Abstract
Background:
In 2023, the diagnostic criteria for gastroesophageal reflux disease (GERD) were updated in the Lyon Consensus 2.0. It is important to investigate the diagnostic accuracy of complementary tests used for GERD diagnosis.
Objectives:
To determine the diagnostic accuracy of mean nocturnal basal impedance (MNBI) measured by pH-metry in patients with suspected GERD compared to the updated 2023 Lyon Consensus Diagnostic Criteria and to identify the optimal cutoff point for diagnosis.
Design:
Diagnostic test study based on a retrospective cohort of patients with suspected GERD.
Methods:
Diagnosis was confirmed using the updated Lyon criteria as the gold standard. We evaluated sensitivity, specificity, and discriminatory ability via area under the receiver operating characteristic curve (AUC-ROC). The best cutoff point was determined using Liu’s method for MNBI, number of reflux episodes, number of acid reflux episodes, DeMeester score, and bolus exposure time. ROC curves were compared using DeLong’s method.
Results:
A total of 55 patients were included, with 26 diagnosed positive and 29 negative for GERD. MNBI yielded an AUC-ROC of 0.77 (95% confidence interval (CI): 0.64–0.89); at 1590 ohms, sensitivity and specificity were 69.2% and 72.4%, respectively. The DeMeester score demonstrated superior discriminatory ability over MNBI (p = 0.03), with an AUC-ROC of 0.90 (95% CI: 0.80–1.00), sensitivity of 92%, and specificity of 93% at a cutoff of 15.6. No significant differences in discriminatory ability were observed when comparing MNBI with other complementary tests (p < 0.05).
Conclusion:
MNBI and other complementary tests showed suboptimal characteristics for GERD diagnosis. These tests should be interpreted alongside clinical, endoscopic, and pH-metry findings. The DeMeester score seems promising, but further prospective studies are needed to prove its utility.
Introduction
Gastroesophageal reflux disease (GERD) has a global age-adjusted incidence of 3793 per 100,000 individuals and an age-adjusted prevalence of 9574 people per 100,000 individuals in 2019.1,2 Associated complications of this disease include dysphagia, chest pain, peptic stricture, esophageal ulceration, Barrett’s esophagus, and esophageal adenocarcinoma. 3
The diagnosis of GERD is based on endoscopic and/or pH-metry findings. In 2023, the Lyon consensus diagnostic criteria for GERD were updated: Los Angeles grade B esophagitis and the requirement of at least 24 h for pH-metry studies were included in these diagnostic criteria. In addition, this consensus proposes complementary tests to support or refute the diagnosis of GERD (Table 1). These complementary tests include the following: symptomatic association of reflux, total number of reflux episodes, mean nocturnal basal impedance (MNBI), the presence of a hypotensive lower esophageal sphincter, hiatal hernia, ineffective esophageal motility/absent contractility, and some histopathological findings on biopsy. 4 Given this update of the GERD diagnostic criteria, it is necessary to evaluate the diagnostic accuracy of the different complementary diagnostic tests, such as MNBI, total number of reflux episodes, number of acidic reflux episodes, bolus exposure time, and the DeMeester score for GERD diagnosis.
Lyon consensus 2.0 for the diagnosis of GERD.
