Abstract
Background:
Bleeding is a serious and frequent adverse event that occurs during and after endoscopic papillectomy (EP). Previous studies have highlighted the effectiveness of preventive clipping closure of the resection site in preventing post-EP bleeding. However, the optimal length of closure remained unclear.
Objectives:
We aimed to clarify the optimal clipping length at the post-EP resection site to prevent delayed bleeding.
Design:
This study was a multicenter retrospective cohort study.
Methods:
We retrospectively analyzed patients who were consecutively admitted to nine high-volume centers for EP between November 2003 and October 2023. The primary outcome was the frequency of delayed bleeding based on the closure length. The optimal closure length rate of the resected site to prevent delayed bleeding was determined using a receiver operating characteristic curve. Secondary outcomes were the incidence, treatment outcomes, and risk factors for post-EP delayed bleeding.
Results:
A total of 130 patients who underwent EP were analyzed. Delayed bleeding was observed in 22 (17%) patients, occurring more frequently in cases without clipping closure than in those with clipping closure (28% (13/47) vs 11% (9/83); p = 0.014). Among 83 patients who underwent clipping closure, delayed bleeding occurred more frequently with a closure length rate <65% than in those with a closure rate ⩾65% (25% (5/20) vs 6% (4/63); p = 0.019). Multivariate analysis showed that a closure rate <65% was the risk factor for delayed bleeding (odds ratio, 6.3; 95% confidence interval, 1.2–33; p = 0.030) in cases with clipping.
Conclusion:
Clipping closure was effective in preventing delayed bleeding, and closure length rate ⩾65% of the resected site significantly reduced post-EP delayed bleeding.

Introduction
Endoscopic papillectomy (EP) is widely used as an endoscopic treatment for ampullary tumors, mainly adenomas of the duodenal papilla. Compared to surgery, EP is a less invasive treatment1,2; however, the occurrence of complications, such as bleeding, pancreatitis, and perforation, is a significant concern.3,4
Among complications arising post-EP, bleeding is a serious and the most frequent adverse event, with an incidence of approximately 20%. 5 Although intraoperative bleeding can be immediately detected and treated using several endoscopic hemostatic treatments, delayed bleeding is more difficult to manage and can be life-threatening. Several prophylactic techniques for delayed bleeding have been reported, including endoscopic clipping closure, epinephrine injection, hemostatic spray, and argon plasma coagulation (APC).6–10 Among these, the clipping closure of ulcers caused by EP significantly reduces the incidence of delayed bleeding.6,7 Ideally, clipping as much as possible at the resection site would help prevent bleeding. However, there exists a risk of pancreatic or bile duct obstruction associated with clipping.
At present, the optimal length to be clipped at post-EP resection sites remains unclear. In fact, the clipping procedure was performed with varying degrees of closure, depending on the discretion of each facility. In this study, to clarify the optimal clipping length at the post-EP resection site to prevent delayed bleeding, we conducted a multicenter retrospective study by enrolling patients who underwent EP and analyzing bleeding events for the association between clipping length and delayed bleeding.
