Abstract
Background:
Upadacitinib (UPA) is a new small-molecule drug for treating inflammatory bowel disease (IBD); however, its therapeutic use in China remains limited.
Objectives:
This study aimed to explore Chinese physicians’ treatment preferences for UPA in IBD and analyze experience-related decision-making differences.
Design:
A nationwide cross-sectional study conducted among Chinese physicians.
Methods:
The study was conducted among Chinese physicians using a 69-item questionnaire on physicians’ backgrounds and UPA preferences, covering (1) Physician backgrounds (12 items), (2) Therapeutic preferences regarding physicians’ attention to different characteristics of UPA and physicians (24 items), and (3) clinical application practices (33 items). Physicians were categorized into experienced and inexperienced groups, and preference differences were compared and analyzed using chi-square or Fisher’s exact tests.
Results:
Of 583 participating physicians, 182 were classified as experienced and 401 as inexperienced. Safety and efficacy were prioritized by 63.8% and 61.6% of physicians, respectively. UPA was preferred for patients with IBD aged 18–60 years (452/583, 77.5%) and with moderate-to-severe disease activity (486/583, 83.4%). For clinical application, experienced physicians preferred efficacy assessments at week 8 (63/182, 34.6%), while inexperienced physicians preferred week 12 (133/401, 33.2%) for patients with ulcerative colitis. Both groups preferred week 12 for efficacy assessments (251/583, 43.0%) and evaluation of perianal involvement (269/583, 46.1%) and extraintestinal manifestations (271/583, 46.5%) in Crohn’s disease. Safety concerns included thrombosis, opportunistic infections, and severe infections, with discontinuation recommended for severe infections, cardiovascular events, or thrombosis.
Conclusion:
This was the first nationwide cross-sectional study to investigate the therapeutic preferences of Chinese physicians for UPA in patients with IBD. These findings provide evidence-based support for UPA applications in China but require further real-world validation.
Plain language summary
This study explores the early-stage application of Upadacitinib (UPA) for IBD treatment in China, highlighting the need for guideline development. UPA, as a highly selective JAK1 inhibitor, has shown good effectiveness in reducing gut inflammation and symptom relief in patients with IBD. Treatment response is evaluated after the induction phase (8 to 12 weeks) and during maintenance (up to 1 year) through symptom relief, biomarkers, and endoscopy and imaging assessment. However, due to potential safety risks, it also requires careful monitoring for potential side effects like infections, rash, thrombosis, etc. Our results reveal that Chinese physicians prioritize UPA’s safety and efficacy, considering patient age, disease severity and type when selecting patients for UPA. Additionally, differences in physicians’ preferences for efficacy and safety assessments are observed based on their experience level. The findings, supported by real-world data, will assist in evidence-based guideline revisions and improve the clinical application of UPA, particularly in safety and efficacy monitoring.
Keywords
Introduction
Inflammatory bowel diseases (IBD), including Crohn’s disease (CD) and ulcerative colitis (UC), are characterized by chronic gastrointestinal inflammation. Studies have shown a continuous global increase in IBD prevalence, with a significant rise in incidence in Asia over the past 30 years, an earlier peak age of onset, and a rapidly growing disease burden. 1 As an emerging industrialized country, China is currently in the epidemiological stage of accelerated onset of IBD, 2 with annual increases in incidence rate; the affected population is estimated to exceed 1.5 million by 2025. 3
With a deeper understanding of the pathogenesis of IBD and rapid advancements in drug research and development, treatment options for IBD have evolved continuously. Traditional treatments such as 5-aminosalicylic acid and corticosteroids reduce inflammation, whereas immunosuppressants and biologics offer alternatives for patients unresponsive to conventional therapies. 4 Despite the high efficacy of biologics, some patients still experience primary nonresponse and secondary loss of response, indicating unmet medical needs. In recent years, small-molecule drugs targeting specific signaling pathways and molecular targets have gradually been developed and have attracted widespread attention. 5 Compared with large-molecule biologics, small-molecule targeted drugs have many advantages, including lower production costs, simpler administration, lack of immunogenicity, and shorter half-lives. Currently, various small-molecule drugs have been approved for IBD treatment or are in the stage of clinical trials, including Janus kinase (JAK) -STAT inhibitors tofacitinib, filgotinib, and upadacitinib (UPA), 6 S1P receptor modulators ozanimod and eltrimod,7,8 SMAD7 inhibitor GED-0301, 9 and PDE4 inhibitor apremilast, 10 etc. Numerous pro-inflammatory cytokines play important roles in the pathogenesis of IBD through the JAK-STAT signaling pathway; therefore, inhibition of this pathway has become a promising target for IBD therapy. 11
As a highly selective JAK1 inhibitor, UPA primarily functions in IBD treatment by blocking downstream cytokine signaling pathways. 12 Multiple clinical trials and real-world studies have demonstrated its therapeutic effects in patients with moderate-to-severe IBD.13–18 Despite substantial evidence supporting the use of JAK inhibitors in the treatment of IBD, their use may increase the incidence of various adverse events, including infection, hyperlipidemia, venous thromboembolism, cardiovascular events, and malignancies.19,20 Based on current clinical trials and real-world observational studies, the overall safety of UPA for the treatment of IBD has been preliminarily recognized. 21 However, the number of real-world studies related to the safety of UPA remains relatively limited, and some rare, serious, and clinically significant adverse effects may require long-term observation. Therefore, strengthening physicians’ awareness regarding safety assessments during UPA treatment is crucial.