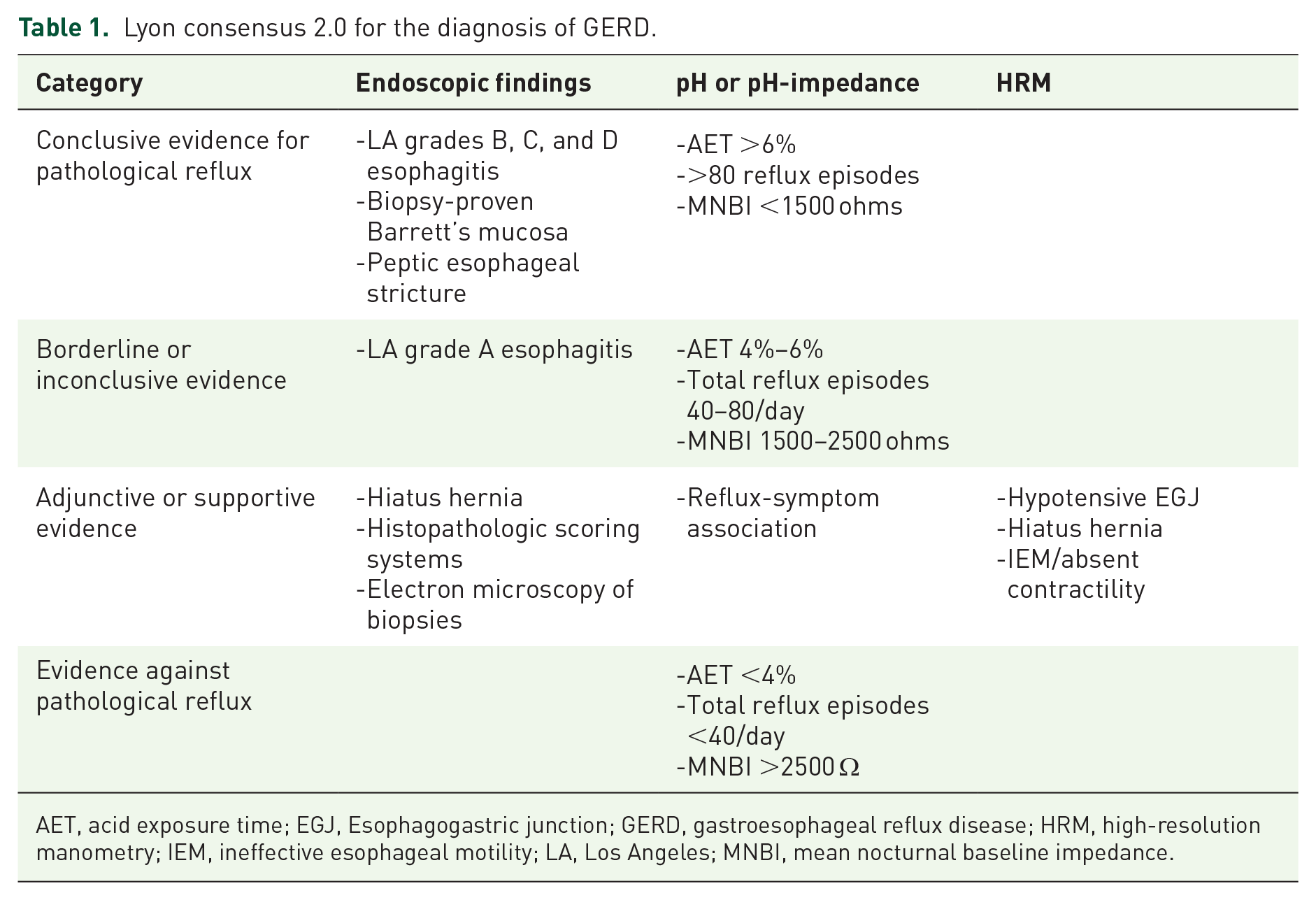
AET, acid exposure time; EGJ, Esophagogastric junction; GERD, gastroesophageal reflux disease; HRM, high-resolution manometry; IEM, ineffective esophageal motility; LA, Los Angeles; MNBI, mean nocturnal baseline impedance.
This study aims to determine the diagnostic accuracy of MNBI measured by pH-metry in patients with suspected GERD compared to the updated 2023 Lyon Consensus Diagnostic Criteria and to identify the optimal cutoff point for diagnosis. In addition, the diagnostic accuracy of other complementary tests, such as the total number of reflux episodes, number of acid reflux episodes, bolus exposure time, and the DeMeester score for the diagnosis of GERD, was evaluated.
Methods
A diagnostic test study was conducted based on a retrospective cohort of patients with suspected GERD who were evaluated at the Hospital Universitario San Ignacio in Bogotá, Colombia, between July 2018 and August 2023. These dates were chosen as they span the period between the publication of the first Lyon consensus and its subsequent update. Inclusion criteria were as follows: patients over 18 years of age with symptoms suggestive of GERD, undergoing pH measurement, and not receiving proton pump inhibitor treatment in the 4 weeks prior to the procedure were included. Typical symptoms (regurgitation, heartburn, or chest pain) and extraesophageal symptoms (cough, laryngitis, asthma, or dental erosions) were considered. In addition, patients had to have an esophagogastroduodenoscopy report. Patients with more than 6 months between the esophagogastroduodenoscopy and pH-metry tests were excluded from this study. This research project was approved by the institutional research committee (FM-CIE-0593-24). The full study protocol is accessible through the institutional repository of Pontificia Universidad Javeriana. The reporting of this study conforms to the Standards for the Reporting of Diagnostic Accuracy Studies (STARD) statement. 5
A consecutive sample of patients who met the inclusion criteria was selected for this study. Sociodemographic characteristics were systematically collected during patient care. Variables related to endoscopic characteristics were systematically gathered prior to the pH-metry test. The pH-metry was performed using a disposable probe (Versaflex Z; Medtronic, Minneapolis, MN, USA) attached to a recorder for 24 h (Digitrapper Recorder, pH-Z; Medtronic) and analyzed using the Reflux Reader Software (Version 6.1.1.11. Medtronic, Minneapolis, USA). The MNBI was calculated by selecting three stable 10-min periods during the night test, avoiding periods with swallowing, reflux, artifacts, or pH drops. The average baseline impedance (BI) for each of these time periods was computed, and the BI values of the three time periods were then averaged to obtain the MNBI value. The DeMeester score was calculated as proposed in the original article, from six parameters number of reflux episodes, number of episodes longer than 5 min, maximal reflux duration, total percentage of time with pH below 4 for the total monitoring, and the percentage of time with pH below 4 in an upright position and supine position, respectively.