Methods
Patients
We retrospectively analyzed patients who were consecutively admitted to nine high-volume centers for EP between November 2003 and October 2023. The inclusion criteria were as follows: (1) patients who had biopsy-proven and morphologically benign-looking ampullary adenoma and (2) patients who had tumors suspected as carcinomas in situ without tolerance for surgery. The exclusion criteria were as follows: (1) patients under 20 years of age, (2) patients with endoscopically unresectable ampullary tumors, (3) patients with tumors with evidence of extension into the pancreatic or bile duct, and (4) patients with tumors of the minor papilla. During the study period, 130 patients who underwent EP met the inclusion criteria. Patient data were collected from the electronic medical records. All patients provided written informed consent for treatment. This study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of our hospital (Approval Number: 2309-028). The reporting of this study confirms the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 11
EP procedure
Experienced endoscopists performed all the EP procedures using side-viewing endoscopes. All lesions were resected with a high-frequency electrosurgical generator (ICC200, VIO300D; Erbe Elektromedizin GmbH, Tubingen, Germany). A blended current from VIO300D (Endocut-I mode, effect 1, duration 4, interval 1) or from ICC200 (Endocut-mode, effect 3) (Output limit 120 W), forced coagulation (Output limit 40 W) were used to transect the lesion. EP was conducted using an oval snare (20–33 mm in diameter, Captivator; Boston Scientific Co., Natick, MA, USA). In procedures where preventive clipping closure was performed, conventional clips (Quick clip pro, EZ clip, HX-610; Olympus Co., Tokyo, Japan; Resolution™; Boston Scientific Co.; Zeo Clip; Zeon Medical Co., Tokyo, Japan) or novel clip devices, namely SureClips (Micro-Tech Co. Ltd, Nanjing, China), which improves rotation and reopening performance, were placed in the area extending from the lower side of the pancreatic orifice to the anal verge of the post-EP ulcer. After clipping closure, biliary and pancreatic stents were inserted to reduce the risk of post-procedural cholangitis and pancreatitis. Clipping closure was applied at a site slightly distal to the pancreatic orifice to avoid blocking it. After placing both a biliary and a pancreatic stent, additional clipping closure was applied proximally if possible. Hemostasis with APC, hemostatic forceps, clipping, or an injection of diluted epinephrine solution was performed in cases where bleeding occurred during the procedure. When performing APC, we limited the ablation to a short duration at a low output (30–40 W) to avoid perforation. Prophylactic hemostasis was not achieved in any patient. Antiplatelet and anticoagulant drugs were either withdrawn or replaced with unfractionated heparin during the EP from 2003 to 2012. From 2012 onward, these agents were administered during EP in accordance with the Japan Gastroenterological Endoscopy Society guidelines; namely, in patients who were at an increased risk of thromboembolism as a result of withdrawing antithrombotic therapy; in this study, antiplatelet agents were not withdrawn, and anticoagulants were replaced by heparin. 12 Withdrawn agents and heparin were generally resumed 24 h after the procedure.
Definitions
The primary outcome of this study was the frequency of delayed bleeding based on the closure length. Two experienced endoscopists, who were blinded to the clinical information of the patients, measured the closure length rate by retrospectively reviewing the endoscopic images, and the mean rate was used as the closure length rate. The closure length rate was computed as a percentage of the actual area clipped within the available area for clipping at the resected site, extending from the lower side of the pancreatic orifice to the anal verge of the post-EP ulcer (Figure 1). The measurement within the above area was performed using the measurement software ImageJ (Fiji; National Institutes of Health, Bethesda, MD, USA). The optimal closure length rate to prevent delayed bleeding was determined using receiver operating characteristic (ROC) curve analysis.

The definition of preventive closure length rate. (a) The area from the lower side of the pancreatic orifice to the anal verge of the post-EP ulcer was regarded as the area available for clip placement (red arrow). The length of this area was defined as 10. (b) The closure length rate was computed as the percentage of the actual clipping closure area (blue arrow) within the area available for clipping closure. This schema demonstrates a closure length rate of 80% (the length of the blue arrow was measured as 8). (c) This schema demonstrates a closure length rate of 30% (the length of the blue arrow was measured as 3). Photos below the schema are representative endoscopic images corresponding to each schema.
The secondary outcomes of this study were the incidence, treatment outcomes, and risk factors for delayed bleeding. Post-EP bleeding was classified as intraoperative bleeding or delayed bleeding based on the timing. Intraoperative bleeding was defined as bleeding that occurred during the endoscopic procedure and required hemostasis. Delayed bleeding was defined as bleeding that required hemostatic treatment after the completion of the endoscopic procedure. Endoscopic surveillance for delayed bleeding was conducted when apparent bleeding symptoms, such as melena or hematemesis, or a decrease of at least 2.0 g/dL in the hemoglobin concentration, were present. Oozing during the scheduled endoscopy before hospital discharge was not considered delayed bleeding.
