Abstract
Background:
Potassium-competitive acid blockers (P-CABs) have shown potential in Helicobacter pylori (H. pylori) eradication, but the efficacy of dual therapy with P-CABs and amoxicillin remains underexplored.
Objectives:
This study evaluated the efficacy, safety, and compliance of a 14-day vonoprazan-amoxicillin (VA) dual therapy compared to a bismuth-based quadruple therapy (BQT) in treatment-naive patients.
Design:
A randomized clinical trial.
Methods:
This single-center, prospective, randomized controlled trial enrolled 250 H. pylori-positive patients from November 2022 to April 2024. Participants were randomly assigned (1:1) to receive 14-day VA dual therapy or BQT (lansoprazole, bismuth, amoxicillin, and clarithromycin). The primary outcomes were eradication rates in intention-to-treat (ITT) and per-protocol (PP) analyses. Secondary outcomes included adverse events and treatment compliance.
Results:
Eradication rates in the ITT analysis were 92.0% for the VA dual group and 88.0% for the BQT group (p = 0.292). In the PP analysis, eradication rates were 95.8% and 91.7%, respectively (p = 0.188). The VA dual group demonstrated statistically significant non-inferiority to the BQT group in both the ITT and PP analyses (both p < 0.001). The incidence of adverse events was significantly lower in the VA dual group compared to the BQT group (11.2% vs 20.8%, p = 0.038), with no severe adverse events reported. The compliance rates of both groups were 97.6%.
Conclusion:
The 14-day VA dual therapy is highly effective and well-tolerated, demonstrating non-inferiority to BQT. Given its reduced antibiotic usage and lower adverse events, it may be a viable first-line alternative for H. pylori in southern China.
Trial registration:
This trial was registered on the Chinese Clinical Trial Registry with the registration number ChiCTR2200055752.
Plain language summary
Helicobacter pylori (H. pylori) is a common bacteria that can cause stomach problems, and treating it is important to avoid complications. One treatment option involves using potassium-competitive acid blockers (P-CABs), which are a newer type of medication. This study tested a 14-day treatment combining vonoprazan (a P-CAB) and amoxicillin (called VA dual therapy) and compared it to a traditional 14-day treatment with four medications (bismuth-based quadruple therapy, or BQT). The study involved 250 patients who were newly diagnosed with H. pylori infection. The patients were randomly assigned to one of two treatment groups: the VA dual therapy group or the BQT group. The main goal of the study was to see how effective the treatments were at eliminating the bacteria. Secondary goals included checking for side effects and how well patients followed the treatment plan. Results showed that both treatments were effective at curing the infection. In the overall analysis, 92% of patients in the VA dual therapy group were cured, compared to 88% in the BQT group. In a more detailed analysis of patients who followed the treatment exactly, 95.8% of the VA dual therapy group were cured, compared to 91.7% in the BQT group. While the VA dual therapy showed slightly better results in this detailed analysis, the difference was not large enough to be considered significantly better overall. However, the VA dual therapy had fewer side effects, 11.2% of patients experienced side effects compared to 20.8% in the BQT group. There were no severe side effects in either group, and both treatments had a high patient compliance rate of 97.6%. Overall, this study suggests that the 14-day VA dual therapy is a safe, effective, and well-tolerated treatment for H. pylori infection. It may be a viable first-line treatment option, especially in southern China, because it uses fewer antibiotics and causes fewer side effects compared to the traditional BQT therapy.
