Abstract
Background:
Endoscopic papillectomy (EP) has emerged as a preferred treatment modality for ampullary adenomas, offering a less-invasive alternative to surgical resection. However, concerns persist regarding the potential for incomplete resection and subsequent recurrence.
Objectives:
This study aims to evaluate the efficacy of EP in treating ampullary adenomas and to identify risk factors associated with recurrence.
Design:
This study was a single-center, retrospective case–control study.
Methods:
We retrospectively analyzed patients who underwent EP at Nanjing Drum Tower Hospital between January 2010 and December 2022. Recurrence rates were assessed, and potential risk factors for recurrence were analyzed using univariate and multivariate Cox proportional hazards models. Hazard ratios (HR) with corresponding 95% confidence intervals (CI) were calculated.
Results:
Among the 137 patients who achieved complete resection, 21 (15.3%) experienced recurrence. The median follow-up period was 17.7 months (interquartile range (IQR) 11.7–37.5), and the median time-to-recurrence was 16.2 months (IQR 9.9–33.9). Of the 21 recurrence cases, 11 (52.3%) were managed successfully with repeat endoscopic intervention, while 6 (28.6%) required pancreatoduodenectomy. Significant risk factors for recurrence included familial adenomatous polyposis (FAP; HR = 9.3; 95% CI: 2.8–30.4; p < 0.001), pancreaticobiliary (PB) subtype (HR = 3.2; 95% CI: 1.5–11.3; p = 0.006), and high-grade dysplasia (HGD; HR = 2.6; 95% CI: 1.1–6.2; p = 0.036). Comprehensive sensitivity and subgroup analyses consistently confirmed the stability of these risk factors.
Conclusion:
EP is an effective treatment for ampullary adenomas; however, recurrence remains a significant concern, particularly among patients with FAP, PB subtype, and HGD.
Plain language summary
In this retrospective case-control study, we aimed to evaluate the efficacy of EP in the treatment of ampullary lesions and to investigate the factors influencing recurrence during long-term surveillance. Among the 137 patients who achieved complete resection, a recurrence rate of 15.3% was observed, with recurrences occurring as late as 126.9 months following the initial procedure. FAP (HR = 9.3; 95% CI: 2.8-30.4; P < 0.001), PB subtype (HR = 3.2; 95% CI: 1.5-11.3; P = 0.006), and HGD (HR = 2.6; 95% CI: 1.1-6.2; P = 0.036) were identified as significant risk factors.
Introduction
Ampullary adenomas are rare gastrointestinal lesions, representing only 0.04%–0.12% of cases according to autopsy studies 1 and accounting for less than 5% of all new digestive tract neoplasms. 2 Over the past two decades, the incidence of ampullary adenomas has increased, particularly among younger adults, partially due to the rise in endoscopic surveillance procedures.3,4 These adenomas are considered precancerous, with a 25%–85% risk of progression to adenocarcinoma in sporadic cases. 5 Given their malignant potential, complete resection is crucial for the effective management of ampullary lesions.6 –8
The traditional approach to treating duodenal papillary tumors is pancreaticoduodenectomy (PD), a highly invasive procedure associated with significant risks, including pancreatic and bile leakage, liver and kidney dysfunction, and severe hemorrhage.9,10 Although mortality rates have decreased, they can still reach up to 5%. 11 Endoscopic papillectomy (EP), first reported in 1983, has increasingly been adopted as the first-line treatment for ampullary adenomas due to its low mortality and high success rates, with previous studies reporting success rates exceeding 70%.12,13 However, since EP is limited to the mucosa and submucosa of the duodenal wall, it does not entirely eliminate the risk of local recurrence when compared to radical surgery. 14 The limitations of endoscopic resection as a curative intervention include the risk of incomplete removal and the potential for recurrence. Prior research has shown that the rate of incomplete resections using EP ranges between 10.6% and 57.1%.15,16 Additionally, recurrence rates vary considerably, ranging from 0% to 58%. 17 Several potential risk factors for recurrence have been identified in previous studies, including familial adenomatous polyposis (FAP), intraductal extension, lesion size, and partial resection.16,18,19 However, these studies have primarily focused on EP procedures, with limited emphasis on pathological characteristics. Moreover, due to the low incidence of these tumors, there are few large-scale studies on long-term outcomes and recurrence.
In this retrospective case–control study, we aimed to evaluate the efficacy of EP in the treatment of ampullary lesions and to investigate the factors influencing recurrence during long-term surveillance.
Methods
Study population
We conducted a retrospective review of all consecutive patients who underwent EP for ampullary adenomas between January 2010 and December 2022. Inclusion criteria comprised consecutive patients with a confirmed diagnosis of ampullary adenoma based on postoperative pathology. Exclusion criteria were as follows: (1) patients who had previously undergone EP or surgical ampullectomy prior to enrollment; (2) patients with incomplete medical records; and (3) patients with incomplete resection. A total of 137 patients with confirmed ampullary adenomas were included, as illustrated in the flow chart shown in Figure 1. We extracted relevant variables from procedural documents and health records. If key demographic or clinical variables were missing, the patient was excluded from the study to maintain data integrity. All patients were thoroughly informed of both endoscopic and surgical treatment options, including the potential risks and benefits of each. Written informed consent was obtained from every patient prior to the procedure. The study adhered to the principles of the Declaration of Helsinki and received approval from the Institutional Review Board of Nanjing Drum Tower Hospital (Approval No. 2024-080-01). The reporting of this study confirms the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 20

Flow chart of study.
