Abstract
Objective:
Indocyanine green has been used in the assessment of the gastric conduit perfusion in thoracoscopic esophagectomy to prevent malperfusion-associated anastomotic leak. This study aims to evaluate the initial results of investigating the gastric conduit perfusion with indocyanine green in the surgical treatment of esophageal cancer.
Patients and methods:
This cross-sectional descriptive study was carried out on 54 esophageal cancer patients undergoing thoracoscopic esophagectomy and gastric conduit reconstruction. The blood flow in the gastric conduit was observed using an infrared camera and indocyanine green after completion of the conduit and after tunneling the conduit through the mediastinum to the neck.
Results:
The gastric conduit width and length were 5.2 ± 0.3 cm, and 31.5 ± 1.6 cm, respectively. The length of the gastric conduit from the junction between the right and left gastroepiploic to the point where the distal end of the gastric conduit still has a vascular pulse was 11.9 ± 4.3 cm. Seventeen patients (31.5%) had poor blood supply at the distal end of the gastric conduit, with indocyanine green appearance time ⩾ 60 s, in whom anastomotic leaks occurred in five patients (9.3%). The lack of connection between the right and left gastroepiploic vessels was associated with poor blood supply of the distal gastric conduit (p = 0.04). Multivariable logistic regression analysis showed association between the time of indocyanine green appearance at the distal gastric conduit and the risk of anastomotic leak (OR = 1.99, 95% CI = 1.10–3.60, p = 0.02).
Conclusion:
Investigation of gastric conduit perfusion using indocyanine green in gastric conduit reconstruction detected 31.5% of patients with poor blood supply at the distal end of the conduit, in whom 9.3% had anastomotic leak. The longer indocyanine green appearance time in the distal gastric conduit (segment BC), was associated with the higher rate of the anastomotic leak.
Keywords
Introduction
Esophageal cancer is the global seventh most common malignancy with poor prognosis. 1 Radical esophagectomy is still the primary treatment for esophageal cancer, and gastric conduit is preferred for replacement of the resected esophagus. 2 Other alternatives, that is, the colon or small intestine, are used when a gastric conduit is not feasible. However, the high rate of anastomotic leak is still the main concern with a gastric conduit, which could be associated with preoperative nutritional status of the patient, conduit tunneling, anastomotic location or techniques and gastric conduit perfusion, in which the latter has been considered the most important and independent cause of anastomotic leak.3–5
Previously, the perfusion of the reconstructed gastric conduit was subjectively determined by direct intraoperative observation of color, peristalsis, pulse rate, bleeding at the cutting edges of the anastomotic site, and the color of the mucosal layer, which are subjective and inaccurate.6,7 Other imaging methods that is, Doppler ultrasound, intraoperative angiography, have been proposed for gastric conduit perfusion assessment, but they are not clinically feasible and, therefore, rarely applied in practice. 8 In 2011, Shimada achieved initial positive results with a near-infrared fluorescence system and ICG green dye to evaluate gastric conduit perfusion. 9 In thoracoscopic esophagectomy, ICG has also been used in guiding lymph node dissection, imaging the thoracic duct, and increasingly applied for gastric conduit perfusion assessment.10,11 Other studies reported that the application of ICG imaging results in significant reduction of anastomotic leak by up to 70%.12–15
In Vietnam, esophageal cancer is the 15th most common cancer, with an incidence of 8.7 per 100,000 people, 2411 new cases and 2222 deaths annually. 16 Thoracoscopic radical esophagectomy with reconstructed gastric conduit and direct visual assessment of stump perfusion is still associated with relatively high rate of postoperative anastomotic leak of 15%–25%. Therefore, this study aims to investigate the efficacy of ICG fluorescence imaging in evaluation of blood flow in the reconstructed gastric conduit for esophageal replacement in Vietnamese patients.
