Abstract
In recent years, common postoperative complications after esophagectomy have received increasing attention. However, the attention paid to rare complications, which often lead to serious consequences if they are not diagnosed in a timely manner, has not been sufficient. In this article, we present both the clinical and imaging features of rare complications following esophagectomy and strategies for their prevention and management. These rare complications are classified into four groups: esophageal substitute-related complications, thoracic duct-related complications, hernia-related complications, and transmural Hem-o-lok clip migration. Esophageal substitute-related complications include redundant conduits and conduit necrosis. We further classify redundant conduits as length-redundant conduits, width-redundant conduits, or a combination of both redundancies. Thoracic duct-related complications include refractory chylothorax and chylomediastinum. Refractory chylothorax is chylothorax refractory to well-established medical, interventional, and even surgical strategies, whereas chylomediastinum is a rare condition characterized by the accumulation of chyle in the mediastinum. Hernia-related complications are subclassified as hiatal hernias, retrocardiac lung hernias, intercostal lung hernias, trocar-site hernias, hernias internal to the retrosternal space or pericardium, and mesenteric defects. Transmural Hem-o-lok clip migration includes penetration of the trachea, conduit, or both. Thoracic surgeons should familiarize themselves with the rare complications of esophagectomy, most of which are serious conditions that require early and accurate diagnosis for proper management. The choice of intervention for rare complications depends on factors such as the patient’s general condition, the specific type of complication, the complication severity, the reconstruction route, the available medical resources, and the surgeon’s preference. To minimize the risk of these complications, esophagectomy and prevention methods need to be standardized.
Introduction
In the treatment of esophageal cancer, esophagectomy can cause considerable physical trauma, resulting in a variety of postoperative complications. These complications, which include postoperative pneumonia, anastomotic leakage, and injury to the recurrent nerve, have been well documented in prior studies. According to the recently published JCOG1109 NExT study, the incidence of these complications in patients undergoing chemoradiotherapy was 12.9%, 13.5%, and 24.2%, respectively. 1 Although the most common complications of esophagectomy are well known to thoracic surgeons, rare complications, which may only be observed postoperatively or during follow-up examinations, have received less attention. We define rare complications as events in our prospective database with an incidence of less than 2% over a 10-year period. Despite their low incidence, however, rare complications have the potential to be devastating2–5 and thus hold considerable clinical significance.
This study aims to review the imaging features and risk factors of and prevention and management strategies for rare complications of esophagectomy. We categorized the complications of interest into the following four categories (Supplemental Figure 1): (1) esophageal substitute-related complications, (2) thoracic duct-related complications, (3) hernia-related complications, or (4) transmural migration of foreign bodies.
Esophageal substitute-related complications
Redundant conduits
After esophagectomy, redundancies in length or width may cause different degrees of conduit dysfunction, manifesting as dysphagia, postprandial fullness, nausea, and reflux. Vagotomy of the gastric tubes or pyloric dysfunction can exacerbate conduit dysfunction. As reported in the literature, the incidence of such complications is less than 2%, 6 whereas that in our center is even lower, at 1.3% (Table 1). We classified redundant conduits as length-redundant conduits, width-redundant conduits, or conduits with a combination of both redundancies (Figure 1). Anatomically, these conduits are characterized by dilation. A length-redundant conduit is likely associated with an inappropriately low esophageal-gastric anastomosis, an inappropriately long gastric tube, or a jejunal loop (Figure 2). In contrast, a width-redundant conduit is likely associated with an inappropriately wide gastric tube.
A brief summary of the rare complications.

Chylothorax after transthoracic esophagectomy.
Acute postesophagectomy herniation.
Only reported in case reports.

Redundant gastric conduit. (a and b) Length-redundant gastric conduit on upper gastrointestinal radiography and chest CT. (c and d) Width-redundant gastric conduit on upper gastrointestinal radiography and chest CT.