The absence of evident bleeding a few minutes after the hemostasis procedure was regarded as successful. Severity grades of bleeding were assessed in accordance with the lexicon and severity grading system of the American Society for Gastrointestinal Endoscopy. 13
To identify the factors associated with delayed bleeding that have been highlighted in previous studies are considered clinically significant, the following variables were considered: age, sex, underlying disease, anti-gastric ulcer drug use, antiplatelet and anticoagulant use, heparin replacement, procedure time, neoplastic tumor, lesion diameter, piecemeal resection, electrosurgical generator, intraoperative bleeding, biliary stent placement, pancreatic stent placement, preventive clipping closure, clip type, post-EP pancreatitis, and post-EP cholangitis.
Statistical analyses
Categorical variables were reported as percentages and continuous variables were reported as medians and interquartile ranges (IQR). Wilcoxon’s rank-sum test and the Kruskal–Wallis test were used to compare continuous data. Fisher’s exact test was used to compare categorical data. Continuous variables were divided into two groups using a cutoff value defined by the ROC curve for univariate and multivariate analyses. To determine the appropriate closure length rate using the ROC curve, the true-positive rate was defined as the proportion of cases with a closure length rate under the cutoff rate in delayed bleeding cases, and the true-negative rate was defined as the proportion of cases with a closure length rate over the cutoff rate in cases without delayed bleeding. Delayed bleeding risk factors were analyzed in the following two steps. First, univariate logistic regression identified potential risk factors among intervenable factors during the procedure. When the odds ratio (OR) could not be calculated due to quasi-complete separation, it was calculated using penalized maximum likelihood logistic regression. 14 Then, multivariate logistic regression was conducted to adjust for confounding effects, with candidate confounders selected according to the modified disjunctive cause criterion. 15 According to this criterion, all variables except gender, age, and those with missing data were included as candidate confounders. The factors actually analyzed were listed below each table. The ORs and their 95% confidence interval (CI) were calculated. Differences with p values <0.05 were considered statistically significant. Analyses were performed using the JMP Pro17 software for Mac (SAS Institute Inc., Cary, NC, USA) and Stata 18/MP (Stata Crop, College Station, TX, USA).
Results
Patient characteristics
In total, 130 patients who underwent EP for ampullary lesions were included in this study. The clinical characteristics of the patients are summarized in Table 1. The median age of the participants was 66 (IQR: 57–74); 63% of the patients were men. The median lesion diameter was 15 mm (IQR: 12–20). The final diagnoses were adenoma in 105 (81%), adenocarcinoma in 9 (6.9%), neuroendocrine neoplasm (grade 1) in 2 (1.5%), non-neoplastic lesion in 13 (10%), and loss of specimen in 1 (0.77%) patient. Underlying diseases were present in 52 (40%) patients, including hypertension in 37 (28%), diabetes mellitus in 20 (15%), cardiac disease in 14 (11%), familial adenomatous polyposis in 14 (11%), liver cirrhosis in 3 (2.3%), hemodialysis in 3 (2.3%), and cerebrovascular disease in 2 (1.5%) patients. Overall, 15 (12%) patients used antiplatelets or anticoagulants.
Clinical characteristics of the patients.