Introduction
Helicobacter pylori (H. pylori) infection remains a significant global health challenge, affecting approximately 50% of the global population, with an estimated 700 million cases reported in China alone.1,2 This pathogen is widely acknowledged as the leading cause of several upper gastrointestinal disorders, including chronic active gastritis, peptic ulcers, gastric cancer, and gastric mucosa-associated lymphoid tissue lymphoma.3,4 Recognizing its carcinogenic potential, the World Health Organization’s International Agency for Research on Cancer classified H. pylori as a Group I carcinogen for gastric cancer as early as 1994. 5 Further emphasizing its clinical significance, the 2015 Kyoto Global Consensus defined H. pylori gastritis as an infectious disease and recommended eradication therapy for all infected individuals, irrespective of symptoms or disease complications. 6
Currently, the eradication of H. pylori faces challenges with declining eradication rates, primarily driven by rising antibiotic resistance (e.g., clarithromycin, metronidazole, and levofloxacin) and reduced effectiveness of acid-suppressing medications. 7 International guidelines and consensus reports recommend 14-day bismuth-based quadruple therapy (BQT) as the first-line treatment in regions where clarithromycin resistance exceeds 15%. 8 Similarly, the fifth and sixth Chinese consensus reports on H. pylori infection endorse BQT as the standard empirical therapy.9,10 While 14-day BQT demonstrates high eradication rates, ranging from 87.5% to 95.2%,11–14 it presents notable limitations. These include the unavailability of key components such as bismuth, tetracycline, and furazolidone in certain regions, as well as challenges associated with regimen complexity, significant adverse effects, poor patient compliance, and high treatment costs. Moreover, the extensive use of multiple antibiotics risks disrupting the intestinal microbiota and promoting secondary antibiotic resistance, thereby narrowing options for rescue therapy in cases of first-line treatment failure. 15 Consequently, there is a pressing need to investigate alternative regimens that maintain high eradication efficacy while reducing antibiotic use and minimizing adverse effects.
In recent years, 14-day dual therapy consisting of a double-dose proton pump inhibitor (PPI) and high-dose, high-frequency amoxicillin (⩾3 g, administered 3–4 times daily) has garnered considerable attention.16–18 This approach leverages the low resistance rate of amoxicillin and the enhanced antimicrobial activity achieved through elevated intragastric pH levels maintained by the double-dose PPI. 18 In Asian populations undergoing initial treatment, this regimen has demonstrated eradication rates exceeding 90%.16–18 However, evidence from Western populations indicates eradication rates below 80%,19–22 highlighting a potential regional disparity. This discrepancy is likely due to the short half-life of PPIs (0.5–2 h) and their metabolism being significantly influenced by CYP2C19 genetic polymorphisms. Western populations predominantly comprise rapid or ultra-rapid metabolizers, for whom double-dose PPIs may fail to sustain the optimal intragastric pH required for effective H. pylori eradication. 23
Vonoprazan (VPZ), a novel potassium-competitive acid blocker (P-CAB), presents an alternative to PPIs with a longer half-life (5.1–9.0 h), faster onset of action, stronger and more sustained acid suppression, and independence from dietary factors or CYP2C19 polymorphisms. 24 These pharmacokinetic advantages position VPZ as a promising candidate for improving eradication outcomes. Despite its potential, current studies on VPZ-amoxicillin (VA) dual therapy are limited, reporting eradication rates ranging from 66.7% to 87.1%.25,26 Given these variable results, further investigation into the efficacy of the VA dual regimen is warranted. This study, therefore, aims to conduct a randomized controlled trial to evaluate the efficacy, safety, and patient compliance of a 14-day VA dual regimen in comparison with the standard 14-day BQT in treatment-naive H. pylori patients.
Methodology
Study design
This study was designed as a single-center, prospective, open-label, randomized controlled trial. It was reported in accordance with the Consolidated Standards of Reporting Trials (CONSORT) guidelines, and the checklist was provided as Supplemental Material. 27
Study population
From November 2022 to April 2024, patients visiting the outpatient gastroenterology department of our hospital were screened for eligibility based on the following criteria: Inclusion criteria: (i) adults aged 18–70 years; (ii) confirmed diagnosis of H. pylori infection through at least one of the following diagnostic methods: 13C-urea breath test (UBT), 14C-UBT, histopathological examination, rapid urease test, or bacterial culture; (iii) no history of previous treatment for H. pylori. Exclusion criteria: (i) documented allergies to study medications; (ii) history of prior upper gastrointestinal surgery; (iii) active gastrointestinal bleeding; (iv) recent use of bismuth, or antibiotics, acid suppressants, including PPIs, P-CABs, or H2 receptor antagonists, within the past 4 weeks; (v) pregnancy or breastfeeding; (vi) serious medical conditions potentially interfering with outcome evaluation, such as hepatic or renal insufficiency or heart failure; (vii) history of alcohol or drug abuse; (viii) inability or refusal to provide informed consent.
