Abstract
Background:
Safety and usefulness of double-balloon enteroscopy (DBE) in patients with short bowel syndrome (SBS) are unknown. DBE could be effective in exploring the residual small bowel (SB) entirely, in monitoring patients receiving teduglutide (TED) treatment and, if necessary, could be operative.
Objective:
This study aimed to assess the safety, feasibility, and efficacy of enteroscopy in SBS patients.
Design:
Single center, retrospective.
Methods:
We collected data on patients with SBS patients undergoing DBE from December 2019 to October 2023. We analyzed the technical success total enteroscopy rates, adverse events, diagnostic yield, and residual SB length measurement.
Results:
A total of 12 SBS patients (8 females, median age at first enteroscopy, 66 years, interquartile range (IQR) 58–79) were included; among them, 10 received TED therapy (median interval from TED prescription to DBE surveillance 9.1 months (IQR 3.2–32.9)). Overall, 15 DBE procedures (3 retrograde) were performed, all of which were technically successful and completed without adverse events. The overall diagnostic yield was 60% for relevant findings, defined as conditions unknown before DBE. No SB malignancies were detected during the study period. In 80% of cases, a complete visualization of the SB was obtained with a reevaluation of the SB length. The median depth of maximal insertion was 85 cm (IQR 50–187).
Conclusion:
DBE is feasible, safe, and effective in SBS patients. Total enteroscopy can be frequently achieved with a single approach in these patients, allowing easy diagnostic and oncologic surveillance for SB and colonic malignancy.
Keywords
Introduction
Short bowel syndrome (SBS) is a clinical condition associated with a small bowel (SB) length of less than 200 cm, usually secondary to extensive surgical resections.1,2 SBS is the main cause of chronic benign intestinal failure, which is defined by the latest European guidelines as the “persistent reduction of the gut function below the minimum necessary for the absorption of macronutrients and/or water and electrolytes, such that intravenous support/supplementation (IVS) is required to maintain health and/or growth, in a patient who is metabolically stable.” 1 As a consequence, parenteral nutrition (PN) is the main therapy for SBS. 1
Beyond PN, teduglutide (TED), a glucagon-like peptide-2 analog, is the only approved pharmacological therapy for SBS and exerts its effect by reducing the need for PN and intravenous fluid support.3,4 Due to its activity as a bowel trophic hormone, there are concerns regarding the increase in colorectal–intestinal cancer risk. 5 Although data on the oncologic risk in TED treated patients are still debated, TED is contraindicated in patients with active oncologic disease and those with a history of malignancy of the gastrointestinal tract. 6 For this reason, a baseline colonoscopy with polyp removal is indicated in patients with a remaining colon (SBS types 2 and 3) before starting TED. 7 A follow-up colonoscopy (or alternative imaging) should also be performed at 1 year and at least every 5 years thereafter, or more frequently if clinically indicated. 7 The detection and removal of polyps prevent the risk of their progression to cancer following the initiation of TED treatment. 7 Although the hypertrophic (and oncogenic) effect of TED are primarily exerted in the SB, to date, there is extremely limited evidence regarding the risk of SB polyps and cancer in patients undergoing TED treatment. Recently, different case reports evidenced the development of SB polyps, respectively adenomas and hamartomas, in patients receiving TED.8–10 Furthermore, there are no data regarding potential surveillance methods for SB malignancies in SBS patients. Thus, discrepancy could appear difficult to justify due to the actual capacity to endoscopically investigate the SB by means of device-assisted enteroscopy (DAE).11,12
This study aimed to assess the safety, feasibility, and efficacy of DAE in SBS patients.
Methods
Study design, study population, and data source
We conducted a retrospective review of all patients with SBS who underwent DAE. All patients included in the study were diagnosed with SBS according to the International Guidelines.
Data were retrospectively extracted from electronic patient files at the Fondazione IRCCS Ca’Granda Ospedale Maggiore Policlinico (Milan, Italy), a tertiary referral center for enteroscopy and SB diseases. Patient demographic characteristics, etiology, and type of SBS, TED treatment, and indication for double-balloon enteroscopy (DBE) were collected. Endoscopic findings included the indication, route of insertion, type of sedation, insertion time, depth of maximal insertion, number of advancements, endoscopic findings, therapeutic maneuvers, and adverse events.
