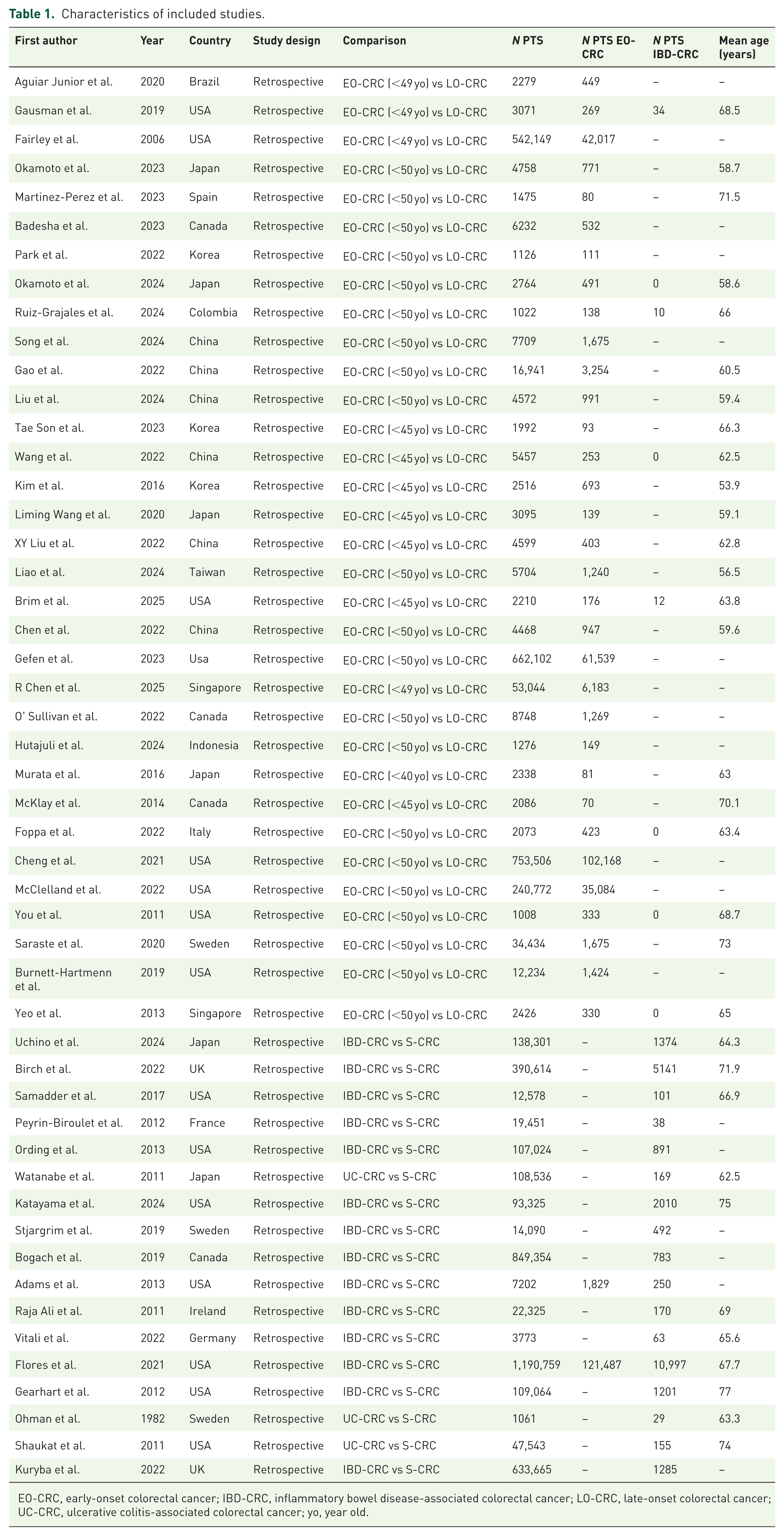
EO-CRC, early-onset colorectal cancer; IBD-CRC, inflammatory bowel disease-associated colorectal cancer; LO-CRC, late-onset colorectal cancer; UC-CRC, ulcerative colitis-associated colorectal cancer; yo, year old.