Randomization and treatment
A computer-generated random allocation sequence was used to assign participants to one of two treatment groups in a 1:1 ratio. They received either a 14-day VA dual therapy or a 14-day BQT. This sequence was concealed from all investigators, and neither patients nor researchers were blinded to the treatment group assignments. All medications were administered by licensed clinical staff at the study site, and no specific eligibility criteria were applied to individuals delivering the interventions beyond standard clinical qualifications.
The VA dual therapy included taking 20 mg of VPZ (Takeda Pharmaceuticals, Tianjin, China) twice daily, 30 min before breakfast and dinner, and 1 g of amoxicillin three times daily, 30 min after each meal. The BQT therapy included taking 30 mg of lansoprazole, 500 mg of clarithromycin, 1 g of amoxicillin, and 240 mg of bismuth potassium citrate twice daily. Lansoprazole and bismuth potassium citrate were taken 30 min before breakfast and dinner, while amoxicillin and clarithromycin were taken 30 min after breakfast and dinner. Both regimens were administered for 14 days.
Follow-up
Upon enrollment, participants received comprehensive verbal and written instructions regarding medication usage and potential side effects. To ensure adherence to the protocol, an independent researcher conducted standardized follow-up calls at the end of the treatment period. Participants were required to report the number of remaining pills, and medication compliance was defined as good if at least 80% of the prescribed doses were taken. Adverse events were documented using standardized questionnaires and classified based on their impact on daily life: “mild” (discomfort without interference in daily activities), “moderate” (discomfort with some interference in daily life), and “severe” (significant disruption to daily life). Four weeks after completing treatment, all participants were reminded to schedule a 13C-UBT. A 13C-UBT result of less than 4.0‰ was interpreted as a negative status for H. pylori.
Outcomes
The primary outcome of the study was the H. pylori eradication rate, evaluated through both intention-to-treat (ITT) and per-protocol (PP) analyses. The ITT analysis included all randomized participants. Patients who were lost to follow-up or did not perform the subsequent 13C-UBT were considered eradication failures. The PP analysis comprised those who adhered to at least 80% of the prescribed medication and completed the follow-up 13C-UBT. Secondary outcomes included patient compliance, as well as the incidence and severity of adverse events.
Sample size calculation
This study was a non-inferiority trial. According to previous studies and our preliminary pilot trial, we assumed that eradication rates for 14-day BQT and 14-day VA dual therapy were 88% and 90%, respectively.13,28,29 Assuming a non-inferiority margin of −10%, a one-sided alpha (α) of 0.025, a power (1−β) of 80%, and a 15% dropout rate, a total of 250 participants (125 in each group) were required.
Statistical analysis
Continuous variables were presented as means and standard deviations (SD) and were compared using Student’s t-test or Mann–Whitney U-test. Categorical variables were expressed as frequencies and percentages (%) and were compared using the chi-square test or Fisher’s exact test. We evaluated non-inferiority of the two groups using a one-sided U-test with a 95% confidence interval (CI). The null hypothesis was rejected if the non-inferiority p value was <0.025. All data analyses were conducted with SPSS (version 26.0; IBM Corp., Armonk, NY, USA) and SAS 9.4 (SAS Institute, Cary, NC, USA). A p value of less than 0.05 was considered statistically significant.