At present, the clinical application of UPA in IBD treatment in China is still in its early stage, and physicians’ experience is relatively limited. Physicians vary in their cognitive and therapeutic preferences for UPA, including the indications, efficacy assessments, and safety evaluations. In addition, physicians’ decision-making and management of patients using UPA will significantly affect the benefits derived from UPA to patients. However, no surveys have been conducted to investigate the therapeutic preferences of physicians and the clinical applications of UPA. This study is the first to assess Chinese physicians’ understanding and preferences regarding UPA applications. It also explores the clinical application preferences of different physicians, offering crucial insights into UPA utilization in the treatment of IBD.
Methods
Calculation of the sample size
The sample size was estimated based on the primary question assessing physicians’ preferred time to evaluate UPA efficacy in CD, assuming 50% would select “12 weeks.” This cross-sectional study aimed to evaluate UPA preference rates among Chinese physicians, with bilateral tests and α = 0.05. The allowable error was set at 0.05. Using PASS 15 software, the calculated sample size was N = 246 participants. Accounting for a potential 20% loss rate (e.g., incomplete responses), at least 308 physicians needed to be included as study subjects to ensure adequate statistical power for detecting differences between experienced and inexperienced groups using chi-square or Fisher’s exact tests. To ensure subgroup comparisons and account for potential nonresponse, we targeted 500 participants originally.
Participants
The survey was distributed to approximately 1000 physicians specializing in gastroenterology across China, with a response rate of 58.3%. We received 583 valid responses from physicians across 29 provinces, municipalities, and autonomous regions. Eligible participants were practicing gastroenterologists or gastrointestinal surgeons in China, and the exclusion criteria were incomplete responses (>20% missing data). Most participating physicians were members of IBD centers, which are specialized units typically comprising multidisciplinary teams working collaboratively under standard operating procedures with established patient monitoring systems. All physicians provided informed consent. This study was approved by the Ethics Committee of Sir Run Shaw Hospital, Zhejiang University School of Medicine (ethical number: 2024-2327-01) and was registered in the Chinese Clinical Trial Registry (registration number: ChiCTR2400085646).
Based on the physicians’ working experience in IBD, the annual number of patients with IBD in their departments, clinical experience with UPA, and self-assessment of UPA understanding, the study divided the participating physicians into two groups: experienced and inexperienced. Physicians were categorized into the experienced group if they met all the following criteria: (1) at least 3 years of experience in IBD diagnosis and treatment, (2) an average of at least 50 patients with IBD admitted to the department annually in the past 3 years, (3) current experience with UPA application in their department, and, (4) a reasonable understanding of UPA. The remaining physicians were classified into the inexperienced group.
Questionnaire design
The questionnaire on “Chinese physicians’ therapeutic preferences and clinical applications of UPA” was designed by the IBD team of Sir Run Run Shaw Hospital, Zhejiang University School of Medicine. The questionnaire’s reliability was assessed by Cronbach’s α (α = 0.80 for patient characteristics, α = 0.78 for efficacy, α = 0.81 for safety), and exploratory factor analysis was performed. Content validity was confirmed by three gastroenterologists (scale-level content validity index = 0.91). The questionnaire, along with an informed consent form, was distributed online via the “Wenjuanxing” questionnaire platform. The reporting of this study conforms to the Checklist for Reporting Results of Internet E-Surveys (CHERRIES) statement. 22 The completed checklist is provided in the Supplemental File 2. It consists of 69 questions and takes approximately 10 minutes to complete. The detailed questions were divided into three sections: (1) Physician background: 12 questions covering demographics such as sex, age, region, and working experience; (2) Therapeutic preferences: 24 questions regarding physicians’ attention to different characteristics of UPA and patients; and (3) clinical application: 33 questions focusing on physicians’ preferences for the clinical application of UPA, including pretreatment infection screening and vaccine injection, efficacy assessment, and safety evaluation.