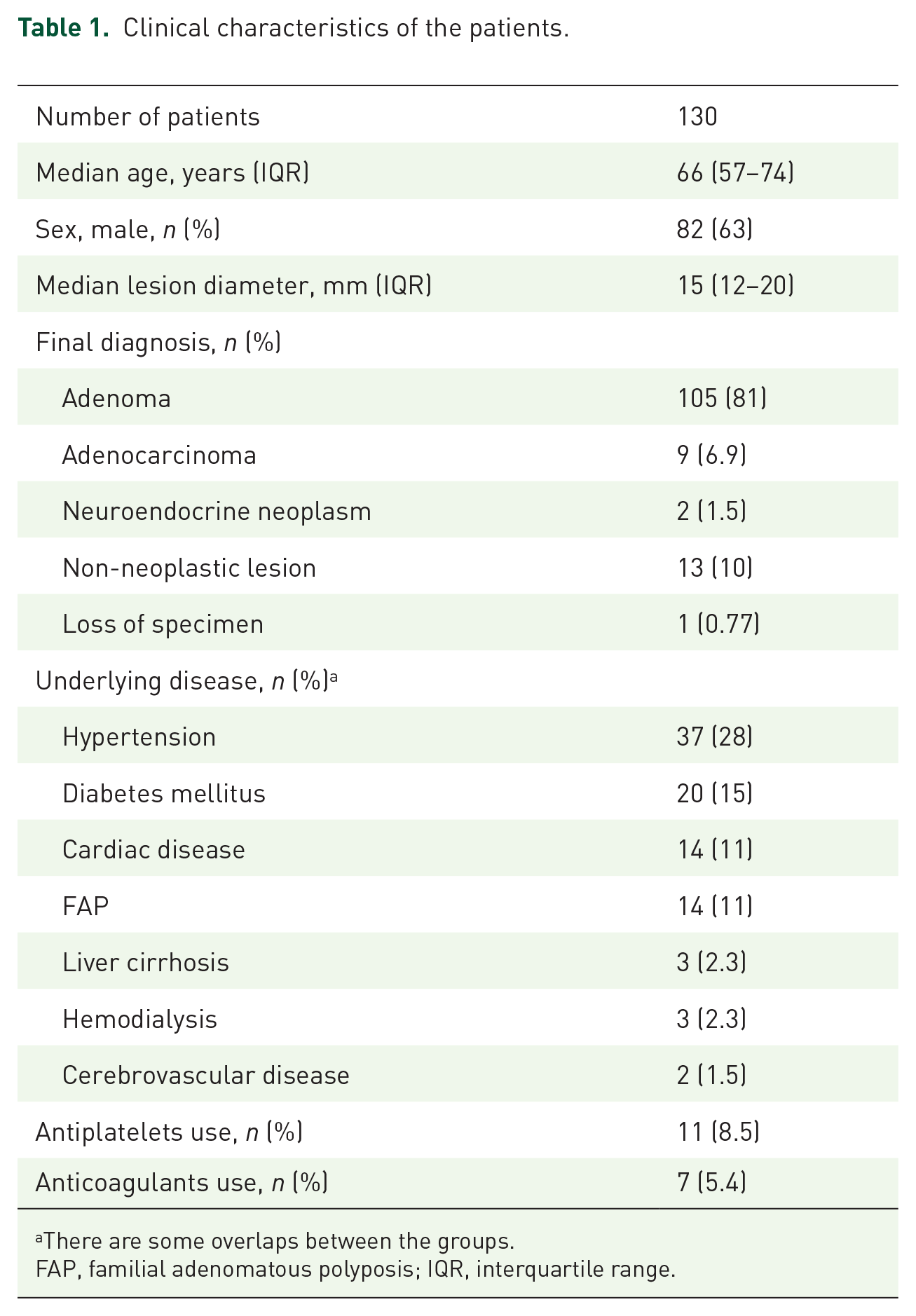
There are some overlaps between the groups.
FAP, familial adenomatous polyposis; IQR, interquartile range.
Procedural details and adverse events
Procedural details and adverse events are presented in Table 2. The median procedure time was 40 min (IQR: 28–55). En bloc resection was performed in 115 (88%) patients. A biliary stent and a pancreatic stent were placed in 119 (92%) and 119 (92%) patients, respectively, and 111 patients (85%) had both biliary and pancreatic stents successfully placed.
Details of the procedures and adverse events.

There are some overlaps between the groups.
EP, endoscopic papillectomy; IQR, interquartile range.
Preventive clipping closure was performed in 83 (64%) patients, with a median clipping closure rate of 71% (IQR: 65–79). The median number of clips used in cases with clipping closure was 3 (IQR: 2–4). A total of 48 (37%) patients reported EP-related adverse events. The median hospital stay was 11 days (IQR: 9–13).