Redundant jejunal loop. (a) Redundant jejunal loop on upper gastrointestinal radiography and (b) redundant jejunal loop on chest CT.
Treatment of redundant conduits is necessary only if symptoms are present. In our institution, the most commonly used methods include conservative strategies (prokinetics, nasogastric drainage, or observation) and endoscopic or interventional methods (pyloric dilatation, botulinum toxin injection into the pylorus; Figure 3). 7 No patients have undergone revision surgery for redundant conduits at our institution, as their symptoms demonstrated varying degrees of improvement following the abovementioned treatments. Lanuti et al. 7 demonstrated that delayed gastric emptying can be adequately treated with pyloric dilatation, yielding a 95% success rate. Although conservative treatment has a high success rate, some patients will still experience suboptimal treatment outcomes, given the abovementioned difference in the degrees of symptom improvement following treatment. In such cases of refractory conduit dysfunction, selective conduit revision can be performed to prevent paroxysmal hernia development, avoid the development of a redundant conduit, tubularize a dilated conduit, and ensure adequate gastric drainage. 8 Studies have revealed substantial symptom relief following gastric conduit revision with controllable complications.6,8

Interventional pyloric dilatation in the redundant conduit. (a) Conduit dysfunction caused by a redundant conduit. (b) Interventional pyloric dilatation.
Conduit necrosis
Although conduit necrosis is uncommon, with an incidence of only 2%–3%, 9 it remains a serious complication. According to the data in our prospective database, the incidence of conduit necrosis in our institution is 0.4% (Table 1). The mortality rate for this complication has been reported to be as high as 90%, 10 while the data from our center indicate a mortality rate of 77% (Table 1). In some cases, conduit necrosis may occur years after esophagectomy. 11 Some scholars believe that anastomotic fistulas represent a special type of conduit necrosis, as the two have similar risk factors.
According to Veeramootoo, gastric conduit failure can be classified into three categories 12 : type I, simple anastomotic leaks; type II, conduit tip necroses; and type III, complete conduit ischemia. Unlike cervical anastomotic leakage, conduit necrosis is a serious clinical event (Figure 4), partly due to the rich vascularization of the conduit, which may lead to difficult-to-control massive hemorrhage. The reconstruction route also significantly affects the clinical outcomes of patients with conduit necrosis. 3 During posterior mediastinal reconstruction, the gastric tube is adjacent to the trachea, bronchi, aorta, azygos vein, pericardium, and other structures. Reports have also documented perforations into the lung, 11 pericardium, atria, and ventricles.3,13,14 Such perforations can lead to tracheo-broncho-esophageal fistula, fatal massive hemorrhage, pericardial perforation, hydropneumothorax, or esophagopulmonary fistula. When the retrosternal route is used, the conduit is close to the pericardium and sternum. Perforations in this location most commonly affect the pericardium, but cases of sternal penetration, atrial fistula, and massive hemorrhage from the left subclavian vein have also been reported.2,3,14,15 Under these circumstances, treatment becomes more complicated, resulting in an increased risk of mortality. 15 Overall, perforations during posterior mediastinal reconstruction are associated with greater risks and require more aggressive management.

A 63-year-old male admitted with a chief complaint of chest pain for 4 days, which became aggravated over the prior 2 h, was later diagnosed with conduit perforation and died of infection and hemorrhagic shock. (a) Chest CT image showing iodine extravasation due to perforation of the gastric conduit. (b) Upper gastrointestinal radiography reveals gastric conduit perforation; a drainage tube was placed into the abscess cavity. (c) Gastric conduit ulceration under gastroscopy. (d) Conduit perforation under a gastroscope.
