Abstract
Background:
Endoscopic resection of extensive esophageal neoplastic lesions is associated with a high rate of esophageal stricture. Most studies have focused on the risk factors for post-endoscopic esophageal stricture, but data on the therapeutic management of these strictures are scarce. Our aim is to describe the management of esophageal strictures following endoscopic resection for early esophageal neoplasia.
Methods:
We included all patients with an endoscopic resection for early esophageal neoplasia followed by endoscopic dilatation at a tertiary referral center. We recorded the demographic, endoscopic, and histological characteristics, and the outcomes of the treatment of the strictures.
Results:
Between January 2010 and December 2019, we performed 166 endoscopic mucosal resections and 261 endoscopic submucosal dissections for early esophageal neoplasia, and 34 (8.0%) patients developed an esophageal stricture requiring endoscopic treatment. The indication for endoscopic resection was Barrett’s neoplasia in 15/34 (44.1%) cases and squamous cell neoplasia (SCN) in 19/34 (55.9%) cases. The median [(interquartile range) (IQR)] number of endoscopic dilatations was 2.5 (2.0–4.0). Nine of 34 (26.5%) patients required only one dilatation, and 22/34 (65%) had complete dysphagia relief following three endoscopic treatment sessions. The median number of dilatations was significantly higher for SCN [3.0 (2–7); range 1–17;
Conclusion:
Refractory post-endoscopic esophageal stricture is a rare event. After a median of 2.5 endoscopic dilatations, 97.0% of patients were permanently relieved of dysphagia. Circumferential endoscopic esophageal resections should be considered when indicated.
Keywords
Introduction
Over the past 20 years, endoscopic resection of early esophageal neoplasia by endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD) has allowed to avoid many esophagectomies.1–3 Although EMR and ESD are effective treatments, widespread endoscopic resection in the esophagus can lead to post-endoscopic esophageal strictures.4–6 Currently, there is no recommended prophylactic treatment of these strictures. 7 Locally injected or systemic corticosteroids are the most promising prophylactic treatment options, but have present safety issues and lack a prospective controlled confirmation of their efficacy; other anti-inflammatory, antifibrotic, or antimitotic treatments have not been implemented in clinical practice due to their insufficient efficacy; preemptive dilatations or esophageal stent placement are associated with repeated endoscopic procedures, general anesthesia, poor quality of life, and esophageal perforations; finally, wound covering techniques with polyglycolic acid sheets or even cell sheets have not been widely used due to their costs and low availability. 7 Predictive factors of post-endoscopic esophageal strictures, such as a resection exceeding the three-fourths of the esophageal circumference and an m2 invasion depth have been well described.8–10 However, few studies have focused on the management of post-endoscopic esophageal strictures in a Western setting,11,12 where endoscopic resections for Barrett’s neoplasia are more frequent, with possible implications in terms of wound healing in the proximal and distal esophagus. Our aim was to describe the management of the post-endoscopic esophageal strictures following ESD or EMR for early esophageal neoplasia at a Western center.
Methods
Study population
Patients were identified from a prospectively maintained database at a tertiary referral center for therapeutic endoscopy. Consecutive patients treated by EMR or ESD for early esophageal neoplasia and further treated by esophageal dilatation or esophageal stents between January 2010 and December 2019 were included in the analysis. Demographic information including age, sex, body mass index, smoking and drinking history, and comorbid conditions were collected. Patients provided written consent for the procedures and the use of their data for the research. The study protocol was reviewed and approved by the Cochin Hospital Institutional review board (CLEP approval no. AAA-2020-08021).
Lesion characteristics including pre- and post-resection pathology, exact location, morphology, lesion height and circumferential extent, circumferential extent of the resection wound, and type and number of endoscopic treatment sessions were recorded. The demographic, endoscopic, and histological characteristics of these patients were collected, as well as treatment outcomes. All patients had a follow-up endoscopy within 6 months of the endoscopic resection.
Post-operative esophageal stricture was defined as the presence of solid food dysphagia occurring after esophageal EMR or ESD, associated with a narrowing of the esophageal lumen impassable by an ordinary gastroscope.