An overview of post-EP bleeding is shown in Figure 2. Post-EP bleeding was observed in 37 (28%) patients; 23 patients exhibited intraoperative bleeding and 22 patients displayed delayed bleeding. Patients with intraoperative bleeding were successfully treated with endoscopic hemostasis, although eight patients still developed delayed bleeding. Details of the delayed bleeding and its management are presented in Supplemental Table 1. The median period of emergent delayed bleeding was 2.5 days (IQR: 1.8–4.5; Supplemental Figure 1). The symptoms of delayed bleeding were melena or hematemesis in 20 (91%), hypotension in 3 (14%), tachycardia in 2 (9.1%), and acute cholangitis in 1 (4.5%) patient. The bleeding sites were the frenulum and ampulla in 14 (64%) and 8 (36%) patients, respectively. The patients with delayed bleeding could have also been managed with endoscopic treatment; however, two to four transfusion units were required in two (9.1%) patients. Seven (35%) patients experienced recurrent bleeding and underwent endoscopic treatments again. Ultimately, all patients achieved endoscopic hemostasis without bleeding.

Overview of the bleeding events after EP.
Among the non-bleeding adverse events, post-EP pancreatitis occurred in 13 (10%) patients, perforation in 3 (2.3%), and cholangitis in 2 (1.5%) patients. The patients diagnosed with post-EP pancreatitis were treated with fluid replacement therapy and pancreatic enzyme inhibitors. All perforations were minor, and treatment consisted of fluid replacement and the administration of antibiotics. Although one patient with cholangitis following stent occlusion, induced by blood clot formation, underwent hemostasis and stent exchange, the other patients were treated conservatively with fluid replacement and/or antibiotics. None of the patients required surgery or were admitted to the intensive care unit.
Association between clipping closure length and delayed bleeding
Among the 130 cases, preventive clipping closure was performed in 83 (64%) patients. With regard to the presence or absence of a preventive clipping closure, delayed bleeding occurred more frequently in cases without preventive clipping closure (28% (13/47) without vs 11% (9/83) with preventive clipping closure; p = 0.014). Univariate analysis showed that no clipping closure (OR, 3.1; 95% CI, 1.2–8.1; p = 0.014) and intraoperative bleeding (OR, 3.6; 95% CI, 1.3–10; p = 0.012) were risk factors for delayed bleeding in all cases (Table 3). And multivariate analysis with a selection of confounding factors according to the modified disjunctive cause criterion identified no clipping closure as a risk factor (OR, 4.1; 95% CI, 1.3–12; p = 0.013). Variance inflation factors (VIFs) for the explanatory variables, calculated using linear regression analysis, ranged from 1.09 to 1.33, indicating that multicollinearity was not a concern. Among the 83 cases with preventive clipping closure, the median value of closure length rate was 71% (IQR: 65–79). The appropriate cutoff rate of closure length was determined to be 65% by the ROC curve (Figure 3). The ROC area under the curve was 0.75. Delayed bleeding occurred more frequently in cases with a closure length rate <65% compared with those having a closure length rate ⩾65% (25% (5/20) vs 6% (4/63), p = 0.019). In the cases with preventive clipping closure, multivariate analyses with a selection of confounding factors according to the modified disjunctive cause criterion identified a closure length rate <65% as the risk factor for delayed bleeding (OR, 6.3; 95% CI, 1.2–33; p = 0.030; Table 4). VIFs for the explanatory variables, calculated using linear regression analysis, ranged from 1.04 to 1.30, indicating that multicollinearity was not a concern.
Univariate and multivariate analyses of the predictive factors for delayed bleeding.

The p-value was calculated using Fisher’s exact test.
The multivariate analysis for preventive clipping closure included the following variables as potential confounders: underlying disease, anti-gastric ulcer drug use, antiplatelets and anticoagulant use, procedure time, neoplastic tumor, lesion diameter, piecemeal resection, intraoperative bleeding, high-frequency electrosurgical generator, biliary stent placement, and pancreatic stent placement.
