Abstract
Background:
Recent changes in the acid-related disease market, particularly the emergence of potassium-competitive acid blockers (P-CABs), require an updated analysis of trends in gastric acid suppressant utilization.
Objectives:
As the use of P-CAB expands globally, a cross-sectional study is needed to assess its association with potential adverse events from a pharmacovigilance perspective.
Design:
Cross-sectional study.
Methods:
We analyzed gastric acid suppressant use at prescription and patient levels using the Health Insurance Review and Assessment National Patient Sample database (2014–2020). Prescription-level analysis included the annual prescriptions for histamine-2 receptor antagonists (H2RAs), proton pump inhibitors (PPIs), and P-CABs, and proportion of visits by acid-related indications. Patient-level analysis examined annual trends in drug use and clinical characteristics. Logistic regression was used to estimate prevalence odds ratios (ORs) for potential adverse events among drug users and non-users according to the drug class.
Results:
In 2020, H2RAs were primarily prescribed for gastritis and duodenitis (64.5%), whereas PPIs (55.4%) and P-CABs (58.6%) were primarily used to treat gastroesophageal reflux disease. H2RA prescriptions declined in 2019, whereas the use of P-CAB increased following the approval of tegoprazan in 2018. Patients aged 40–64 years and female users were predominant across all classes of gastric acid suppressants. P-CAB use was significantly associated with osteoporosis (OR = 1.17; 95% confidence interval (CI): 1.13–1.20), pneumonia (OR = 1.65, 95% CI: 1.55–1.75), and acute kidney injury (OR = 1.39, 95% CI: 1.20–1.61).
Conclusion:
P-CABs have reshaped the prescription landscape of gastric acid suppressants. Further longitudinal cohort studies are required to evaluate long-term safety in routine clinical practice.
Plain language summary
•
○ Gastroesophageal reflux disease (GERD) and peptic ulcers are commonly treated with acid-suppressing medications like histamine-2 receptor antagonists (H2RAs) and proton pump inhibitors (PPIs). Recently, a new class of acid blockers called potassium-competitive acid blockers (P-CABs) has become available. P-CABs inhibit gastric acid secretion by reversibly and competitively binding to the potassium site of the H⁺/K⁺-ATPase pump, providing rapid acid suppression. Although P-CABs are increasingly prescribed, there is limited real-world information about their overall use and how they compare to older medications. This study was conducted to understand how P-CABs have changed prescription patterns and to explore potential safety considerations for different acid-suppressing drugs.
•
○ Researchers examined nationwide health data from South Korea between 2014 and 2020, focusing on people treated for various acid-related disorders. We looked at how often different types of acid-suppressing medications were prescribed each year. We also assessed demographics, comorbidities, concomitant medications, and investigated whether certain events were associated with using these medications.
•
○ Over the study period, P-CABs became more widely used while the prescriptions for some older drugs decreased. Many patients receiving gastric acid suppressants had other common conditions such as hypertension or dyslipidemia. The study also noted that while different acid-suppressants may be linked to certain adverse events, more detailed research is needed to confirm these findings. The overall trend shows that P-CABs are becoming a more common choice in treating acid-related diseases at least in South Korea.
•
○ These results highlight a shift in treatment practices. Because P-CABs are relatively new, researchers and healthcare professionals should continue to investigate their long-term safety and effectiveness.
Keywords
Introduction
Gastric acid suppressants, such as histamine-2 receptor antagonists (H2RAs) and proton pump inhibitors (PPIs), are widely used to treat gastrointestinal disorders such as gastroesophageal reflux disease (GERD) and peptic ulcer disease. H2RAs and PPIs are among the most commonly prescribed medications because of the high prevalence of these conditions and the favorable safety profiles of gastric acid suppressants.1–3 However, concerns regarding their appropriate use have persisted, prompting numerous studies on their prescribing patterns and trends.4–6 Nationwide studies have provided valuable insights into the rational use of gastric acid suppressants and supported evidence-based policymaking for decision-makers.