Patients and methods
In this cross-sectional, prospective descriptive study, 54 esophageal cancer patients undergoing thoracoscopic esophagectomy and gastric conduit reconstruction for esophageal cancer from June 2022 to December 2023 were enrolled, including 46 patients with preoperative chemoradiation therapy. This study was approved by the Ethics Council of Military Hospital 103, Vietnam on 9th December 2022, approval number 193/CNChT – HĐĐĐ.
Patients who met the following criteria were included in the study (a) diagnosis of carcinoma in the thoracic esophagus and undergoing thoracoscopic radical esophagectomy for the preoperative stage: cT1b-cT2, N0 or cT1b-cT3, N+ after preoperative chemotherapy, (b) undergoing ICG imaging for evaluation of the blood supply of the replacement gastric conduit (c) ASA-PS ⩽ 3. Patients undergoing thoracoscopic esophagectomy for noncancerous causes or those with tumor invasion at T4b level according to the classification of the American Joint Committee on Cancer were excluded.
All patients underwent thoracoscopic esophagectomy and 2—field lymphadenectomy. Briefly, the patient was placed on a semiprone position, the thoracic esophagus was resected, and mediastinal lymph nodes, fat layer and lymph nodes groups of 106R, 106L, 107, 108, 109, 110, and 120 were removed via thoracoscopic approach. A gastric conduit which had been created via the abdominal approach was passed through the chest for anastomosis with the proximal esophagus in the left neck after resection of the cervical esophagus. In the abdominal phase, the greater omentum was dissected between the stomach and the colon beyond the greater curvature vascular arch to the right omental gastric vascular pedicle. The abdominal lymph node groups of 1, 2, 3, 4, 5, 6, 7, 8, 9 or any suspected of metastasis were removed.
The reconstructed gastric conduit was approximately 5 cm wide, retaining the right and left gastroepiploic arteries, some of the first branches of the right gastric artery, and the arcade between the right and left gastroepiploic arteries. The purpose of this arcade preservation is to ensure better blood supply of the entire gastric conduit, using an open stapler for laparoscopically assisted or open surgery (Figure 1), or an endoscopic stapler for totally laparoscopic surgery (Figure 2).

Gastric tube shaping with stapler-assisted laparoscopic abdominal surgery or open surgery. (a) The entire gastric tube has not been cut. (b–d) gastric tube is shaped with two Stapler size 80 mm

Gastric tube shaping with stapler, laparoscopic abdominal surgery. (a–c) gastric tube is shaped with two Stapler size 60 mm. (d) The stomach tube is completely formed.
After the reconstruction completion, all the dimensions of the conduits were measured as in Figure 3: the length of the gastric conduit (AD) was measured from the pylorus to the distal end of the gastric conduit; the length of conduit supplied by the right gastroepiploic artery (AB) was measured from the pylorus to the connection between the right and left gastroepiploic vessels; the length of conduit with adequate perfusion (AC) was measured from the pylorus to the point in the distal gastric conduit where the vasculature was still visible after ICG injection. (BC) was measured from the connection between the right and left gastroepiploic vessels to the distal gastric conduit abbreviated as “the distal gastric conduit.” All measurements were performed along the greater curvature of the conduit. The width of the gastric conduit was the distance from the small to the large curvature, measured at the middle of the gastric conduit.

How to measure indicators of gastric tube size. (a) Measuring stomach size during laparoscopic-assisted or open abdominal surgery. (b) Measuring stomach size during laparoscopic abdominal surgery. (c) Location of landmark to measure stomach size.
Fluorescence imaging of the gastric conduit was acquired using ICG (Verdye™, Diagnostic Green GmbH, Aschheim-Dornach, Germany). The ICG solution was prepared before injection in distilled water (2.5 mg/ml) and injected rapidly through the internal jugular vein or the basilic vein (within 3 s) at 2 time points: the 1st dose was injected after completion of the gastric conduit to evaluate the perfusion status of the gastric conduit without tension (Figures 4 and 5); the 2nd dose was injected after passing the gastric conduit through the posterior mediastinum to the neck in order to re-examine the status of gastric conduit perfusion. The perfusion of the gastric conduit was evaluated using a near-infrared light camera and the acquired images were viewed in color and monochromatic modes.

