Abstract
Endoscopic submucosal dissection (ESD) has emerged as a cornerstone therapy for early-stage esophageal cancer due to its minimally invasive characterization and high efficacy. However, postoperative esophageal stricture remains a critical complication, particularly in cases involving circumferential mucosal defects, with reported incidence rates exceeding 80%. Conventional preventive strategies, including local or systemic steroid administration, esophageal stent placement, and repeated balloon dilation, are limited by side-effect concerns, procedural complexity, and suboptimal long-term outcomes. Recent advancements in regenerative medicine have highlighted hydrogels as innovative tissue-shielding materials with significant potential to mitigate post-ESD stenosis. These hydrophilic polymer networks exhibit unique advantages such as biocompatibility, tunable mechanical properties, and the ability to transfer therapeutic agents directly to the wound. This review synthesizes current evidence on synthetic and natural hydrogels, emphasizing their mechanisms in promoting epithelial regeneration, reducing fibrosis, and synergizing with current therapies (e.g., steroid-loaded formulations or stent placement). Key findings suggest that hydrogel-based interventions significantly lower stricture rates and minimize the need for invasive procedures, offering a promising paradigm shift in postoperative management. By bridging material science and clinical practice, this work underscores the transformative potential of hydrogels in enhancing patient quality of life following esophageal ESD.
Keywords
Introduction
Esophageal cancer (EC) remains a major global health burden, with an estimated 604,100 new cases and 544,076 deaths worldwide in 2020, ranking seventh in cancer incidence and sixth in mortality.1,2 Notably, over 85% of EC cases occur in low- and middle-income countries, with squamous cell carcinoma dominating in regions such as Eastern Asia and Sub-Saharan Africa. 1 Despite advancements in early detection and treatment, the 5-year survival rate for advanced EC remains dismal (<20%), underscoring the critical importance of early intervention. 3
Endoscopic submucosal dissection (ESD) has emerged as the first-line treatment for superficial esophageal dysplasia and early-stage EC, owing to its minimally invasive nature, high resection rate (90%–95%), and anatomical preservation benefits.3 –5 Recent epidemiological studies indicate that ESD is increasingly adopted globally, particularly in Japan and China, where early EC detection programs have significantly improved outcomes. 5 However, this technique is not without challenges. Post-ESD esophageal stenosis, particularly in cases with mucosal defects exceeding three-quarters of the esophageal circumference, remains a devastating complication, with an incidence exceeding 80% and reaching 100% in circumferential resections.6,7 The clinical ramifications of post-ESD stenosis are profound. Patients typically experience progressive dysphagia, leading to malnutrition and weight loss.
Studies have shown that esophageal stricture occurs within 2–4 weeks following ESD. 8 The underlying mechanism of post-ESD stenosis in early-stage EC is predominantly speculated to be fibrous scar formation.9 –11 This process can be delineated into three successive phases (shown in Figure 1).9,11 In the early stage following ESD, mucosal damage initiates an inflammatory response, serving as the body’s intrinsic defense mechanism against tissue injury.11,12 During this phase, inflammatory cells are activated and migrate to the mucosal defects, releasing a cascade of cytokines and molecules that set the stage for wound healing. 12 As inflammation subsides, epithelial cells begin to actively proliferate and migrate to cover and repair the mucosal defects, which is critical for wound healing, as the rapid proliferation of epithelial cells facilitates the restoration of the integrity of the esophageal mucosa. 13 Following the proliferation of epithelial cells, the extracellular matrix (ECM) undergoes a remodeling process. This phase involves the synthesis and degradation of collagen and other ECM components, resulting in permanent alterations to tissue structure, including fibrosis scar formation.11,12 These three stages are interrelated and collectively facilitate the development of post-ESD stenosis. 11 Consequently, esophageal stricture can be effectively prevented by inhibiting the inflammatory response, promoting epithelial regeneration, and suppressing fibrosis formation.14,15

Mechanism of post-ESD esophageal stenosis.
Typical preventive strategies, including systemic or local steroid administration, botulinum toxin injection, esophagus stent placement, and endoscopic balloon dilatation (EBD) have shown promising results in preventing post-ESD esophageal stenosis.7,16 –18 However, these methods are not without limitations (as shown in Table 1). Local steroid injections have been associated with an increased risk of perforation, 19 while oral steroids can lead to secondary diabetes and infections. 20 Although technically recommended, repeated endoscopic balloon dilation (EBD) sessions (median 3–6) are required in 60%–70% of cases to achieve optimal outcomes, and repeated EBDs increase the risk of bleeding and perforation.8,21 –24 Self-expanding endoscopic metal stent (SEMS) placement and biodegradable esophageal stents have been reported for the prevention of post-ESD esophageal stenosis. However, reports of adverse events such as perforation, infection, restenosis, and stent migration have significantly hindered their application.18,25 –30 Therefore, there is an urgent need for a simple, minimally invasive, and reliable strategy to prevent post-ESD esophageal stenosis.
Typical preventive strategies for preventing post-ESD esophageal stricture.

BTX-A, botulinum toxin type A; EBD, endoscopic balloon dilatation; SEMS, self-expanding endoscopic metal stent.
Hydrogels, a type of hydrophilic polymer network that swells in water without dissolving, have emerged as promising tissue-shielding materials in regenerative medicine. 38 They can be categorized into synthetic and natural hydrogels based on their original materials. Functional groups including amine groups (NH2), hydroxyl groups (–OH), amide groups (–CONH–, –CONH2), and sulfonic acid groups (–SO3H) determine the hydrogel’s bibulousness.39,40 Herein, we outline some of the promising hydrogels and look ahead to their future (summarized in Tables 2 and 3).
Summary of preclinical studies of hydrogel on the prevention of post-ESD esophageal stricture.

MCR (%) = [1 − (length of the short axis at the site of maximal constriction)/(length of the short axis at a normal mucosal site on the upper side + length of the short axis at a normal mucosal site on a lower side)/2] × 100. Stricture index: Defined by the ratio between the narrowest and the widest esophageal diameter proximal to the stricture.
ADM, acellular dermal matrix; ADSC, adipose tissue-derived stromal cells; bFGF, basic fibroblast growth factor; ChGelMSC-Exo, chitosan/gelatin (ChGel) sponge encapsulating the adipose mesenchymal stem cells-derived exosomes; CMC, carboxymethylcellulose; ESD, endoscopic submucosal dissection; EV, extracellular (nano) vesicles; FCSEMS, fully covered self-expanding metal stent; hMP, hydrophobized microparticles; HPF, high-power field; IQR, interquartile range; MCR, mucosal constriction rate; PEG, polyethylene glycol; PF-127, pluronic F-127; PGA, polyglycolic acid; SAP, self-assembling peptide; TGF-β1, transforming growth factor β1; Tβ4, thymosin β4; α-SMA, α-smooth muscle actin.
Summary of clinical studies of hydrogel on the prevention of post-ESD esophageal stricture.