Clinical evaluation of dysphagia severity was performed using a scale introduced by Atkinson
Endoscopic treatment
While ESDs were conducted under general anesthesia with endotracheal intubation, EMR and esophageal dilatation were conducted under deep sedation using propofol, and monitored by an anesthesiologist and a specialized nurse. All procedures required CO2 insufflation. Antiplatelet agents other than aspirin and anticoagulant therapy were discontinued before the procedure. High-definition gastroscopes with narrow-band imaging (GIF-H180J, GIF-2TH180, GIF-1TH190 or GIF-HQ190, Olympus, Japan) were used.
Esophageal EMRs were performed as previously described using a therapeutic gastroscope, a multiband mucosectomy device (Duette® Multi-Band Mucosectomy, Cook Medical, USA), without submucosal lifting, and using the forced coagulation current. 14
Esophageal ESDs were performed as previously described. 15 Briefly, procedures were carried out using a soft distal attachment cap and waterjet. The choice of ESD knives was left to the operator, among the Dual knife (Olympus, Japan), the Flush knife (Fujifilm, Japan), and the Triangle tip (TT) knife (Olympus, Japan). After delineation of the lesion with soft coagulation dots positioned 2–3 mm external to the lateral margins of visible lesions, submucosal injection of indigo-carmine-stained lifting solution (a mixture of 5% fructose and 10% glycerol with saline) was performed, followed by the incision in endocut mode and the dissection in swift coagulation mode.
All patients were prescribed double dose esomeprazole for 3 months after endoscopic resection. Locally injected triamcinolone in the residual submucosa or oral prednisone administration following established protocols were left at the discretion of the endoscopist.
Endoscopic dilatations were conducted using a hydrostatic balloon (CRE, Boston Scientific, Boston, MA, USA) of 12–20 mm, or wire-guided endoscopic bougies (Savary-Gilliard dilatators, Cook, Cook Medical, USA), with the following principles: dilatations were conducted in the left lateral position, under fluoroscopy, after performing an esophagogram to estimate the length of the stricture. In the case of long (>2 cm) strictures, we favored initial bougie dilatation, while balloon dilatators were used in all other cases. Bougienage was stopped when sensing a resistance against two consecutive bougies. Balloon dilatation was typically started at 12 mm, 13.5 mm, and 15 mm, provided the endoscopic control between two dilatations did not show a major mucosal tear. The next endoscopic dilatation session, 2–4 weeks later, aimed at reaching 18 mm with a 15–18 mm balloon.
Dilatations were repeated every 2–4 weeks until reaching an 18 mm dilatation diameter, or in the case of symptom recurrence. In the case of late stricture recurrence (i.e. >3 months), we started again the dilatation from 12 mm on, preceded by submucosal injection of 80 mg triamcinolone in the esophageal stricture.
Endoscopic stenting was performed in supine position and under fluoroscopic guidance, using fully covered self-expandable metallic stents 22 mm × 80 mm (Hanarostent EBN22080-Z070, MI tech, Gyeonggi-do, Korea), and indicated in the case of early (<3 months) recurrence after esophageal dilatation up to 18 mm with triamcinolone injection, or impossibility to reach 18 mm without esophageal perforation.
Endoscopic dilatations were conducted as outpatient procedures: after 2 h fasting, liquid intake was resumed and patients were allowed to resume soft food and leave the hospital in the absence of pain or clinical symptoms suggesting esophageal perforation. All patients were prescribed double dose proton-pump inhibitors for 4 weeks after the dilatation.
Statistical analysis
Data are presented as median and interquartile range (IQR), or percentage, as appropriate. All statistical analyses were two-sided and a
Results
Patient characteristics
Between January 2010 and December 2019, 427 esophageal endoscopic resections were performed, among which 166 were EMR and 261 ESD. Thirty-four (8.0%) patients developed a postoperative esophageal stricture requiring endoscopic treatment: 7/166 (4.2%) after EMR and 27/261 (10.3%) after ESD (Figure 1).