GERD was diagnosed according to the updated Lyon criteria: Endoscopy showing esophagitis grade B, C, or D according to the Los Angeles classification, Barrett’s esophagus, peptic stricture, or pH-metry with Acid Exposure Time (AET) >6% in a 24-h study. GERD diagnosis was excluded in patients who did not meet these endoscopic criteria and presented an AET <4% in a 24-h study.
It is important to note that clinical information and reference standard results (GERD diagnosis according to updated Lyon criteria) were available to the performers and readers of each of the index tests (MNBI, total reflux episodes, acidic reflux episodes, bolus exposure time, and the DeMeester score). Since the pH-metry test is derived from the same examination process, it was not feasible to remain unaware of the reference standard results.
Statistical analysis
Qualitative variables were reported using absolute and relative frequencies. Quantitative variables were reported using mean and standard deviation if the assumption of normal distribution was met, or median and interquartile range (IQR) if it was not. The Kolmogorov–Smirnov test with a significance level of 5% (p < 0.05) was used to assess this assumption. Groups were compared based on the presence or absence of GERD using Student’s t-test, Mann–Whitney U test, or Chi-squared test, depending on the variable’s characteristics. All data collected for the index test (pH-metry) and the reference standard (endoscopic findings) were fully accounted for.
MNBI discriminative ability for GERD diagnosis was evaluated using the area under the receiver operating characteristic curve (AUC-ROC). The optimal cutoff point was defined using Liu’s 6 method. In addition, the sensitivity and specificity of the cutoff points of 1500 and 2500 ohms were evaluated as they were recommended in the updated Lyon criteria. 4 The same procedure was used to assess the operating characteristics and discriminative ability of total reflux episodes, acidic reflux episodes, bolus exposure time, and the DeMeester score, using the updated Lyon criteria as the reference standard for GERD diagnosis. Discriminative ability of each test compared to the MNBI was evaluated using the method proposed by DeLong et al. 7 The statistical analysis was performed using STATA software (Stata Statistical Software: Release 16; StataCorp LLC, College Station, TX, USA).
Results
The flow chart for patient selection is shown in Supplemental Figure 1. A total of 55 patients were included, 29 without GERD according to the new Lyon criteria and 26 with a positive diagnosis. Importantly, all data collected for the index test and the reference standard were fully accounted for, ensuring a robust analysis of the diagnostic performance. Of these 26 patients, 3 met the diagnosis only with endoscopic criteria, 21 only with pH-metry criteria, and 2 met the diagnosis by both methods. Two patients had pH-metry values between 4% and 6% but met endoscopic criteria that confirmed GERD diagnosis.
The median age was 62 years (IQR, 52–68), with a higher proportion of males in the GERD group (34.6% vs 10.3%, p < 0.05). Body mass index (BMI) was not different between the groups (26.8 vs 26.7, p = 0.87). Grade B esophagitis was more frequent in the same group (19.2% vs 0%, p < 0.05). As for the pH-metry findings, the GERD group had a higher median AET (9.2% vs 1.4%, p < 0.05), as well as a higher number of reflux episodes (114 vs 55, p < 0.05), number of acidic reflux episodes (93 vs 27, p < 0.05), DeMeester score (36.5 vs 6.6, p < 0.05), and bolus exposure time (2.3 vs 1, p < 0.05). No statistically significant differences were found in the presence of positive symptom association (p = 0.12; see Table 2). MNBI was lower in GERD patients (930 vs 2130 ohms, p < 0.05).
Description of sociodemographic, endoscopic, and pH-metry characteristics according to GERD diagnosis.