ICG appearance of the gastric tube when placed in front of the chest wall. (a) ICG appears to point a (origin of the right gastroepiploic artery). (b) ICG appears to point b (the junction between the right and left gastroepiploic arteries). (c) ICG appears up to point c (the farthest point of the gastric tube with a pulse on the greater curvature).(d) ICG appears to point d (furthest part of the gastric tube).
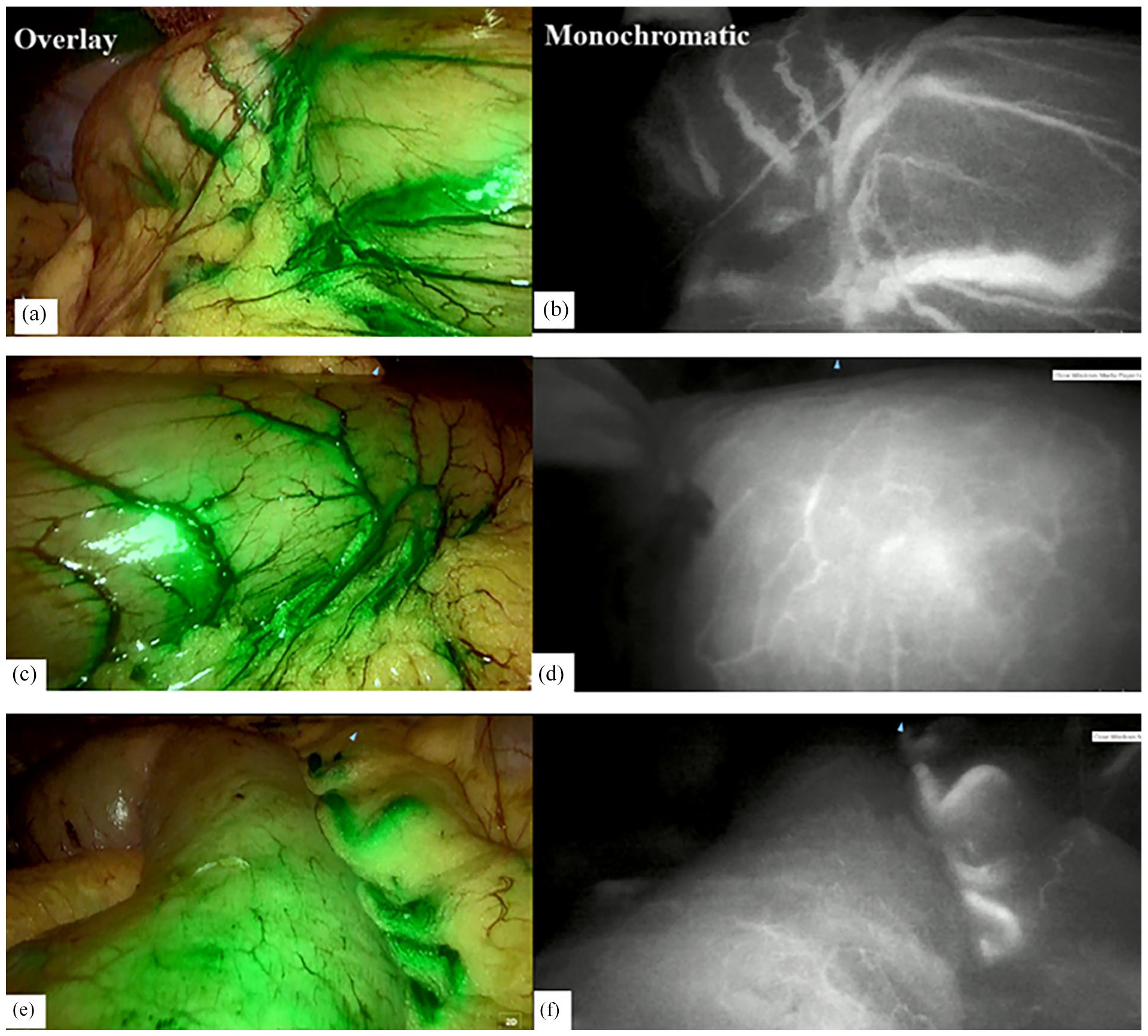
Image of gastric tube showing ICG during laparoscopic abdominal surgery. (a, c, e) images of ICG appearance of the gastric tube in overlay mode.(b, d, f) images of ICG appearance of the gastric tube in monochromatic mode.