Significant changes have recently occurred in the acid-related disease market. First, potassium-competitive acid blockers (P-CABs) have emerged as a novel class of gastric acid suppressants for GERD, competitively inhibiting the potassium-binding site of the H+/K+-ATPase, unlike PPIs.7,8 Vonoprazan was the first P-CAB approved for GERD, receiving approval in Japan in 2014 and in the United States in 2023.9,10 Although vonoprazan was not introduced in Korea, tegoprazan was launched in 2019. 11 Based on its superior clinical efficacy including reduced food effect, faster onset of action, and sustained acid suppression,12,13 P-CAB use has rapidly increased and been incorporated into updated treatment guidelines in Japan and Korea.14,15 Second, ranitidine, a second-generation H2RA, was withdrawn in 2020 following the detection of the probable carcinogen N-nitrosodimethylamine. 16 Given these dynamic shifts, an up-to-date study on the evolving utilization pattern of gastric acid suppressants and physicians’ prescribing preferences is warranted.
Furthermore, although the global use of P-CABs is expected to increase, safety data remain limited owing to insufficient clinical experience. As a potential alternative to PPIs, the safety profile of P-CABs should be evaluated and compared with that of PPIs. Previous studies have associated PPIs with various adverse events, including an increased risk of fractures, gastric cancer, pneumonia, and chronic kidney disease.17–20 A cross-sectional study investigating the associations between P-CAB drugs and potential adverse events is needed to generate hypotheses for further pharmacoepidemiologic research and systematically assess potential safety concerns related to P-CABs.
Therefore, the aim of this study was to analyze the utilization trends of gastric acid suppressants at both the prescription and patient levels from 2014 to 2020. Additionally, we examined the characteristics of patients prescribed H2RAs, PPIs, or P-CABs each year and assessed the association between these drugs and specific adverse events in 2020, the most recent year in our dataset.
Methods
Data source
This study utilized Health Insurance Review and Assessment service-National Patient Sample (HIRA-NPS) data from 2014 to 2020. The HIRA-NPS is a nationally representative claims database, consisting of a 2%–3% sample (approximately 1,000,000 individuals) selected annually from 98% of the Korean population using sex- (two strata) and age-stratified (16 strata) random sampling. 21 The HIRA database provides comprehensive information such as patient demographics (sex, age group, and type of insurance), diagnoses, and prescription records including drug ingredients, prescription duration, and dosage. 21
Drugs of interest
Gastric acid suppressants used in this study were H2RAs (cimetidine, ranitidine, famotidine, nizatidine, roxatidine, and lafutidine), PPIs (omeprazole, pantoprazole, lansoprazole, rabeprazole, esomeprazole, ilaprazole, dexlansoprazole, and s-pantoprazole), and P-CABs (tegoprazan and revaprazan). Only oral and injectable formulations were analyzed, and drug identification was based on the active ingredient codes issued by the Ministry of Health and Welfare of South Korea (Table S1).
Prescription-level utilization analysis
We identified prescriptions of gastric acid suppressants from the database for the prescription-level analysis. We restricted the prescriptions to those with diagnostic codes for acid-related conditions to exclude off-label use. These included esophagitis (K20), GERD (K21), other diseases of the esophagus (K22 and K23), peptic ulcers (K25, K26, K27, and K28), and gastritis and duodenitis (K29). The diagnostic codes were based on the 7th Korean Standard Classification of Diseases, corresponding to the 10th revision of the International Classification of Diseases. We examined the total number of visits for gastric acid suppressant prescriptions in this analysis, and the proportions of visits by patient age group, sex, and annual acid-related diagnoses were calculated.