ADM, acellular dermal matrix; CMC, carboxymethylcellulose; EBD, endoscopic balloon dilatation; ESD, endoscopic submucosal dissection; ETI, endoscopic local regional triamcinolone acetonide injection; IT + ST, intralesional steroid injection and systemic steroid; OHA, oral hydrocortisone sodium succinate and aluminum phosphate gel; PGA, polyglycolic acid.
Synthetic hydrogel
Synthetic hydrogels are typically composed of synthetic hydrophilic polymers that are crosslinked through either physical or chemical pathways. 41 These hydrogels offer several advantages, including high purity, good reproducibility, controllable degradation, stable mechanical properties, and cost-effectiveness. 42
Polyglycolic acid hydrogel
Polyglycolic acid (PGA) is one of the most widely utilized synthetic polymer hydrogels, known for its excellent biocompatibility and low cytotoxicity. It has been applied in the regeneration of various tissues, such as nerve, articular cartilage, and bladder tissue. 43 PGA hydrogel is biodegradable and can gradually degrade into water and carbon dioxide in the human body. 44
Nishimura et al. 45 revealed through a porcine model that PGA reduces inflammatory exudation via its physical barrier effect. Furthermore, the combination of PGA with basic fibroblast growth factor (bFGF) inhibits the activation of myofibroblasts (as evidenced by a 33.3% reduction in α-smooth muscle actin (α-SMA) expression, p < 0.01) and promotes the orderly arrangement of collagen fibers, thereby minimizing the thickness of the fibrous layer (467.3 vs 726.2 μm in the control group, p = 0.02). 45
Several studies have demonstrated comparable efficacy between PGA-fibrin gel and local steroid injections in preventing post-ESD strictures.46,47 Sakaguchi et al. 46 pioneered the application of PGA hydrogel to cover esophageal ESD wounds. In a pilot study involving eight patients with mucosal defects exceeding three-quarters of the esophageal circumference, post-ESD strictures occurred in 37.5% of the patients, requiring an average of 0.8 ± 1.2 sessions of EBD. Iizuka et al. 47 further reported in their study that at 6 weeks postoperatively, 3 of 33 patients (9.1%) in the PGA hydrogel group developed strictures, compared to 3 of 29 patients (10.3%) in the local steroid injection group (p = 1.00). The mean number of EBD sessions required to resolve the strictures was 0.057 ± 0.24 in the PGA group versus 1.9 ± 5.1 in the control group (p = 0.95). 47 While the precise mechanism by which PGA hydrogel mitigates post-ESD strictures remains unclear, these studies affirmed its safety and efficacy in esophageal wound repair.
Subsequent research validated the synergistic effects of combining local steroid injections with PGA hydrogel.48,49 In a follow-up study by Sakaguchi et al., 48 nine patients received local triamcinolone acetonide injections (5 mg/mL) and PGA hydrogel application concurrently after more than 75% circumferential esophageal ESD. Only one case (11.1%) developed esophageal stricture, with no additional patients reporting dysphagia at the 12-week follow-up. 48 In another study, 10 patients undergoing more than 75% circumferential esophageal ESD were treated with both local steroid injection and PGA tablets combined with fibrin glue postoperatively. 49 Esophageal stenosis was observed only in 50% of these patients (5/10), requiring a median EBD session of 0.5 (range 0–16). 49 Sakaguchi et al. 50 then retrospectively analyzed 500 cases of esophageal ESD. After excluding independent risk factors for esophageal stricture, such as cervical EC, the postoperative stricture rates in the “steroid injection + PGA shielding” group, the “PGA shielding” group, and the “control group” were 18.9%, 41.4%, and 51.7%, respectively (p = 0.015). 50 This indicates an outstandingly synergistic effect in combining steroid injection with PGA hydrogel shielding to prevent post-ESD stenosis (odds ratio (OR), 0.30; 95% confidence interval (CI), 0.10–0.78; p = 0.009). 50
The endoscopic clip-pull technique was introduced to ensure procedural feasibility.46,51 Notably, the additional endoscopic procedure time for PGA hydrogel placement remained clinically acceptable.46,48 The few reported cases of perforation were attributed to endoscopic examination and balloon dilation procedures.48,49 No adverse reactions related to the application of PGA hydrogel after esophageal ESD have been reported. However, Iizuka et al. 47 highlighted the critical issue of PGA hydrogel detachment in their study. By 1-week postoperative endoscopic follow-up, PGA detachment occurred in 10/33 cases, showing a significant correlation with stricture formation (p = 0.021). 47 More importantly, PGA sheets that had been clipped during application exhibited a higher detachment rate than intact sheets (55.6% vs 20.8%). 47
The probability of PGA detachment can be reduced when combined with esophageal stent placement following ESD. Chai et al. 52 conducted a multicenter randomized controlled trial (RCT) using a stratified randomization design with a 1:1 allocation ratio. Their study conclusively demonstrated that the combination of PGA and stenting could independently predict a lower risk of stenosis (odds ratio (OR) = 0.197, p = 0.026). The excellent biocompatibility, carrier properties, and extensive preclinical and clinical studies render PGA one of the most promising hydrogels.
Polyethylene glycol hydrogel
Polyethylene glycol (PEG) is known for its excellent stability, lubricity, film-forming properties, plasticity, and dispersibility. The active hydroxyl groups at the termini of PEG molecular chains readily participate in chemical reactions to form functional monomers, enabling the preparation of hydrogels with diverse structures and properties. 53 PEG hydrogels demonstrate favorable plasticity and fluid characteristics, making them suitable for providing optimal coverage and adhesion to irregular wounds. These attributes underscore the potential of PEG hydrogels in managing post-ESD wounds.
In vitro testing of PEG hydrogel may elucidate its mechanism in preventing post-ESD stenosis. Wei et al. 54 developed a tetra-armed polyethylene glycol (Tetra-PEG) hydrogel and investigated its efficacy in a porcine model. Six domestic pigs underwent 3/4 circumference esophageal ESD and were randomly assigned to either the PEG group (n = 3) or the control group (n = 3). Histological examination via hematoxylin and eosin staining revealed that the PEG group demonstrated significant epithelialization compared to the control group (4.10 ± 0.76 vs 1.14 ± 0.40 cm, p = 0.037). 54 Furthermore, on day 14 post-ESD, the submucosal layer of the control group showed more spindle-shaped myofibroblasts, whereas the experimental group demonstrated more neovascularization. 54 Tetra-PEG hydrogel promotes healing of ESD-induced wounds, enhances epithelial regeneration, and mitigates inflammation and fibrosis by regulating transforming growth factor β (TGF-β)/Smad2 signaling. 54
Thymosin β4 (Tβ4) is a water-soluble acidic peptide that promotes angiogenesis and endothelial migration and inhibits fibrosis. 55 Wang et al. 56 utilized a PEG–polylactic acid–glycolic acid copolymer hydrogel as a carrier for Tβ4, which was injected into the circumferential ESD wounds in Bama minipigs. Although both the experimental group (n = 4) and the control group (n = 4) eventually developed esophageal stricture, the experimental group treated with Tβ4 hydrogel required fewer sessions of esophageal balloon dilation (EBD) to resolve the stricture (8.5 ± 2.08 vs 17.25 ± 2.75, p = 0.002). However, compared with PGA hydrogels, PEG hydrogels have not been widely used in the prevention of early EC, which may be due to the relatively weaker biodegradability of PEG in the human body.