Endoscopic technique
EP was performed by endoscopists with over 5 years of experience in endoscopic retrograde cholangiopancreatography (ERCP). An example of the EUS evaluation and EP procedure for ampullary adenoma is shown in Figure 2. Prior to the EP, endoscopic ultrasound (EUS) was recommended to assess the depth of tumor invasion and intraductal involvement. At the outset of the procedure, the ampulla of Vater was meticulously examined for size, extent, and any signs of malignancy. If the lesion was deemed resectable, a papillectomy was performed. Mucosal lifting with saline was employed as needed. An en bloc resection of the ampullary adenoma was the primary goal, with piecemeal resection as a secondary approach if en bloc resection was not feasible.
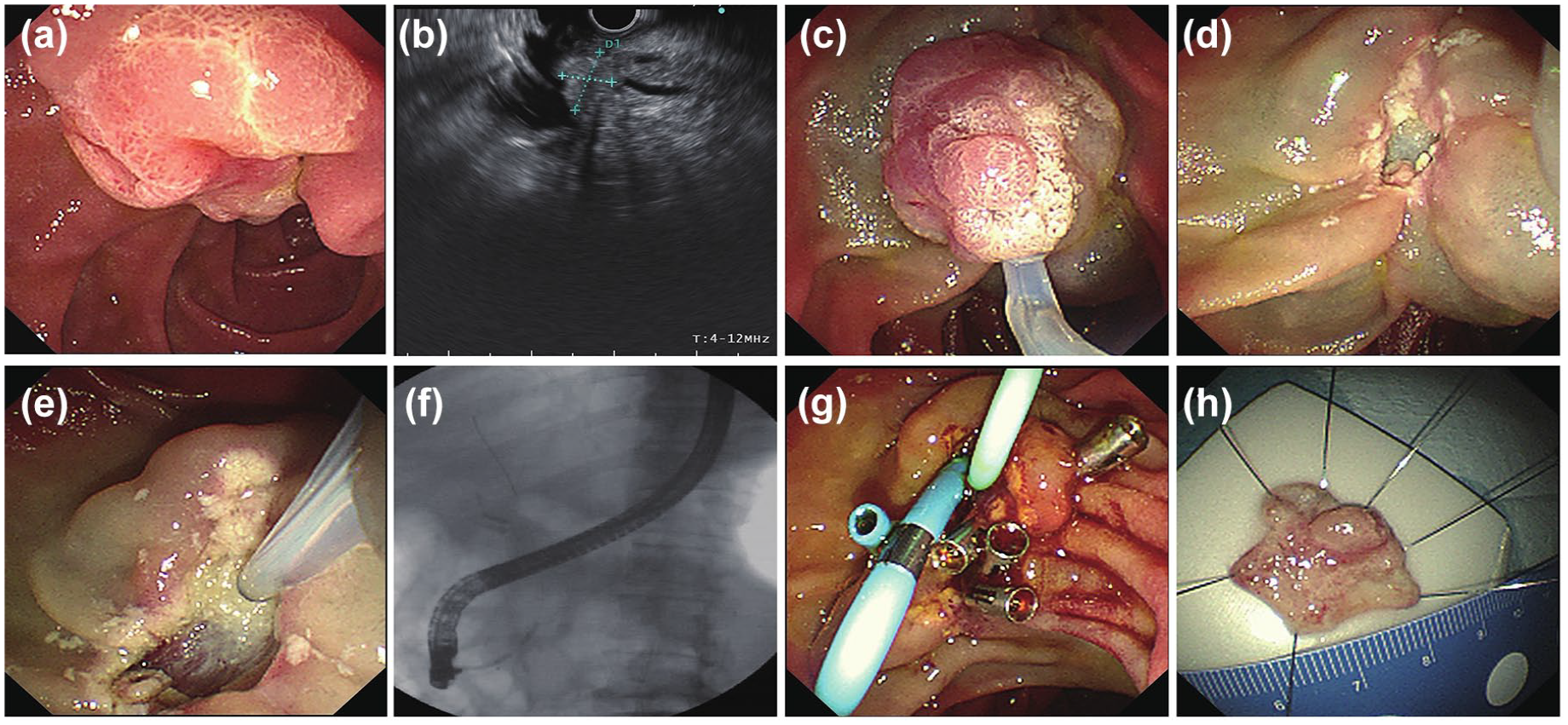
Example of the EUS evaluation and EP procedure for ampullary adenoma. (a) Preoperative EUS carefully evaluates the lesions of the duodenal papilla. (b) Inspection of the tumor site and diameter by EUS. (c) EP was performed using a conventional polypectomy snare. (d) Careful inspection of the resected area. (e, f) Cholangiogram is done to confirm clear bile duct. (g) Bile duct stenting and pancreatic duct stenting was performed. (h) The resected specimen was retrieved and sent for pathological evaluation.
