Abstract
Background:
Mucosal histological activity is increasingly valued as a treatment endpoint in inflammatory bowel diseases (IBD). In the Asia Pacific region, the utility and acceptability of IBD histology as a treatment endpoint are uncertain due to the heterogeneity of IBD prevalence, resourcing and level of knowledge among practitioners. There is an opportunity to engage clinicians to harmonise histology reporting and collaborate with pathologists in this field.
Objectives:
We aimed to develop consensus statements through anonymous voting on histological features, processing, reporting and relevance to treatment outcomes in IBD, including ulcerative colitis (UC) and Crohn’s disease (CD).
Design:
The consensus document was developed through a comprehensive literature review, followed by a deliberation process among experts in the field.
Methods:
Representatives of the Asia Pacific Association of Gastroenterology, in collaboration with pathologists, voted anonymously in accordance with modified Delphi methodology on statements relevant to IBD and histology. Domains of interest were histological features of UC and CD, relevance to clinical management and the potential utility of artificial intelligence (AI) in grading histological disease severity. Level of evidence and recommendation grade were included in accordance with the National Health and Medical Research Council, Australia guidelines of Australia.
Results:
Consensus was reached on 37 out of 38 statements concerning definitions, pathology processing and reporting, scoring system and relevance to clinical outcomes. Knowledge gaps were identified with uncertainty over the role of AI.
Conclusion:
These consensus statements provide recommendations, with specific relevance to the Asia Pacific region, on the role of histology in IBD to harmonise its use. The statements will promote understanding and applicability in research and in the routine management of IBD.
Plain language summary
A group of international experts from the Asia Pacific region reviewed the latest literature and developed consensus statements to improve the use of histology in inflammatory bowel disease care. They agreed on 37 key statements covering the important topics such as the features indicating disease activity, how histology can guide treatment decisions, the role of artificial intelligence in improving diagnostic accuracy, among other topics. These consensus guidelines will help doctors and pathologists work together to make better choices for patients care.
Introduction
Inflammatory bowel diseases (IBD) are chronic inflammatory conditions encompassing ulcerative colitis (UC) and Crohn’s disease (CD). The natural history of IBD is variable but may result in the need for medical therapy, hospitalisation and surgery.1,2 Evaluating mucosal histological inflammatory activity is an effective tool in the assessment of IBD. As an objective measure, histology has the potential to assist in diagnosis, assess disease severity, monitor treatment response and provide prognostication. 3 However, it has not been adopted as a treatment target by the Selecting Therapeutic Targets in Inflammatory Bowel Disease-II (STRIDE-II) consensus. 4 While it was considered a potential future therapeutic target in UC, the lack of prospective data confirming the role of histology in IBD care, the absence of agreement on a grading system and questions about generalisability and acceptability were limitations to application in current practice. Nonetheless, histology may be especially relevant in the Asia Pacific region, where clinicians may have less experience in IBD and face a greater need to distinguish it from infectious enterocolitis.5–7 Raising the awareness of IBD histology in this region might also promote routine use as an assessment tool. Harmonisation of histological terminology, appropriate processing of data and samples and synaptic reporting may also improve care and foster collaborative research across countries. Artificial intelligence (AI) might further enhance the diagnostic accuracy, yet this technology has not been adopted in the region for analysing IBD.
Consensus statements on histology in IBD are essential, given its potential as a unifying management tool. Agreement among representative gastroenterologists and pathologists across the region could establish standardised definitions, reporting, activity measurement and treatment approaches. There may be relevance with machine learning and AI in diagnosing IBD, excluding infectious enterocolitis, and in interpreting histological disease severity.
Methods
The Asia Pacific IBD consensus working group was established in 2006 to coordinate research and increase the awareness of IBD in a highly populous region of the world with a rising disease incidence. In response, the Asia Pacific Association of Gastroenterology (APAGE) has funded a project to determine histological role in IBD. Voting members were selected based on demonstrable expertise in IBD, including leadership roles, clinical experience, research authorship and guideline development. The working group meeting involved 20 gastroenterologists and 6 histopathologists from Australia, China, Hong Kong, India, Japan, Malaysia, Singapore, South Korea, Taiwan and Thailand.