Data collection
The online questionnaire was available from June 19, 2024, to July 6, 2024. All participating physicians completed the survey voluntarily and anonymously. During the survey, all participants were required to acknowledge a mandatory pop-up notification before accessing the questionnaire, explicitly prohibiting communication or accepting temporary guidance to ensure objectivity and independence. Once the questionnaire is submitted, it is prohibited to make any secondary modifications. The participants completed questionnaires, which were collected and compiled by the IBD team of Sir Run Run Shaw Hospital, Zhejiang University School of Medicine. Two individuals independently cleaned, scrutinized, and analyzed the data.
Statistical analysis
Continuous variables were expressed as mean ± standard deviation or median, and categorical variables were expressed as frequency and percentage. Comparisons between the experienced and inexperienced groups regarding UPA clinical application and therapeutic preferences were conducted using t-tests or Wilcoxon signed-rank tests for continuous variables and chi-square tests or Fisher’s exact tests for categorical variables. All tests were two-sided, and a p-value of <0.05 was considered statistically significant. Data analysis was performed using SPSS software (version 23.0, IBM Corp., Armonk, NY, USA) and R software (version 4.4.1, R Foundation for Statistical Computing, Vienna, Austria), and graphs were plotted using GraphPad Prism software (version 10, GraphPad Software, Boston, MA, USA) and the ggplot2 package in R software.
Results
Study population
We collected valid online questionnaires from 583 physicians representing all provincial-level administrative regions in mainland China (except Qinghai Province and Tibet Autonomous Region). Among them, 402 were female (69.0%), and the majority were aged 30–50 years (436, 74.8%). A total of 455 (78.0%) physicians held a master’s degree or higher, and 349 (59.9%) held the title of associate chief physician or higher. Most physicians (540, 92.6%) worked in tertiary hospitals, and most physicians (481, 82.5%) were members of IBD centers. Nearly half of the physicians (279, 47.8%) had more than 5 years of work experience in IBD diagnosis and treatment. In addition, more than half of the physicians (382, 65.5%) worked in departments that treat more than 50 patients with IBD per year, and the majority (377, 64.7%) had experience with UPA utilization. The physicians’ characteristics are detailed in Table 1. A comparison between the experienced and inexperienced groups revealed significant differences in age, professional title, educational background, hospital grade, and medical team (p < 0.001). However, there was no significant difference in sex composition (p = 0.148) (Table 1).
Characteristics of participating physicians in China.

IBD, inflammatory bowel disease; UPA, upadacitinib.
Therapeutic preferences
Physicians’ therapeutic preferences based on different characteristics of UPA
We investigated the concerns of Chinese physicians regarding the characteristics of UPA, focusing on key features, such as efficacy, onset of action, and safety. The questionnaire shows that Chinese physicians are particularly concerned about the safety, efficacy, and approved indications of UPA in China, with 61.6%, 63.8%, and 54.9% of physicians, respectively, considering these aspects of high importance. Following this, treatment continuity of UPA was noted by 54.0% of physicians, onset of action by 47.7%, immunogenicity by 46.0%, route of administration by 44.3%, expert recommendations by 43.6%, and improvement in extraintestinal manifestations by 40.2%. In addition, 36.7% and 33.6% of physicians attached great importance to UPA dosing frequency and cost, respectively. The specific numbers and proportions of physicians concerned about the different characteristics of UPA are shown in Figure 1(a) and Supplemental Table 1.

Physicians’ attention to different characteristics of UPA (a) and physicians’ preferences for patient selection based on different comorbidities (b).
Physicians’ therapeutic preferences based on patient characteristics
To investigate whether Chinese physicians prefer certain patients suitable for UPA treatment, we set options in the questionnaire based on patient characteristics such as sex, age, disease type, lesion location, IBD duration, disease severity, and history of biologics. Physicians were asked to identify the patient groups they believed would be appropriate for UPA treatment. As shown in Table 2, 77.5% of physicians preferred to use UPA only for patients aged 18–60 years, and 80.6% reported no sex preference. With regard to disease duration, 87.6% believed that UPA was suitable for patients with an IBD history of less than 5 years. Regarding the type of IBD, 72.4% of the physicians had no preference between patients with CD and UC. In terms of disease severity, 83.4% physicians preferred to use UPA only for patients with moderate-to-severe disease activity. Only 7.7% preferred UPA for the biologic-naïve patients. For different locations, 58.7%, 69.8%, 83.7%, and 35.2% of physicians preferred UPA for patients with CD who were involved in L1, L2, L3, and L4, respectively; and 17.5%, 46.3%, and 94.3% of physicians preferred UPA for patients with UC who were involved in E1, E2, and E3, respectively.