TED has been administered in cases of SBS and intestinal failure, after a period of postsurgical intestinal adaptation and following the international guidelines. 13
The study protocol was reviewed and approved by the “Comitato Etico Milano Area 2” (no. 137-2021). Patients gave their written consent to the use of anonymized clinical data to research purposes. This study was reported following the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines 14 (Supplemental File 1).
Procedure
The endoscopic procedures were performed using standard, short, or pediatric DBEs (EN580BT, EI580BT, and EN580XP enteroscopes; Fujifilm, Fujinon, Saitama, Japan) as the DAE technique. The insertion route (antegrade or retrograde) of DBE was chosen based on the remaining anatomy after surgery and the results of previous endoscopic and/or radiological examinations. Usually, the antegrade approach has been preferred because it allows a deeper insertion and a broader exploration of the SB. 15 Preparation for anterograde DBE consisted of 8–12 h of fasting from solid food and 4–6 h of fasting from liquids before the procedure.11,16 Patients received a standard bowel preparation for the retrograde DBEs.11,16 DBE procedures were performed under procedural sedation with continuous hemodynamic monitoring during and immediately after the procedure.
In cases where complete SB exploration was not achieved using the antegrade approach, if indicated, India ink tattooing or through-the-scope clips were placed at the site of maximal insertion, and a retrograde enteroscopy was performed during the same or subsequent session. 11
SB biopsies were performed with standard biopsy forceps for colonoscope (Boston Scientific, Natick Massachusetts, USA). Biopsies were oriented on adhesive filter paper, fixed in a 10% formalin buffer, and paraffin embedded. Sections were stained with hematoxylin/eosin. Intraepithelial lymphocytes were counted by means of anti-CD3 immunohistochemical staining (monoclonal mouse anti-human CD3 clone F7.2.38; Dako, Santa Clara, California, USA). 17 To define hypertrophy, villous/crypt ratio was considered normal when below 4:1. 18
Outcome variables
The primary outcome variables were technical success, adverse events, diagnostic yield, and total enteroscopy rates (i.e., the successful visualization and examination of the entire remaining SB). Secondary outcome variables included the incidence of SB polyps, other findings, and residual SB length.
Statistical analysis
The statistical analyses were performed using SPSS statistics software version 25 (IBM Corp., Armonk, NY, USA). Continuous variables were reported as mean ± standard deviation or median with interquartile range (IQR). Categorical variables were expressed as a number of patients and percentages. Quantitative variables were compared using the students’ two-tailed t test for independent variables assuming two different variances; for the qualitative variables, the Chi-squared test or Fisher’s exact tests were used.
Results
From December 2019 to October 2023, 12 patients (8 (66%) females, median age at first enteroscopy 66 years, IQR 58–79) were included in the study. Demographic and clinical characteristics of the patients are reported in Table 1.
Characteristics of patients with SBS undergoing DBE.