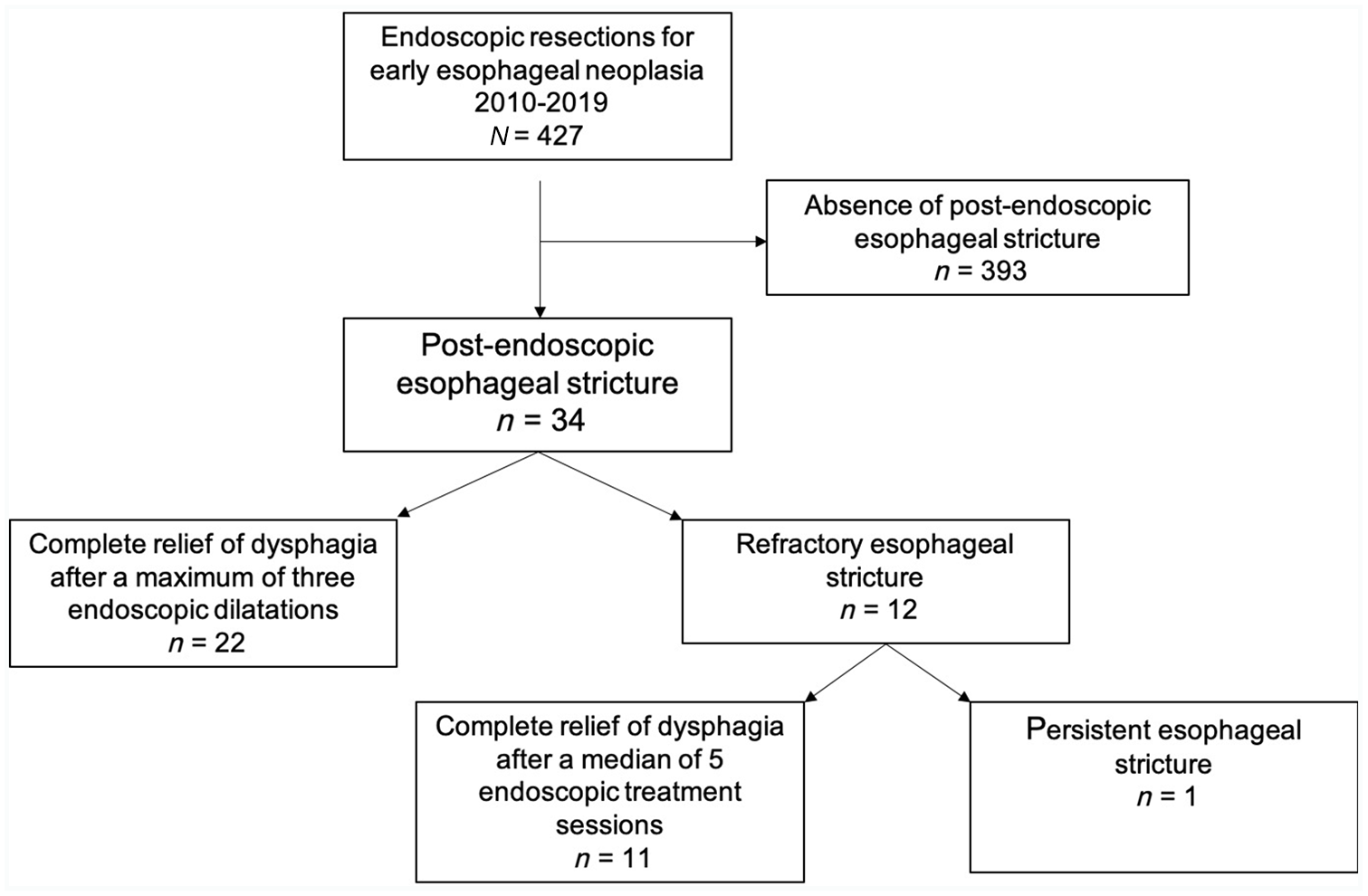
Study flowchart.
The median (IQR) age of the patients was 67.0 (59.0–74.0) years. The indication for endoscopic resection was Barrett’s neoplasia in 15/34 (44.1%) cases and squamous cell neoplasia (SCN) in 19/34 (55.9%) cases. The patients and the endoscopic characteristics of their lesions are presented in Table 1.
Patient characteristics.