Conduit necrosis may result from conduit torsion or intrinsic vascular insufficiency. 16 Furthermore, delayed conduit necrosis has been reported to be associated with radiotherapy, chemical or drug stimulation (alcohol abuse or nonsteroidal anti-inflammatory drug (NSAID) use), gastric stasis, bile juice regurgitation, or Helicobacter pylori infection. 14
According to the severity of the necrosis, a conservative, interventional, or surgical approach may be necessary. 12 In general, conservative treatment, such as anti-inflammatory and nutritional support, is recommended for type I gastric conduit failure, whereas surgical removal of the gangrenous conduit is strongly recommended for type II and III gastric conduit failure. Patients with conduit necrosis and perforations are often in a poor general condition and are unable to tolerate surgical treatment; consequently, interventional therapy, such as drainage of the pus cavity, may be used as a transitional treatment. Studies have revealed that conservative treatment may be safer than emergency surgery for patients with type II necrosis.11,17 However, emergency radical surgery should also be recommended for massive necrotic conduits if they are discovered early to prevent further progression of the disease. Under such conditions, conservative management failure may cause the infection to progress and lead to massive fatal hemorrhage (Figure 4).
To prevent conduit necrosis, other comorbid diseases of the stomach, the ischemic condition of the conduit, or the reconstruction approach should be evaluated. Specifically, for patients with severe gastric ulcers, the use of a gastric tube for reconstruction could help avoid potential perforations. However, gastric tubes should be chosen with caution if the patient has a short right gastroepiploic artery or intraoperative gastroepiploic artery injury. In cases of uncertain R0 resection with a high risk of local recurrence or an anticipated need for adjuvant/radical radiotherapy postoperatively, the selection of a retrosternal or subcutaneous reconstruction route warrants careful consideration. Finally, periodic endoscopic examination may be necessary to detect an early ulcer, and timely intervention can help prevent existing ulcers from progressing to conduit necrosis, thereby avoiding severe clinical consequences.
Thoracic duct-related complications
Refractory chylothorax
Chylothorax caused by injury to the thoracic duct or the surrounding collateral lymphatic duct is a relatively rare but serious complication of esophagectomy. Chylothorax can lead to severe metabolic, nutritional, and immunologic derangements and even postoperative mortality. Although this condition is usually treated effectively, some cases are refractory to well-established medical, interventional, or even surgical approaches. 18 The incidence of refractory chylothorax is less than 2% in the literature and 0.3% in our center (Table 1).
Studies have revealed that preoperative radiation, squamous histology, a large body surface area, and intraoperative fluid imbalance are risk factors for chylothorax. 19 Except for a few cases in which the causes of the refractoriness are well known, such as an unenclosed esophageal hiatus (chylothorax caused by chylous abdomen) 20 and anatomical variation of the thoracic duct,18,21 the etiology of refractory chylothorax remains unclear.
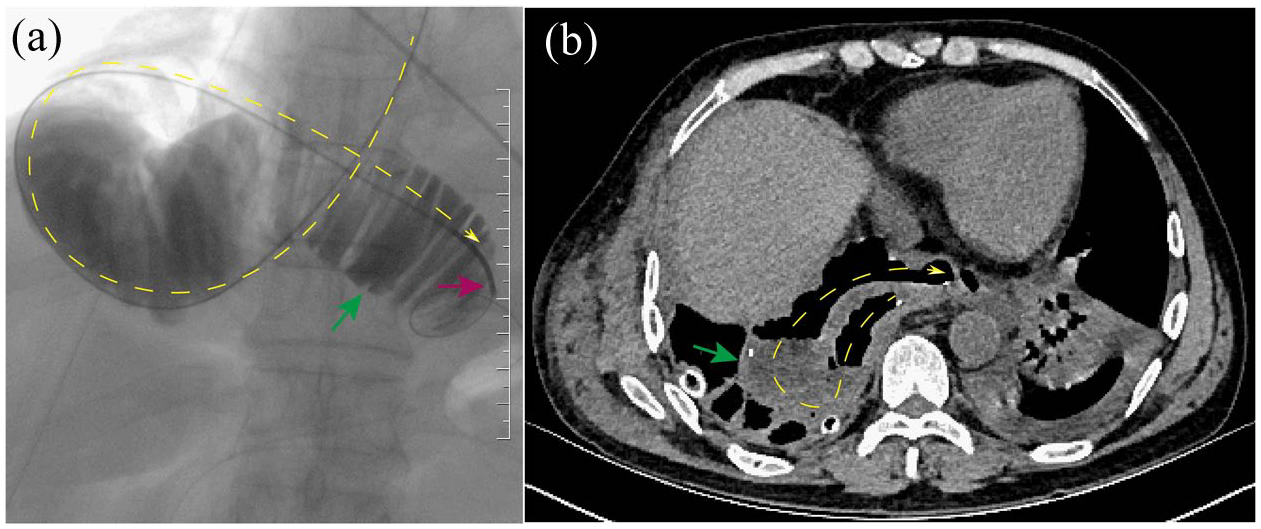