Physicians’ therapeutic preferences based on patient characteristics.

CD, Crohn’s disease; IBD, inflammatory bowel disease; UC, ulcerative colitis; UPA, upadacitinib.
In addition, the physicians’ preferences for patient selection based on different comorbidities were investigated (Figure 1(b)). A total of 36.4%, 40.5%, 45.1%, and 44.1% of physicians clearly indicated a preference for UPA for patients with fibrosis, fistula, perianal involvement, and a history of gastrointestinal surgery, respectively, which were lower than those expressing non-recommendation or neutrality. Notably, a greater number of physicians (60.5%) tended to recommend UPA for IBD patients with extraintestinal manifestations.
Comparative analyses were conducted to further explore the differences in therapeutic preferences based on patient characteristics between experienced and inexperienced physicians. The numbers and proportions of physician preferences for different patient selections are presented in Table 2. The results of the one-way ANOVA indicated no significant differences in age preferences between the two groups (p = 0.553), but significant differences were found in preferences for patient characteristics, such as age (p < 0.001), disease type (p = 0.004), disease location (p < 0.001), disease severity (p < 0.001), and history of biologics (p < 0.001). Regarding preferences related to the location of IBD, significant differences were observed between the groups regarding the inclination toward L2 (p = 0.012) and L3 (p < 0.001) in patients with CD, as well as E1 (p = 0.038), E2 (p = 0.014), and E3 (p = 0.009) in patients with UC. Experienced physicians showed a higher preference for UPA in L2 and L3 in patients with CD and E2 and E3 in patients with UC.
The subgroup analysis based on physicians’ preferences for UPA in E2 or E3 type patients (categorized as preferring E2, preferring E3, or having no preference for E2/E3) reveals significant differences among physicians of varying experience levels (χ2 = 16.181, p < 0.001). The proportion of physicians with no preference for E2/E3 (45.3%) was significantly higher in the experienced group than in the inexperienced group (27.3%). Conversely, the proportion of physicians who preferred E3 (52.8%) was significantly lower in the experienced group than in the inexperienced group (68.3%) in patients with UC. Another subgroup analysis of physicians who preferred using UPA for patients with a history of at least one biologic was conducted (categorized as only preferring those who had been treated with one biologic previously, two or more, or considering all as suitable), and the results indicated significant differences in physicians’ perspectives on the applicability of UPA as a second-line, third-line and beyond, or both second- and third-line therapy. The results showed that 64.7% of experienced physicians believed that UPA would be suitable as a second- and third-line treatment, compared to 43.2% of the inexperienced group. Conversely, a higher proportion of inexperienced physicians (34.1%) prefer UPA only as a third-line or beyond treatment, significantly higher than 20.0% in the experienced group (χ2 = 18.446, p < 0.001). For patients with various comorbidities, inexperienced group shows significantly higher tendency of UPA for patients with intestinal fibrosis (χ2 = 26.481, p < 0.001), fistula (χ2 = 20.111, p < 0.001), perianal involvement (χ2 = 23.989, p < 0.001), and gastrointestinal surgery history (χ2 = 18.966, p < 0.001), while the experienced group tends to adopt a neutral stance. There was no significant difference between the two groups in the tendency to use UPA for patients with extraintestinal manifestations (χ2 = 1.541, p = 0.463).
Clinical application
Pretreatment infection screening and vaccine injection
Both first- and second-generation JAK inhibitors have received black-box warnings from the Food and Drug Administration (FDA) owing to safety concerns. As a highly selective JAK1 inhibitor, UPA can cause similar adverse events. While the 2021 European Crohn’s and Colitis Organisation (ECCO) guidelines provide recommendations on infection screening and vaccination prior to immunosuppressive therapy in IBD patients, 23 the real-world implementation remains unclear, particularly for UPA treatment. Therefore, this study investigated the physicians’ preferences for pretreatment infection screening and vaccination recommendations (Supplemental Figure 1). Most physicians supported the completion of screening for tuberculosis (97.8%), viral hepatitis (97.1%), Epstein-Barr virus (93.5%), cytomegalovirus (92.6%), and herpes zoster (87.14%) before UPA initiation. Some physicians also mention screening for HIV, syphilis, and Clostridium difficile infections. Regarding the prevention of related infections, most physicians recommended completing the hepatitis B vaccine (88.3%) and shingles vaccine (for those over 50 years old) (76.1%) before UPA treatment, with some suggesting the flu vaccine (44.6%). While offering the above suggestions, a few physicians reflected on the clinical challenges of implementing vaccination. These results provide preliminary insights into the preventive measures that Chinese physicians prioritize for safe use of UPA in clinical practice.