A comprehensive literature search was conducted through PubMed and EMBASE up to January 2023, supplemented by a manual search of relevant references relating to histology and IBD. Keywords used included ‘inflammatory bowel disease’, ‘ulcerative colitis’, ‘Crohn’s disease’ combined with terms such as ‘histology’, ‘histopathology’, ‘histological activity’, ‘histological index’, ‘biopsy’, ‘multidisciplinary’ and ‘artificial intelligence’. Studies that were not published in English were excluded. The steering committee (R.W.L., H.L., T.C.) drafted the initial consensus statements and divided the statements into domains of interest. Where relevant, the Patient Intervention Comparator Outcomes approach in generating statements was taken, especially where controlled data were available. 8 Subsequently, the statements and corresponding literature were distributed to the panel members to provide comments. Voting was performed through a personalised online survey in accordance with the RAND/UCLA modified Delphi method.9,10 This is a formal group consensus process that systematically and quantitatively combines expert opinion and evidence by asking panellists to rate, discuss then re-rate items. The IBD consensus working group has adopted this process in prior clinical practice consensus guidelines.11–15 The panellists rated the level of agreement for each statement as: (a) agree completely, (b) agree with some reservation, (c) neither agree nor reject, (d) reject with reservation, (e) reject completely. The threshold for acceptance was 75% agreement. Draft statements were revised based on feedback. Up to three iterative rounds of voting were conducted if statements needed clarification or consensus was not initially reached. Each accepted statement was then graded to indicate the quality of evidence and classification of recommendations according to the National Health and Medical Research Council, Australia (NHMRC; Tables S1 and S2). 16 A final in-person meeting of all the panel members was convened in Bangkok, Thailand, in December 2023. Members reviewed statements lacking consensus, refining or rejecting them as needed. The final consensus was presented to global IBD experts for external validation, with independent voting to ensure no discrepancy. This consensus was reported in accordance with the ACCORD (Accurate COnsensus Reporting Document) guideline. 17
Results
Following a literature review, 37 statements were developed covering the domains of histology of UC and CD, pathology reporting, histological indices for measuring disease activity, endoscopic biopsy methods and tissue processing, biopsy indications, defining mucosal healing, pathologist team membership and role of AI (Table 1 and Table S3). There was uncertainty on escalating treatment when histological remission is not achieved, preventing consensus on this statement. Overall, there was strong support for adopting histology in IBD evaluation in the Asia Pacific region and agreement on using AI to improve throughput and accuracy.
Summary of statements, the status of consensus agreement and the NHMRC designation of the level of evidence and grade of recommendation.

AI, artificial intelligence; GS, Geboes score; H&E, hematoxylin and eosin; IBD, inflammatory bowel disease; IHC, immunohistochemistry; NHMRC, National Health and Medical Research Council, Australia; NI, Nancy index; RHI, Robarts Histopathology Index.