Patient-level utilization analysis
Patients who received at least one prescription of H2RAs, PPIs, or P-CABs per year were identified from the prescription-level data. We examined annual records for hypertension, diabetes, hyperlipidemia, and chronic obstructive pulmonary disease to assess the prevalence of comorbidities. We also analyzed various co-medications including glucocorticoids, non-steroidal anti-inflammatory drugs (NSAIDs), thiazide diuretics, renin-angiotensin-aldosterone system inhibitors, calcium channel blockers, antipsychotics, antifungals, antibiotics, benzodiazepines, and anticoagulants.
Fractures, osteoporosis, pneumonia, Clostridioides difficile infection, enteric infections, chronic kidney disease, acute kidney injury, acute interstitial nephritis, myocardial infarction, stroke, pancreatic cancer, gastric cancer, colon cancer, inflammatory bowel disease, hypomagnesemia, and gastric mucosal atrophy were defined as potential adverse events. The adverse events of interest were selected based on frequently studied safety issues associated with PPIs, as identified in a previous study that reviewed 42 systematic reviews. 22 The corresponding diagnostic codes are listed in Table S2.
Patients who were prescribed gastric acid suppressants were categorized annually based on the class of medications used in the patient-level analysis. Demographic characteristics (age, sex, and type of insurance), comorbidities, co-medications, and the prevalence of potential adverse events were evaluated by drug class.
Associations between drug use and potential adverse events
Given the increased use of P-CABs in 2020, the prevalence odds ratio (OR) was estimated using only 2020 data. Because the dataset was sampled annually, patient-level identification across the entire study period was not feasible. Therefore, even in the estimation of prevalence ORs using data from 2020, the temporal relationship between drug use and adverse events could not be clearly established. Logistic regression analyses were performed to estimate both crude and adjusted ORs with 95% confidence intervals (CIs) to assess the association between drug use and prevalence of potential adverse events by drug class. Statistical analyses were conducted using the SAS Enterprise Guide 7.1 (SAS Institute Inc., Cary, NC, USA). Data visualization was performed using Python with Matplotlib version 3.4.3. 23 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Table S3). 24
Ethical statement
The Institutional Review Board (IRB) of Jeonbuk National University exempted this study from IRB review because the data were anonymized and retrospectively generated, precluding individual identification (JBNU202404019-UE002).
Results
Prescription trends of gastric acid suppressants between 2014 and 2020
At the prescription level, H2RAs were primarily prescribed for gastritis and duodenitis (78.5%), whereas PPIs (88.4%) and P-CABs (97.6%) were most commonly prescribed for GERD in 2020 (Table 1). The baseline characteristics of the prescriptions according to the drug class per year are shown in Tables S4–S6.
Baseline characteristics of prescriptions, including gastric acid suppressants issued in 2020.

The total number of prescriptions by ICD-10 code exceeds the overall number of prescriptions because multiple codes can be assigned to a single prescription.
GERD, gastroesophageal reflux disease; H2RA, histamine-2 receptor antagonist; ICD, International Classification of Diseases; P-CAB, potassium-competitive acid blocker; PPI, proton pump inhibitor.
Figure 1(a)–(d) presents the annual prescription trends of gastric acid suppressants in South Korea from 2014 to 2020, including class-specific patterns for H2RAs, PPIs, and P-CABs. Detailed patient-level data corresponding to gastric acid suppressant use are also provided in Figure 1(a) and Table S7. The proportion of patients with acid-related disorders who received at least one prescription of gastric acid suppressants was highest (56.2%) in 2018. Prescriptions of H2RAs consistently accounted for the largest percentage of prescriptions among gastric acid suppressants; however, the number of H2RA prescriptions decreased to 30.3% in 2020. The prescription of PPIs showed an increasing trend, rising from 15.1% in 2014 to 24.3% in 2020. The number of prescriptions for P-CABs was <1% between 2014 and 2018. However, the number of P-CAB prescriptions showed a 10-fold increase in 2019 after the approval of tegoprazan in 2018.