To prevent pancreatitis, pancreatic duct stenting was attempted in all patients, with plans for stent removal at the next follow-up. A cholangiogram was typically performed at the conclusion of the procedure to ensure a clear bile duct. Biliary stents were inserted in patients with insufficient biliary drainage postresection to mitigate the risk of postprocedural cholangitis. The EP site was closely monitored for residual lesions and immediate adverse events. To manage potential residual tissue, thermal ablation was performed using argon plasma coagulation (APC), radiofrequency ablation, the tip of the snare, coagulation graspers, or hot biopsy forceps as needed. In cases of active bleeding, epinephrine was initially administered, with APC as a supplementary measure if hemorrhaging persisted. In the event of a duodenal perforation, endoscopic hemoclips were applied for primary closure, with surgery as a subsequent option if necessary. The procedure was concluded once no residual tissue was observed and in the absence of immediate adverse events. Postprocedure, patients were observed in the ward, with physical examinations, vital sign monitoring, and laboratory tests conducted to detect any early adverse events. The specifics of each endoscopic procedure were determined by the endoscopist.
Follow-up
All patients underwent routine surveillance following EP. This monitoring regimen included periodic endoscopic evaluations and biopsies, which were conducted every 3–6 months during the first year, followed by annual assessments thereafter. Stent replacement or removal was typically performed 6 months postoperation. During routine follow-up duodenoscopy, the identification of a polyp-like growth raised concerns for potential tumor recurrence. In such cases, an endoscopic biopsy was performed. If the biopsy confirmed a tumorous lesion, additional therapeutic options, such as APC or surgical intervention, were considered by the endoscopist in consultation with the patient.
Outcomes and definitions
Residual tissue was defined as the presence of adenomatous tissue, either macroscopic or microscopic, indicating incomplete initial resection. Complete resection was characterized by a clear resection margin and the absence of residual tissue during the procedure. Recurrence occurred after complete resection, was defined as histologically confirmed adenomatous tissue identified through both endoscopic visualization and directed biopsies during follow-up.5,21 To ensure accurate assessment of recurrence, a minimum follow-up duration of 6 months was established. Intraductal extension was defined as the spread of the ampullary lesion into the bile duct and/or pancreatic duct.
Preoperative EUS images were evaluated by an experienced digestive physician. Ampullary neoplasms were classified into three subtypes based on anatomical positioning: peri-AMP type, intra-AMP type, and extended type, with categorization determined by the tumor’s location relative to the duodenum and associated ducts.
Endoscopic adverse events and their severity were graded according to the criteria established by the American Society for Gastrointestinal Endoscopy (ASGE). 22 Post-ERCP pancreatitis was defined as the onset or worsening of abdominal discomfort, leading to an extended hospital stay of at least 2 days, accompanied by a rise in serum amylase levels exceeding three times the upper limit of normal within 24 h postprocedure. ERCP-related bleeding was identified as either immediate bleeding during the procedure requiring endoscopic intervention, or as evidenced by hematemesis, melena, or a hemoglobin drop of more than 2 g/dl, along with clinical signs indicative of gastrointestinal bleeding. Perforation was classified as guidewire-induced perforation, periampullary perforation during sphincterotomy, or luminal perforation at other sites.
Histology
All pathological features were independently reviewed and confirmed by a specialist pancreatic histopathologist who was blinded to the clinical outcomes. Tumors were classified into two histopathologic subtypes based on morphological characteristics. 23 All the lesions were classified according to the revised Vienna classification of gastrointestinal epithelial neoplasia. 24 The intestinal subtype was characterized by the presence of tall columnar cells forming elongated glands, with deeply stained columnar cells often arranged in a pseudostratified manner. The pancreatobiliary (PB) subtype, originating from the epithelium of the bile and pancreatic ducts, features tumor cells that form complex, thin, branched papillary structures similar to those observed in biliary papillary tumors. The epithelium in this subtype is primarily arranged in a single layer and is cuboidal in shape (Figure 3).

Representative histologic images of different histopathologic subtype (hematoxylin and eosin stain). (a, b) Intestinal subtype (×5; ×20). (c, d) Pancreaticobiliary subtype (×5; ×20).
Statistical analysis
Descriptive statistics were utilized to summarize patient demographics, procedural details, and lesion pathology. Patients who achieved total eradication were included in the analysis of recurrence. Continuous variables were presented as mean ± standard deviation, while categorical variables were expressed as frequencies and proportions, with comparisons made using the Chi-square test or Fisher’s exact test. Time-to-recurrence was analyzed using Kaplan–Meier survival curves. Differences in recurrence-free survival between groups were compared using the log-rank test. Univariate and multivariate Cox proportional hazards models were employed to identify risk factors for recurrence. Proportional hazards assumptions were verified via Schoenfeld residual analysis. A post hoc power analysis was conducted to assess whether our study sample was sufficient. Hazard ratios (HR) were estimated with 95% confidence intervals (CI). A significance level of p < 0.05 was applied. All statistical analyses were conducted using R software (version 4.3.3, R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics
Over the 12-year study period, a total of 203 consecutive patients were screened (Figure 1). Of these, 18 patients were excluded due to insufficient follow-up data, and 5 were excluded due to missing clinical and/or pathological information. Additionally, 43 patients who underwent incomplete resection were also excluded. Thus, the final analysis included 137 patients.