Polyacrylamide hydrogel
Polyacrylamide (PAM) is a synthetic polymer that can be synthesized by crosslinking acrylic monomers. 57 Acrylamide-based hydrogels show significant volume changes in response to physical and chemical stimuli. 57 The hydrolytic stability of these hydrogels can be enhanced by incorporating alkyl and hydroxyalkyl groups in acrylamide. 57 A highly adhesive alginate-PAM hydrogel, tunable tough adhesive drug delivery system (TADDS), was developed for the controlled release of triamcinolone acetonide, showing excellent adhesion and slow degradation properties. 58 The TADDS combines high mechanical toughness and strong tissue adhesion, enabling sustained drug release over 2 weeks while maintaining structural integrity in dynamic tissue environments. 58 Key innovations include the incorporation of stimulus-responsive mechanisms: ultrasound pulses disrupted ionic crosslinks to accelerate drug release, while temperature modulation (e.g., 37°C vs 25°C) altered dissolution rates, achieving up to 50% burst release reduction at lower temperatures. 58 In addition, embedding laponite nanoparticles or PLGA microparticles extended release duration by sequestering drugs through hydrogen bonding or hydrolytic degradation, respectively. TADDS functionality can be preserved by lyophilization, demonstrating shelf-life potential without compromising drug bioactivity. 58 This platform addresses limitations of conventional hydrogels, such as low drug loading and poor mechanical stability, offering a versatile strategy for localized anti-inflammatory therapy. These advancements underscore the potential of hydrogel-based bio-adhesives in addressing clinical needs for tissue repair and drug delivery. Their tunable mechanical properties, multifunctionality, and biocompatibility make them promising candidates for applications ranging from internal tissue sealing to localized drug administration. Future research should focus on in vivo validation and clinical translation of these bio-adhesives to fully realize their therapeutic potential.
Poloxamer hydrogel
Poloxamer hydrogel, also known as Pluronic F-127 or PF-127, is a synthetic and non-biodegradable hydrogel. 59 Poloxamer is composed of hydrophilic PEG and hydrophobic polypropylene oxide, and it remains in a liquid state at low temperatures while transitioning to a gel state at body temperature (~37°C).59,60
Substantial research has reported the potential applications of poloxamers in the biomedical field, primarily characterized by their thermos-responsiveness, wound healing properties, and inherent antibacterial activity. Poloxamers accelerate autolytic debridement by promoting the degradation of damaged collagen while protecting intact collagen. 61 In addition to this, poloxamers can stimulate the expression of vascular endothelial growth factor (VEGF) and TGF-β, thereby enhancing the wound healing process, reducing blood viscosity, 62 and preventing red blood cell aggregation and adhesion to the vascular endothelium. Poloxamers possess certain antibacterial properties, forming water spheres on bacterial surfaces to inhibit bacterial adhesion, 63 and can also enhance the removal of bacterial biofilms. 64
However, the mechanical properties of poloxamer hydrogels are relatively weak, such as their low tensile strength and Young’s modulus, which restricts their practical applications. 65 Researchers have improved the mechanical properties and adhesiveness of poloxamer hydrogels by incorporating other biocompatible polymers (e.g., hydroxypropyl methylcellulose, xanthan gum) into the formulation. 59 These additives can modulate the gelation temperature, viscosity, and drug release rate of the hydrogel. Coffin et al. 66 evaluated the efficacy of a combination treatment involving PF-127 and extracellular (nano) vesicles (EVs) from pig adipose tissue-derived stromal cells (ADSCs) for the prevention of esophageal strictures following extensive ESD. The combination treatment of PF-127 hydrogel with ADSC-EVs was more effective in reducing the severity of esophageal strictures compared to PF-127 or EVs alone. 66 Furthermore, the PF-127 gel appeared to promote wound healing, recruit various cytokines, and promote micro-vessel and granulation tissue formation, 66 making it a promising strategy for the prevention of esophageal strictures. Zhou et al. designed a dual-component hydrogel, in which the colloidal solution is composed of sodium alginate and poloxamer, while the fixing solution contains calcium chloride. The incorporation of sodium alginate and calcium chloride enhanced the mechanical properties of the poloxamer hydrogel. 67 On the 14th day after ESD, a follow-up gastroscopy revealed that the healing rate in the experimental group was significantly higher than that in the control group (83.89% ± 9.51% vs 73.28% ± 17.81%, p = 0.0013), demonstrating the hydrogel’s ability to promote mucosal healing. 67 As one of the few human studies, this research provides significant reference value. However, whether patients with circumferential esophageal lesions can benefit from this treatment requires further investigation.
Overall, despite the many advantages of poloxamer hydrogels, several major challenges remain in their clinical application. For instance, the relatively low gel strength may limit its application in peristaltic movement organs like esophagus. In addition, the relatively poor biodegradability of poloxamer hydrogels may restrict their use in long-term applications.
Aluminum phosphate hydrogel
Yan et al. 68 reported an easily synthesized and orally hydrocortisone sodium succinate and aluminum phosphate gel (OHA), a strong ion buffer system in an acidic environment. They first explored the feasibility and efficacy of OHA in reducing the incidence of restenosis after EBD for benign esophageal strictures. The results indicated that the restenosis rate was significantly lower in the EBD/oral hydrogel (EBD/OHA) group compared to the EBD alone group (5/20, 25.0% vs 8/12, 66.7%; p = 0.025). 68 They further evaluated the feasibility and prophylactic effects of OHA compared to endoscopic intralesional steroid injection and systemic steroid (IT + ST) administration after ESD for the early EC in 27 cases with mucosal defect involving 3/4 of the esophageal circumference. 69 Stricture rates in the IT + ST and OHA groups were 53.8% and 7.1% (p < 0.05), respectively. 69 In addition, the requirement for balloon dilations was significantly reduced in the OHA group compared with the IT + ST group (p < 0.05). 69 In another RCT, OHA was safe and significantly reduced the incidence of esophageal strictures compared to endoscopic local regional triamcinolone acetonide injection plus oral prednisone (p = 0.011). 70 OHA eases the endoscopist’s operation compared with endoscopically sprayed hydrogel and could also resist attacks from non-acid reflux. 70 The positive outcomes observed in these studies indicate that OHA are relatively biocompatible, easy to synthesize, and convenient for periodic administration, demonstrating significant potential in preventing post-ESD stenosis for early-stage EC.
Natural-sourced hydrogel
Natural-sourced hydrogels are composed of natural polymers produced by living organisms, derived from fundamental components such as polysaccharides and peptides, with appropriate modifications. 71 Previous studies have identified several key components, such as collagen, gelatin, hyaluronic acid (HA), alginate, chitosan, xanthan gum, pectin, cellulose, and their derivatives. 71 These hydrogels exhibit superior biocompatibility and biodegradability compared to synthetic hydrogels, making them highly suitable for various biomedical applications, including drug delivery systems, tissue engineering, wound healing, and scaffolds for cell growth.71 –77 Their inherent resemblance to the ECM environment is irreplaceable by chemically synthesized hydrogels.