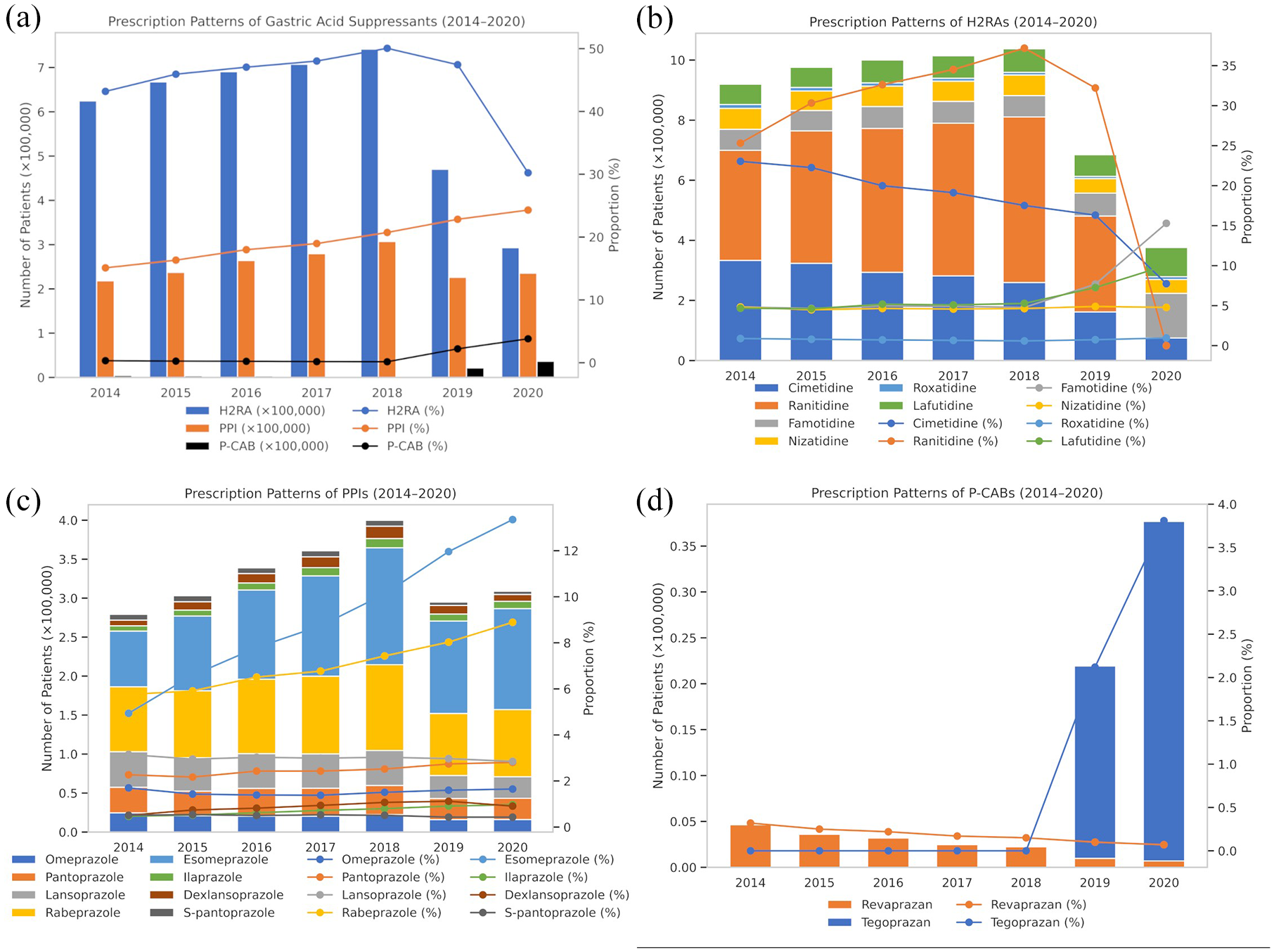
Prescription patterns of (a) gastric acid suppressants, (b) H2RAs, (c) PPIs, and (d) P-CABs from 2014 to 2020. The number of patients by ingredient within the class of gastric acid suppressants by year.