AET, acid exposure time; BMI, body mass index; GERD, gastroesophageal reflux disease; IQR, interquartile range.
MNBI discriminative ability for GERD diagnosis was suboptimal (AUC-ROC = 0.77; 95% confidence interval (CI): 0.64–0.89). The cutoff point with the higher simultaneous sensitivity and specificity was 1590 ohms (sensitivity = 69.2%, specificity = 72.4%). MNBI cutoff points of <1500 and >2500 ohms showed a sensitivity of 61% and 72.4%, and a specificity of 88% and 41.4%, respectively (see Figure 1). The number of reflux episodes had a sensitivity of 69% and specificity of 72% (AUC-ROC = 0.80; 95% CI: 0.67–0.92) for a cutoff point of 88 reflux episodes. The number of acidic reflux episodes had a sensitivity of 77% and specificity of 83% (AUC-ROC = 0.85; 95% CI: 0.74–0.96) for a cutoff point of 87 acidic reflux episodes. The DeMeester score had a sensitivity of 92% and specificity of 93% (AUC-ROC = 0.90; 95% CI: 0.8–1.0) for a cutoff point of 15.6 points. Finally, for bolus, exposure time showed a sensitivity of 69% and a specificity of 66% (AUC-ROC = 0.73; 95% CI: 0.59–0.86) for a cutoff point of 1.6%.

ROC curve for mean nocturnal basal impedance for GERD diagnosis.
When comparing the AUC-ROC, it was evident that the AUC-ROC for the DeMeester score was superior to MNBI (p = 0.03). This difference was not statistically significant when comparing MNBI with the total number of reflux episodes (p = 0.67), number of acid reflux episodes (p = 0.21), and bolus exposure time (p = 0.71; Figure 2 and Table 3).

ROC curve for different complementary tests for GERD diagnosis.
Cutoff values and operating characteristics of different diagnostic tests for GERD diagnosis.

p-Value calculated using DeLong’s statistic comparing the ROC area under the curve between the different tests and nocturnal baseline impedance.
AUC-ROC, area under the receiver operating characteristic curve; CI, confidence interval; GERD, gastroesophageal reflux disease.
Finally, a sensitivity analysis, including only patients with an interval between the endoscopy and pH-metry of up to 90 days, showed similar result, demonstrating that DeMeester score had a better area under the curve (AUC-ROC = 0.86; 95% CI: 0.71–1.00) compared to the MNBI (AUC-ROC = 0.78; 95% CI: 0.62–0.94), but did not achieve statistical significance (p = 0.35).
Discussion
MNBI showed suboptimal operational characteristics for the diagnosis of GERD. Other adjunctive tests, such as total number of reflux episodes, number of acid reflux episodes, and bolus exposure time, also showed similar suboptimal operational characteristics. The DeMeester score stands out for its high sensitivity and specificity for GERD diagnosis in our study, with an AUC-ROC superior to that of the MNBI.
In our study, MNBI exhibited suboptimal operational characteristics for GERD diagnosis with the updated Lyon criteria. MNBI is a surrogate measure of esophageal barrier integrity. Gastric acid exposure to the esophagus causes dilation of the intercellular spaces, resulting in a decrease in impedance. 8 Therefore, a lower MNBI value indicates a higher probability of GERD. A multicenter study in healthy controls found that an MNBI measurement of ⩽1500 ohms was within the 5th percentile of the population, while an MNBI measurement of ⩾2500 ohms corresponded to the 95th percentile or higher. 9 Although there is geographic and commercial variability in the MNBI normality curve, these values were set as complementary criteria for GERD diagnosis with the updated Lyon consensus. 4 However, this curve, assumed as “normal,” has not been validated in different settings or across different commercial brands, which could explain its suboptimal operational characteristics. In our study, the best cutoff point was 1590 ohms, showing sensitivity and specificity close to 70%. For 1500 and 2500 ohms cutoff points, sensitivity was 61% and 88%, and specificity was 72.4% and 41.4%, respectively, when compared with the new Lyon criteria as the gold standard. Similar data have been reported in other studies. Cutoff points of 1500 and 2500 ohms, using the initial Lyon criteria as the gold standard, showed sensitivities ranged from 70% to 80% and specificities from 53% to 91%.10,11 Therefore, it is suggested to interpret the MNBI in conjunction with other clinical, endoscopic, and pH-metry findings to support or refute the diagnosis of GERD, just as recommended in the updated Lyon consensus.