Results
Baseline characteristics
The patient recruitment flowchart is presented in Figure 1. From November 2022 to April 2024, a total of 282 patients infected with H. pylori were screened. Of these, 12 subjects met the exclusion criteria, and 20 declined to participate. Ultimately, 250 patients were enrolled in the study and randomized into the VA dual group (n = 125) or the BQT group (n = 125). In the VA dual group, 119 patients were included in the PP analysis, as 3 patients were lost to follow-up and did not undergo the 13C-UBT, and 3 were classified as having poor compliance due to adverse events leading to treatment discontinuation or not having post-treatment 13C-UBT results. In the BQT group, 120 patients were included in the PP analysis, with 2 patients lost to follow-up and 3 classified as non-compliant for analogous reasons.
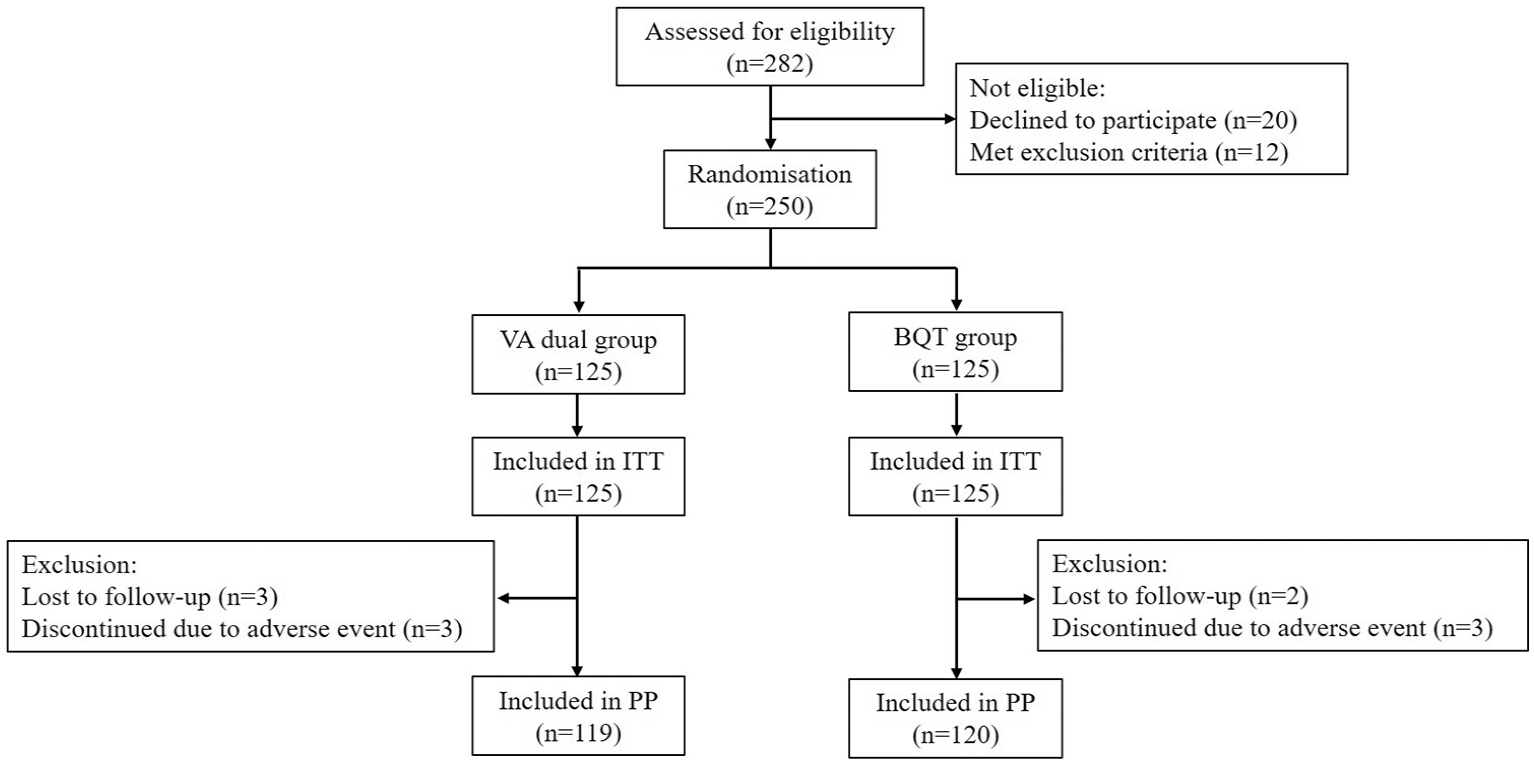
Flow chart of patient enrollment.