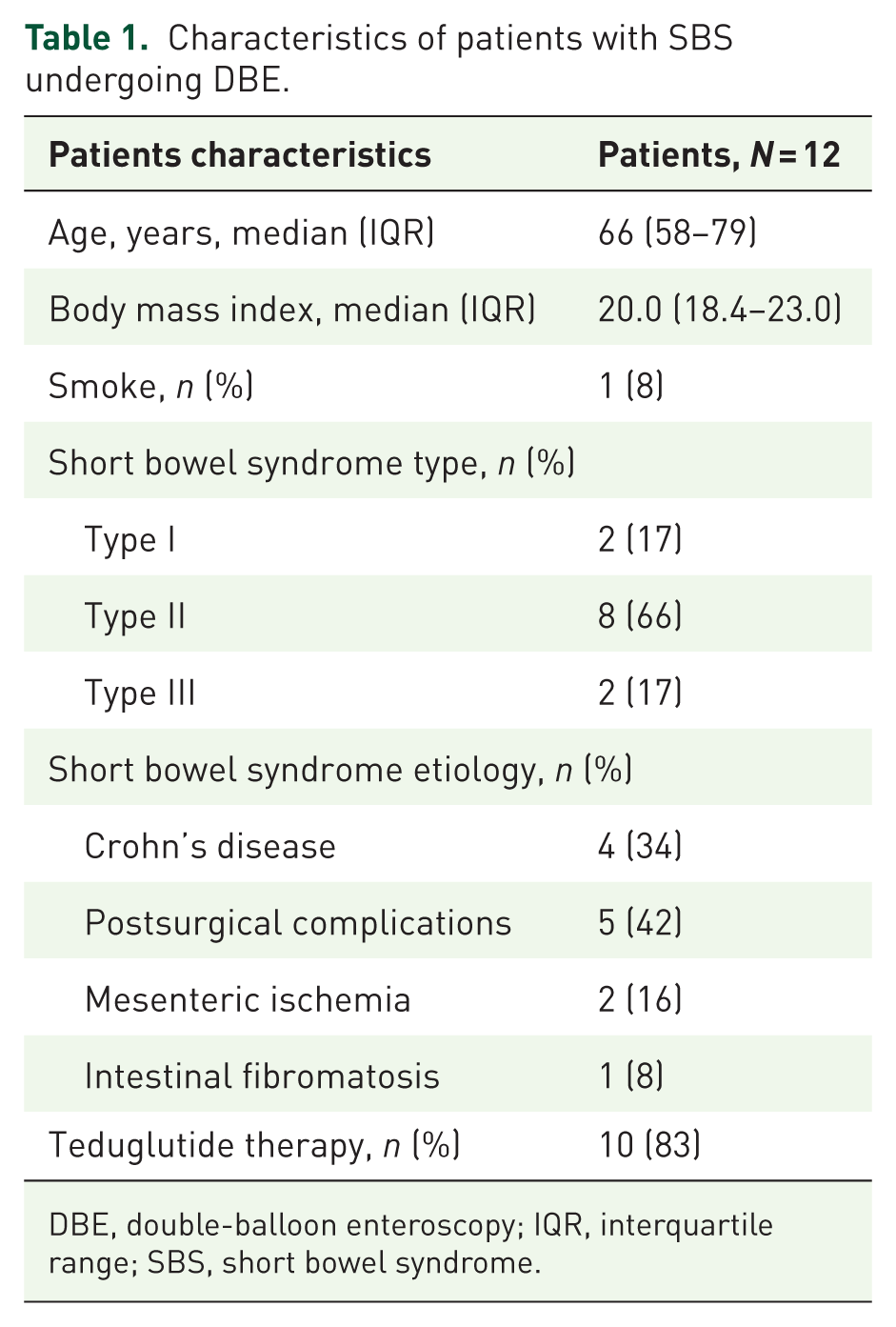
DBE, double-balloon enteroscopy; IQR, interquartile range; SBS, short bowel syndrome.
Fifteen DBEs (12 anterograde, 3 retrograde) were performed in SBS patients with different indications. The indications for SB endoscopy were Crohn’s disease (33%) and unexplained symptoms or suspect SB diseases on radiologic imaging (63%). Enteroscopic data are detailed in Table 2.
Enteroscopic details of patients with SBS.

Difference between DBE and previous estimation made during the surgery of residual small bowel length.
Negative numbers mean that TED therapy started before DBE.
DBE, double-balloon enteroscopy; SB, small bowel; SBS, short bowel syndrome; TED, teduglutide.
The technical success rate was 100%, with no adverse events during any of the procedures. The median depth of maximal insertion in the SB was 85 cm (IQR 50–187 cm). Ten (66%) procedures were performed with the pediatric enteroscope (EN580XP). Complete visualization of the SB was obtained in 12 (80%) procedures: 10 antegrade and 2 retrograde. In 8 (53%) procedures, the length of the remaining SB was recalculated after DBE. Of note, in seven cases, the SB length measured by DBE was longer compared to the previously known measure. The known SB length was 88 ± 55 cm compared to 113 ± 67 cm after DBE (p = 0.34).