The interval between the time of appearance of ICG at any location and at point A (the origin of the right gastric artery) was calculated to evaluate blood supply of the distal gastric conduit. The gastric conduit segment was considered adequately perfused if the interval was less than 60 s, and poorly perfused if the interval was equal or greater than 60 s. The demarcation line between the perfused and nonperfused areas as identified by ICG dye was marked (Figure 6).

Image of the poor blood supply boundary of the distal gastric tube through ICG appearance. The red arrow is the boundary between poor blood supply and good blood supply marked with an electric knife.
Statistical analyses were carried out using SPSS version 26.0 software for Windows (SPSS Inc., Chicago, IL, USA) and a p-value < 0.05 was considered significant. Quantitative variables with normal distribution were presented as mean and standard deviation. For quantitative variables that did not conform to a normal distribution, median values with interquartile ranges (Q1–Q3) were reported. Categorical and continuous variables were compared using the Chi-square or Fisher tests and Student’s t-test when appropriate. Multivariate logistic regression models were used to analyze factors responsible for anastomotic leakage.
Results
As shown in Table 1, the age of our patients was 58.8 ± 7.7 years. All patients were male. A total of 85.2% of patients received preoperative chemo-radiation therapy. Anastomotic leakage was 9.3%. Among other complications, respiratory complications were the most common, accounting for 16.7%. The surgical time was 258.3 ± 34.6 min, and intraoperative blood loss was 80 (50–90) ml. The postoperative hospital stay was 14.7 ± 9.7 days. Except for a longer hospital stay in five cases (9.3%) with anastomotic leakage and nine cases with respiratory complications (16.7%), most patients had a hospital stay of 8–10 days.
General patient characteristics.

The gastric conduit width was 5.1 ± 0.3 cm, length (A–D) 31.5 ± 1.6 cm, length (A–B) 18.9 ± 1.5cm, length (A–C) 29.2 ± 1.3 cm, and length (B–C) 11.9 ± 4.3 cm. The connection between the right and left gastroepiploic vessels was present in 25 patients (46.3%). Conduit distal-end ischemia was observed at the first check in eight patients (14.8%), and at the second check in 11 patients (20.4%).
ICG was administered via the central venous line in 33 patients (61.1%) and peripheral venous line in 21 patients (38.9%). 96.3% of patients received two ICG injections, two patients had to receive three ICG injections. The systolic blood pressure at the first injection and second injection was 118.6 ± 8.4 mmHg and 117.8 ± 7.7 mmHg, respectively.
The ICG appearance time of the entire gastric conduit (A–D) in the first and second injections were 24.5 (21.0–63.5) seconds and 26.0 (22.8–63.3) seconds, respectively. The ICG flow rate of segment A-B at the first injection was the fastest (3.2 ± 1.2 cm/s). The overall ICG flow rates of the whole gastric conduit in the two injections were 1.1 ± 0.6 cm/s and 0.9 ± 0.4 cm/s, respectively (Table 2).
Time and rate of appearance of ICG (n = 54).