Baseline characteristics of the participants are summarized in Table 1. The mean (SD) age was 44.6 (12.6) years in the VA dual group and 41.5 (12.3) years in the BQT group (p = 0.053). Both groups were comparable with respect to baseline variables, including sex, endoscopic findings, family history of gastric cancer, smoking, alcohol consumption, tea drinking, and coffee drinking, with no significant differences observed.
Baseline characteristics of patients with Helicobacter pylori in two groups.
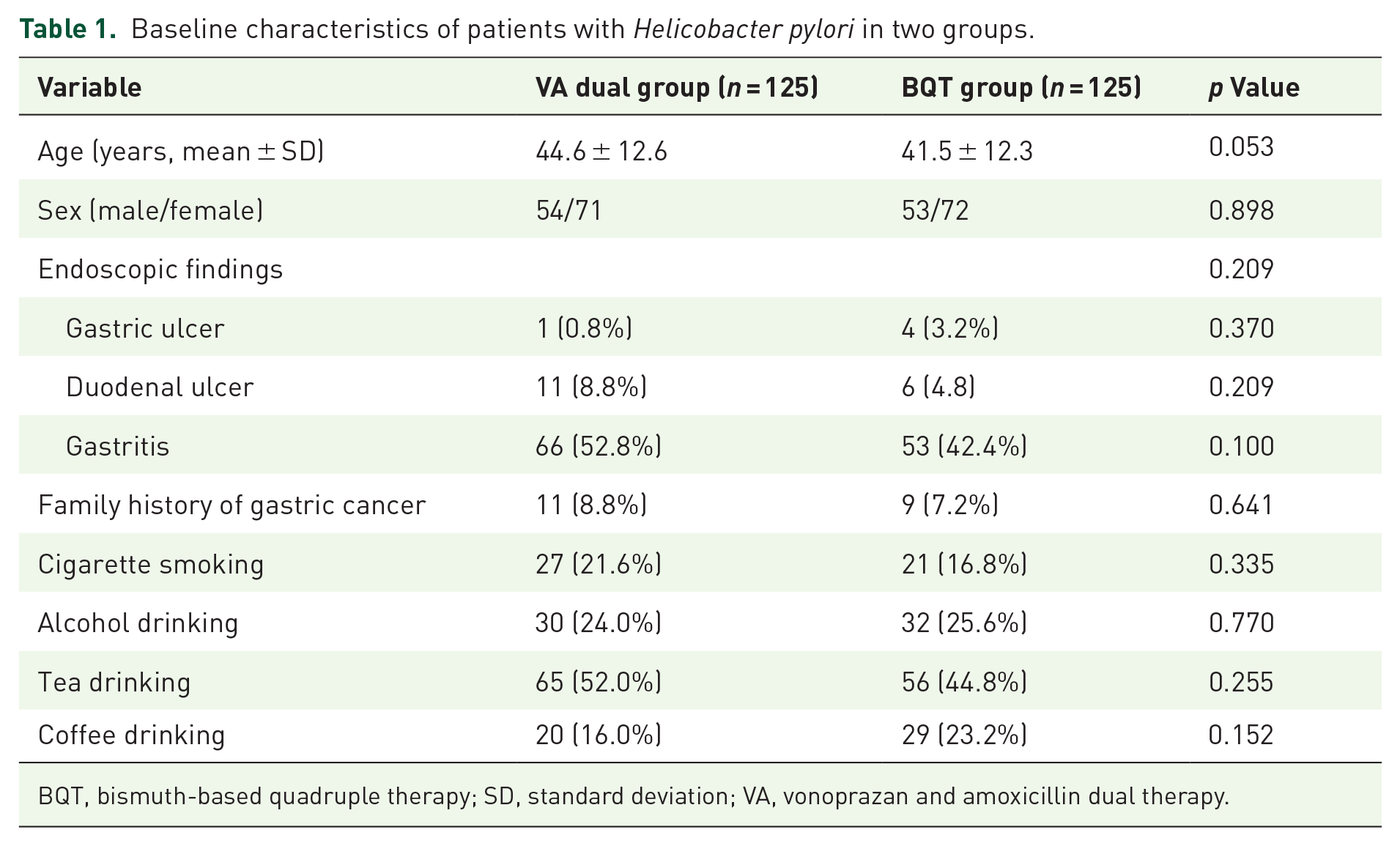
BQT, bismuth-based quadruple therapy; SD, standard deviation; VA, vonoprazan and amoxicillin dual therapy.
Efficacy
As shown in Table 2, in the ITT analysis, the eradication rates were 92.0% (115/125; 95% CI 87.2% to 96.8%) in the VA dual group and 88.0% (110/125; 95% CI 82.3% to 93.7%) in the BQT group, yielding in a difference of 4.0% (95% CI −3.4% to 11.4%). In the PP analysis, eradication rates were 95.8% (114/119; 95% CI 92.2% to 99.4%) for the VA dual group and 91.7% (110/120; 95% CI 86.7% to 96.6%) for the BQT group, with a difference of 4.1% (95% CI −2.0% to 10.2%). The differences in eradication rates between the groups were not statistically significant (p = 0.292 for ITT analysis and p = 0.188 for PP analysis). The lower limit of the 95% CI for the difference of eradication rates in VA dual therapy from BQT was above the non-inferiority margin of −10% in both analyses. The non-inferiority test confirmed that VA dual therapy was statistically not inferior to BQT (both p < 0.001).
Eradication rates of each therapy group.