Defined by a tumor infiltration >200 µm for squamous cell carcinoma and >500 µm for esophageal adenocarcinoma.
COPD, chronic obstructive pulmonary disease; EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; HGD, high-grade dysplasia; IQR, interquartile range; LGD, low-grade dysplasia; RFA, radiofrequency ablation.
Post-endoscopic esophageal stricture management
Nineteen of 34 (55.9%) strictures occurred in the lower third of the esophagus, following ESD in 27/34 (79.4%) of cases. The median dysphagia score was 3.0 (2.0–4.0). The median (IQR) time before the first dilatation was 31.0 (21.3–78.0) days. The median (IQR) number of dilatation procedures was 2.5 [(2.0–4.0), range (1–17)], allowing for a clinical success with complete relief of the dysphagia in 33/34 (97%) patients after a mean ± SD follow-up of 25.3 ± 22 months. The median (IQR) number of dilatations was 2.0 (1–3)
Outcomes of the post-endoscopic esophageal strictures depending on the histological subtype.

EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; IQR, interquartile range.

Representative case of the management of a post-endoscopic stricture.
Description of the first four endoscopic treatment sessions for the 34 post-endoscopic esophageal strictures.

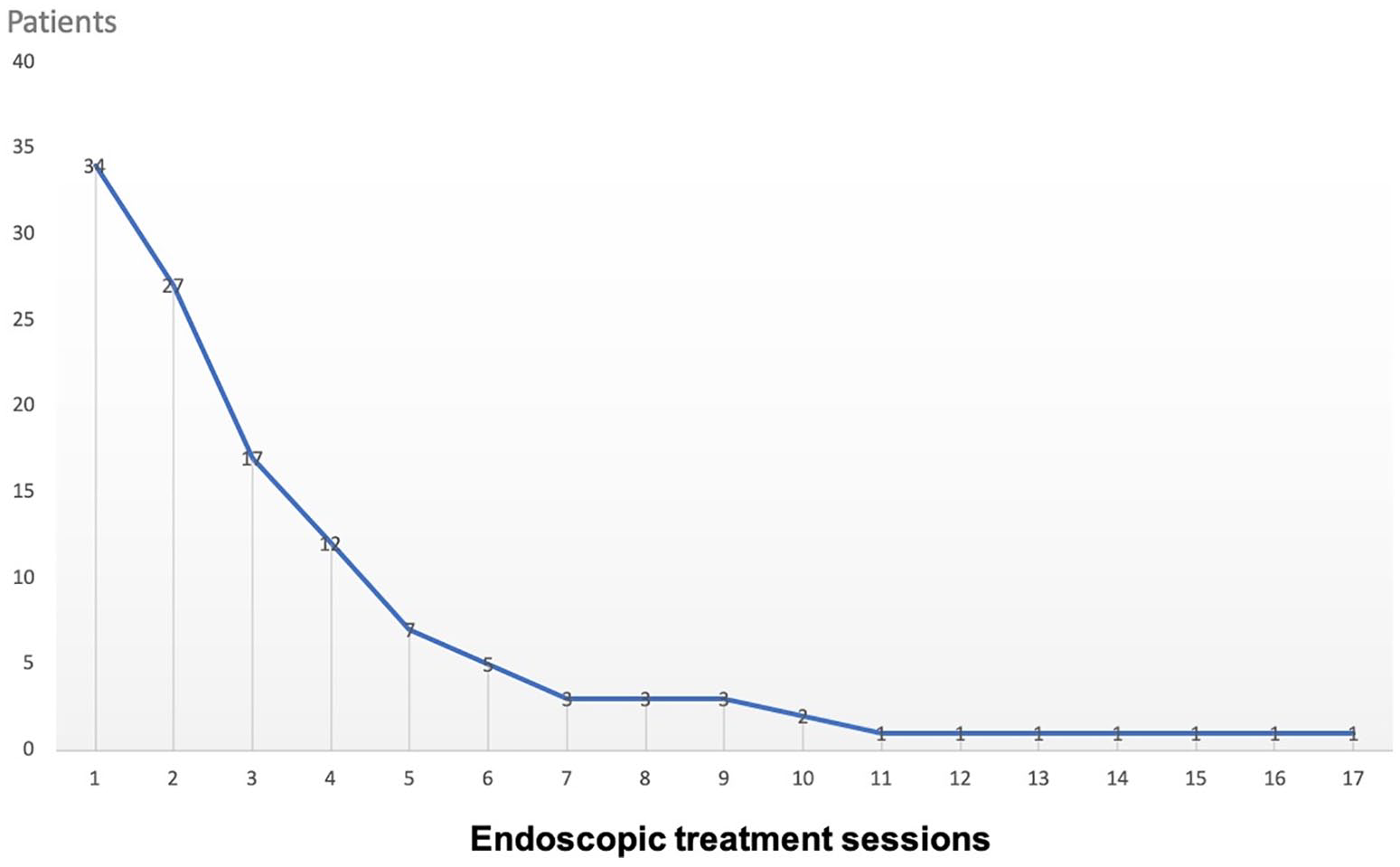
Evolution of the post-endoscopic esophageal stricture rate with the number of endoscopic treatment sessions.