The baseline characteristics of the patients are presented in Table 1. Among these patients, 21 (15.3%) experienced recurrence. The results indicated no significant differences between the two groups in terms of sex (p = 0.809), tumor size (p = 0.591), or clinical symptoms (p = 0.799). Preoperative EUS was performed in 85.4% of patients, with similar frequencies observed between the two groups. There was also no significant difference in the rates of intraductal involvement (9.9% vs 12.5%; p = 0.750) or location subtype (89.1% vs 87.5%; p = 0.849).
Baseline characteristics of all patients.

EUS intraductal involvement and location subtype in EUS is the proportion of patients who underwent EUS before endoscopic papillectomy.
APC, argon plasma coagulation; EUS, endoscopic ultrasound; peri-AMP, periampullary; OS, overall survival.
Table 1 also details the EP techniques employed. Submucosal lifting with normal saline was performed in 115 patients (83.9%). Thermal ablation was applied in 10 patients (7.3%) postresection to address remnant tissue or immediate bleeding, while en bloc resection was achieved in 78 patients (56.9%). Pancreatic and biliary stents were placed in 84.7% and 16.8% of cases, respectively, with the primary reason for omitting intubation being failed cannulation of the duct. No significant differences were observed between the two groups in the rates of submucosal injection (82.8% vs 90.5%; p = 0.573), en bloc resection (57.8% vs 52.4%; p = 0.647), biliary stent placement (15.5% vs 23.8%; p = 0.350), or pancreatic stent placement (85.3% vs 81.0%; p = 0.607).
Histologically, 76.7% of the resected lesions were identified as ampullary adenomas with at most low-grade dysplasia (LGD), while 23.4% exhibited high-grade dysplasia (HGD). These lesions were further classified histologically into intestinal (89.8%) and PB (10.2%) subtypes. Morphologically, the resected adenomas were categorized as villous/tubulovillous (51.1%), tubular (40.1%), with 12 cases of unknown morphology. The results indicated that patients with the PB subtype were more likely to experience recurrence (6.9% vs 28.6%; p = 0.003). Additionally, patients with HGD exhibited a higher recurrence rate (19.8% vs 42.9%; p = 0.022). The median follow-up duration in our cohort was 17.7 months (interquartile range (IQR) 11.7–37.5), and it did not significantly differ between the two group (p = 0.626).
Risk factors for recurrence
Among the 137 patients who underwent complete resection, 21 (15.3%) experienced recurrence. Of the 21 recurrence cases, all recurrences remained adenomatous, with 11 cases (52.4%) successfully managed by repeat endoscopic intervention. During subsequent follow-up, two patients experienced re-recurrence at 6 and 10 months postoperatively. Six patients (28.6%) underwent pancreatoduodenectomy. In the Cox proportional hazards univariate analysis, the identified risk factors for recurrence included FAP, PB subtype, and adenomas with HGD. Multivariate analysis further confirmed these factors as significant (Table 2). Specifically, FAP (HR = 9.3; 95% CI: 2.8–30.4; p < 0.001), PB subtype (HR = 3.2; 95% CI: 1.5–11.3; p = 0.006), and HGD (HR = 2.6; 95% CI: 1.1–6.2; p = 0.036) were all significant predictors of recurrence in the Cox proportional hazards multivariate model (Table 2).
Univariable and multivariable analysis.

EUS intraductal involvement and location subtype in EUS is the proportion of patients who underwent EUS before endoscopic papillectomy.
APC, argon plasma coagulation; CI, confidence interval; EUS, endoscopic ultrasound; FAP, familial adenomatous polyposis; HR, hazard ratio.
The Kaplan–Meier curve illustrating the cumulative probability of recurrence is presented in Figure 4. In the survival analysis stratified by histologic subtype (Figure 5(a)), recurrence was observed in 6 (42.9%) of the 14 patients with the PB subtype, compared to 15 (12.2%) of the 123 patients with the intestinal subtype. A log-rank test confirmed that this difference was statistically significant (HR = 3.6; 95% CI: 1.4–9.3; p = 0.004). Additionally, a significant difference in recurrence rates was observed between patients with HGD and those with LGD, as indicated by the log-rank test (Figure 5(b); HR = 3.1; 95% CI: 1.3–7.4; p = 0.008). The Kaplan–Meier analysis of cumulative recurrence according to FAP history also revealed significant differences between the two groups (Figure 5(c); HR = 7.3; 95% CI: 2.4–22.6; p < 0.001).