Gelatin hydrogel
Gelatin, a protein biomaterial derived from type I collagen, is known for its biodegradability, biocompatibility, and non-toxicity. 78 Type A gelatin hydrogel, which is derived from pig and fish skin, features a simple crosslinked network, with an isoelectric pH of 8–9, 79 while type B gelatin hydrogel has a highly crosslinked structure with an isoelectric pH of 4.8–5.5. 79 Type A gelatin exhibits higher molecular weights (approximately 100 kDa) and more elasticity compared to Type B gelatin. 80 Amide bonds form between carboxyl groups and amino groups, which improve the stability and mechanical properties of gelatin, resulting in a network compound that absorbs water through hydrogen bonds establishment. 81
Hydrophobic modification of gelatin can produce temperature-sensitive, sprayable hydrogels with strong tissue adhesion. For example, Nishiguchi et al. 82 developed a hydrophobically modified Alaskan pollock gelatin that demonstrated significant underwater stability and adhesion to submucosal tissues. The gelatin was capable of self-assembly and thermal crosslinking in water-ethanol mixed solvents. Hydrophobic modification of gelatin with aliphatic aldehydes significantly enhanced its underwater stability and adhesion to submucosal tissues. In a rat skin wound healing model, hydrophobically modified gelatin hydrogel reduced the expression of α-SMA in the dermis, thereby inhibiting fibrosis and inflammation after ESD. 82 On this basis, Yano et al. 83 investigated hydrophobized microparticles (hMPs) derived from swine gelatin. Six miniature pigs were randomly divided into hMPs-sprayed and non-hMPs-sprayed groups, undergoing circumferential esophageal ESD. The hMPs group received hydrophobic particle spray after ESD on days 0, 3, and 7. Histological results showed significantly less esophageal constriction in the hMPs group compared to the non-hMPs group (76.1% vs 90.6%, p < 0.05), and the hMPs group demonstrated a reduction in inflammatory cell infiltration (p < 0.01) and muscle layer thickening (p < 0.01) in the submucosa. 83 This tissue-shielding microparticle holds substantial potential for gastrointestinal wound healing following ESD.
Gelatin possesses inherent advantages that make it suitable for an ECM skeleton or a drug delivery vehicle. Lai et al. 84 sprayed a chitosan/gelatin (ChGel) sponge encapsulating the adipose mesenchymal stem cell-derived exosomes (ChGelMSC-Exo) onto post-ESD wounds on day 2 and day 7 following ESD in a porcine model. The ChGelMSC-Exo treatment significantly reduced mucosal contraction, inhibited fiber accumulation and inflammatory infiltration, and enhanced angiogenesis by day 21, in comparison to the sponge control group (ChGelPBS) and the negative control group. 84 This hydrogel provides a suitable carrier for exosomes. The MSC-Exo delivered by ChGel hydrogel alleviates esophageal stricture by enhancing angiogenesis and reducing fibrosis and inflammation after circumferential ESD.
A novel hydrogel system has been developed through the integration of gelatin, alginate, calcium ions (Ca2+), and transglutaminase (TG), leveraging dual enzymatic and ionic crosslinking mechanisms. Schwarz et al. 85 pioneered a double-crosslinked alginate/gelatin hydrogel for cartilage tissue regeneration, where TG-mediated enzymatic bonds and Ca2+-induced ionic networks synergistically ensured structural stability in 3D-printed constructs. Similarly, Pilipenko et al. 86 engineered alginate/gelatin hydrogel microspheres using the same crosslinking strategy, achieving enhanced swelling capacity and mechanical resilience. To expand these concepts, Pan et al. 87 designed an endoscopic-compatible in situ crosslinkable powder composed of alginate and gelatin. This formulation rapidly absorbs bodily fluids to form an adhesive hydrogel upon contact with Ca2+ and TG, maintaining submucosal adherence in porcine models for over 2 weeks. Remarkably, the hydrogel demonstrated exceptional biocompatibility and therapeutic efficacy: in rat models, body weights remained stable by postoperative day 14 (POD14), while epithelialization rates reached nearly 100% at POD7 and POD14. Furthermore, histological analysis revealed reduced collagen deposition and decreased α-SMA-positive cell counts, indicating attenuated fibrotic responses following hydrogel administration. 87
Alginate hydrogel
Alginate, a by-product of the extraction process from brown algae, is a polysaccharide composed of β-D-mannuronic acid (M) and α-L-guluronic acid (G). 88 The molecular chain of sodium alginate contains a significant number of hydroxyl and carboxyl groups, which engage in complex reactions with various divalent or trivalent metal cations in aqueous solution, leading to the formation of sodium alginate gel. 88 A notable advantage of sodium alginate hydrogel is that its carboxyl groups exhibit polyanionic behavior in aqueous solutions, resulting in the production of a viscous adhesive substance. 89 In addition, alginate gels serve as excellent carriers for proteins and cells, and the mild extracellular milieu under which these gels are formed helps preserve the activity of sensitive substances, such as drugs, proteins, cells, and enzymes. 89
Lei et al. 90 reported a two-component in situ hydrogel composed of maleimide-based oxidized sodium alginate and carboxymethyl chitosan. This hydrogel exhibits a short gelation time, exceptional tissue adhesion, favorable hemostatic properties, and good biocompatibility. An in vivo/in vitro rabbit liver hemorrhage model demonstrated the hydrogel’s effectiveness in achieving rapid hemostasis and preventing delayed bleeding. 90 In addition, the canine esophageal ESD model confirmed that the in situ hydrogel promotes significant mucosal uplift and wound closure effects, while also markedly accelerating wound healing, re-epithelialization, and ECM remodeling following ESD. 90 Furthermore, it has been established that there is a strong interaction between alginate and growth factors with a heparin-binding domain, such as VEGF, facilitating sustained and controlled drug release. 91 Zhou et al. 92 reported an in situ crosslinked Konjac Glucomannan (KGM)/sodium alginate (SA)/ε-poly-L-lysine (ε-PLL) hydrogel. The innovative incorporation of KGM effectively enhanced the adhesion strength of the hydrogel, addressing the limitation of traditional hydrogels such as PGA, which tend to detach from wounds. The colloidal solution was composed of KGM and SA, while the fixative solution contained ε-PLL and calcium chloride. Experiments on Bama pigs demonstrated that the hydrogel promotes wound healing. Immunohistochemistry and immunofluorescence staining results showed that the hydrogel inhibits inflammatory cell infiltration, promotes revascularization, and reduces fibrosis in the esophagus. Although less commonly utilized for post-ESD stricture, Fan et al. 93 reported a gelatin-oxidized alginate hydrogel for submucosal liquid pad creation during esophageal ESD. This hydrogel demonstrated good histocompatibility and excellent endoscopic injectability in porcine and rat models. 93
Despite the excellent biocompatibility of alginate and the straightforward ionic crosslinking process for gel formation hold significant potential for various applications, alginate hydrogels are susceptible to degradation in highly acidic environments. 94 Therefore, further experiments are necessary to enhance the physical properties of alginate hydrogels to better adapt to the esophageal environment.