The endoscopic treatment for esophageal strictures was always an outpatient procedure. No patient required surgery for esophageal stricture.
Outcome of refractory strictures
At the last follow-up, one patient had persistent dysphagia related to an esophageal stricture. This patient had a history of pharyngeal squamous cell carcinoma treated by chemoradiotherapy 9 years before endoscopic resection. During follow-up, two esophageal SCNs were diagnosed, one in the upper third and the second in the middle third of the esophagus. Both lesions were treated by ESD in two different sessions: the most proximal lesion was treated by circumferential ESD and, 2 months later, an endoscopic balloon dilatation of the strictured scar was required in order to perform the ESD of the distal lesion. The patient required 14 endoscopic treatment sessions including balloon and bougie dilatations, local steroid injection, and stent placement. The total follow-up time was 63.6 months after the first ESD treatment. The patient died from an extra esophageal cause without resolution of his esophageal stricture.
Outcomes according to the circumferential extent of the resection
Table 4 summarizes the outcomes of post-endoscopic strictures depending on the circumferential extent of the endoscopic resection. A complete (100%) circumferential resection was associated with a median (IQR) 4.0 (3.0–7.0) endoscopic treatment sessions,
Outcomes of the post-endoscopic esophageal strictures depending on the circumferential extent of the mucosal defect following endoscopic resection.

EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; IQR, interquartile range.
Of note, we performed 15 circumferential resections over the study period, with a 100% stricture rate. Stricture preventive methods used in 26.7% of Barrett’s neoplasia cases (locally injected triamcinolone,
Discussion
The occurrence of post-endoscopic esophageal stricture, reported as high as 90% after resection of lesions exceeding 75% of the circumference, is regarded as one of the main limitations of endoscopic resection for early neoplasia. 16 Post-endoscopic esophageal strictures can be refractory 10 and require numerous endoscopic dilatations, with a 1% risk of esophageal perforation at each session, 17 significantly impairing the patients’ quality of life.
In our experience, clinically meaningful esophageal stricture following endoscopic resection was an uncommon adverse event, occurring in 8% of all esophageal endoscopic resections.