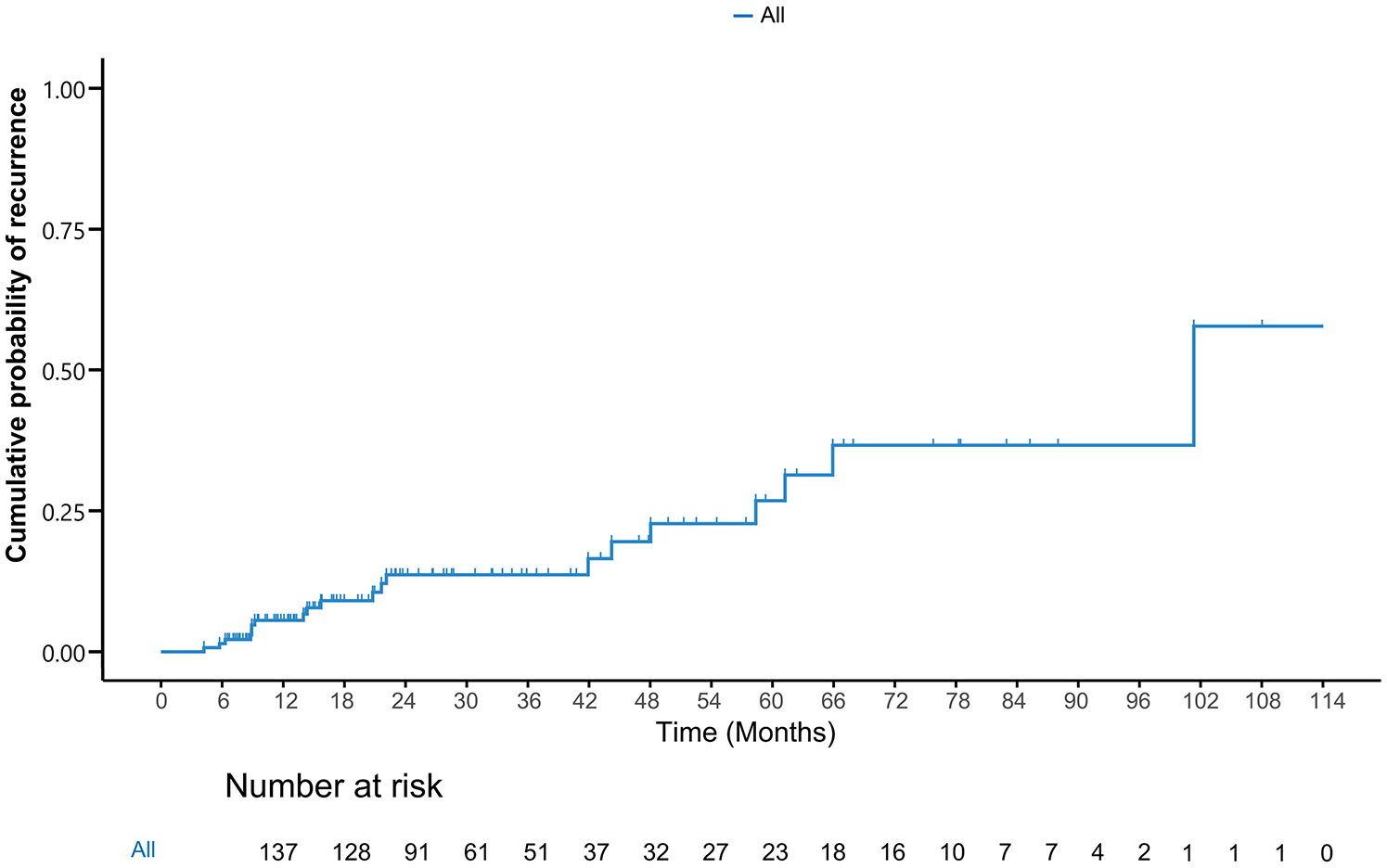
Kaplan–Meier for cumulative probability of recurrence.

Kaplan–Meier for cumulative probability of recurrence based on subgroup. (a) Kaplan–Meier curve for cumulative probability of recurrence according to histopathological subtype. (b) Kaplan–Meier curve for cumulative probability of recurrence according to histology. (c) Kaplan–Meier curve for cumulative probability of recurrence according to FAP history. (d) Kaplan–Meier curve for cumulative probability of recurrence according to histology and histopathological subtype.
Comparison of recurrence in subgroups
To assess the impact of potential confounding factors on the recurrence rate of ampullary adenomas, a comprehensive subgroup analysis was conducted. The analysis indicated no significant interaction between the assessed factors, as evidenced by a p value greater than 0.05. As depicted in Figures 6 and 7, the forest plots for both the PB subtype and HGD show HRs significantly greater than 1, identifying these factors as substantial predictors of recurrence. Specifically, the PB subtype was associated with a markedly increased risk of recurrence, as illustrated in Figure 6, while adenomas with HGD exhibited a higher likelihood of recurrence compared to those with LGD, as shown in Figure 7.