Chitosan hydrogel
Chitosan, a copolymer composed of D-glucosamine and N-acetyl-D-glucosamine, is derived from the deacetylation of chitin. 95 Chitin is naturally abundant in the exoskeletons of crustaceans, the cuticles of insects, and the cell walls of algae and fungi. 96 As a cationic polymer, chitosan exhibits variable charge density, which is influenced by the degree of acetylation and the pH of the surrounding medium. 97 The solubility of chitosan is highly dependent on both pH and molecular weight. Specifically, high-molecular-weight chitosan hydrogels are soluble only under acidic conditions and exhibit poor solubility at neutral and alkaline pH values. 97 Xia et al. 98 evaluated the feasibility of delivering oxidized dextran (ODA) and chitosan hydrochloride (CS) hydrogels to esophageal lesions via an endoscopic catheter in a porcine model. In this model, ODA-CS hydrogels were delivered to wounds in esophageal tissue following ESD using a custom-made catheter device. The homogeneous mixing of ODA and CS resulted in the in situ formation of a bio-hydrogel that adhered firmly to the esophageal wound surface. After 2 weeks, the hydrogel remained structurally intact and continued to adhere to the esophageal wound surface, indicating that chitosan hydrogel can serve as a potential drug carrier. 98
Chitosan hydrogels can be developed as thermo-sensitive, photo-sensitive, or pH-sensitive hydrogels. 99 β-Glycerophosphate is a common thermo-sensitive material that facilitates sol-gel transition by thermally inducing the movement of protons from chitosan to glycerophosphate, decreasing electrostatic repulsion, and promoting the formation of hydrogen bonding between chitosan chains. 99 Liu et al. 100 designed a novel high pH chitosan-polyvinylpyrrolidone-glycerophosphate (HpHCS-PVP-GP) thermo-sensitive hydrogel, which is currently employed in the creation of submucosal fluid pads during ESD. The HpHCS enhances the injectability of hydrogel while maintaining the capacity to gel rapidly at low concentrations. The modification of PVP through hydrogen bonding with HpHCS significantly improved the stability of the low-temperature hydrogel precursor solution, as well as the integrity of the hydrogel formed at 37°C. Experiments conducted in Bama piglets demonstrated that the pad remained approximately 80% 1 h after the injection of the HpHCS-PVP-GP hydrogel. 100 Wang et al. 101 developed a thermo-sensitive chitosan-based hydrogel designed for application in post-ESD wounds. This hydrogel remains in a liquid state at low temperatures, facilitating its injection through an endoscopic biopsy channel directly into the wound site. Upon reaching body temperature, the hydrogel rapidly transitions to a solid state and adheres firmly to the wound surface as a protective barrier. When loaded with 10 mg of triamcinolone, the hydrogel demonstrated an extended-release profile, sustaining drug release for over 10 days. In vitro studies in a rat skin defect model demonstrated that this chitosan-based thermosensitive hydrogel inhibited the proliferation and fibrogenesis of human keloid fibroblasts. 101 The temperature sensitivity of the hydrogel enables its potential for endoscopic injection while ensuring prolonged triamcinolone release, making it particularly suitable for post-esophageal ESD management.
Chitosan-based hydrogels have also been developed in other responsive forms. Li et al. 102 developed a hydrogel comprising caffeic acid-grafted chitosan, gallic acid-grafted chitosan, and oxidized microcrystalline cellulose. This hydrogel exhibits blood-responsive behavior, providing a synergistic combination of tissue adhesion, antimicrobial properties, and tissue repair capabilities through carboxyl, hydroxyl, phenolic hydroxyl, and aldehyde groups. 102 This hydrogel not only facilitates rapid and effective hemostasis but also significantly promotes wound healing in a rat skin defect model. As a blood-responsive ternary hydrogel, it presents a promising alternative to conventional wound management materials due to its superior performance and efficacy across all phases of wound healing. However, it is worth noting that gastroesophageal acid reflux can irritate the esophagus and may cause the dissolution of chitosan hydrogels, which could potentially compromise the efficacy of chitosan hydrogels. Therefore, chemical modification of chitosan hydrogels and sufficient in vitro testing are essential, and further investigation into the long-term adhesion under acidic environments is warranted.
HA hydrogel
HA is an acidic mucopolysaccharide, specifically an advanced polysaccharide composed of D-glucuronic acid and N-acetylglucosamine, and functions as a polymer. 103 HA is a naturally occurring macromolecular material that serves as a crucial component of the ECM in various tissues. 103 It is recognized for its natural compatibility, biodegradability, and non-immunogenicity, making it an ideal building block for the fabrication of hydrogels intended for diverse biomedical applications. HA hydrogels can be synthesized via chemical crosslinking, physical crosslinking, and enzymatic crosslinking to achieve different mechanical properties, swelling behaviors, and degradation rates. 39 Higher molecular weight HA hydrogels typically exhibit increased viscosity and slower degradation rates. 39 Given that HA is a vital component of the ECM in many tissues, HA hydrogels can effectively replicate the structure and function of the ECM, thereby providing an optimal environment for cell growth and maturation, which is essential for tissue engineering applications. 104
Kim et al. 105 injected human mesenchymal stem cell spheres (MSC-SP) encapsulated in catechol-functionalized hyaluronan acid hydrogel into the damaged esophageal layers of mice. Masson trichrome staining on 4 weeks and 12 weeks post-injection indicated that the inner esophageal layer was significantly thinner in the MSC-SP + HA gel group compared to the other experimental groups. 105 In addition, qPCR analysis demonstrated an increase in epithelial gene expression and a decrease in the expression of most fibrosis-related genes in the MSC-SP + HA gel group over time. 105 Chung et al. 106 introduced an injectable and self-crosslinking HA hydrogel that serves as an in situ scaffold for stem cell therapy. In a porcine model following ESD (75% of circumference, 5 cm in length), a mixture of adipose-derived stem cells and the hydrogel was shown to significantly alleviate esophageal strictures by regulating the regenerative process. On POD21, the stenosis rates were recorded at 79.5% ± 2.0%, 62.8% ± 1.7%, and 37.9% ± 2.9% for the control, stem cell-only, and stem cell-hydrogel groups, respectively (p < 0.05). 106 Nevertheless, HA hydrogels are more commonly used for the creation of a submucosal fluid cushion during ESD, 107 with fewer reports of their application in preventing post-ESD stricture formation, an area that merits further investigation.