Efficacy assessment
The efficacy of UPA has attracted significant attention, and a subsequent survey focused on physicians’ preferences for the initial time point of UPA efficacy assessment using various indicators, including clinical symptoms, laboratory indicators (Supplemental Figure 2), endoscopic and imaging evaluations (Supplemental Figure 3), assessment of perianal involvement, and extraintestinal manifestations. The numbers and proportions of each time point that the physicians preferred to evaluate were recorded and analyzed (Table 3 and Figure 2).
Physicians’ perspectives on the timepoint of initial assessments of UPA efficacy.
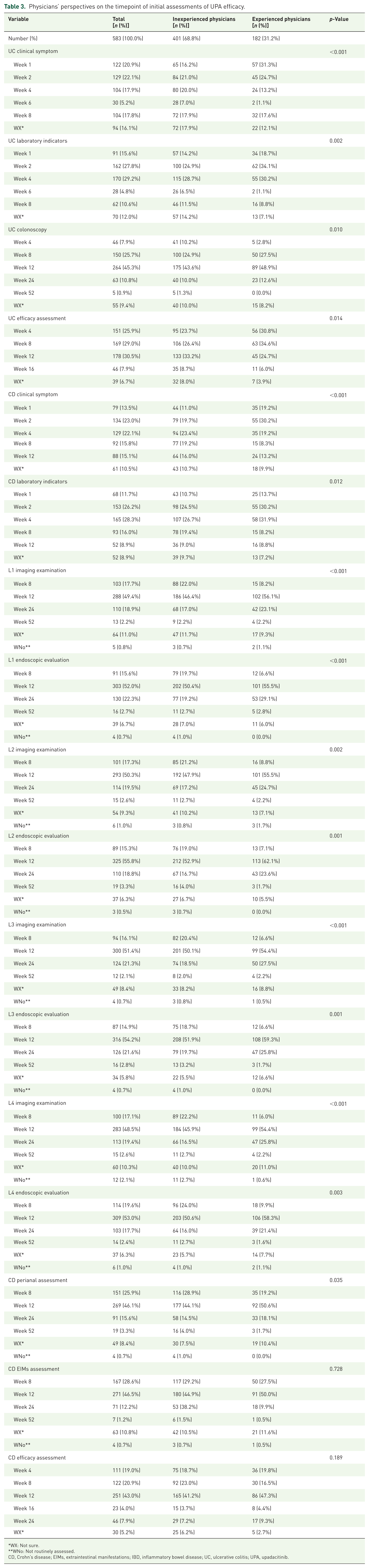
WX: Not sure.
WNo: Not routinely assessed.
CD, Crohn’s disease; EIMs, extraintestinal manifestations; IBD, inflammatory bowel disease; UC, ulcerative colitis; UPA, upadacitinib.

Physicians’ perspectives on the timepoint of initial assessments of UPA efficacy, including clinical symptoms of UC (a), clinical symptoms of CD (b), laboratory indicators of UC (c), laboratory indicators of CD (d), efficacy of UC (e), efficacy of CD (f), perianal involvement of CD (g), extraintestinal manifestations of CD (h), endoscopic assessments in different locations of CD (i), laboratory indicators assessments in different locations of CD (j).
Specifically, for patients with UC, a relatively high proportion of physicians preferred to conduct the first clinical symptom assessment at 1 and 2 weeks after treatment (20.9% and 22.1%, respectively). A higher preference for laboratory indicator assessments was noted at 2 and 4 weeks (27.8% and 29.2%, respectively), whereas the highest proportion (45.3%) favored an initial endoscopic assessment at 12 weeks. Regarding the efficacy assessment of UPA in UC therapy, 30.5% of physicians believed that the effectiveness of the treatment can be judged 12 weeks after medication, which is higher than the 29.0% who preferred 8 weeks. For patients with CD, the preference for the timing of the initial clinical symptom assessment was 23.0% at 2 weeks and 22.1% at 4 weeks. Similarly, a higher proportion of physicians favored laboratory indicator assessments at 2 and 4 weeks (26.2% and 28.3%, respectively). Notably, the physicians’ preference for the time point of the first endoscopic or imaging evaluation was not influenced by the disease location. For all CD locations, the physicians preferred the first imaging and endoscopic evaluation at 12 weeks, which was also the most common choice for assessing perianal involvement (46.1%) and extraintestinal manifestations (46.5%). When evaluating the effectiveness of UPA treatment for patients with CD, the proportion of physicians who chose 12 weeks was highest, at 43.0%.