BQT, bismuth-based quadruple therapy; CI, confidence interval; ITT, intention-to-treat; PP, per protocol; VA, vonoprazan and amoxicillin dual therapy.
Safety and compliance
Adverse events are summarized in Table 3. The overall incidence of adverse events was significantly lower in the VA dual group compared to the BQT group (11.2% vs 20.8%, p = 0.038), corresponding to a risk difference of −9.6%. Notably, dysgeusia occurred in 0% of the VA group compared to 12.8% in the BQT group (p < 0.001; risk difference: −12.8%). Other adverse events, such as dizziness, headache, skin rash, diarrhea, bloating, abdominal pain, and nausea, were comparable between the groups. Most adverse events were mild (41/50, 82.0%), with a smaller proportion being moderate (18.0%), and no severe adverse events occurred in either group.
Adverse events in each therapy group.

AE, adverse event; BQT, bismuth-based quadruple therapy; VA, vonoprazan and amoxicillin dual therapy.
The rates of good compliance were identical between the VA dual and BQT groups (97.6% each, p = 1.000). In both groups, three patients failed to adhere to at least 80% of the prescribed medications. In the VA dual group, one patient discontinued treatment due to a skin rash, another due to dizziness, and a third missed 7 days of medication because of mild nausea. Despite the missed doses, the third patient underwent a 13C-UBT, which confirmed successful eradication. In the BQT group, two patients discontinued treatment due to dysgeusia, and one failed to adhere to the regimen due to forgetfulness. None of these patients in the BQT group underwent 13C-UBT.