Statements reached by consensus
Domain 1: Histological features of UC
In UC, lymphocytes and plasma cells infiltrate the mucosa and superficial submucosa. An early diagnostic feature is basal plasmacytosis, which is non-specific, characterised by predominant plasma cells found between crypts and the muscularis mucosae. 11 A sign of histological activity is marked by the presence of neutrophils, with the severity progressing from cryptitis and crypt abscesses to erosions and ulcers. The diagnostic value of eosinophils is limited due to the lack of a specified threshold. 18
A chronic feature of UC is characterised by widespread crypt architectural distortion, along with crypt branching and atrophy. 19 Other features include hypertrophy of the muscularis mucosae and submucosal fibrosis.20–22 Furthermore, Paneth cell metaplasia and goblet cell depletion, sometimes referred to as ilealisation, also reflect sustained mucosal injury.23,24 Histological remission is characterised by the absence of mucosal erosion or ulcer, accompanied by the absence of neutrophilic infiltration and paucity of mononuclear cells.20,25 Even with resolution of active inflammation, features of chronicity, such as crypt irregularity, may persist.19,26 The presence of both histological activity and chronic changes is associated with a significant risk of relapse.27–31 In a comprehensive meta-analysis, individual histological features, basal plasmacytosis (odds ratio [OR]: 1.95; 95% confidence interval [CI]: 1.10–3.46), neutrophilic infiltrations (OR: 2.30; 95% CI: 1.14–4.63), mucin depletion (OR: 2.05; 95% CI: 1.12–3.73) and crypt architectural irregularities (OR: 2.22; 95% CI: 1.30–3.80) were predictors of disease relapse. 27
Domain 2: Histological features of CD
CD is characterised by transmural inflammation and skipped bowel segments that can occur anywhere along the gastrointestinal tract, with the terminal ileum being the most frequently affected location.32,33 Neutrophilic infiltration in microscopic view indicates active disease, while focal crypt architectural distortion and chronic inflammatory cellular infiltration represent disease chronicity. Small, poorly formed, non-caseating and non-necrotic granulomas formed by epithelioid histiocytes may be observed.34,35 However, this is not specific and can be found in other conditions such as sarcoidosis and tuberculosis. 36 Eosinophilic infiltration does not necessarily support CD histological activity due to its lack of specificity. 37 The absence of a single pathognomonic feature makes diagnosing CD a complex challenge. The term Inflammatory Bowel Disease Unclassified (IBD-U) may be used when endoscopy shows evidence of colitis but does not clearly demonstrate specific features of either CD or UC. The use of biomarker signatures might help differentiate CD from UC. 38
Defining histological remission after therapy involves improvement of features, including the absence of ulceration, erosion and mucosal neutrophil.39–41 However, the absence of histologic inflammation does not exclude the possibility of active disease due to the patchy nature of disease and potential sampling error. Currently, there is insufficient evidence to indicate that histological chronic features can predict relapse in CD, primarily due to the limited number of studies investigating this prognostic value. Histological remission, however, can predict decreased risk of clinical relapse.42,43
Domain 3: Standardised histopathology reporting
Pathologists are essential team members to help diagnose IBD, evaluate disease extent and identify infection, dysplasia and other complications. A growing role is in the objective evaluation of IBD inflammatory activity. Histopathological reports generated by the pathologist should be structured and standardised to include a validated histological scoring system.44–46 Histological evaluation and interpretation rely on the clinical context and information provided by clinicians. The pathology request form should, therefore, include clear and relevant clinical information, including the indication of biopsy, location in the bowel, the nature and duration of IBD, endoscopic inflammatory activity (with the formal endoscopy report included if available), suspicion for infections, biochemical inflammatory activity, prior dysplasia and any visible dysplasia. We recommend that IBD pathology be interpreted by pathologists who sub-specialise in gastroenterology to generate expertise, reduce intra- and inter-observer disagreement.
Histopathological reports should include both qualitative and quantitative values. Key elements to describe include the extent of disease location, crypt architecture, type, pattern, severity of inflammation, identification of complications, other relevant findings, comparison with previous histological reports and the adequacy of biopsy material.25,47,48 Additional information of relevance includes the use of special stains for the diagnosis of infection and dysplasia, which are complications of IBD. 49 The terms ‘non-specific colitis’ and ‘indeterminate colitis’ should be avoided where possible.
Domain 4: Histological indices for measuring disease activity in IBD
Several histological indices have been developed to assess disease activity and promote data harmonisation in IBD. Some have undergone at least partial validation and are now employed in both clinical trials and routine practice.50–54 Most indices that were used in UC drug trials share common elements, including degree of architectural change, chronic inflammatory cell infiltration in the lamina propria, neutrophil infiltration in the lamina propria and epithelium, as well as epithelial damages. Variations between indices have been observed in the weighting and endpoint values of the elements included in the indices. The panel recommended utilising validated indices established for clinical ease and easy interpretability, including the GS, 55 RHI 56 and NI. 57 These indices showed strong correlation with each other, with high intra-observer reproducibility and interobserver agreement.52,58–61 Table 2 demonstrates standard cutoff scores for histological response and remission for each index.