A one-way ANOVA was also conducted to examine the differences in efficacy assessment timing preferences between the experienced and inexperienced groups. As shown in Table 3, significant differences were observed between the two groups in the timing of the first clinical symptom assessment (p < 0.001), laboratory evaluation (p = 0.002), and colonoscopy assessment (p = 0.010) in patients with UC. There were also significant differences in the preference for the first clinical symptom assessment (p < 0.001), laboratory evaluation (p = 0.012), perianal assessment (p = 0.035), and endoscopic and imaging assessments of various locations for patients with CD. However, no significant difference was observed in the timepoint for CD extraintestinal manifestation assessment (p = 0.728) or the final determination of effectiveness for CD patients (p = 0.189), and both experienced and inexperienced physicians generally agreed that 12 weeks was the appropriate time for evaluation.
We further explored the timing preferences for the first clinical symptom assessment among experienced and inexperienced physicians in patients with CD and UC. The intergroup analysis reveals significant differences in the preferences for assessing UC patients at week 1 and 4 between the two groups (χ2 = 28.778, p < 0.001). The proportion of experienced physicians who preferred to conduct the first clinical symptom assessment at week 1 after medication was 31.3%, which was significantly higher than that of the inexperienced group (16.2%). Conversely, the proportion of those who preferred week 4 was 13.2%, which was significantly lower than that of the inexperienced group (20.0%). For patients with CD, significant differences were observed between the two groups regarding first clinical symptom assessments at weeks 1, 2, and 8 (χ2 = 23.580, p < 0.001). Compared with week 8, experienced physicians preferred weeks 1 and 2 post-treatment, with proportions of 19.2% and 30.2%, respectively, both significantly higher than those of the inexperienced group of 11.0% and 19.7%, respectively. These results indicate that experienced physicians tended to conduct clinical symptom assessments earlier.
Regarding the final timepoint to determine UPA efficacy, the intergroup analysis indicates a significant difference in the proportion of physicians in the experienced and inexperienced groups who choose to assess the efficacy of UPA in patients with UC at weeks 8 and 12 (χ2 = 12.575, p = 0.014). Previous results indicate that, compared with the earlier 4 weeks after medication, physicians are more inclined to make decisions at 8 and 12 weeks after medication. Furthermore, experienced physicians were more inclined toward the 8-week point (34.6%), whereas inexperienced physicians favored the 12-week point (26.4%). For CD patients, both experienced and inexperienced physicians prefer week 12 for efficacy evaluation, accounting for 47.3% and 41.2%, respectively, showing no significant preference difference between the two groups (χ2 = 7.456, p = 0.189).
Safety assessment
Given the possible adverse events associated with JAK inhibitors and the relatively short period of UPA use for IBD, Chinese physicians should pay special attention to the safety of UPA. In the questionnaire, we listed adverse events, including hypersensitivity reactions, opportunistic infections, gastrointestinal perforation, and cardiovascular events, and asked physicians to select prioritized events during UPA treatment, which should lead to discontinuation if they occur, and the adverse events encountered in clinical practice (Table 4).
Physicians’ perspectives on the safety profile of UPA.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; N, number; P, percentage.
We also investigated the laboratory and imaging indicators used by physicians to monitor the safety of UPA treatment. Over half of the physicians recommended monitoring blood routine (95.0%), liver function (94.0%), renal function (91.3%), myocardial enzymes (87.3%), coagulation function (87.3%), blood lipids (73.9%), stool routine (54.4%), urine routine (50.8%), and other laboratory indicators during medication. The tumor marker and D-dimer levels were also within the recommended limits. Regarding imaging examinations, most physicians recommended regular examinations of lower extremity deep vein ultrasound (90.6%), electrocardiogram or echocardiogram (84.1%), chest CT scan (76.5%), small intestinal CT/MRI or abdominal CT, or intestinal ultrasound (72.4%) during medication to evaluate adverse reactions.
Discussion
To the best of our knowledge, this is the first cross-sectional study among Chinese physicians to investigate the therapeutic preferences and clinical applications of UPA in patients with IBD. This study investigated physicians’ concerns regarding the characteristics of UPA and patients. In terms of clinical application, this study investigated physicians’ preferences for efficacy and safety assessments, including pretreatment infection screening and prevention, timing of efficacy evaluations, and monitoring and management strategies for potential adverse events during treatment. In addition, we summarized the differences in preference between experienced and inexperienced physicians, aiming to provide more evidence-based support for the application of UPA in IBD management.