The stricture rate was 4.2% after EMR and 10.3% after ESD, and the endoscopic treatment, mainly relying on balloon dilatations, resulted in a 97% prolonged clinical success after a median (IQR) of 2.5 (2.0–4.0) treatment sessions. No esophageal perforation was observed in our patient cohort. Despite having included 15 strictures following complete circumferential endoscopic resections, the overall esophageal stricture rate in our cohort is amongst the lowest reported, with a 3% rate of refractory strictures. Other works report stricture rates ranging from 6% to 27%, and up to 88–90% for resections involving >75% of the esophageal circumference.8,9,12,18–20 Most of these studies were performed in Eastern Asia, and address SCN in the proximal esophagus. A higher proportion of lesions of the lower esophagus (wider than the proximal esophagus), or ethnical differences in wound healing properties, could explain the low rates observed in our study. Also, Ono
The management of post-endoscopic esophageal strictures has been scarcely reported: Takahashi
Considering that differences in patient characteristics, endoscopic resection techniques, or location in the esophagus might impact the management of post-endoscopic resection strictures, we compared Barrett’s neoplasia and SCN. Unsurprisingly, the use of EMR was significantly higher in the Barrett’s neoplasia group, and strictures were more frequently located at the lower esophagus. However, strictures seemed less severe in the Barrett’s neoplasia group, as suggested by the significantly lower number of dilatations required to achieve clinical remission, and the absence of refractory strictures in this patient group. Although the proportion of mucosal defects >75% of the esophageal circumference was smaller in the Barrett’s neoplasia group, a larger esophageal diameter at the distal esophagus and persistence of more residual submucosa after EMR than after ESD could explain these results. Lewis
While 32% of the lesions extended over less than 50% of the esophageal circumference, none of the mucosal defects following endoscopic resection did. The remaining esophageal mucosa tends to retract during an endoscopic resection in the esophagus, and can lead to overestimation of the circumferential extent of the resection. Therefore, the size of the resected specimen or the endoscopically estimated size of the lesion is probably a more reliable predictor of the risk of post-endoscopic esophageal stricture than the size of the mucosal defect after endoscopy.
We performed hydrostatic balloon dilatation in two-thirds of the cases, and bougie dilatation in one-third of the cases. No stricture was amenable to incisional therapy. Only two patients with refractory strictures were treated with esophageal stents, both developing recurrent strictures within 3 months of stent removal. Given these disappointing results, also reported by others,11,25 we abandoned this therapeutic option and returned to repeat dilatations. The efficacy of locally injected or systemic administration of corticosteroids remains controversial, with no randomized controlled data,7,10,26,27 and no clinically demonstrated effect after circumferential endoscopic resections.27,28 Esophageal stricture prevention using locally injected or systemic administration of corticosteroids was conducted in four patients with circumferential endoscopic resection. All four eventually developed esophageal stricture.
The question of circumferential endoscopic resections is important. The possibility to safely achieve these procedures could allow for a radical treatment of Barrett’s neoplasia with a thorough histological analysis. We found that a circumferential endoscopic resection was significantly associated with a higher number of dilatation sessions, with a median (IQR) of 4.0 (3.0–7.0)
Our study is the first to focus on the management of post-endoscopic esophageal strictures, comparing the two histological subtypes of esophageal neoplasia. The strengths of this study include the inclusion of consecutive patients from a prospectively collected database, and the availability of long-term follow-up data for the study patients, all followed up endoscopically at our center.
The main limitation of our study is the absence of standardized measurement of the esophageal strictures, in terms of diameter or length. Second, the quality of life and nutritional management of the patients throughout the study was not available. Third, the relatively small number of patients did not allow us to perform a multivariate analysis on the significant variables identified in the univariate analysis. We did not study the predictive factors for post-endoscopic esophageal strictures, previously widely described.8–10,12,18,19,28 We chose to focus on the management of these strictures, and demonstrated that most of them are amenable to remission with a limited number of endoscopic treatment sessions.
Conclusion
Post-endoscopic esophageal strictures occur infrequently in our experience, even after endoscopic resections exceeding 75% of the esophageal circumference. Most patients (97%) had a complete and permanent relief of their dysphagia and esophageal stricture after endoscopic treatment with a median of 2.5 dilatations. In conclusion, the fear of post-endoscopic esophageal stricture should not lead endoscopists to forgo extensive or circumferential resections of early esophageal neoplasia when indicated.
Footnotes
Acknowledgements
The authors thank Dr. O. Cerles for his help with the English proofreading and editing of the manuscript.
Author contributions
Study design: Einas Abou Ali; Maximilien Barret
Analysis and interpretation of data: Einas Abou Ali; Maximilien Barret
Collection of samples: Einas Abou Ali
Drafting of the manuscript: Einas Abou Ali; Maximilien Barret
Revision of the manuscript and approval of the final version of the manuscript: Einas Abou Ali; Maximilien Barret; Arthur Belle; Rachel Hallit; Sarah Leblanc; Benoit Terris; Frédéric Beuvon; Mahaut Leconte; Anthony Dohan; Solène Dermine; Lola-Jade Palmieri; Romain Coriat; Stanislas Chaussade
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