Histological endpoints of commonly used scoring systems.
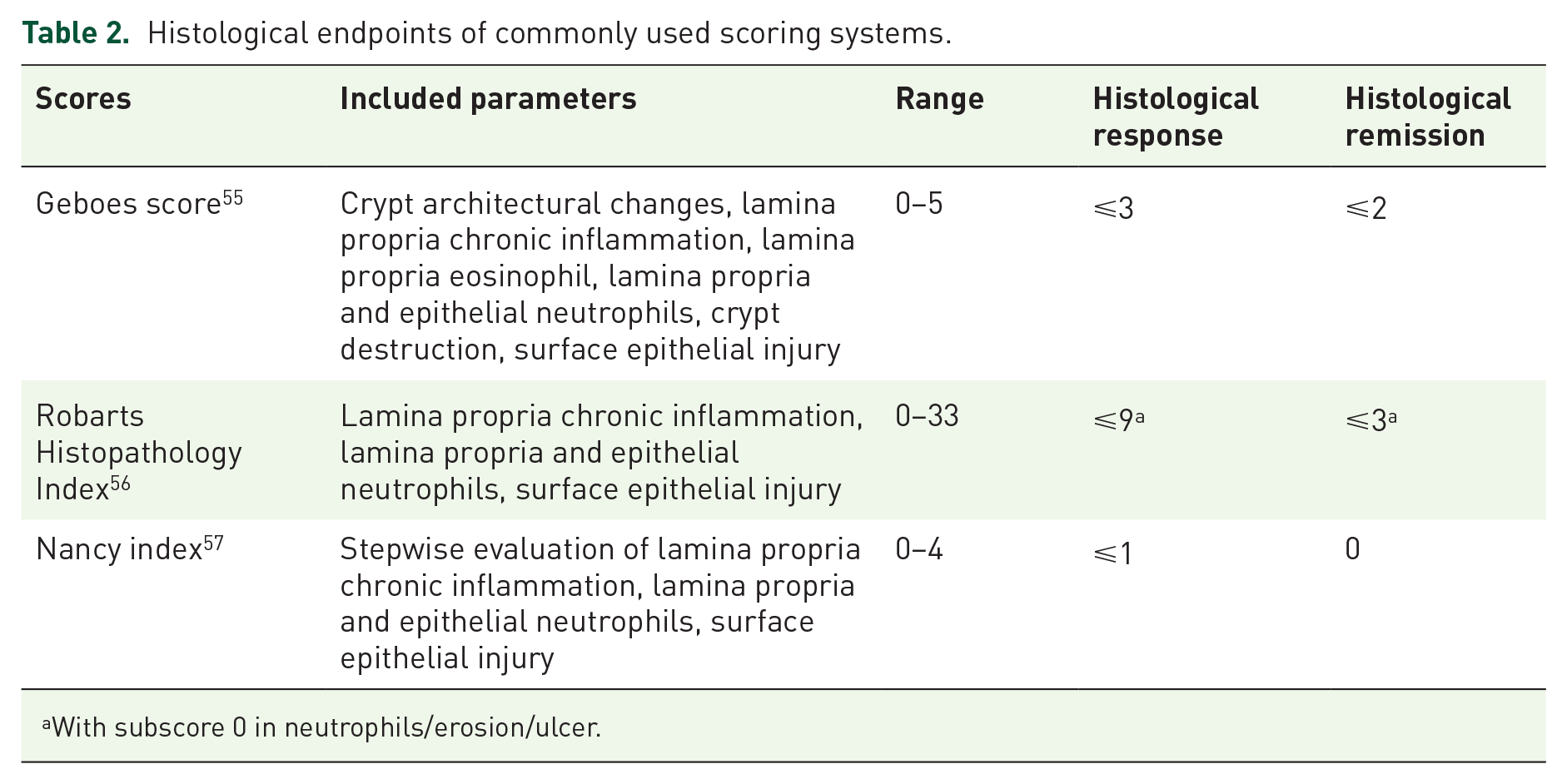
With subscore 0 in neutrophils/erosion/ulcer.