Figure 1(b) illustrates the annual prescription patterns of H2RAs. Ranitidine showed the highest use between 2014 and 2019. However, the number of patients dropped to nearly 0% in 2020. After ranitidine, cimetidine was the next most frequently used H2RA. The number of patients prescribed cimetidine in 2020 (75,010 (7.74%)) was reduced to one-third of that in 2014 (333,241 (23.04%)). Famotidine maintained a relatively stable level of use from 2014 to 2019. However, the number of patients prescribed famotidine increased in 2020, whereas prescriptions for both ranitidine and cimetidine decreased. The most commonly used PPIs were rabeprazole and esomeprazole, and the proportion of these ingredients increased from 2014 to 2020 (Figure 1(c)). The proportion of patients prescribed with other PPIs remained relatively consistent throughout the study period. Tegoprazan was introduced in 2019 and has been gradually increasing, with prescriptions increasing from 20,965 (2.1%) in 2019 to 36,920 (3.8%) in 2020 (Figure 1(d)). Revaprazan was used exclusively from 2014 to 2018, with the number of patients decreasing from 4635 (0.3%) in 2014 to 693 (0.07%) in 2020.
Baseline characteristics of the study population
Table 2 presents the baseline characteristics of the patients prescribed H2RAs, PPIs, or P-CABs at least once in 2020. For all three drug classes, the highest proportion of patients were aged between 40 and 64 years. Males constituted 41%–45% of drug users. Hyperlipidemia was the most prevalent comorbidity in each drug class (38.2% for H2RA, 47.6% for PPI, and 49.2% for P-CAB). NSAIDs, antibiotics, and glucocorticoids were the most commonly prescribed co-medications in all groups. Fractures and osteoporosis were the most frequently observed potential adverse events. The baseline characteristics of the drug users for each year are presented in Tables S8–S10. The process of identifying prescriptions and patients who used gastric acid suppressants from the HIRA-NPS database is presented in Figure S1.
Baseline characteristics of patients prescribed gastric acid suppressants at least once in 2020.

C. difficile, Clostridioides difficile; COPD, chronic obstructive pulmonary disease; H2RA, histamine-2 receptor antagonists; NSAIDs, non-steroidal anti-inflammatory drugs; P-CAB, potassium-competitive acid blocker; PPI, proton pump inhibitor; RAAS, renin-angiotensin-aldosterone system.
Prevalence OR of potential adverse events
Although the direction of significance varied among the potential adverse events analyzed, most events, including fractures, osteoporosis, and gastric mucosal atrophy, were significantly associated with the use of gastric acid suppressants (Table 3). Osteoporosis, gastric mucosal atrophy, and fractures were the most frequent adverse events associated with the use of these medications. The use of gastric acid suppressants was significantly associated with osteoporosis, with adjusted ORs of 1.07 (95% CI: 1.05–1.08) for H2RAs, 1.44 (95% CI: 1.42–1.45) for PPIs, and 1.17 (95% CI: 1.13–1.20) for P-CABs. The adjusted OR for gastric mucosal atrophy was notably high at 10.11 (95% CI: 9.98–10.24) for PPI use. Conversely, adjusted ORs for fractures were <1 across all classes of gastric acid suppressants, with 0.84 (95% CI: 0.83–0.86) for H2RAs, 0.84 (95% CI: 0.83–0.86) for PPIs, and 0.55 (95% CI: 0.51–0.58) for P-CABs.
Prevalence ORs of potential adverse events associated with gastric acid suppressants in 2020 data.