CI, confidence intervals; NA, not available; OR, odds ratio.

Determining the optimal closure length rate after EP by ROC curve.
Univariate and multivariate analyses of the predictive factors for delayed bleeding in cases with preventive clipping closure.

The p-value was calculated using Fisher’s exact test.
The multivariate analysis for preventive clipping closure rate included the following variables as potential confounders: underlying disease, anti-gastric ulcer drug use, antiplatelets and anticoagulant use, neoplastic tumor, lesion diameter, intraoperative bleeding, high-frequency electrosurgical generator, biliary stent placement, and SureClip use.
CI, confidence intervals; NA, not available; OR, odds ratio.
Discussion
To the best of our knowledge, this is the first study to investigate whether clipping closure length is correlated with post-EP delayed bleeding. Delayed bleeding occurred more frequently in patients with a closure length rate <65% than in those with a closure length rate ⩾65% (25% (5/20) vs 6% (4/63), p = 0.019). In addition, a closure length rate of <65% was the sole risk factor for delayed bleeding (OR, 4.7; 95% CI, 1.1–21; p = 0.039) in the multivariate analyses. These results clarify the extent of clipping needed to prevent delayed bleeding and are beneficial in clinical practice.
While many trials have tried to prevent post-EP delayed bleeding using various endoscopic techniques, such as clipping closure, epinephrine injection, hemostatic spray, and APC,6–10 a previous study reported that post-EP clipping closure was an effective and safe method for preventing delayed bleeding without increasing the risk of post-EP pancreatitis or cholangitis. 6 The study reasoned that preventive closure of the frenulum would be effective due to its rich blood supply. Indeed, the predominant delayed bleeding site in this study was the frenulum in 14 (64%) patients. In addition, clipping closure directly sutures the post-EP ulcer and prevents exposure to bile and pancreatic juice. Therefore, clipping closure is a good option for preventing post-EP delayed bleeding. Among the three cases of perforation, one case was a delayed perforation that occurred in a patient without the application of clipping closure. Perforation is a rare complication, and the efficacy of clips for preventing delayed perforation remains unclear.
Although previous studies have indicated the effectiveness of preventive clipping closure for post-EP delayed bleeding, the optimal degree of closure remains unclear. Although clipping may be ideal for preventing bleeding, it may also be a risk factor for pancreatic or bile duct obstruction. Therefore, clinicians should appropriately use clipping sutures such that they do not block the pancreatic orifice after placing the pancreatic stent to prevent pancreatitis. In clinical practice, achieving complete clipping closure is sometimes challenging because stents interfere or the resection surface is too large to be adequately closed with clips. However, the results of this study suggest that a closure length rate of at least 65% is necessary to prevent post-EP delayed bleeding, providing valuable guidance for the clinicians performing EP procedures.
While previous studies focusing on the risk factors of post-EP bleeding are limited, a few studies have listed these risk factors, including the use of anticoagulants, large-sized ampullary tumors, hemorrhage during EP, and non-endoscopic clipping closure.16–18 Our study revealed the lack of preventive clipping closure and the presence of intraoperative bleeding as significant risk factors for delayed bleeding. After intraoperative bleeding, 35% of cases also developed delayed bleeding (OR, 4.0; 95% CI, 1.3–12; p = 0.015), while all intraoperative bleeding was controlled by immediate endoscopic hemostasis in our study. Previous reports have suggested that the premature shedding of blood scabs during intraoperative bleeding may be related to delayed bleeding. 11 In addition, thermal coagulation of endoscopic hemostasis during intraoperative bleeding may also cause further ulcer formation. Clinicians should pay particular attention to patients with intraoperative bleeding on post-EP days to reduce that risk or, at least, to be able to manage it in a safe setting, such as an extended fasting period or systematic prolonged hospitalization.