Other complementary tests (total number of reflux episodes, acid reflux episodes, and bolus exposure time) also showed suboptimal operational characteristics for GERD diagnosis in our study. These findings are difficult to compare with previous evidence because the different cutoff points and outcomes are reported in each report.
In our study, the best cutoff point for the total number of reflux episodes was >88, with a suboptimal diagnostic accuracy. This is a similar value to that reported in the updated Lyon consensus (>80 total reflux episodes) as evidence supporting GERD. Other studies have shown similar findings with a cutoff point >80, with a sensitivity of 50%, and a specificity of 71% for predicting non-response to Proton Pump Inhibitors (PPIs). 12
For acid reflux number and bolus exposure time, there is low-quality evidence from studies with an inappropriate disease spectrum (i.e., comparing healthy controls with patients with confirmed GERD diagnosis). Therefore, it is difficult to reliably determine the operational characteristics of these tests.13,14 Thus, like MNBI, our data suggest that the total number of reflux episodes, acid reflux episodes, and bolus exposure time should be interpreted in conjunction with other clinical, endoscopic, and pH-metry findings to support or refute GERD diagnosis.
In our study DeMeester score was notable, considering its sensitivity and specificity exceeding 90% and its superior AUC-ROC compared to MNBI. Other studies have shown similar results, with a cutoff point of 14.7, with a sensitivity of 96% and specificity of 100% for GERD. 15 DeMeester score is not mentioned as a complementary test in the Lyon consensus. However, our results suggest the need for clinical studies evaluating the operational characteristics of this score with the updated Lyon criteria.
We found overweight patients in our population, but no obese patients. In addition, we found no differences in BMI between the two groups. However, BMI is associated with GERD symptoms, and even moderate weight gain can worsen reflux symptoms, 16 with an increased number of proximal refluxes in obese patients. 17 Further studies with larger sample sizes may provide additional information on the effect of BMI and weight loss on reflux. However, weight loss is recommended for GERD control. 18
This study describes the clinical, endoscopic, and pH-metry characteristics of a retrospective cohort of patients with clinical suspicion of GERD and confirmed or ruled-out GERD diagnosis. Strengths of the study include the selection of patients with suspected GERD, encompassing the full spectrum of the disease, and the adequate endoscopic and pH-metric characterization of the patients.
Some limitations must be acknowledged: Our methodology is a retrospective design with a relatively limited sample size. However, it was sufficient to demonstrate significant differences in key diagnostic parameters. In addition, there was a large time interval between the endoscopic and pH-metric studies, but we believe that the impact on the results was limited, as sensitivity analysis with a smaller time window of 90 days between the two tests maintained the conclusions, including the superiority of the AUC-ROC for the DeMeester score over the MNBI. Furthermore, this study includes only two patients with borderline AET in which the GERD diagnosis was confirmed by endoscopy, so recommendations regarding diagnostic tests for patients with inconclusive GERD diagnoses are not possible. Moreover, this study did not evaluate the operational characteristics of MNBI in specific subgroups, such as those with equivocal acid exposure, atypical symptoms, obese patients, or non-responders to PPI therapy. Complementary studies focused on these populations are needed to assess the utility of MNBI in such contexts. Given these limitations, our results should be interpreted as a useful study for hypothesis generation rather than proving the superiority of any complementary test over another. Therefore, further prospective studies are needed to confirm our results.
Conclusion
Our results suggest that MNBI has suboptimal operational characteristics for GERD diagnosis. Other complementary tests, including the total number of reflux episodes, acid reflux episodes, and bolus exposure time, seem to have similar limitations. For now, these tests should be interpreted alongside clinical, endoscopic, and pH-metry findings to support or refute the diagnosis of GERD, as suggested by the updated Lyon Consensus. The good diagnostic accuracy observed for the DeMeester score is noteworthy, but prospective studies are needed to further assess its potential utility and to confirm our findings.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251340495 – Supplemental material for Diagnostic accuracy of mean nocturnal basal impedance and other complementary tests for the diagnosis of gastroesophageal reflux disease according to the new Lyon criteria
Supplemental material, sj-docx-1-tag-10.1177_17562848251340495 for Diagnostic accuracy of mean nocturnal basal impedance and other complementary tests for the diagnosis of gastroesophageal reflux disease according to the new Lyon criteria by Carlos Lombo-Moreno, David La Rotta, Manuelita Pardo-Ortiz, Fredy Alexander Avila, Raul Antonio Cañadas, Óscar Muñoz and Albis Cecilia Hani in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