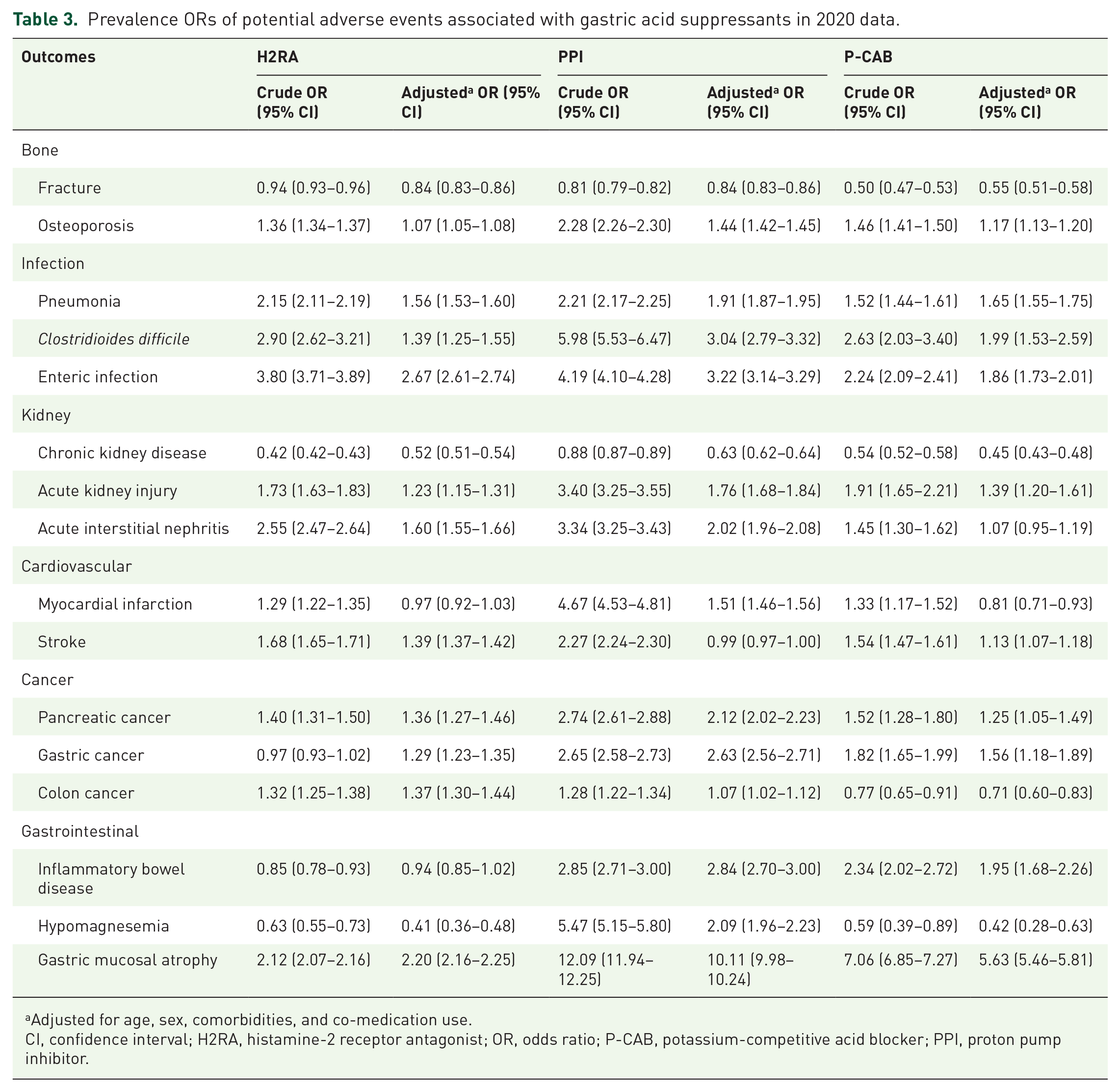
Adjusted for age, sex, comorbidities, and co-medication use.
CI, confidence interval; H2RA, histamine-2 receptor antagonist; OR, odds ratio; P-CAB, potassium-competitive acid blocker; PPI, proton pump inhibitor.