S-CRC versus IBD-CRC
Seventeen studies provided data on the comparison between S-CRC and IBD-CRC. 3,748,665 patients with CRC were included, of whom 25,149 had IBD (0.55%, 95%CI 0.543–0.558,
Out of them, 61.23% (95%CI 60.589–61.860,
The OS of patients with S-CRC was longer than that of patients with IBD-CRC (61.88 (range 41.3–78.7) vs 55.54 (51.9–80.9) months). The mean age at diagnosis of S-CRC patients was 69.1 (40–78) years, while that of IBD-CRC patients was 63.5 (45–78) years. In both S-CRC and IBD-CRC, colon cancer was more frequent than rectal cancer, RR 2.144 (95%CI 2.39–2.150,

Meta-analysis on colon versus rectum localization in S-CRC and IBD-CRC. Forest plots comparing colic and rectal localization of CRC in patients with sporadic (left) and colitis-associated (right). The effect size is expressed as relative risk.

Meta-analysis on risk of stage IV in S-CRC versus IBD-CRC. A forest plot comparing sporadic CRC versus colitis-associated CRC on risk of diagnosis with stage IV. The effect size is expressed as odds ratio.
Mucinous tumours were more frequent in IBD-CRC versus S-CRC, OR 3.150 (95%CI 2.797–3.548,
EO-CRC versus LO-CRC
Thirty-three studies provided data comparing EO-CRC and LO-CRC, with 2,400,186 patients included in the analysis; of them, 265,450 (10.87%, 95%CI 10.831–10.910,
The mean age of the EO-CRC patients was 41.6 (36–44.9) years versus 64.5 (60–73) years in LO-CRC.
OS was longer in EO-CRC than in LO-CRC (79.42 (54–96) vs 77.58 (32–92) months).
In both EO-CRC and LO-CRC patients, the colon was the most common site compared to the rectum: in EO-CRC, the RR was 2.112 (95%CI 2.097–2.127,
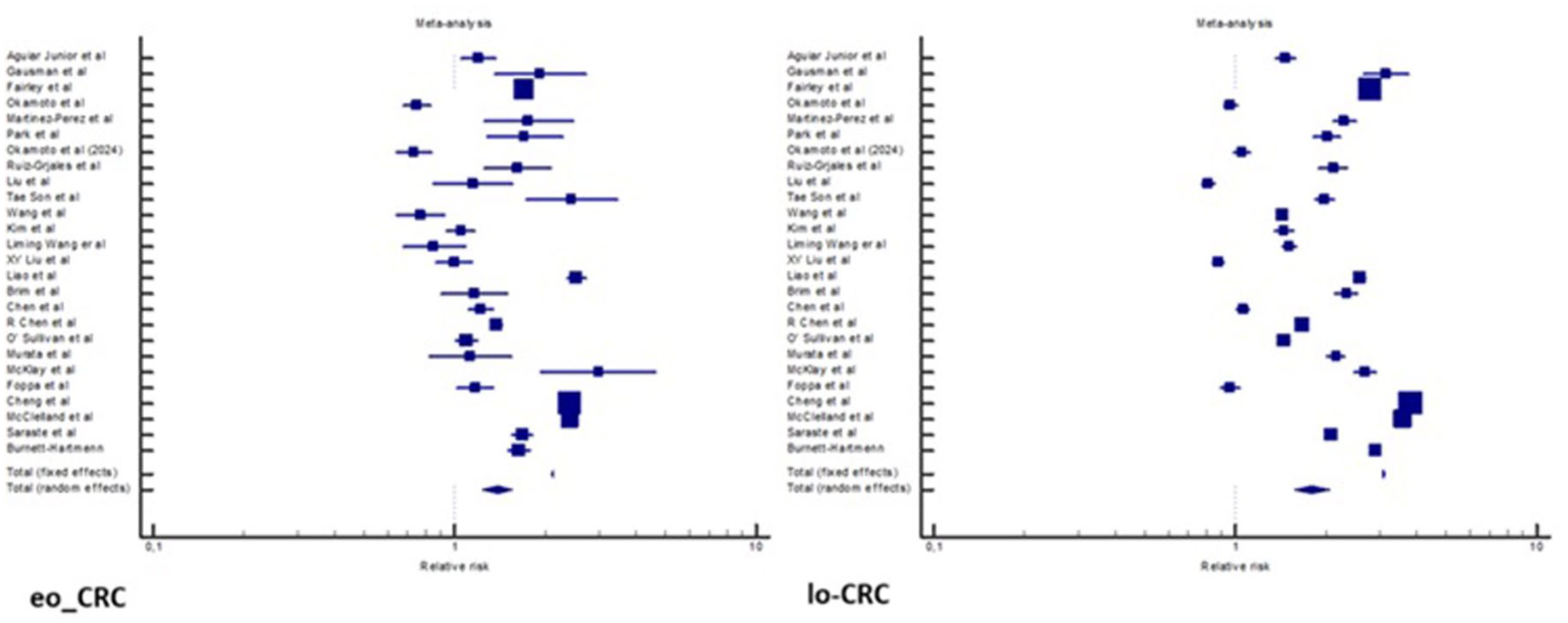
Meta-analysis on colon versus rectum localization in EO-CRC and LO-CRC. Forest plots comparing colic and rectal localization of CRC in patients with early onset (left) and late onset (right). The effect size is expressed as a relative risk.