Self-assembling peptide hydrogel
A novel self-assembling peptide (SAP), called PuraStat (3-D Matrix Europe SAS, Caluire-et-Cuire, France), RADA16, has been developed as a hemostatic agent. The RADA16 solution, upon exposure to and buffering in biological fluids, facilitates the physical crosslinking of nanofibers into a stable, interwoven three-dimensional hydrogel matrix. This matrix closely resembles the natural ECM and promotes wound healing and tissue regeneration. 108 As the most intensively used and researched self-assembled material, RADA16 has been shown to be a suitable matrix for cell cultures as well as a wound dressing material, promoting wound healing in injured skin and mucosa.108 –110 It was first investigated by Barret et al., 110 who performed a 5-cm circumferential ESD of the lower esophagus in 10 pigs that were randomly assigned to either the SAP group or the control group. On day 14, the esophageal stricture rate was 40% in the SAP group, compared to 100% in the control group (p = 0.2). 110 The median esophageal diameter was 8 mm (interquartile range (IQR) 2.5–9) in the SAP-treated group, compared to 4 mm (IQR 3–4) in the control group (p = 0.13). 110 Despite no significant differences in fibrosis, granulation tissue, and neoplastic epithelium between the groups, the application of the SAP matrix following ESD delayed the onset of esophageal stricture. 110 In another study, Oumrani et al. 108 assessed the efficacy of RADA-16 (4 Arg-Ala-Asp-Ala) gel in preventing esophageal strictures following circumferential ESD in a porcine model. Pigs treated with SAP exhibited a significantly lower rate of esophageal strictures on day 14 (33% vs 100%; p = 0.045) compared to the control group (n = 5). Histological analysis revealed that the neoplastic epithelium was longer in the SAP-treated group than in the control group (3075 vs 1155 μm; p = 0.014). Furthermore, immunohistochemical results indicated reduced expression of α-SMA in the SAP-treated group. By promoting re-epithelialization of the resected area, the application of the self-assembled peptide matrix decreased the occurrence of post-ESD strictures by 67% on day 14. 108
In a recent multicenter prospective study involving 43 patients (median age 71 years; 81.4% male) who underwent at least semi-circumferential esophageal ESD, SAP gel was applied postoperatively. 109 Strictures developed in 20.9% of cases (9 out of 43), all of which were resolved with endoscopic treatment. 109 Postoperative bleeding occurred in 6.9% of cases (3 out of 43) but was successfully managed endoscopically. 109 These findings demonstrate that the application of SAP (RADA16) gel is simple and rapid, and it is associated with a low rate of stenosis.
Acellular dermal matrix hydrogel
Acellular dermal matrix (ADM) is a biodegradable scaffold composed of ECM components, including collagen, and retains its original three-dimensional structure. Derived from natural skin, ADM undergoes a decellularization process to remove cellular components of the dermal tissue while preserving the collagenous meshwork and other ECM components. ADM represents a promising approach for preventing esophageal stricture after ESD, as its ability to promote rapid tissue regeneration and minimize fibrosis has been demonstrated in both preclinical and clinical settings.
Several reports of preclinical trials and clinical translations have provided a comprehensive validation of ADM hydrogel for the prevention of esophageal stricture. In a porcine model, Han et al. 111 found that ADM grafts placed over mucosal defects after semi-circumferential ESD significantly reduced stricture formation compared to controls. The study reported complete epithelial coverage and organized collagen fibers in the ADM group, with fewer inflammatory cells infiltrating the wound site. These findings suggest that ADM promotes rapid re-epithelialization and organized tissue remodeling, which are crucial for preventing stricture formation. 111 To address the challenge of fixing ADM hydrogel following circumferential ESD, Zhang et al. 112 attempted an improved therapeutic approach that combines ADM hydrogel with a fully covered self-expanding metal stent (FCSEMS). Twelve Bama piglets were randomly divided into two groups to undergo full circumferential ESD in the distal esophagus. The experimental group received FCSEMS with ADM placed at the mucosal defect, while the control group received a standard FCSEMS placement. Two weeks after ESD, the scaffolds were removed by gastroscopy. Another follow-up gastroscopy 4 weeks after ESD assessed local healing and stenosis. More re-epithelialization (99.13% ± 0.98% vs 96.63% ± 1.64%, p = 0.009), reduced submucosal fibrosis (1117.53 ± 188.83 vs 1834.69 ± 421.99 µm, p = 0.003), and decreased inflammatory infiltrate (121.00 ± 30.66 vs 188.17 ± 64.92, p = 0.045) were observed in the experimental group, 112 so as serum CRP levels compared to the control group, 112 which underlined combination of FCSEMS and ADM significantly promoted re-epithelialization during wound healing and mitigated esophageal stenosis after circumferential ESD. Zhou et al. 113 investigated seven patients who received ADM shielding to the mucosal defects using titanium clips plus FCSEMS. The esophageal stent was removed at a median of 27 days post-ESD. At follow-up, a total of three patients developed strictures, with a mean onset time of 87 days (ranging from 42 to 140 days). The EBD sessions needed to resolve esophageal stenosis were 2 in medium, ranging from 2 to 9. 113 By contrast, traditional local steroid therapy may necessitate dozens of EBD sessions to resolve esophageal stricture. 114
Despite the encouraging results, several challenges remain in the clinical application of ADM. In another study, Schomisch et al. 115 investigated ADM-covered stents in a porcine model of circumferential esophageal mucosectomy, and highlighted the challenges of delivering ADM hydrogels effectively in the dynamic environment of the esophagus. The study suggested that the method of ADM application, rather than the material itself, might be a critical factor in its efficacy. 115 The technique of graft fixation is critical, as improper placement can lead to graft migration or failure. Recent studies have explored various methods, including metal clips and surgical adhesives, to secure ADM grafts effectively.111,113 In addition, the optimal duration of stent placement and the choice of ADM material need further investigation. Future studies should also focus on long-term outcomes and the potential for ADM to promote functional recovery in addition to preventing stricture formation. ADM represents a promising approach for preventing esophageal stricture after ESD. Its ability to promote rapid tissue regeneration and minimize fibrosis has been demonstrated in both preclinical and clinical settings. Further research is needed to optimize the technique and explore the full potential of ADM in esophageal healing.
Fibrin patch hydrogel
Cellulose is a polysaccharide, a long-chain polymer composed of glucose molecules linked by β-1,4-glycosidic bonds. Tang et al. 116 investigated the efficacy of carboxymethylcellulose (CMC) flakes for preventing esophageal strictures following ESD in the porcine model. Fourteen pigs were randomized into a control group (n = 7) and a CMC group (n = 7) and received circumferential esophageal ESD. The CMC sheet was completely applied over the mucosal defect after ESD, while the control group underwent only conventional ESD. 116 In the second week after ESD, esophageal stenosis was significantly slighter in the CMC group. 116 Histological assessment revealed that CMC-treated pigs experienced less fibrosis in the submucosal layer, reduced intrinsic muscle damage, and enhanced re-epithelialization, with significantly higher serum TGF-β1 levels compared to the control. 116 Clinical research reported seven patients who received CMC sheets shielding immediately after esophageal ESD. Postoperative strictures were observed in 57% (4/7) of the patients, 117 relieved by 2.8 ± 2.2 times of EBD on average. No serious postoperative adverse events were reported. 117 The use of CMC tablets appears to be a safe and effective prophylactic treatment for esophageal strictures following extensive ESD.