Clinically significant findings at DBE were reported in nine cases, resulting in an overall diagnostic yield of 60%. These findings were: SB dilatation in one case, ulcers, and erosions compatible with active Crohn’s disease in three cases, anastomotic substenosis in four, and patchy inflammation of the SB mucosa in one case (examples of DBE findings are shown in Figure 1). In all nine cases, therapy and follow-up were modified based on the DBE results.

Examples of enteroscopic findings in patients with short bowel syndrome: (a) jejunal–ileal anastomotic stenosis; (b) anastomotic ulcers in Crohn’s disease; (c) normotrophic villous in teduglutide (underwater image); and (d) pylorus reached with retrograde DBE.
Ten out of 12 patients were treated with TED. Seven cases had positive DBE findings (Table 2); the median interval from TED prescription to DBE surveillance of 9.1 months (IQR 3.2–32.9). No SB malignancies were detected during the study period. All TED patients underwent colonoscopy surveillance: No colon malignancies or polyps were detected. Oriented biopsies of the SB did not reveal any alteration of the villous/crypt ratio in terms of hypertrophy, also in TED-treated patients. Sex and age were not associated with DBE outcomes.
In Supplemental File 2, the video of a complete SB exploration from a retrograde DBE in an SBS patient is shown (see DBE-SBS-.avi for the video).
Discussion
The present study describes for the first time the feasibility, efficacy, and safety of DBE in patients with SBS (Table 3). DBE is an advanced endoscopic technique used for diagnostic and therapeutic procedures of the SB. 11 It is the most used DAE technique and utilizes two balloons, one on the endoscope and another on an overtube, to achieve deep intubation of the SB. DBE can explore extensively the SB, providing access that was previously unattainable with standard endoscopes. 11 This technique is usually adopted to investigate obscure gastrointestinal bleeding, detecting, and treating SB tumors, polyps, strictures, and other pathologies. 11 DBE has significantly improved the diagnostic yield and therapeutic options for SB disorders, offering a less invasive alternative to surgical exploration. Furthermore, DBE has a good safety profile, even in fragile patients. 19 DBE safety has been confirmed in our study, showing an optimal safety profile and no registered adverse events in a group of patients with previous major, often multiple, abdominal surgeries such as in the case of SBS patients. The extensive use of the pediatric enteroscope (EN580XP), characterized by both smaller scope and overtube diameters and increased flexibility, could be a reason for the absence of adverse events. The safety issue is particularly relevant in patients with a severe condition such as SBS, a malabsorption disorder caused by the surgical removal of a significant portion of the SB. 20 These patients often require long-term PN and present a high number of comorbidities and a relevant mortality rate. 20
Summary of enteroscopy efficacy in patients with SBS.

SB, small bowel; SBS, short bowel syndrome.
Furthermore, DBE in SBS showed a diagnostic yield of 60%, comparable to that seen in more common indications like SB bleeding and immune enteropathies.12,21 Findings at DBE were clinically relevant and previously unknown, especially in cases of erosions and/or severe inflammation, leading to changes in the therapeutic approach and monitoring. Erosions and alterations of the SB mucosa are hardly detected by means of imaging techniques (as computed tomography, CT, or magnetic resonance, MR). Moreover, capsule endoscopy is not recommended in SBS patients due to the risk of retention due to enteric anastomosis, and intestinal obstruction could be an excessively dangerous condition to manage in SBS patients. For these reasons, DBE represents the most suitable tool for SB exploration in these patients. 11
The reference therapy for SBS with intestinal failure is PN. However, a novel drug, TED, has recently been introduced to reduce PN dependence in SBS patients. 22 TED is a recombinant analog of human glucagon-like peptide-2 (GLP-2). By binding to GLP-2 receptors on the surface of intestinal epithelial cells, TED activates downstream signaling pathways that stimulate mucosal growth, enhancing the proliferation of enterocytes, increasing villous height, and expanding the crypt cell population. Additionally, TED promotes blood flow to the SB and decreases gut permeability aiding in nutrient absorption and reducing fluid loss. 23 Although without a pre–post-TED evaluation, in our study, no evidence of hypertrophy (villous/crypt ratio >3) was observed during the follow-up biopsies in patients undergoing TED therapy. Administered via subcutaneous injection, TED is generally well tolerated, with the most common adverse effects being abdominal pain, injection site reactions, and nausea. TED is clinically effective in reducing PN dependence and holds great potential to improve health-related quality of life and clinical outcomes of patients with SBS. 6 A recent meta-analysis showed a response rate to TED of 64% at 6 months, 77% at 1 year, and 82% at ⩾2 years. 24