Discussion
Drugs for peptic ulcers and GERD have historically been limited to H2RAs and PPIs; however, more potent P-CABs have recently emerged. The prescription of P-CABs has steadily increased, with approval from multiple countries, including approval by the United States Food and Drug Administration (FDA), in July 2024. Clinical guidelines recommend using P-CABs for Helicobacter pylori eradication, suggesting that their prescription is likely to continue rising in the future.25,26 Our study, utilizing the HIRA-NPS database, is the first to analyze the use of gastric acid suppressants before and after the introduction of P-CABs. Our study focused on the baseline characteristics of the prescriptions and patients using different classes of gastric acid suppressants, trends in drug use, and the association between drug use and potential adverse events.
One notable finding in our study was the change in the number of patients who received gastric acid suppressants from 2018 to 2019. As shown in Figure 1(a), the number of patients who used both H2RAs and PPIs decreased from 2018 to 2019, whereas the number of patients who used P-CABs increased. This phenomenon appeared to be associated with the approval of tegoprazan in South Korea in July 2018, which led to an increase in the number of patients who started using tegoprazan in 2019. Vonoprazan was introduced in Japan in 2015 and its prescription increased noticeably from 2016. 5 The introduction of vonoprazan did not affect the overall prescription volume of other PPIs in Japan. 5 However, a notable decrease in the number of patients prescribed H2RAs was observed from 2018 to 2019 in Korea. In 2014, the two most commonly used H2RA ingredients were cimetidine (n = 333,241) and ranitidine (n = 366,326). Although ranitidine was used by 319,273 patients in 2019, its use decreased sharply by 2020. This sharp decline was related to the discontinuation of ranitidine production in 2019. 27 As a result, famotidine levels showed an increasing trend from 70,904 patients in 2018 to 76,093 in 2019 and 148,304 in 2020. However, the overall number of H2RA users decreased. The number of patients prescribed commonly used PPIs, including rabeprazole and esomeprazole, decreased from 2018 to 2019. This decline is also suspected to have been influenced to some extent by an increase in tegoprazan use. 28
Regardless of the introduction of P-CABs, most patients receiving prescriptions for gastric acid suppressants were aged 40–64 years. A slightly higher proportion of females was consistently observed across all years. This suggested that our analysis of comorbidity and co-medication patterns may be more representative of the 40–64 age group and the female population. Hypertension and hyperlipidemia represented a major proportion of comorbidities, suggesting that these two chronic diseases are likely to affect clinical outcomes associated with the use of gastric acid suppressants. Glucocorticoids, NSAIDs, and antibiotics were the most frequently used co-medications. Other studies also found that glucocorticoids or NSAIDs are commonly used with gastric acid suppressants.29,30 The pattern of co-medications did not change significantly after the approval of tegoprazan in 2018.
As indicated by several studies, we examined whether using gastric acid suppressants is associated with certain clinical outcomes, such as fractures, osteoporosis, and pneumonia.31–33 When examining all gastric acid suppressants over the years, fractures were frequently prevalent during the study period when examining all gastric acid suppressants. Fractures are a known adverse event that could occur with the use of gastric acid suppressants. 32 According to a previous study, there is a modest link between PPI use and fracture risk, while H2RAs do not show such an association. 34 In our study, the association between the use of gastric acid suppressants and the occurrence of fractures was low. Our analysis also showed that the use of H2RAs is not associated with an increased risk of fractures (adjusted OR 0.84 (95% CI: 0.83–0.86)). However, unlike the above study, our findings indicated that PPI use does not significantly affect fracture risk (adjusted OR 0.84 (95% CI: 0.83–0.86)). Moreover, P-CABs demonstrated a lower association with fracture risk, suggesting that P-CABs could be a safer alternative for patients concerned about fractures (adjusted OR 0.55 (95% CI: 0.51–0.58)). A randomized controlled trial comparing vonoprazan and lansoprazole reported fractures in approximately 1% of patients, 35 while no data on fractures were available from clinical trials of tegoprazan. 36 More research is needed to explore the association between fracture risk and the use of P-CABs.