Recommendations for the management of refractory chylothorax are lacking. Studies have revealed that α1-adrenergic agonists, such as midodrine and etilefrine,22,23 and microwave ablation 24 can be beneficial. However, clinical experience with these methods is limited. Lymphangiography is a minimally invasive procedure that has been shown to have a high technical success rate in the management of refractory chylothorax and an 83% clinical success rate after the surgeon has performed one or two procedures. 25 Injury to the thoracic duct as a result of inappropriate positioning of fine ligature threads may also cause refractory chylothorax. Theoretically, thoracic duct ligation can be performed at any segment through cervical, thoracic, or abdominal approaches. However, in the context of extensive lymph node dissection and an anatomic variation of the thoracic duct, ligation just above the diaphragm is advised. Surgeons tend to prefer to ligate the thoracic duct through the right transthoracic approach. However, repeated ligation can damage the tissue above the diaphragm, resulting in ligation failure. Therefore, other researchers prefer ligating the thoracic duct at a lower position through a transhiatal approach 26 (Figure 5). This approach is advantageous because its associated morbidity rate is lower than that of the second transthoracic approach; it allows easy identification of anatomical variations of the thoracic duct at the level of the hiatus, and bilateral ligation can be completed in a single operation. 27

Refractory chylothorax after bilateral transthoracic ligation cured by transhiatal ligation. (a) Chest CT image showing bilateral pleural effusion with one chest drainage tube on each side of the thoracic cavity (thoracic drainage > 2000 ml per day in total). (b) Surgical inspection of the hiatus of the diaphragm. (c) Severance of the right diaphragmatic crus with an endoscopic stapler. (d) Exposure of the descending aorta and thoracic duct. (e) Ligation of an anatomical variation of the thoracic duct. (f) Chest CT 1 month after transhiatal ligation.
Chylomediastinum
Chylomediastinum, or a loculated chylothorax, is a rare condition characterized by the accumulation of chyle in the mediastinum. To date, only seven cases have been reported worldwide, two of which have been reported at our center.28–33 Similar to chylothorax, chylomediastinum can develop during any procedure that might damage the thoracic duct or its tributaries, regardless of whether the pleura is breached. Unlike chylothorax, chylomediastinum often occurs in the mediastinal region and does not require significant drainage of pleural fluid; thus, it does not usually cause fluid imbalance and is usually detected during routine follow-up or after the emergence of compressive symptoms.
Differentiating chylomediastinum from mediastinal infections is important because the latter can lead to serious consequences (purulent exudate can erode the airway or arteries, potentially leading to life-threatening complications). Chylomediastinum is diagnosed on the basis of clinical symptoms, upper gastrointestinal radiography findings, density of the fluid collection on CT scan, CT-guided puncture results, and lymphangiography findings. Notably, anastomotic leakage and chylomediastinum may coexist, thus complicating clinical assessment. 28
Treatment for chylomediastinum is similar to that for chylothorax and includes dietary control measures, interventional treatments (Figure 6), and surgery. Our preliminary research revealed that chyle can be intermittently drained in cases of chylomediastinum. 28 Similarly, Wang et al. demonstrated that clamping the chest tube intermittently decreased the duration of chest tube placement and the total drainage volume after lung cancer surgery in patients without air leakage. 34 Therefore, intermittent drainage to relieve extrinsic compression of the esophagus or airway can alleviate clinical symptoms, and positive pressure drainage of chyle can promote thoracic duct healing. Negative pressure drainage should be avoided because it may delay healing and cause significant fluid loss.