Our results revealed physicians’ concerns about the characteristics of UPA, including its safety, efficacy, and approved indications. The non-immunogenicity and administration routine of UPA are also aspects of concern for many physicians. Owing to its smaller molecular size and usually having only one antigenic epitope, UPA does not induce immunogenicity, thereby reducing the occurrence of immune-related side effects and drug interactions. 24 In addition, oral administration also offers more convenience for patients and potentially improves patients’ medication compliance, which is associated with good disease control outcomes and lower relapse risk.25,26
In terms of patient selection for UPA, Chinese physicians prefer using UPA for patients with severe disease activity, extensive lesions, and prior biologic use. Experienced physicians prioritize disease severity over factors such as sex and disease type when determining the suitability of UPA. Moreover, our results indicated that Chinese physicians prefer UPA as a second- or third-line treatment for IBD rather than for patients newly diagnosed with IBD. Nevertheless, experienced physicians view UPA as suitable not only for third-line use, but also for those who have used only one biologic, showing greater flexibility in its application. Approximately 60% of physicians favor UPA for patients with CD with extraintestinal manifestations. Previous studies have shown that specific doses of UPA improve extraintestinal manifestations in patients with IBD,14,17 but most studies have focused on overall disease control rather than specific improvements. Half of the physicians remained neutral regarding other comorbidities. This may be explained by the limited evidence on UPA use in patients with specific comorbidities, highlighting the need for more research data. This individualized approach is reflected in physician decision-making patterns, where a neutral stance might suggest that physicians prioritize evaluating the comprehensive patient condition over relying solely on the presence of specific comorbidities.
Compared with experienced physicians, inexperienced physicians more frequently prescribed UPA for fibrosis, fistulas, perianal lesions, or post-gastrectomy patients. For CD fibrosis, anti-inflammatory pharmacotherapy alone appears insufficient for long-term benefit, with endoscopic intervention and surgery remaining to be the primary treatment options.27,28 Furthermore, the ECCO guidelines consider stenosis or enterocutaneous fistula as clear surgical indications. 29 This may explain why experienced physicians adopted a more neutral stance regarding UPA use, as they likely weighed surgical options more comprehensively. Regarding perianal disease, while current guidelines note insufficient evidence to recommend UPA for complex fistulae, 29 some clinical trial data suggest potential efficacy. 30 On this point, the clinical decisions of inexperienced physicians seem to demonstrate better concordance with research evidence. For postoperative patients, ECCO recommends prophylactic medication for high-risk patients, 29 though UPA’s specific role remains unestablished. The lack of evidence, combined with the likely need for larger sample sizes to detect intergroup differences in decision-making patterns, may have contributed to the observed neutral therapeutic preferences among experienced physicians.
Because of the potentially increased risk of JAK inhibitor infections, physicians generally emphasize infection screening and prevention before UPA initiation, as recommended by the ECCO guidelines. Recent studies have shown that UPA increases the rate of herpes zoster infection, 31 whereas the ECCO guidelines recommend a recombinant vaccine (RZV) before JAK inhibitor treatment, and real-world data confirm the safety and effectiveness of RZV in patients with immune-mediated inflammatory diseases. 32 Most physicians in our study held the same view but also stated challenges, such as the timing of vaccination, vaccine accessibility, and patient compliance. Therefore, the actual vaccination rates and incidence of herpes zoster infection require further validation in clinical practice.
For the application of UPA in patients with IBD, the timing of the efficacy evaluation of UPA in patients with IBD is crucial, enabling timely treatment adjustments and making it a key clinical decision point for further exploration. In clinical trials, the key assessment time points are 12 weeks for induction and 52 weeks for maintenance, with CDAI/SES-CD for CD and Mayo clinical/endoscopic scores for UC as reference indicators.14,15 According to our results, physicians prefer to evaluate UPA efficacy for patients with CD primarily at 12 weeks, whereas for patients with UC, decisions are often made at 8 or 12 weeks. Therefore, we hypothesized that physicians’ preferences for these time points may be influenced by the timing of the assessment established in clinical trials. In addition, physicians tend to assess clinical symptoms earlier than they do during endoscopic or imaging evaluations. In real-world studies, early follow-up is often arranged at 2, 4, and 8 weeks after initiation, 17 with some reporting symptom relief from UPA as early as the first day of treatment. 33 Experienced physicians also tended to conduct earlier clinical symptom assessments and laboratory follow-ups than inexperienced physicians. Therefore, clinical symptom assessments should be conducted sooner to detect patient responses promptly, if permitted.
The observed differences in therapeutic preferences between experienced and inexperienced physicians appear closely associated with the availability of definitive guideline recommendations. The newest ECCO guideline consensus recommended UPA as induction and maintenance therapy in moderate-to-severe CD, 34 a preference also shown in our data, reflected by 83.4% of physicians (87.9% experienced vs 81.3% inexperienced). Compared to other decision-making factors, this relative consistency highlights the significant influence of clinical guidelines on physician preferences. However, current guidelines lack clear recommendations for managing complications or specifying detailed treatment assessment methods and timepoints. Our findings reveal differing decision-making between experienced and inexperienced physicians in these situations, highlighting a critical area for future guideline refinement.