The application of CMC sheets is technically feasible and straightforward. In the study by Lua et al., 117 the average time required for CMC sheet deployment was 12.6 ± 4.0 min. The sheets rapidly dissolve and adhere to the mucosal defect within 1–2 min, forming a sturdy protective barrier without the need for additional materials such as clips or fibrin glue. 117 This ease of use makes CMC sheets a practical option for clinical practice. The safety profile of CMC sheets is also noteworthy. No major adverse events were reported in either study, suggesting that CMC sheets are well tolerated by patients.116,117 The biodegradable nature of CMC sheets ensures that they are completely absorbed via hydrolysis within 7–14 days, minimizing the risk of long-term complications. 117
Despite the promising results, there are still limitations to the current studies. Both studies had small sample sizes and short follow-up periods, which may limit the generalizability of the findings. In addition, the exact mechanism by which CMC sheets prevent stricture formation is not fully understood and requires further investigation.116,117 Future research should focus on larger, multicenter RCTs to validate the efficacy and safety of CMC sheets in diverse patient populations. Long-term follow-up studies are also needed to assess the durability of the protective effect and potential late-onset complications.
In conclusion, CMC sheets offer a safe, effective, and cost-efficient method for preventing esophageal strictures following ESD. Their ability to reduce fibrosis and promote wound healing makes them a valuable addition to the current preventive strategies. With further research and validation, CMC sheets could become a standard approach in the management of post-ESD esophageal strictures.
Discussion
The prevention of post-ESD esophageal stricture remains a critical challenge in the management of early EC. While ESD offers curative potential with organ preservation, extensive mucosal defects (particularly those exceeding three-quarters of the esophageal circumference) carry a staggering stenosis risk exceeding 80%.7,26 Conventional strategies, such as steroid injections and repeated balloon dilations, are limited by adverse effects, procedural complexity, and suboptimal long-term outcomes.19,25,27 Recent advancements in hydrogel-based therapies, however, present a paradigm shift by addressing the pathophysiological mechanisms of stricture formation—excessive inflammation, delayed epithelialization, and dysregulated ECM remodeling.10,12,13 This review synthesizes compelling evidence that hydrogels, whether synthetic or natural, mitigate fibrosis, accelerate mucosal healing, and synergize with existing therapies, offering a transformative approach to postoperative management (Figure 2(a)).
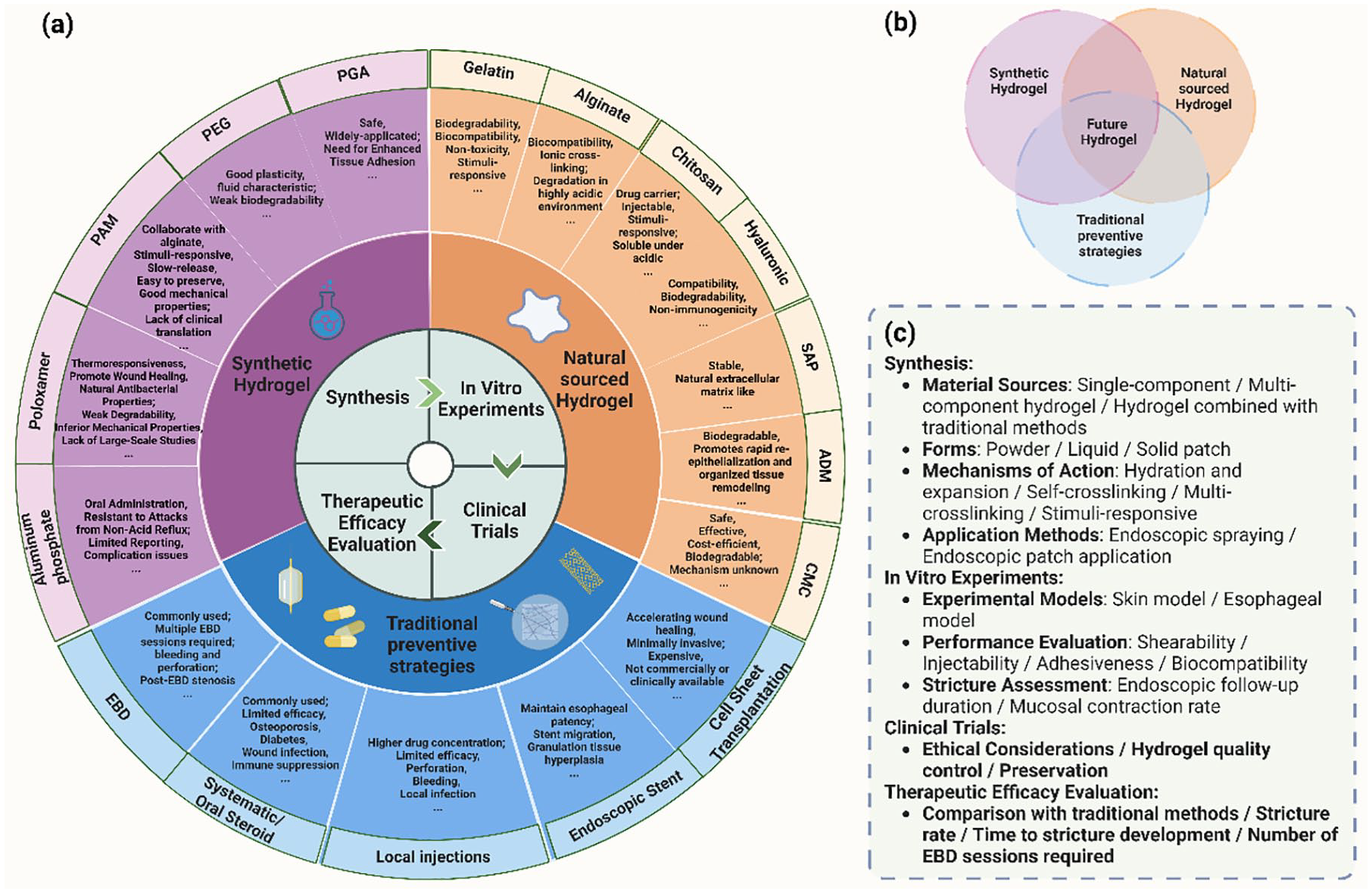
Summary and outlook of hydrogel in the prevention of esophageal ESD postoperative stricture. (a) Summary of the characteristics of reported hydrogels and traditional preventive strategies (including the most significant advantages and disadvantages). (b) The integration of natural hydrogels/synthetic hydrogels/traditional preventive strategies represents an innovation for the development of future novel hydrogels. (c) The synthesis and validation process of future hydrogels.
Mechanistic insights and comparative efficacy of hydrogels
Synthetic hydrogels, such as PGA and PEG, demonstrate robust mechanical properties and tunable drug-release profiles. PGA sheets, when combined with fibrin glue, reduce stricture rates to 9.1%–37.5% in clinical studies,46,47 outperforming steroid monotherapy. The physical barrier effect of PGA shields wounds from inflammatory exudates, while its degradation into water and CO2 ensures biocompatibility.43,44 However, detachment remains a limitation, with studies reporting detachment rates up to 55.6% when clips are used. 47 This challenge is partially resolved by combining PGA with stents, as demonstrated by Chai et al., 52 where PGA-stent co-application reduced stricture rates to 20.5% versus 46.9% with stents alone (p = 0.024). Furthermore, PGA functionalized with bFGF enhances collagen alignment and suppresses α-SMA expression, directly targeting fibrogenesis. 45 Such innovations highlight the potential of drug-eluting hydrogels to achieve dual anti-inflammatory and pro-regenerative effects.