The occurrence of osteoporosis, pneumonia, and gastric mucosal atrophy has also been documented in association with using gastric acid suppressants.31–33 Our analysis demonstrated a statistically significant association between using gastric acid suppressants and both osteoporosis and pneumonia. According to a study by Li et al., 31 there is a significant association between PPI use and the risk of gastric mucosal atrophy risk. This finding is consistent with our results, indicating a similar trend across all three drug classes.
Our results showed a significant association between the use of gastric acid suppressants and the risk of cancer, particularly gastric cancer. A population-based retrospective cohort study conducted in Japan demonstrated that users of vonoprazan had a significantly higher hazard ratio for gastric cancer compared to users of H2RAs. 37 Notably, prolonged duration and higher doses of vonoprazan were associated with an increased incidence of gastric cancer. 37 Alterations in the gut microbiota and the development of hypergastrinemia secondary to potent gastric acid inhibition may contribute to the observed increased risk.38,39 In our study, the use of H2RAs was associated with a lower risk of gastric cancer, which may be attributable to the less potent acid-suppressive effect of H2RAs compared to other agents.
Our study had several limitations. First, the HIRA-NPS database used in this research contains data from 2014 to 2020, allowing only 2 years of observation of the impact of P-CAB introduction. However, given the nature of the institution, which collects nationwide data and provides sample databases, the 2020 data were the most recently available at the time of analysis. The data available up to 2020 enabled us to analyze trends for approximately 2 years after the approval of tegoprazan. Second, there was ambiguity regarding the timing of disease occurrence in the prevalence analysis of the outcomes. The HIRA-NPS database, provided for each year from 2014 to 2020, does not allow for patient-level identification across years, precluding longitudinal follow-up and limiting the ability to establish temporality between drug exposure and outcome occurrence. Therefore, prevalence ORs were used to assess associations, which do not reflect incident cases or imply causality. As a result, we could not calculate cumulative treatment duration nor evaluate the relationship between duration of use and response. Future studies using longitudinal cohorts are warranted to address this gap. Third, although both revaprazan and tegoprazan were classified as P-CABs in this study, revaprazan has pharmacodynamic differences compared to tegoprazan. The inclusion of both agents under the same category may have introduced some heterogeneity; however, given the small number of revaprazan users, the impact on the results is likely minimal. In addition, unmeasured confounding factors that are not captured in the HIRA-NPS database such as lifestyle factors, use of over-the-counter medications, and utilization of non-reimbursed treatments were not accounted for in the analysis. Therefore, the possibility of residual confounding cannot be excluded, and the results should be interpreted with caution.
Despite these limitations, our study had several strengths. First, the data used in this study span 7 years from the HIRA-NPS database and cover approximately 98% of the South Korean population, which helps minimize bias due to the large sample size and allows for relatively accurate statistical analysis. 21 Additionally, to our knowledge, this study is the first nationwide study to analyze changes in the trends of gastric acid suppressant use before and after the introduction of P-CABs and generate hypotheses for further pharmacovigilance studies.
Conclusion
This study revealed a shift in the prescription patterns of gastric acid suppressants after the introduction of P-CABs. Although P-CABs appear to share some potential adverse effects with PPIs and H2RAs, further epidemiological studies using longitudinal cohorts might help establish temporal relationships. These findings provide baseline statistics on comorbidities and co-medications that may influence patient outcomes, and offer a landscape for future research on gastric acid suppressants.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251356406 – Supplemental material for Prescription trends of gastric acid suppressants and association with potential adverse events in Korea: a real-world cross-sectional study
Supplemental material, sj-docx-1-tag-10.1177_17562848251356406 for Prescription trends of gastric acid suppressants and association with potential adverse events in Korea: a real-world cross-sectional study by Min Jeong Je, Jeong Min Go, Shin Yeong Kim, Min-Gul Kim, Hankil Lee, Ha-Lim Jeon and Jae Hyun Kim in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