ICG injection detected 17 patients with poor blood supply to the distal end of the gastric conduit in both first and second injections, as shown by ICG appearance time at distal end of the gastric conduit ⩾60 s (31.5%), in which gastric conduit ischemia was visually detected by color change to blue in the serosa and mucous in eight patients. All these 17 patients had the distal end removed to an area of good perfusion, and the length of removed part was 2.4 ± 0.6 cm.
Anastomotic leakage occurred in 5/17 patients, in which three patients were completely stabilized with medical treatment. The other two patients required surgical interventions. One patient was managed with continuous suction of the gastric conduit combined with pleural cavity drainage and was stable after 2 weeks. One patient had peritonitis that required re-do surgery to close the gastric conduit fistula. This patient was stable after 1 week of intensive treatment and discharged home.
As shown in Table 3, the presence of connection between the right and left gastroepiploic vessels was associated with better blood supply in distal gastric conduit (p = 0.04).
Risk factors of delayed ICG appearance time of the gastric conduit.

Table 4 shows that there was no association between the anastomosis at 1 month after surgery and intraoperative blood supply condition.
Relationship between ICG appearance time and the condition of the anastomosis at 1 month after surgery.

Multivariable logistic regression analysis shows the association between ICG appearance time of segment (B-C) and the rate of anastomotic leak (OR = 1.99, 95% CI = 1.10–3.60, p = 0.02) (Table 5).
Anastomotic leak predictors.

Discussion
In this study, we managed to construct a large gastric conduit of 5.1 ± 0.3 cm in width, larger than the expected width of 5 cm. The average gastric conduit length (A–D) was 31.5 ± 1.6 cm, which is slightly shorter than that reported by Koyanagi et al. 6 This showed that the average length of the gastric conduit is of 34.9 ± 3.6 cm, regardless of the patient’s height and weight. The length from point (A–B) (from the pylorus to the connection between the right and left gastroepiploic vessels) was 18.9 ± 1.5 cm. This was the gastric conduit section that was best perfused with blood and the average length of (A–C) segment was 29.2 ± 1.3 cm. We preserved the right and left omental vascular arches to nourish the stomach well. Twenty-five patients (46.3%) had the connection between the right and left gastroepiploic vessels. As shown in Table 3, the connection between the right and left gastroepiploic vessels was significantly related to ischemia at the distal end of the gastric conduit. When the connection between the two arteries was present in the replacement gastric conduit, the distal gastric conduit ischemia rate was lower (p = 0.04). Possibly due to more blood from the right gastroepiploic, although collaterals to the left gastroepiploic supplies blood to the distal part of the gastric conduit. This finding is similar to that by Koyanagi et al., 6 who found the distance (A–B) of 23.1–3.1 cm, and patients with connections between the two arteries had better distal blood supply to the gastric conduit.
Investigation of gastric conduit perfusion with ICG showed that ICG appearance time (A–D) in the first injection and the second injection were 24.5 (21.0–63.5) seconds and 26.0 (22.8–63.3) seconds, respectively. Univariate analysis of the relationship between the time of ICG appearance and several factors showed that gastric conduit had a connection between the right and left gastroepiploic arteries and had better perfusion. Our data showed that poor blood supply was not associated with preoperative chemotherapy, radiotherapy, disease stage, surgery time, and postoperative complications. Multivariable logistic regression showed that the longer time of ICG appearance in the B–C segments (from the junction between the right and left gastric arteries to the distal end of the gastric tube with a pulse) had a higher rate of anastomotic leak (OR = 1.99, 95% CI = 1.10–3.60, p = 0.02). The distal gastric conduit segments B–C depend on the blood supply from the artery on the greater curvature and the vascular network in the stomach wall, especially the distal part of the gastric tube. According to Kumagai et al., 18 the shorter time of ICG appearance at the distal end of the gastric conduit brought about the better perfusion of the gastric conduit