A patient who complained of gradually increasing chest tightness was ultimately diagnosed with chylomediastinum, which was cured by lymphangiography. (a) Chest CT after oral intake of iodine reveals no fistula 1 month after esophagectomy. (b) Increased mediastinal collection was mistakenly diagnosed as a cervical anastomotic fistula combined with intrathoracic infection in a local hospital 3 months following esophagectomy. (c) Lymphangiography revealed no clear leakage of the thoracic duct. (d) After the chest tightness disappeared, CT reexamination revealed absorption of the mediastinal collection 3 days after lymphangiography.
To prevent thoracic duct-related complications, intervention during the perioperative period is necessary. First, preoperative magnetic resonance thoracic ductography, 35 preoperative or intraoperative administration of oral cream, 36 olive oil, 37 lipid-rich nutrition 38 and intraoperative thoracic duct lymphography via near-infrared indocyanine green fluorescence imaging 39 can aid in the identification of the thoracic duct to prevent thoracic duct-related complications. Second, a low-fat diet after esophagectomy may reduce thoracic duct-related complications by reducing lymphatic pressure, facilitating the healing of lymphatic defects due to lymph node dissection. 40 Third, some scholars have recommended performing prophylactic thoracic duct ligation in patients with risk factors such as a difficult mediastinal dissection. 19
Hernia-related complications
Hiatal hernia
A hiatal hernia is a rare complication that can occur postoperatively or even years after esophagectomy. The colon, small bowel, greater omentum (Figure 7), and even other organs such as the duodenum and pancreatic head 41 can be herniated; the herniations may be asymptomatic and identified only at routine follow-up examinations or on the basis of different symptoms according to the herniated organ and degree of severity.

Hiatal hernia after esophagectomy. (a) Small bowel hernia 3 days after esophagectomy without clinical symptoms. (b) The hernia disappeared 3 months after esophagectomy; clinical observation was continued. (c) Colon hernia 7 days after esophagectomy with constant abdominal pain. (d) CT image 1 month after surgical repair. (e) Omental hernia compressing the lungs and mediastinum in a patient suffering from persistent chest tightness. (f) CT image 1 month after surgical repair.
According to the literature, the incidence of acute postoesophagectomy herniation is less than 1%. 42 Large-scale retrospective data indicate an incidence of 2.6% among all patients and 4.5% in patients who have undergone minimally invasive esophagectomy. 43 In contrast, our institution reports a 0.3% incidence of hiatal hernia. Because some cases of herniation are asymptomatic, the incidence of this complication may be underreported; one study estimates a true incidence of 3%–15%. 44 Several risk factors may contribute to hiatal herniation; these include anatomical disruption of the crura diaphragmatica and enlargement of the hiatus to facilitate esophageal mobilization, considered major predisposing factors; physiological factors such as negative intrathoracic pressure and increased intra-abdominal pressure; and a BMI < 25 kg/m2 (Ref. 45) and minimally invasive esophagectomy. 46
The clinical characteristics and treatment principles of hiatal hernias after esophagectomy differ from those of other types of hernias. 47 Patients with symptoms and large, progressive hernias should be considered for surgical treatment; however, the surgical indications for asymptomatic hernias are inconsistent.44,48,49 Price et al. 50 suggested that hiatal hernias should be repaired routinely. Patients with asymptomatic hernias may have a sudden onset or exacerbated symptoms, leading to intestinal ischemic necrosis, hydrothorax, and fecopneumothorax 5 without previous clinical signs. For such patients, emergency surgery is associated with a high rate of perioperative complications and should be avoided. 49 However, Barron et al. revealed that the “watch-and-wait” strategy is not associated with emergency complications and recommended close clinical follow-up for patients with asymptomatic paraconduit hernias after esophagectomy, 44 consistent with findings of other studies. 45 Moreover, the potential complications of emergency repair surgery and the long-term prognosis should be considered in surgical decision-making for asymptomatic patients.