The panel rated the NI as the most preferred index for clinical practice in UC. This index is graded on a scale of 0–4, with a higher score indicating greater histological inflammation. A score of 0 indicates no to mild increase in chronic inflammatory cell infiltrates, signifying histological remission; 1 indicates a moderate increase in chronic inflammatory cell infiltrate; 2–3 indicates moderate to severe degrees of neutrophilic infiltration and 4 indicates the presence of erosion or ulceration. The index is widely recommended by several international IBD working groups due to its comprehensive validation, robust correlation with other scores and simplicity.53,62,63 Alternative validated indices with a finer scale may be more suitable for research settings, where smaller incremental changes need to be measured. For these reasons and the fact that the index has been adopted by major IBD centres, the APAGE agreed to recommend the adoption of the NI.
In CD, histological platforms might be adopted in a similar way to UC.40,64,65 However, they are not currently well-defined and await further validation.
Domain 5: Endoscopic biopsies in IBD
Indications for endoscopic mucosal biopsies for IBD include making the diagnosis, exclusion of alternative diagnoses or infectious complications, diagnosing discrete lesions, visible and invisible dysplasia and scoring of histological activity.11,25,47,66–68 We recommend implementation of a standardised protocol of segmental mucosal biopsies during the index colonoscopy, which are collected in separate jars. Not only will this assist in making the initial diagnosis of IBD and help exclude alternative pathologies, but it may also help determine histological activity changes over time. 69 Identifying the maximal histological disease extent prior to commencing effective treatment might help assess colitis-associated colorectal dysplasia in the future, with histological extent potentially greater than the endoscopic extent. For grading of active disease severity, biopsies should be taken from the most severely affected area of each colonic segment.25,70–72 In the presence of an ulcer, biopsies should be taken from the ulcer edge. Standard biopsy forceps are suitable for obtaining two tissue samples with a single pass from each ileocolonic segment.47,73 On follow-up colonoscopy, in addition to sampling from the most-inflamed areas, biopsies should ideally target locations closely correlating to the baseline procedure to detect changes in disease activity.74–76 Biopsies are recommended during endoscopic remission to assess for histological activity and detect for invisible dysplasia.
Domain 6: Histological processing of endoscopic mucosal biopsies
Generally, H&E stain is sufficient for evaluation of disease activity. However, special stains and IHC should be considered based on the clinical context and at the request of the physician. For refractory colitis or on detection of cytopathic inclusion bodies, cytomegalovirus IHC is recommended. Ziehl-Neelsen stain may be requested for suspicion of tuberculosis.20,77 Additional microbiological testing, including PCR testing or immunofluorescence stains, can assist in the diagnosis of infectious complications.
Domain 7: Histological disease activity assessment in IBD
Long-term treatment goals of IBD include endoscopic remission, which is associated with improved long-term clinical outcomes.4,78,79 Histological remission and combined histo-endoscopic remission appear to be additional factors associated with improved clinical outcomes, and may be even more specific than endoscopic remission alone. Performing a histological assessment during endoscopy will ensure more comprehensive disease activity evaluation. 27 Histological activity is believed to drive the development of colitis-associated dysplasia, further strengthening the goal of eventual histological remission.27,39,80–82 A meta-analysis of 10 studies involving patients with UC revealed a 63% reduction in the risk of clinical relapse among patients achieving histological remission compared to those with persistent histologic activity (relative risk; [RR]: 0.37; 95% CI: 0.24–0.56). Patients who achieve histological remission demonstrated a decrease in the need for corticosteroids (OR: 0.26; 95% CI: 0.11–0.56; p = 0.002). 83 Active histological disease is associated with a higher need for surgery (OR: 3.14; 95% CI: 1.53–6.45; p < 0.01), and hospitalisation (OR: 2.52; 95% CI: 1.59–4.00; p < 0.01). 84 In UC, NI score of 0 is associated with improved clinical outcomes, with studies showing a reduced risk of hospitalisation and clinical relapse.63,85–89 Therefore, an NI of 0 may offer significant benefits. These findings underscore the importance of maintaining adequate treatment in the presence of ongoing histological inflammation.