and vice versa. Noma et al. 19 reported that if ICG enhanced gastric conduit perfusion within 20 s, the gastric conduit was considered well-perfused at that location. The anastomotic leak rate in patients using ICG as perfusion indicator was significantly lower than in patients not using ICG (8.8% vs 22%, p = 0.03) 8 . Luo et al. 8 found that the gastric conduit was considered well perfused when the ICG appearance time was within 60 s. According to the 60 s rule, the anastomosis was performed at the site where ICG appeared in less than 60 s, and the poorly perfused part was removed. The rate of anastomotic leak in esophagectomy for esophageal cancer was 10.4% in the group without ICG injection and significantly higher than in the group with ICG injection (1.2%). This result suggests that a reduction in the anastomotic leak rate of approximately 9% can be achieved when using ICG. This evidence strongly suggests that adequate gastric conduit perfusion is important. Kumagai et al. 20 proposed a 90-s rule to confirm good perfusion at the anastomotic site. All anastomoses are created in the area of the ICG-enhanced gastric tube within 90 s of the initial ICG injection into the right gastroepiploic artery stump. Based on this rule, the distal end of the gastric conduit needed to be revised at a rate of 50%, and the anastomosis site was changed in 18 out of 35 cases of ischemia. No patient underwent anastomosis creation at a site with delayed ICG enhancement after 90 s. The anastomotic leak rate in this study was 1.4%.
When investigating the speed of ICG appearance in gastric conduit perfusion, we found that the speed of ICG appearance at segments (A–B) was the fastest at 3.2 ± 1.2 cm/s. Meanwhile, the ICG appearance in gastric conduit in two injections was 1.1 ± 0.6 cm/s and 0.9 ± 0.4 cm/s, respectively. ICG appearance in the proximal part of the gastric conduit was faster than in the distal part of the gastric conduit. The faster ICG appearance may be associated with better blood perfusion of the gastric conduit. Koyanagi et al.6,21 demonstrated that ICG fluorescein blood flow velocity in the intraoperative gastric conduit is a valuable tool for predicting the risk of anastomotic leak. The rate of ICG flow in the gastric conduit wall is not always the same as that of the larger curvilinear vessel. The ICG flow rate in the gastric conduit wall is critical in influencing perfusion of the distal portion of the tube. Based on the ICG flow rate in the gastric conduit wall and the vascular loop on the greater curvature, we divided the flow rate into two groups: an “equivalent” group with similar ICG fluorescence speed in the gastric conduit wall and in the omental vessels of the stomach, and a “slow” group in which the ICG fluorescence flow rate in the gastric conduit wall is slower than the omental gastric vessels. No anastomotic leak developed in any of the patients (0/25) in the “equivalent” group, but in 46.7% (7/15) of cases in the “slow” group (p < 0.001). Based on the results of univariate analysis, Koyanagi et al. 6 chose the ICG fluorescence flow rate on the gastric conduit wall as a parameter for multivariate analysis to evaluate the risk factors of anastomotic leak after esophagectomy. The analysis also included the reconstruction route, operative time, and intraoperative blood loss, which were considered risky factors for anastomotic leak. Based on the receiver operating characteristic (ROC) curve of the ICG flow rate in the gastric conduit wall, the area under the curve was 0.96, and the threshold value was determined as 1.76 cm/s according to the ROC curve. Therefore, logistic regression analysis determined that the only independent significant predictor of anastomotic leak was an ICG fluorescence flow rate in the gastric conduit wall of 1.76 cm/s or less (rate odds, 36.5; 95% confidence interval, 4.02–905.7; p = 0.004). 6