Regarding antithrombotic agents, previous studies have reported differing results. One study demonstrated that patients administered anticoagulant agents had a significantly increased risk of delayed bleeding. 17 Conversely, another study did not observe any association between the use of antithrombotic agents and the incidence of post-EP adverse events. 19 In our study, the use of antithrombotic agents and heparin replacement showed no significant differences in delayed bleeding, even when restricted to patients using anticoagulants. Further studies with larger sample sizes are warranted to examine the association between antithrombotic agents and delayed bleeding.
This study had some limitations. First, although this was a multicenter retrospective study, the number of patients with delayed bleeding was small. Second, some treatment choices for hemostasis of intraoperative bleeding were not standardized but were chosen on a case-by-case basis. There is a possibility of postoperative bleeding induced by hemostasis for intraoperative bleeding, and the influence of delayed bleeding may vary depending on the hemostasis method used. Third, the study included patients who had been treated for more than two decades. Some techniques have changed, new knowledge has been introduced, and new endoscopic devices have been introduced during the study period. We compared the improved clips (SureClip) with the conventional clips, but no differences were observed in the preventive hemostatic effects. In addition, the high-frequency device has been upgraded from ICC to VIO300, but no differences in bleeding rates were noted. As for future study designs, efforts to control these factors will be necessary, such as limiting the study duration or including only cases that utilize specific techniques and devices. Fourth, the patients were included before and after the release of the current guidelines regarding the use of antithrombotic agents. Therefore, the management of patients using antithrombotic agents varied depending on the study period. In future studies, it is necessary to standardize the management and monitoring of antithrombotic therapy according to the latest clinical guidelines to more accurately assess the impact of antithrombotic therapy on the risk of delayed bleeding. Fifth, this was a multicenter study, and it is possible that differences in the technical level of endoscopists and postoperative care between different hospitals may have influenced the results. Although these are indirect metrics, it would be necessary to investigate the relationship between the results and factors such as the annual number of EP procedures in each hospital and hospital size.
Conclusion
Clipping closure is effective in preventing post-EP delayed bleeding. Although achieving complete clipping closure is sometimes challenging in clinical practice; however, a closure length rate of at least 65% of the resected site is important to reduce post-EP delayed bleeding.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251326450 – Supplemental material for Clipping closure length is a crucial factor for delayed bleeding after endoscopic papillectomy: a retrospective multicenter cohort study
Supplemental material, sj-docx-1-tag-10.1177_17562848251326450 for Clipping closure length is a crucial factor for delayed bleeding after endoscopic papillectomy: a retrospective multicenter cohort study by Yuki Fujii, Kazuyuki Matsumoto, Kiyoaki Ochi, Hitomi Himei, Ichiro Sakakihara, Eijiro Ueta, Tatsuya Toyokawa, Ryo Harada, Taiji Ogawa, Takeshi Tomoda, Hironari Kato, Ryosuke Sato, Taisuke Obata, Akihiro Matsumi, Kazuya Miyamoto, Daisuke Uchida, Shigeru Horiguchi, Koichiro Tsutsumi and Motoyuki Otsuka in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-2-tag-10.1177_17562848251326450 – Supplemental material for Clipping closure length is a crucial factor for delayed bleeding after endoscopic papillectomy: a retrospective multicenter cohort study
Supplemental material, sj-jpg-2-tag-10.1177_17562848251326450 for Clipping closure length is a crucial factor for delayed bleeding after endoscopic papillectomy: a retrospective multicenter cohort study by Yuki Fujii, Kazuyuki Matsumoto, Kiyoaki Ochi, Hitomi Himei, Ichiro Sakakihara, Eijiro Ueta, Tatsuya Toyokawa, Ryo Harada, Taiji Ogawa, Takeshi Tomoda, Hironari Kato, Ryosuke Sato, Taisuke Obata, Akihiro Matsumi, Kazuya Miyamoto, Daisuke Uchida, Shigeru Horiguchi, Koichiro Tsutsumi and Motoyuki Otsuka in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The authors would like to express our gratitude to Dr Toshiharu Mitsuhashi from the Center for Innovative Clinical Medicine in Okayama University Hospital for conducting the statistical analysis for this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