Preventing the formation of hiatal hernias after esophagectomy is even more important. This can be achieved through strategies that involve manipulation of the crura diaphragmatica, including routine assessment and tightening of the hiatus to accommodate the conduit and fixation of the gastric conduit to the crura diaphragmatica. In addition, the anterior division rather than the lateral division of the crura can be performed, which allows adequate passage of the conduit. 51 Finally, the most effective prevention method may be closure of the esophageal hiatus combined with retrosternal or subcutaneous reconstruction, especially for patients at high risk of hiatal hernia.
Other hernias
Every area involved in esophagectomy is a potential site of herniation. In addition to hiatal hernias, retrocardiac lung hernias,52,53 intercostal lung hernias, 54 trocar-site hernias, internal hernias to the retrosternal space 55 and pericardium 56 and mesenteric defects 57 may also occur after esophagectomy. Each of these hernias is associated with unique risk factors related to the surgical procedure; however, most patients present with clinical symptoms and need surgery.
Dissection of the pleura is necessary for retrocardiac lung hernias that develop after esophagectomy; several factors may contribute to this complication, such as partial contralateral lung resection, aspiration via a drainage tube, or reconstruction via the nonretromediastinal route. 52 The herniated lung should be reintegrated into the thoracic cavity, and the hernial orifice should be closed properly by suturing the mediastinal pleura. An intercostal lung hernia is caused by defects or weakness of the chest wall and has been reported in two-stage minimally invasive esophagectomy. 54 Intercostal lung hernias may cause coughs, chest pain, or dyspnea; however, most hernias are asymptomatic and differ from retrocardiac lung hernias. Asymptomatic cases may be managed conservatively with observation, and symptomatic cases can be managed via surgical closure of the intercostal and thoracic wall muscles. 54
Although trocar-site hernias have not been reported as a postesophagectomy complication, some cases have been reported after laparoscopic surgery. Usually, suturing abdominal fascia defects at trocar sites measuring ⩽8 mm is unnecessary, 58 whereas fascial defects at trocar sites measuring ⩾10 mm require surgical closure. 59 Moreover, the incidence of complications is greater for defects located at the umbilical trocar than for defects located at the lateral trocar. Trocar-site hernias can lead to small bowel obstruction or even life-threatening strangulation if not recognized in a timely manner. We observed a left lower abdominal wall hernia at the site of a 5-mm trocar 2 days after esophagectomy, which led to small bowel obstruction. This hernia was treated with emergency surgery (Figure 8).

Hernia at a 5-mm trocar site 3 days after esophagectomy. (a) Abdominal pain suddenly occurred 3 days after esophagectomy; physical examination revealed a mass at the site of the 5-mm trocar in the left lower abdomen. (b) Abdominal CT scan revealed an abdominal wall hernia. (c) Operative inspection revealed small bowel herniation. (d) Proximal small intestine dilatation.
Retrosternal hernias and hiatal hernias may share similar risk factors; among them, extensive dilation of the retrosternal orifice may be the most important. 55 Intrapericardial hernias usually develop as a result of the opening of the pericardium during esophagectomy. 56 An internal hernia of the small intestine may develop through a mesenteric defect that forms after esophagectomy with interposition of the small or large intestine. 57 All three types of hernia may lead to intestinal obstruction, similar to trocar-site hernias, and require treatment with emergency surgery. To effectively prevent the development of such internal hernias, surgeons should properly close sites prone to hernia development.