While evidence for CD is more limited, one study highlighted the significance of histological remission in ileal CD (as opposed to endoscopic healing), which was associated with a reduced risk of clinical relapse (hazard ratio (HR): 2.05; 95% CI: 1.07–3.94; p = 0.031), medication escalation (HR: 2.17; 95% CI: 1.2–3.96; p = 0.011) or corticosteroid requirement (HR: 2.44; 95% CI: 1.17–5.09; p = 0.018). 42 The definition, sensitivity and specificity of histological remission in CD remain to be determined. In particular, the incremental gain of histological remission in CD beyond endoscopic remission or transmural remission using prospectively acquired data remains an area for future research.
Domain 8: Combined histological and endoscopic outcome assessments in IBD
Observational and controlled studies have demonstrated that combined endoscopic- and histological-remission is associated with a lower risk of relapse in IBD patients, compared to achieving either endoscopic or histologic remission alone.3,88,90 A meta-analysis of 17 studies, incorporating data of 2608 UC patients in clinical remission, revealed that patients achieving a Mayo endoscopic subscore (MES) of 0 had a 52% lower risk of relapse compared to those achieving MES 1. Moreover, even achieving MES 0, patients with histological remission had a lower risk of clinical relapse than those with persistent histological activity. 3 Additionally, patients with histological remission demonstrated favourable outcomes, including lower rates of hospitalisation and cancer risk. 91 Combined histological and endoscopic remission, therefore, has been implemented in several clinical trial settings.92–96 Upadacitinib maintenance trial in moderate-to-severe UC defined histologic endoscopic mucosal improvement as MES 0–1 and GS ⩽ 3.1 and histologic endoscopic mucosal remission as MES 0 and GS < 2. 94 These outcomes could be achieved even during induction therapy and were associated with a greater likelihood of improved clinical outcomes. 97 We would recommend documentation of histological and endoscopic outcomes in standard clinical practice, and encourage standardisation of these definitions in clinical trials. Mucosal healing in UC was formerly defined by a MES 0–1, and the term should be replaced with endoscopic improvement.98,99
Domain 9: Gastroenterology pathologist as member of an IBD MDT
The management of IBD should involve a gastroenterology pathologist as a member of the multidisciplinary team (MDT). 100 The MDT framework for interdisciplinary discussions to diagnose and treat patients with complex conditions should be standard. The gastroenterology pathologists would work alongside the gastroenterologist, colorectal surgeon, gastroenterology radiologist, nurse, dietician, psychologist, pharmacist and support personnel.100–103 The incorporation of a dedicated pathologist in MDTs resulted in changes in the diagnosis or management in 51% of cases discussed, and 70% of patients achieved clinical response or remission following the management plan instituted during the MDT meeting. 104 Beyond IBD case management, a dedicated MDT promotes collaboration, positive work culture, professional education, the adoption of best practices and audit or research opportunities.