However, despite its promise, TED is associated with potential oncological risks. To the best of our knowledge, the feasibility of endoscopic surveillance of the SB has not yet been investigated. In the present study, DBEs were not performed with the aim of oncologic surveillance; however, in most cases, the entire surface of the remaining SB length was observed and screened. Therefore, total enteroscopy rates may be even higher in procedures performed with a surveillance indication. SB surveillance in patients treated with TED could be warranted, as the hypertrophic (and potentially oncogenic) effect of TED targets the SB, as suggested by some previous case reports.9,23 However, endoscopic SB surveillance is not considered in guidelines 1 ; therefore, the timing of repeat enteroscopy is determined individually, considering the patient’s underlying disease and clinical picture. However, in our case series, no benign or malignant tumors were detected.
Before definitive conclusions can be drawn regarding the effectiveness of DBE in SBS patients, several unmet needs should be addressed. In fact, the risk of SB polyps and cancer development in SBS patients, especially those receiving TED treatment, remains theoretical. In the literature, there are only a few case reports describing the development of SB polyps after TED treatment, and a causal relationship cannot be ascertained.8,9 However, owing to its mechanism of action as a trophic hormone analog and consistent with preclinical study findings, TED has the potential to cause hyperplastic changes in the gastrointestinal and hepatobiliary tracts. 6 In the long-term open-label extension of the phase III trial to evaluate the safety and efficacy of TED (STEPS2), colorectal polyps were reported in 9 of 51 patients who underwent colonoscopies during or at the end of the 24-month treatment period; no cases of intestinal dysplasia or malignancy were indentified. 25 Pooled safety data from clinical trials showed no major safety concerns, including bowel cancer risk, related to TED use in clinical practice. 26 Therefore, high-quality long-term prospective data from the ongoing observational, multinational SBS registry are warranted to further clarify the possible increased risk of bowel polyps and cancer in this population. 27
While meaningful, the study has several limitations: Its retrospective nature and the limited follow-up of SBS patients represent the two major weaknesses of the study. Furthermore, DBE is not available in all centers and thus proposing an exploration of the SB to all SBS patients could be difficult.
In conclusion, DBE in SBS patients is feasible, safe, and effective. The DBE diagnostic yield is significant, revealing previously unknown notable findings of clinical relevance. Total enteroscopy can be frequently achieved with a single approach, allowing easy surveillance. However, long-term data are required to evaluate the eventual incidence of SB polyps and cancer in SBS patients, especially in cases of TED therapy.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251346272 – Supplemental material for Double-balloon enteroscopy evaluation and surveillance in patients with short bowel syndrome
Supplemental material, sj-docx-1-tag-10.1177_17562848251346272 for Double-balloon enteroscopy evaluation and surveillance in patients with short bowel syndrome by Lucia Scaramella, Andrea Sorge, Gian Eugenio Tontini, Elia Fracas, Veronica Smania, Andrea Costantino, Tommaso Pessarelli, Marco Michelon, Matilde Topa, Ettore Corradi, Paola Onida, Annalisa Mascheroni, Livia Gallitelli, Daniela Milani, Nicoletta Nandi, Maurizio Vecchi and Luca Elli in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
Luca Elli, Maurizio Vecchi and Gian Eugenio Tontini acknowledge that the Department of Pathophysiology and Transplantation, University of Milan, is funded by the Italian Ministry of Education and Research (MUR): Dipartimenti di Eccellenza Program 2023 to 2027.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