In this study, the anastomotic leak rate was 9.3%. All of these cases were in the group of 17 patients detected with distal ischemia by ICG with the time of first ICG appearance ⩾ 60 s. Although all these patients had the ischemic distal portion of the gastric conduit removed, the anastomotic leak still occurred in 5/17 patients. There were three patients with anastomotic leaks who received medical treatment, and their anastomotic leaks were completely stabilized. Two patients had a gastric conduit leak. The first patient had a continuous suction gastric conduit combined with pleural cavity drainage. After 2 weeks, the patient was stable. The second patient showed signs of peritonitis, so he had surgery again to stitch up the gastric conduit leakage. After 1 week, the patient was stable. Campbell et al., 14 showed a significant reduction in anastomotic leak rate from 20% to 0% after performing intraoperative perfusion assessment using ICG images. A meta-analysis by Slooter found that the application of ICG in assessing gastric conduit perfusion reduced anastomotic leak complications by 70%. The reported anastomotic leak rate was 11.1%. 12 Luo et al. 8 studied on 192 patients, 86 in the ICG group, and 106 in the non-ICG group. As a result, 12 patients had anastomotic leakage, of which the rate of anastomotic leakage in the group not using ICG was 10.4%, significantly higher than in the group using ICG (1.2%). However, a meta-analysis by Deggett’s showed 14% anastomotic leak in patients with ICG assessment. 22 Zehetner et al. 23 recently demonstrated that anastomotic leaks can frequently occur when gastroesophageal anastomosis is placed in an area without ICG fluorescence. These findings strongly suggest that adequate perfusion of the gastric conduit wall is important for complete tissue healing at the gastric anastomosis site. Gastric conduit leakage and gastric conduit necrosis are very dangerous complications that often lead to death. In our study, we encountered two patients with gastric conduit leaks at the site of the stapler cut for shaping. All use endoscopic staplers, so there was no stitching to increase the cutting area. In the above two cases, one case needed re-surgery due to peritonitis. After suturing the fistula, the patient was stable and discharged from the hospital. According to research by Masaki Ohi, out of 120 patients, one case had gastric conduit necrosis (accounting for 0.83%). 15
The mortality in this study was 1.9%, which was associated with postoperative respiratory complications. Some other authors showed mortality from 1.4% to 8.3%.24–26 Respiratory complications are still the most common and severe complications in esophageal cancer surgery and are also complications that often lead to operative mortality. In the study, nine patients had respiratory complications, accounting for 14.8%, most commonly pneumonia, effusion, pneumothorax, and empyema. These complications could be reduced with antibiotics, early chest tube insertion, and removal
This study has the following limitations
The study had small sample size and lack of control group. Moreover, the application of both open and endoscopic staplers during gastric conduit construction may confuse the interpretation of anatomostic leak data. ICG-based assessment of gastric conduit perfusion is a semiquantitative method, and therefore less reliable.
Conclusion
Indocyanine green for the assessment of gastric conduit perfusion in thoracoscopic esophagectomy to treat esophageal cancer is a safe and feasible method to help reduce leaking complications, with an anastomotic leak rate of 9.3%. ICG helped detect 31.5% of patients with distal ischemia. The longer ICG appearance time at the distal end of the gastric conduit (segment BC) was associated with a higher risk of anastomotic leak.
Footnotes
Acknowledgements
We would like to express our gratitude to all officers and staff of the Gastrointestinal Surgery Department, the Digestive Surgery Center, Military Hospital 103 and the Department of Gastrointestinal Tract Surgery, Central Military Hospital 108, who made a great contribution to the success of the study.
Author contributions
All authors made substantial contributions to this study. Tiep Nguyen Van, Son Le Thanh, and Tuan Nguyen Anh were responsible for the study concept, design, data analysis; manuscript revision. Hoai Nguyen To and Hiep Pham Van were responsible for analysis and manuscript preparation. Hoe Nguyen Trong was responsible for manuscript revision. Khanh Ngo Gia and Du Nguyen Van were responsible for data collection. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Research ethics approval
This study was conducted in accordance with the declaration of Helsinki, approved by the Ethics Council of Military Hospital 103, Vietnam on 9th December 2022, approval number 193/CNChT – HĐĐĐ.
Informed consent
Participants fully agreed and voluntarily participated in the study. Written informed consents were obtained with full signatures.
Trial registration
Not applicable.