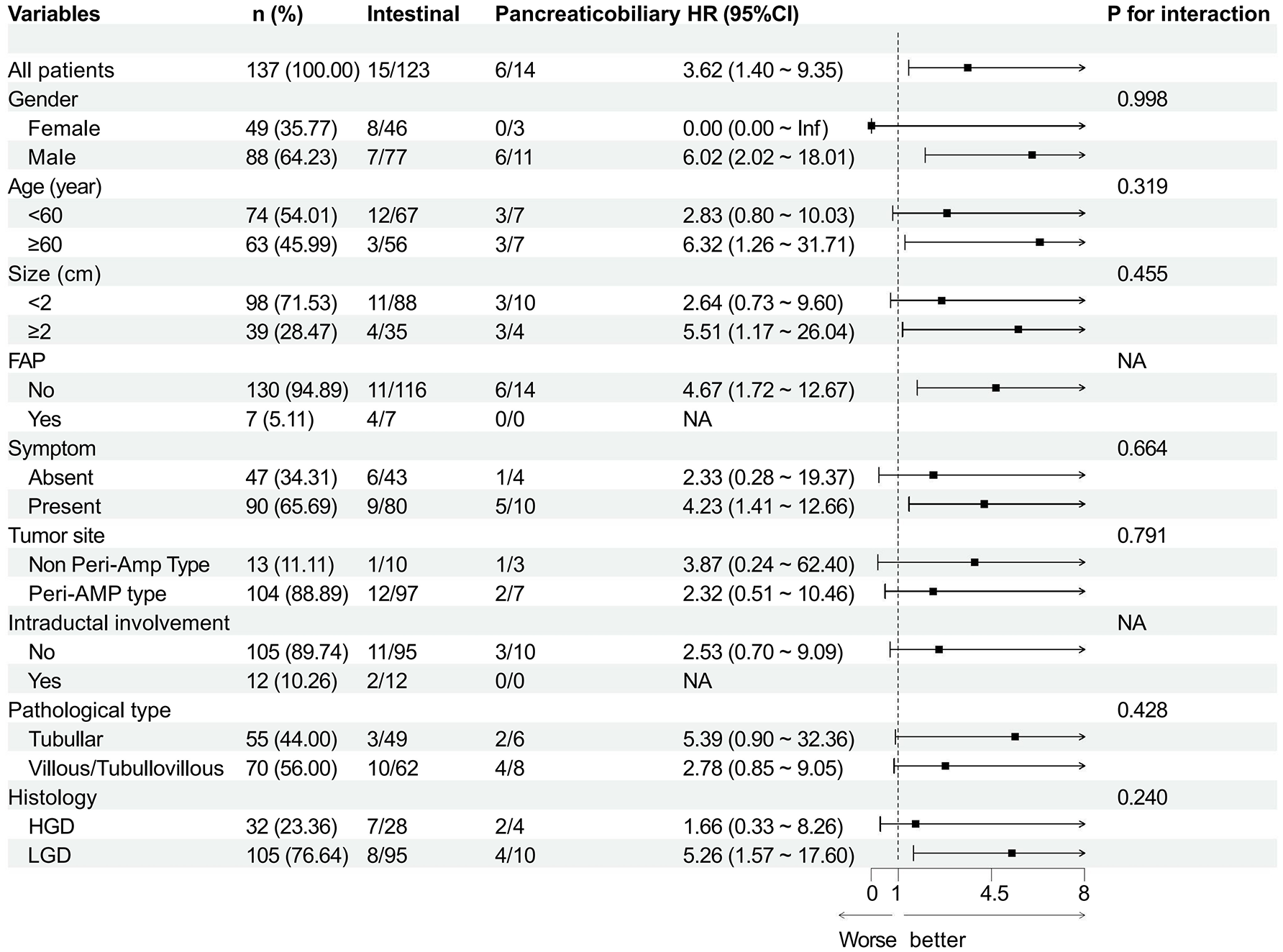
Subgroup analyses of the recurrence on histopathological subtype according to baseline characteristics. HRs were estimated in a Cox proportional hazards regression model.
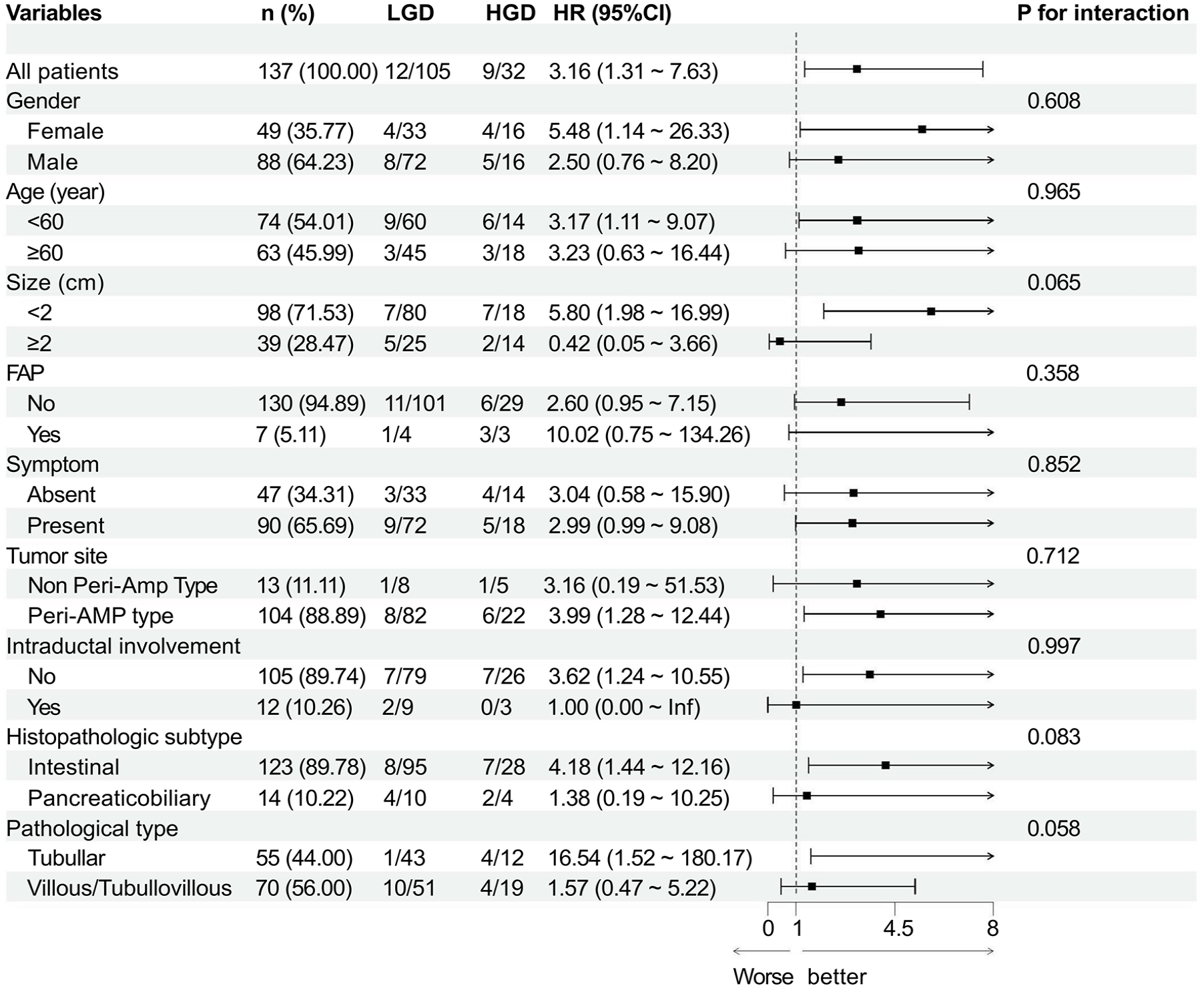
Subgroup analyses of the recurrence on histology according to baseline characteristics. HRs were estimated in a Cox proportional hazards regression model.
Discussion
EP has been established as a safe and effective treatment for ampullary adenoma, serving as a viable alternative to surgical resection for select ampullary lesions. 17 However, challenges associated with endoscopic resection include the potential for incomplete resection and recurrence. 5 This single-center retrospective case–control study aimed to identify the risk factors associated with recurrence. Among the 137 patients who achieved complete resection, a recurrence rate of 15.3% was observed. The median time to recurrence in our cohort was 16.2 months (IQR 9.9–33.9), consistent with the heterogeneous recurrence patterns reported in prior studies.25,26 FAP (HR = 9.3; 95% CI: 2.8–30.4; p < 0.001), PB subtype (HR = 3.2; 95% CI: 1.5–11.3; p = 0.006), and HGD (HR = 2.6; 95% CI: 1.1–6.2; p = 0.036) were identified as significant risk factors for the recurrence of duodenal papillary adenomas. Notably, the impact of PB subtype and HGD on recurrence rates persisted across various subgroups.
The collection of biopsy samples prior to papillectomy is crucial in developing appropriate treatment plans for patients. The ASGE recommends obtaining biopsy specimens from all ampullary lesions suspected of neoplasia before proceeding with endoscopic resection. 27 However, the diagnostic concordance between preoperative biopsies and subsequent ampullectomy findings varies significantly, with agreement rates ranging from 45% to 85%, and a relatively high rate of false negatives. 28 In some studies, the inclusion of lesions was based on biopsy specimens, which may introduce bias. In this retrospective case–control study, all lesions were diagnosed based on postoperative specimens, thereby minimizing potential diagnostic inaccuracies.