Transmural Hem-o-lok clip migration
The migration of Hem-o-lok clips and stitches into the surrounding hollow viscera following esophagectomy is incredibly rare, and therefore the underlying pathophysiological mechanism is poorly understood,4,60 although it is relatively common after laparoscopic surgery.61,62 During routine esophagectomy, thoracic surgeons divide the azygos arch and then double clip it with Hem-o-lok clips proximally and distally. As a result, the Hem-o-lok clips remain close to both the anterior wall of the conduit and the posterior wall of the trachea during esophageal bed reconstruction, making it possible for the clips to migrate transmurally. We believe that this migration is due to erosion of the wall of the conduit or trachea while eating, breathing, or coughing, an inflammatory response, or injury to the conduit or trachea during the operation. To date, only two cases of Hem-o-lock clip migration have been reported in esophagectomy,4,60 whereas three cases have occurred at our center, potentially attributable to our routine usage of Hem-o-lok clips.
Interestingly, simple penetration of the trachea or conduit may not cause serious symptoms, 60 possibly because the process of penetration is slow and accompanied by tissue destruction, regeneration, and fibrosis; consequently, a curative outcome is potentially attainable through conservative therapy. Under these conditions, patients typically do not present with symptoms or have causes for concern; thus, further intervention, such as endoscopic or surgical clip removal, may be unnecessary. However, simultaneous penetration of the trachea and conduit may induce development of a tracheoesophageal fistula, 4 which can be addressed with interventional or surgical treatment. In contrast, the transmural migration of Hem-o-lok clips is potentially fatal and should be given special consideration by thoracic surgeons, particularly when it occurs in the early postoperative period. In such circumstances, meticulous drainage coupled with cavity obliteration is imperative (Figures 9 and 10). Theoretically, in addition to the use of absorbable Hem-o-lok clips and staple or suture ligation without clipping, measures to prevent iatrogenic injury to the conduit and trachea can include reconstruction via a route other than through the esophageal bed (retrosternal or subcutaneous reconstruction).

Transmural Hem-o-lok clip migration causing a gastric conduit fistula. (a) Pneumomediastinum combined with a partial defect in the gastric tube (3 days after esophagectomy). (b) Gradual absorption of pneumomediastinum (2 weeks after esophagectomy). (c and d) The Hem-o-lok clip is shown and was removed under gastroscopy. (e) Mucosal defect after Hem-o-lok clip removal. (f) Upper gastrointestinal radiography revealed no fistula after Hem-o-lok clip removal.

Transmural Hem-o-lok clip migration causing a tracheal fistula. (a) Detection of pneumomediastinum. (b) Partial defect of the intermediate bronchial membrane. (c) A Hem-o-lok clip is shown under bronchoscopy. (d) Fistula healing after conservative tube drainage.
Conclusion
Esophagectomy remains a complex procedure with significant risks of rare but life-threatening complications. This review highlights four categories of underrecognized complications (Table 1): (1) esophageal substitute-related complications, (2) thoracic duct-related complications, (3) hernia-related complications, and (4) transmural Hem-o-lok clip migration. Thoracic surgeons should familiarize themselves with these rare complications of esophagectomy, most of which are serious conditions that require early and accurate diagnosis for proper management. However, given the rarity of these complications, the findings of this study should be cautiously interpreted.
Supplemental Material
sj-jpg-1-tag-10.1177_17562848251366238 – Supplemental material for Rare complications after esophagectomy: incidence, clinical features, risk factors, management, and prevention
Supplemental material, sj-jpg-1-tag-10.1177_17562848251366238 for Rare complications after esophagectomy: incidence, clinical features, risk factors, management, and prevention by Guoqing Zhang, Jia Jiao, Long Qiu, Zheng Ding, Jia Zhao, Xue Pan and Xiangnan Li in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