Meta-analysis on risk of stage IV in EO-CRC versus LO-CRC. A forest plot comparing early-onset CRC versus late-onset CRC on risk of diagnosis with stage IV. The effect size is expressed as odds ratio.
The risk of mucinous cancer was higher in EO-CRC than in LO-CRC, OR 1.0142 (95%CI 1.015–1.070,
Overall comparisons: IBD-CRC versus EO-CRC versus LO-CRC
Table 2 shows the main findings, comparing the features of IBD-CRC, EO-CRC and LO-CRC. IBD-CRC had the shortest OS (61.88 months), the highest rate of mucinous cancer (13%), and the highest rate of emergency diagnosis (24%). EO-CRC was associated with the highest risk of metastatic disease at diagnosis (22.6%).
Features of EO-CRC, LO-CRC and IBD-CRC among the included studies.

Worse features are in red; intermediate in yellow; best in green.
IBD-CRC versus S-CRC, OR 3.150 (95%CI 2.797–3.548,
EO-CRC versus LO-CRC, OR 1.471 (95%CI 1.456–1.486,
IBD-CRC versus S-CRC, OR 1.598 (95%CI 1.509–1.693,
EO-CRC, early-onset colorectal cancer; IBD-CRC, inflammatory bowel disease-associated colorectal cancer; LO-CRC, late-onset colorectal cancer; OR, odds ratio; S-CRC, sporadic colorectal cancer.
Quality of studies
The overall strength of evidence is summarized in Supplemental Table 2. Low risk of bias was found for 90% of the studies (risk scores 8–10). Five studies had a medium risk with scores between 5 and 7.
Discussion
This systematic review and meta-analysis aimed to provide a comprehensive comparison of clinical, histopathological, and prognostic factors between S-CRC and IBD-CRC, as well as between EO-CRC and LO-CRC. The findings underscore the heterogeneity among these CRC subtypes, with practical implications. Regarding differences between S-CRC and IBD-CRC, the meta-analysis found that IBD-CRC is diagnosed at a younger mean age than S-CRC (63.5 years vs 69.1 years). This observation may be explained by two factors: first, the chronic inflammatory process in colitis may accelerate carcinogenesis; second, patients with long-standing colitis undergo regular surveillance, enabling earlier CRC detection. Notably, metastatic disease at diagnosis was more prevalent in the S-CRC group than in the IBD-CRC group (OR 1.091, 95%CI 1.031–1.155,
Despite this, OS was lower in IBD-CRC than in S-CRC, EO-CRC and LO-CRC which may be attributed, among other causes, to a more aggressive histopathological phenotype. IBD-CRC was more frequently associated with mucinous histology and had a higher prevalence of synchronous lesions, previously reported in 1.1%–8.1% of cases. 71 Additionally, IBD-CRC was more commonly located in the right colon, which could be explained by both specific epigenetic modifications,72,73 and sustained inflammation in the right colon, more commonly affected together with the terminal ileum in CD.
Emergency diagnoses were found to be higher in the IBD group (OR 1.598,
The current study also seems to support the hypothesis that EO-CRC is a distinct entity from LO-CRC.15,74 According to the meta-analysis, EO-CRC was associated with a higher risk of mucinous cancers and a higher likelihood of metastatic disease at diagnosis, emphasizing the need for improved screening strategies in younger populations, as the majority of cases are currently uncovered by these screening programmes. 75
Of note, EO-CRC showed better OS than LO-CRC. This finding could be attributed to a better frailty profile of EO-CRC patients, who can undergo more extensive surgery and chemotherapy. 76 Colonic location was more frequent than rectal location in both EO-CRC and LO-CRC, with an RR of 2.112 and 3.115, respectively, suggesting that flexible sigmoidoscopy is not an appropriate screening tool in young patients.