Ampullary lesions detected after EP are typically classified as remnant, residual, or recurrent. Distinguishing between remnant and recurrent lesions can be challenging, and clinical differentiation often depends on the timing of lesion discovery. 28 To ensure accurate assessment of recurrence, the minimum follow-up duration was set at 6 months, with all patients undergoing at least one endoscopic examination within this period.
FAP was the most significant risk factor for recurrence in our study. Although the HR was high (HR = 9.3), the wide CI (95% CI: 2.8–30.4) reflects the small size of this subgroup. Ma et al. 29 reported a recurrence rate of 58.3% among FAP patients, attributing this elevated risk to the genetic nature of FAP, which predisposes patients to the development of multiple adenomas. Han et al. 26 suggests that while initial recurrence risks may be similar between FAP and non-FAP patients, those with FAP are more susceptible to recurrence over long-term follow-up. A retrospective multicenter study analyzed the outcomes of EP in matched cohorts of FAP-related and sporadic ampullary lesions, although the difference was not significant in recurrence rates, patients with FAP relapsed later than sporadic lesions. 30 Current guidelines recommend visualization of the ampulla during esophagogastroduodenoscopy for patients with FAP 31 support endoscopic surveillance at intervals based on the appearance of the ampulla. Endoscopic surveillance and biopsy sampling of the papilla to assess for histologic progression may be sufficient for most patients. 32 Our data support this recommendation that recurrences in FAP patients were observed late in follow-up, emphasizing the need for indefinite, risk-adapted surveillance in this group.