Domain 10: The role of AI in histological assessment of IBD
AI encompasses machines that possess cognitive functions resembling those of humans, which might be applicable to the field of IBD. 105 These problem-solving and learning capabilities include aiding disease diagnosis, risk prediction, assessing disease severity and predicting treatment responses and clinical outcomes. In distinguishing between UC and CD, one deep learning model trained on 29,414 endoscopic images from 1358 patients achieved high diagnostic accuracy, with per-patient and per-lesion accuracies of 96.2% and 91.6%, respectively. The model outperformed endoscopists and demonstrated excellent discriminatory performance. 106 Furthermore, in endoscopic evaluation either by colonoscopy or capsule endoscopy, retrospective and prospective image datasets have guided the rapid and accurate grading of inflammatory activity.107–113 With receiver operator characteristic area under the curve of 0.86, AI demonstrated a high degree of accuracy in diagnosing endoscopic remission in UC as defined by MES of 0. 111 Another study reported an accuracy of 0.91 in classifying CD ulcer severity using capsule endoscopy. 107 A computer-aided diagnosis system was able to detect histologic activity during endocytoscopy in UC with sensitivity of 74% (95% CI: 65–81) and specificity of 97% (95% CI: 95–99) when compared to histology of biopsy. 114
Machine learning is applicable to histology interpretation in IBD, especially in UC.114–120 Using 535 digitalised UC biopsy images with the NI as reference, AI modelling could determine histological activity with a sensitivity of 89% (95% CI: 81–94) and specificity of 79% (95% CI: 73–83). 115 The PICaSSO histologic remission index, based on the detection of neutrophils by deep learning, could identify UC activity with a sensitivity of 78% and specificity of 91.7% and correlated with endoscopic scores and clinical outcomes. 116 AI yielded higher diagnostic capability versus endoscopists in the detection and differentiation of dysplasia, with sensitivity and specificity of 72.5% and 82.9%, respectively. 121 The role for AI in the detection of colitis-associated dysplasia is of high potential benefit, especially given the potential in diagnosing commonly missed flat dysplastic lesions by conventional white-light endoscopy. However, cost effectiveness and the incremental gain in value to pathologists need to be evaluated at a local level. Future prospective trials are necessary to validate the benefit of AI on endoscopic and histological assessment.
The integration of AI into IBD practice faces challenges, including the need to digitise histological slides, the availability and cost of AI devices, medicolegal concerns over accuracy and misdiagnosis, patient acceptance and the requirement for site-specific regulatory approvals. However, these barriers can be addressed through cost-sharing collaborations between institutions and industry, clinician and patient education programmes to build trust and understanding and partnerships with regulatory bodies to streamline approvals and ensure compliance. Importantly, AI is not intended to replace pathologists but to enhance their capacity and provide complementary support.
Statements for which consensus agreement were not achieved
The following statement did not achieve consensus.
Level of agreement: (a) 6%, (b) 0%, (c) 12%, (d) 76%, (e) 6% (EL: IV. Grade: D).
This statement did not achieve consensus agreement and was considered a major knowledge gap. Currently, there is insufficient support for switching effective therapies in the presence of histological activity alone without endoscopic activity. Whilst optimisation, adherence assessment and dose-increase of lower-efficacy treatments were deemed acceptable, escalation to advanced therapy in resource-poor countries was deemed impractical. Apart from the additional cost, the risk of treatment-induced toxicity needs to be considered. Most delegates agreed that histological remission might lag behind endoscopic remission, indicating that further time might eventually lead to histological remission. A prospective study is needed to further validate the potential benefits of treatment intensification based on histological activity alone. The Consensus Working Group recommends repeating histological evaluation at a later time to determine if delayed histological remission might occur, identifying non-adherence, and institution of simple dose-optimisation strategies.
Discussion
As histological outcomes become an important therapeutic endpoint in the management of IBD, 4 this consensus provides practical and relevant recommendations for histopathological assessment, with particular focus on clinical practice across the Asia Pacific region. We propose unified histological definitions for IBD, where neutrophilic infiltration signifies active inflammation and chronic changes, such as crypt architectural distortion, indicates chronicity. Clarifying these features may enhance the understanding and application of histology among gastroenterologists. Effective communication between gastroenterologists and pathologists is strongly encouraged to ensure that pathology reports are clinically relevant and actionable. The use of validated histological scoring indices, particularly the NI for UC, is recommended to promote consistency and shared interpretation. Segmental biopsies using standard forceps are advised to accurately assess disease activity in both conditions, and H&E staining remains the standard for histological evaluation.