The current study highlights the need to at least consider to lower the screening age in European countries, as has already been implemented in the United States. 77 A “one-size-fits-all” approach may not be an optimal strategy for CRC screening; this applies not only to patients with IBD but also to individuals with risk factors for EO-CRC, such as obesity, red meat consumption, Caucasian ethnicity, smoking habits and, mainly, family history of CRC.78,79 As for IBD-CRC prevention, EO-CRC deserves a tailored approach for appropriate prevention and early intervention.
Regarding the histopathological findings, both IBD-CRC and EO-CRC had a high prevalence of mucinous cancers, associated with a more aggressive pattern. Mucinous tumours are less responsive to standard chemotherapy, particularly 5-fluorouracil-based regimens, but seem to benefit from targeted therapy or immunotherapy, especially in cases with microsatellite instability or high CpG island methylator phenotype. 80 This is important when considering the ideal multimodal treatment in IBD-CRC and EO-CRC and warrants further investigation in the coming years.
However, the current study could not provide a meta-analysis comparing EO-CRC in IBD-related versus non-IBD-related CRC, as no studies were identified that provided data for this analysis. A recent study by Arif et al., which compared sporadic, hereditary, and IBD-related EO-CRC, showed that patients with IBD have a lower age of onset (
Although EO-CRC shows a heterogeneous molecular basis, it is important to note that some of them show alterations related to chronic inflammation processes, suggesting a potential common pathway with IBD-CRC.82,83
Study limitations and strengths
The findings of the present meta-analysis should be interpreted with due consideration to the substantial heterogeneity observed in most comparisons, largely attributable to the retrospective design of the included studies and to variations in patient populations and data reporting. An additional limitation is that the available data did not allow distinct analyses for CD versus UC. Given their distinct inflammatory patterns and potentially different carcinogenic pathways, this inability to stratify by disease type may have impacted the results.
Nevertheless, this study has several strengths. It included a large sample of over 6 million CRC patients, which extensively represents the global population and analyses the features of different CRC subtypes using a meta-analytical approach where feasible, highlighting differences in presenting features, outcomes and tumour biology. Finally, it ensures direct clinical implications which can be translated into practice.
Conclusion
CRC remains a significant clinical challenge worldwide, despite notable advances in diagnosis and management. This comparative analysis of S-CRC, IBD-CRC, EO-CRC and LO-CRC provides a comprehensive overview of the current state of knowledge in the field. Key differences in age and stage at diagnosis, tumour location and histological features highlight the need for tailored screening strategies and targeted prevention programmes. Enhancing early detection efforts could facilitate timely diagnosis and optimise treatment
Supplemental Material
sj-jpg-1-tag-10.1177_17562848251379961 – Supplemental material for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis
Supplemental material, sj-jpg-1-tag-10.1177_17562848251379961 for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis by Giacomo Fuschillo, Olga Maria Nardone, Giulio Calabrese, Marc Martí-Gallostra, Francesco Selvaggi, Eloy Espín-Basany, Gianluca Pellino and Jose Perea in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-2-tag-10.1177_17562848251379961 – Supplemental material for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis
Supplemental material, sj-jpg-2-tag-10.1177_17562848251379961 for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis by Giacomo Fuschillo, Olga Maria Nardone, Giulio Calabrese, Marc Martí-Gallostra, Francesco Selvaggi, Eloy Espín-Basany, Gianluca Pellino and Jose Perea in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-3-tag-10.1177_17562848251379961 – Supplemental material for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis
Supplemental material, sj-jpg-3-tag-10.1177_17562848251379961 for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis by Giacomo Fuschillo, Olga Maria Nardone, Giulio Calabrese, Marc Martí-Gallostra, Francesco Selvaggi, Eloy Espín-Basany, Gianluca Pellino and Jose Perea in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-4-tag-10.1177_17562848251379961 – Supplemental material for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis
Supplemental material, sj-jpg-4-tag-10.1177_17562848251379961 for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis by Giacomo Fuschillo, Olga Maria Nardone, Giulio Calabrese, Marc Martí-Gallostra, Francesco Selvaggi, Eloy Espín-Basany, Gianluca Pellino and Jose Perea in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-5-tag-10.1177_17562848251379961 – Supplemental material for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis
Supplemental material, sj-jpg-5-tag-10.1177_17562848251379961 for Comparative analysis of sporadic, IBD-associated, early-onset and late-onset colorectal cancer: a systematic review and meta-analysis by Giacomo Fuschillo, Olga Maria Nardone, Giulio Calabrese, Marc Martí-Gallostra, Francesco Selvaggi, Eloy Espín-Basany, Gianluca Pellino and Jose Perea in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