PEG-based hydrogels, such as tetra-PEG, exhibit unique advantages in mucosal coverage due to their fluid adaptability. Wei et al. 54 demonstrated that tetra-PEG promotes epithelialization (4.10 ± 0.76 vs 1.14 ± 0.40 cm in controls, p = 0.037) and modulates TGF-β/Smad2 signaling to attenuate fibrosis in porcine models. Similarly, PAM hydrogels engineered as TADDS enable sustained corticosteroid release over 2 weeks, combining high mechanical toughness (>1 kJ m−²) with strong tissue adhesion (>1000 J m−²). 58 These systems exemplify the convergence of material science and pharmacology, addressing the shortfalls of transient drug delivery in conventional therapies.
Among natural hydrogels, gelatin and alginate derivatives stand out for their ECM-mimetic properties. Hydrophobically modified gelatin microparticles (hMPs) reduced submucosal fibrosis by 14.5% (p < 0.05) in swine models, while alginate/gelatin dual-crosslinked hydrogels achieved near-complete epithelialization within 7 days in rats.82,87 Chitosan-based thermosensitive hydrogels further exemplify innovation, transitioning from injectable liquids to adhesive gels at body temperature. Wang et al. 101 reported that triamcinolone-loaded chitosan hydrogels suppress keloid fibroblast proliferation and extend drug release beyond 10 days, offering a localized anti-fibrotic strategy. Nevertheless, chitosan’s pH-dependent solubility in acidic environments necessitates structural modifications to ensure durability in the esophagus. 102
Hydrogel optimization
The integration of hydrogels with established therapies amplifies their efficacy. For instance, combining PGA with intralesional triamcinolone injections reduced stricture rates to 11.1% in high-risk patients, compared to 51.7% in controls.48,50 Similarly, aluminum phosphate gel (OHA), a hybrid of hydrocortisone and ion-buffering agents, achieved a stricture rate of 7.1% versus 53.8% with steroid injections alone (p < 0.05). 69 These combinations leverage hydrogels’ capacity to localize therapeutics, minimize systemic toxicity, and prolong drug retention. Hybrid hydrogels that integrate synthetic polymers with natural components offer a balanced approach to optimizing mechanical properties and biocompatibility. For instance, alginate-PGA composites can combine the tunable mechanical strength of PGA with the ECM-mimetic properties of alginate, potentially enhancing both tissue integration and wound healing. 80 Such hybrid systems can leverage the strengths of both material classes, providing robust mechanical support while promoting a favorable biological response. In addition, the incorporation of bioactive molecules directly into hydrogel matrices represents another key innovation pathway. For example, hydrogels functionalized with growth factors, such as bFGF, can directly target fibrogenesis and promote collagen alignment, thereby reducing fibrosis and enhancing tissue regeneration. 45
Challenges, clinical translation, and future directions
Despite promising results, several hurdles impede widespread adoption. First, hydrogel detachment remains a persistent issue, particularly for synthetic variants like PGA. Solutions such as stent fixation 52 or chemical modifications (e.g., catechol-functionalization for adhesion 105 ) are under exploration. Second, long-term biocompatibility and degradation kinetics require rigorous evaluation. For instance, non-biodegradable PF-127 hydrogels, while effective in recruiting cytokines, 66 may pose risks of chronic inflammation. Wounds shielded by hydrogel may complicate hemostasis, as the adhesion of the hydrogel can interfere with the accurate identification of true bleeding points. 118 The cost-effectiveness of hydrogels is another issue that needs to be considered. Taking the relatively mature PGA as an example, the total cost of applying PGA sheets with fibrin is approximately 40,000 Japanese yen. 45 While this cost may appear acceptable, it is predicated on the condition that it does not need to be repeated multiple times. Meanwhile, bFGF may promote cancer cell growth in most malignancies. 119 In cases where it cannot be guaranteed that ESD has completely resected the tumor tissue, this may raise potential concerns about the use of novel materials. In addition, the limited application of novel biologically derived hydrogels may be related to more complex preparation steps and stricter ethical regulations. Some hydrogels require the development of specific devices for spraying, and the vast majority of hydrogels need to undergo repeated validation of tissue compatibility, biodegradability, sterility, and storage conditions, which may extend the research and development cycle. As heterogeneity in study designs (e.g., retrospective analyses, small sample sizes, and variable follow-up periods) limits cross-trial comparability, large-scale, RCTs are essential to validate efficacy across diverse populations. Comprehensive in vitro performance testing, preclinical experiments, and clinical studies are required before hydrogels can be widely applied to the prevention of post-ESD esophageal stricture.
The development of smart hydrogels that can respond to specific stimuli within the esophageal microenvironment represents a transformative innovation. Future research should prioritize multifunctional hydrogels that integrate stimuli-responsive drug release, real-time monitoring, and enhanced biomechanical resilience (Figure 2(b)). These hydrogels can be engineered to release therapeutic agents in response to pH changes, temperature fluctuations, or enzymatic activity. For example, pH-responsive hydrogels could release anti-inflammatory or anti-fibrotic drugs specifically in the acidic environment of the esophagus, thereby maximizing local efficacy while minimizing systemic exposure.58,99 Similarly, temperature-sensitive hydrogels could transition from a liquid to a gel state upon contact with body temperature, providing immediate mechanical support and sustained drug delivery.
To better translate these innovations into clinical practice, preclinical models must be refined to more accurately mimic human pathophysiology. For instance, incorporating acid reflux simulations into animal models can help assess the stability and efficacy of hydrogels under physiological stress. 70 In addition, large-scale, RCTs are essential to validate the long-term safety and efficacy of hydrogel-based therapies across diverse patient populations (Figure 2(c)). These trials should focus on optimizing hydrogel formulations, delivery methods, and treatment protocols to maximize clinical benefits. Future hydrogel systems could be equipped with real-time monitoring capabilities, allowing for adaptive therapies tailored to individual patient needs. For example, hydrogels embedded with biosensors could detect changes in tissue inflammation, fibrosis, or epithelialization, triggering the release of therapeutic agents as needed. This adaptive approach would enable more precise and personalized treatment, potentially improving outcomes for high-risk patients.
Conclusion
Hydrogels represent a transformative tool in preventing post-ESD esophageal stricture, bridging the gap between regenerative medicine and clinical practice. Synthetic variants offer precision in drug delivery and mechanical customization, while natural hydrogels excel in biocompatibility and ECM mimicry. Their synergy with steroids, stents, and growth factors underscores the potential for personalized, multimodal therapies. However, translational success hinges on overcoming material limitations, standardizing application techniques, and validating outcomes in robust clinical trials. By fostering interdisciplinary collaboration (spanning material science, gastroenterology, and pharmacotherapy), hydrogels may soon redefine postoperative management, enhancing quality of life for patients undergoing curative ESD.