Discussion
In this single-center, non-inferiority, open-label, and prospective randomized controlled trial, we evaluated the efficacy and safety of the 14-day VA dual regimen compared to the standard 14-day BQT as first-line treatment for H. pylori infection. The results demonstrated satisfactory eradication rates of 92.0% in the ITT analysis and 95.8% in the PP analysis for the VA dual regimen, which were comparable to those of the BQT regimen. In addition, the VA therapy was associated with a lower incidence of adverse events, particularly dysgeusia.
Over the past decade, the eradication rate of the PPI/amoxicillin/clarithromycin triple therapy has gradually declined due to the increasing prevalence of clarithromycin-resistant H. pylori strains. 30 In countries or regions with high clarithromycin resistance, the BQT regimen, which includes a PPI, bismuth, and two antibiotics, is recommended as the first-line treatment.8–10 Although BQT achieves relatively high eradication rates, its complexity—involving multiple medications—is associated with notable adverse effects, high cost, and reduced patient adherence, which may compromise treatment outcomes. In recent years, dual therapy combining a PPI with amoxicillin has gained increasing attention.16–18 The latest (sixth edition) Chinese consensus on H. pylori recommends a high-dose PPI (double-dose) and amoxicillin regimen (⩾3 g, 3–4 times daily) as a first-line treatment. 10 However, the efficacy of PPI-amoxicillin dual therapy varies due to differences in acid inhibition and CYP2C19 genetic polymorphisms.16–22 Vonoprazan, a novel P-CAB, offers faster onset, more potent, and sustained acid suppression compared to PPIs and is unaffected by diet or CYP2C19 polymorphisms. These properties make it a promising candidate for improving H. pylori eradication rates. Multiple randomized controlled trials conducted in Japan, China, Europe, and the United States have demonstrated the potential effectiveness of VA dual therapy for H. pylori eradication.25,31,32 However, reported eradication rates have varied considerably, likely due to heterogeneity in treatment duration, dosing regimens, study populations, and sample sizes. In addition, regional differences in endemic H. pylori strains and antimicrobial resistance patterns may further contribute to these discrepancies, emphasizing the importance of generating local data. For instance, a study conducted in Fujian, China, found that a 14-day VA dual regimen achieved significantly higher eradication rates than a 14-day BQT regimen comprising ilaprazole, bismuth, amoxicillin, and clarithromycin, based on ITT analysis. 33 In contrast, another randomized trial from a different region in China reported comparable eradication rates between the 14-day VA regimen and a 14-day BQT regimen containing esomeprazole, bismuth, amoxicillin, and metronidazole: 88.7% versus 91.8% in the ITT analysis and 95.6% versus 96.7% in the PP analysis, respectively, with no statistically significant differences observed. 34 These divergent findings underscore the need for robust, high-quality clinical trials to rigorously evaluate the comparative efficacy of 14-day VA dual therapy versus standard BQT regimens. Moreover, the potential influence of regional strain variability and antimicrobial susceptibility highlights the necessity of localized evidence to guide optimal treatment strategies.
In the present study, the 14-day VA dual therapy suggested significant advantages over the 14-day BQT in several aspects. First, the twice-daily administration of vonoprazan ensures potent and sustained acid suppression. This robust acid inhibition enhances antibiotic efficacy by lowering the minimum inhibitory concentration, improving chemical stability, and increasing antibiotic concentration in gastric fluid. 35 Second, amoxicillin, as one of the treatment drugs, provides significant benefits. It is readily available, demonstrates low resistance rates, and rarely induces secondary resistance. Moreover, it has a favorable safety profile with few side effects and minimal impact on gut microbiota. Consequently, when utilizing the VA dual therapy, antibiotic susceptibility testing is unnecessary, and even in cases of treatment failure, a wide range of antibiotic options remains available. Lastly, cost-effectiveness emerged as a critical advantage of VA dual therapy. With fewer medications required, the VA regimen cost only 400 RMB (57.1 USD) per patient compared to 746 RMB (106.6 USD) for the BQT regimen, representing a 46.4% cost reduction. This financial benefit underscores its potential as a sustainable option for population-based H. pylori screening and eradication programs in China.