The ampullary region comprises two distinct epithelial types: the ampulla-duodenum, formed by intestinal epithelium, and the surrounding areas, covered by mucinous epithelium derived from the PB duct. 33 It is widely accepted that adenomas and carcinomas in these regions originate from their respective epithelial tissues. Several studies have highlighted the histological similarities between adenomas and adenocarcinomas. 34 Previous research has examined the prognostic implications of histopathological subtypes in carcinoma, noting that the PB subtype is associated with worse outcomes.35,36 Bhandare et al. 37 identified that margin involvement, perineural invasion, poor tumor differentiation, and PB subtype were associated with poor outcomes in a cohort of 572 patients with ampullary adenocarcinoma. To date, no other studies have evaluated the differential impact of histopathological subtypes on adenoma recurrence. In our study, the PB subtype exhibited a higher recurrence rate compared to the intestinal subtype. To rule out potential confounding factors, we conducted a subgroup analysis, and the impact of histopathological subtypes on recurrence was consistent across all subgroups.
Our study demonstrated a significant disparity in recurrence rates between patients with HGD and those with LGD, with HGD patients exhibiting a notably higher recurrence rate. HGD is characterized by increased cellular atypia and architectural distortion, indicating that the adenomatous tissue is at a more advanced stage of neoplastic progression. According to our data, the recurrence rate for patients with HGD was 28.1%, significantly higher than the 11.2% observed in LGD cases, consistent with findings from previous studies,8,38 which emphasize closer postresection surveillance in cases with HGD. Kim et al. 39 have suggested that patients with preprocedural HGD may benefit from more radical surgical interventions, such as PD, rather than relying solely on EP, unless contraindicated by high-operative risks. This approach could help achieve complete resection and reduce the likelihood of recurrence. Some studies have reported that lymphovascular invasion or lymph node metastasis was not found in patients with focal cancer within the main adenoma or carcinoma in situ after EP,40,41 suggesting that EP remains a viable option for T1a ampullary carcinoma without local lymphatic or distant metastasis. Our study found a positive association between HGD and recurrence, underscoring the importance of careful preprocedural assessment and vigilant postprocedural monitoring. These findings highlight the importance of tailoring treatment strategies according to the grade of dysplasia to improve patient outcomes and reduce recurrence risk.
Reported recurrence rates after EP vary widely, ranging from 1.1% to 58.3%, 17 influenced by diagnostic criteria and follow-up duration. Our recurrence rate of 15.3% is consistent with prior studies.25,26,42 Prior studies have reported considerable heterogeneity in surveillance durations, the optimal timing, and duration of endoscopic monitoring following EP remain uncertain.26,34 We observed recurrence as late as 126.9 months postresection, underscoring the limitations of surveillance protocols with short follow-up windows. While ESGE suggests a minimum of 5 years for high-risk cases, 28 our findings advocate for potentially extended or lifelong surveillance in patients with FAP, PB subtype, or HGD, and suggest that surveillance intervals and duration may need to be stratified based on individual risk profiles.
While our study provides valuable insights into the outcomes and risk factors associated with EP for ampullary adenomas, the histological and molecular similarities between adenomas and adenocarcinomas emphasize the importance of understanding their pathological features to effectively guide clinical practice. To our knowledge, this study is the first to investigate the recurrence patterns of different histological subtypes in ampullary adenoma patients at risk of recurrence. Furthermore, we demonstrated that a significant number of recurrences are detected only during long-term follow-up, underscoring the necessity of continued surveillance for these patients.
There are several limitations to this study that warrant consideration. First, the retrospective design inevitably carries the risk of selection bias, particularly due to the absence of standardized indications for EP. However, all procedures were performed in a high-volume tertiary center by experienced endoscopists, and we employed strict inclusion and exclusion criteria to minimize potential biases. Additionally, while the retrospective nature may raise concerns regarding data quality and completeness, our dataset was derived from comprehensive electronic medical records and supplemented by telephone follow-ups to ensure accuracy and continuity. Second, there was considerable variation in follow-up duration, with the longest follow-up extending to 126.9 months, while some patients were monitored for only a few months. To mitigate this, we used Kaplan–Meier survival analysis and Cox proportional hazards modeling to account for censoring and time-to-event differences, thereby enhancing the reliability of recurrence-related findings. Third, although the overall sample size was acceptable, certain subgroups—such as patients with FAP—were relatively small, potentially limiting the statistical power to detect meaningful differences or associations within these specific populations. As such, the findings regarding these subgroups should be interpreted with caution. Despite these limitations, the study offers new insights into the risk factors for recurrence, supported by its statistical power. Future research should focus on prospective, multicenter studies to validate our findings and to develop optimal surveillance strategies post-EP.
Conclusion
In conclusion, this study suggests that while EP remains an effective treatment for ampullary adenomas, there may be a significant risk of recurrence, particularly among patients with FAP, PB subtype, and HGD. These findings underscore the potential importance of careful patient selection and vigilant long-term follow-up, especially for those identified as high-risk. However, given the retrospective nature of our study, these conclusions should be interpreted with caution, and further prospective studies are needed to validate these associations and optimize post-EP surveillance strategies.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848251343419 – Supplemental material for Risk factors for recurrence after endoscopic papillectomy in ampullary adenomas: a retrospective case–control study
Supplemental material, sj-pdf-1-tag-10.1177_17562848251343419 for Risk factors for recurrence after endoscopic papillectomy in ampullary adenomas: a retrospective case–control study by Kun Liu, Yuhang Zhuang, Ningjing Gao, Muhan Ni, Xiang Zhang, Peng Yan, Quan Zhou, Zhao Shi, Pin Wang, Dehua Tang, Ying Lv, Lei Wang and Shanshan Shen in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