While the prognostic value of achieving histological remission is well supported in UC, evidence in CD remains limited and warrants further investigation. There is an increasing trend towards using combined endoscopic-histological assessment as a routine metric of disease activity, which may soon become part of standard practice. The inclusion of gastrointestinal pathologists as part of the MDT is strongly supported to enhance diagnostic precision and interdisciplinary communication. Lastly, the role of AI in IBD is rapidly evolving. AI tools have shown high accuracy in evaluating histological activity; however, these technologies remain adjunctive and cannot replace the expertise of trained pathologists.
A key strength of this work is that it represents the first consensus on IBD histology developed specifically for the Asia Pacific region, with input from a diverse panel of gastroenterologists and pathologists across multiple settings. The use of a modified Delphi method ensures methodological rigour and transparency. Importantly, the involvement of gastrointestinal pathologists throughout the consensus process enhanced mutual understanding. The statements address both well-established practices and emerging innovations, including AI. However, several limitations should be noted. First, the available evidence regarding the prognostic and therapeutic roles of histology in IBD remains limited in this region. Second, although experts from multiple countries participated, each representative may not fully reflect the diversity of practice within their respective countries. Third, variability in local healthcare infrastructure, expertise and access to specialised pathology or AI-based tools may affect implementation and generalisability of the recommendations.
Conclusion
Histological assessment is an important component in the management of IBD. This consensus will help gastroenterologists and pathologists unify practices related to tissue acquisition, processing, histological descriptions and the use of histological indices, thereby standardising IBD care across the Asia Pacific region. This harmonisation may foster interdisciplinary research and enable cross-national collaborations. Incorporating AI into histological evaluation is potentially beneficial in streamlining diagnostic workflows by quickly identifying key histological features and enhancing precision care for patients with IBD.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251363703 – Supplemental material for Asia Pacific association of gastroenterology consensus statements on histopathological evaluation of inflammatory bowel diseases
Supplemental material, sj-docx-1-tag-10.1177_17562848251363703 for Asia Pacific association of gastroenterology consensus statements on histopathological evaluation of inflammatory bowel diseases by Rupert W. Leong, Thanaboon Chaemsupaphan, Huiyu Lin, Wee Chian Lim, Choon Jin Ooi, John D. Chetwood, Ren Mao, Hsin Yun Wu, Shu Chen Wei, Govind Makharia, Vineet Ahuja, Rupa Banerjee, Raja Atreya, Julajak Limsrivilai, Satimai Aniwan, Pises Pisespongsa, Ida Hilmi, Raja Affendi Raja Ali, Wai Keung Leung, Siew C. Ng, Byong Duk Ye, Taku Kobayashi, Katsuyoshi Matsuoka, Cora Chau, Anapat Sanpavat, Chia-Tung Shun, Pavitratha Puspanathan, Richard B. Gearry, Silvio Danese, Christopher Ma, Aviv Pudipeddi and Sudarshan Paramsothy in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251363703 – Supplemental material for Asia Pacific association of gastroenterology consensus statements on histopathological evaluation of inflammatory bowel diseases
Supplemental material, sj-docx-2-tag-10.1177_17562848251363703 for Asia Pacific association of gastroenterology consensus statements on histopathological evaluation of inflammatory bowel diseases by Rupert W. Leong, Thanaboon Chaemsupaphan, Huiyu Lin, Wee Chian Lim, Choon Jin Ooi, John D. Chetwood, Ren Mao, Hsin Yun Wu, Shu Chen Wei, Govind Makharia, Vineet Ahuja, Rupa Banerjee, Raja Atreya, Julajak Limsrivilai, Satimai Aniwan, Pises Pisespongsa, Ida Hilmi, Raja Affendi Raja Ali, Wai Keung Leung, Siew C. Ng, Byong Duk Ye, Taku Kobayashi, Katsuyoshi Matsuoka, Cora Chau, Anapat Sanpavat, Chia-Tung Shun, Pavitratha Puspanathan, Richard B. Gearry, Silvio Danese, Christopher Ma, Aviv Pudipeddi and Sudarshan Paramsothy in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We gratefully acknowledge the Asia Pacific Association of Gastroenterology (APAGE) for their generous support through the project grant, which made this research possible.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