Patient compliance and safety are also key factors in optimizing treatment regimens. In our study, compliance rates for both regimens were similar, exceeding 95%, but the safety profile of VA dual therapy was superior to that of BQT, with a significantly lower overall incidence of adverse events. Adverse effects associated with VA dual therapy, such as dizziness and rash, were generally mild and rare. Despite penicillin skin testing, some patients still experienced mild rashes, likely due to individual variability, but no severe reactions such as anaphylactic shock were reported. In contrast, the BQT regimen had a higher incidence of adverse events, particularly dysgeusia (12.8%), which may be related to clarithromycin. 36 Clarithromycin can alter the bacterial resistance spectrum, including H. pylori, 25 and acts as a potent inhibitor of p-Gp and CYP3A4, necessitating caution in patients on concomitant medications. Furthermore, clarithromycin may stimulate intestinal peristalsis, increasing the risk of diarrhea during eradication therapy. 30 However, no significant difference in diarrhea incidence was observed between the two regimens in this study. Although no severe adverse events related to clarithromycin were reported, its potential risks still warrant careful consideration.
This study has several limitations. First, gastric acid pH monitoring was not conducted, so we cannot determine the differences in acid suppression between the two groups. In addition, CYP2C19 genotyping was not performed. Given the influence of CYP2C19 polymorphisms on the metabolism of acid-suppressing agents, particularly PPIs, the absence of genotypic data may limit pharmacodynamic comparisons. Nevertheless, this concern is partially mitigated by the use of vonoprazan, a P-CAB, whose efficacy is largely unaffected by CYP2C19 status. Second, the absence of antibiotic susceptibility testing may limit the ability to tailor treatment based on individual resistance profiles. However, given the low resistance rate of amoxicillin in China, this omission likely may not significantly affect the applicability of the VA dual therapy regimen. 32 Third, the VA dual therapy is not suitable for patients with penicillin allergies or in regions with high amoxicillin resistance rates. This highlights the need for further research to develop effective, alternative regimens that do not rely on amoxicillin. Fourth, to provide robust evidence for standardized and optimized treatment regimens, this study employed an RCT with a 14-day eradication protocol. Nevertheless, additional studies are required to investigate whether shorter treatment durations can achieve comparable eradication rates while reducing adverse events. Finally, although this study was a well-designed randomized trial, its single-center, open-label nature may still introduce potential bias, especially in the reporting of adverse events. This limitation necessitates cautious interpretation of the results. Future multicenter, double-blind studies are warranted to validate our findings, minimize subjective bias, and enhance generalizability across diverse populations.
Conclusion
In summary, this study demonstrates that 14-day VA dual therapy is non-inferior to 14-day BQT for H. pylori eradication, achieving a high eradication rate of 95.8% with significantly fewer adverse events in a population from southern China. These results support the VA dual regimen as a safe, effective, and cost-efficient alternative, particularly suitable for regions where bismuth-based regimens are less accessible or associated with tolerability concerns.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251354868 – Supplemental material for Fourteen-day vonoprazan-amoxicillin dual therapy versus 14-day bismuth-based quadruple therapy for Helicobacter pylori treatment: a randomized clinical trial
Supplemental material, sj-docx-1-tag-10.1177_17562848251354868 for Fourteen-day vonoprazan-amoxicillin dual therapy versus 14-day bismuth-based quadruple therapy for Helicobacter pylori treatment: a randomized clinical trial by Jin-Yan Zhang, Jin-Hai Chen, Yu-Lin Huang, Ji Li, Dong Xu, Zhong Xu and Xiao-Yi Lei in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors would like to thank all the patients who participated in this study. The authors would also like to thank the many staff members at the medical institutions who supported this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