Some adverse events (AEs) have been reported in previous clinical studies, with limited real-world experience. A meta-analysis combining real-world studies and clinical trials reported an overall incidence rate of AEs of 69% with UPA treatment for IBD, with rates of 65% for UC and 75% for CD. 21 Another meta-analysis of JAK inhibitors for treating IBD and other autoimmune diseases reported incidence rates of serious infections, herpes zoster infections, malignancies, and major cardiovascular events of 2.81, 2.67, 0.89, and 0.48 per 100 person-years, respectively. 35 Clinical data regarding another JAK inhibitor, tofacitinib, for UC treatment indicated that the commonly reported AEs included infections and vascular disorders, 20 which were broadly comparable to the prevalent AEs identified in our study. Research evidence indicates that JAK inhibitors increase the risk of infection as well as cardiovascular events and malignancies in IBD, especially in those over 50 years old with risk factors or a disease history, warranting cautious screening and considering the balance of medication risks and benefits.36–38 Rashes, anemia, headache, and upper respiratory tract infections are also common AEs. 14 These events usually do not require discontinuation but require appropriate interventions for safety. Therefore, AE monitoring and management should be continuously improved to provide reliable support in clinical practice.
This study had several strengths. It involved a substantial sample of physicians from nearly all provincial-level administrative regions in mainland China to ensure data representativeness. In addition, the content of the questionnaire was comprehensive and covered the physicians’ understanding of UPA, concerns about UPA attributes, patient selection preferences, efficacy evaluation, and safety monitoring. Furthermore, the questionnaire provided diverse options, enabling the participants to accurately express their clinical experiences and preferences. In addition, the questionnaire was refined through pre-filling and feedback before distribution to enhance its quality.
This study has some limitations. Although this study covers a wide area of China, its scope is confined to Chinese physicians, limiting its applicability to other countries due to differences in drug approval status, insurance coverage, clinical guidelines, and patient backgrounds elsewhere. While these Chana-specific data may have limited generalizability to Western populations, as a major developing country in Asia, the findings could offer valuable insights for other developing countries in Asia. However, the observed variation in physician decision-making based on experience levels suggests a need for a standard operating procedure in UPA-inexperienced regions. In addition, the online format of distributing and collecting questionnaires (via Wenjuanxing platform) inevitably introduces selection bias, as respondents were primarily from referring hospitals and previously collaborating institutions, potentially limiting the representation of participants. Information bias may also exist as physicians’ responses were based on their subjective cognition rather than on objective clinical data. Besides, the grouping criteria of physicians into “experienced” and “inexperienced” groups, while based primarily on our own experience-based assessment and sample size requirements for subgroup analysis, were not externally validated. This limitation should be considered when interpreting the between-group comparisons in our analysis. Finally, as a cross-sectional rather than prospective cohort study, the study captures physicians’ perspectives at a single time point but cannot determine how the preference variations affect patient outcomes or reveal potential gaps between physicians’ stated preferences and actual clinical practice. Future studies with wider geographical coverage, more participant recruitment, and stricter questionnaire designs are expected to enhance the comprehensiveness and scientific validity of this study.
Conclusion
This was the first nationwide cross-sectional study to investigate the therapeutic preferences of Chinese physicians for UPA in patients with IBD. First, it demonstrates significant variation in prescribing patterns based on physician experience levels and regional practice settings. Second, it identifies key decision-making factors, including safety concerns and efficacy expectations. While these results provide valuable insights into current UPA utilization in China, they also highlight the need for standardized treatment protocols and further real-world effectiveness studies.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251375851 – Supplemental material for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians
Supplemental material, sj-docx-1-tag-10.1177_17562848251375851 for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians by Lingya Yao, Yining Xu, Wenhao Xia, Kaituo Huang and Qian Cao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-3-tag-10.1177_17562848251375851 – Supplemental material for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians
Supplemental material, sj-docx-3-tag-10.1177_17562848251375851 for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians by Lingya Yao, Yining Xu, Wenhao Xia, Kaituo Huang and Qian Cao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-4-tag-10.1177_17562848251375851 – Supplemental material for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians
Supplemental material, sj-docx-4-tag-10.1177_17562848251375851 for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians by Lingya Yao, Yining Xu, Wenhao Xia, Kaituo Huang and Qian Cao in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-pdf-2-tag-10.1177_17562848251375851 – Supplemental material for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians
Supplemental material, sj-pdf-2-tag-10.1177_17562848251375851 for Therapeutic preferences of upadacitinib for patients with inflammatory bowel diseases: a nationwide study among Chinese physicians by Lingya Yao, Yining Xu, Wenhao Xia, Kaituo Huang and Qian Cao in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
