Abstract
Background:
Vitamin D deficiency is prevalent and linked to chronic diseases; its association with advanced liver disease progression requires clarification.
Objectives:
To investigate the association between vitamin D levels and risks of liver cirrhosis, hepatocellular carcinoma (HCC), and mortality, and assess risk changes after achieving sufficiency post-supplementation.
Design:
This was a retrospective cohort study.
Methods:
Utilized TriNetX US data (3,905,594 patients, 2000–2024). Adults with vitamin D deficiency (20.00–30.00 ng/mL) were compared with those with sufficient levels (30.01–80.00 ng/mL). Follow-up was initiated from the first vitamin D test or start of supplementation to minimize immortal time bias. Propensity score matching (1:1) balanced >20 baseline confounders.
Results:
After matching, 1,204,760 patients with vitamin D deficiency and 1,204,760 with sufficient vitamin D levels were included. Vitamin D deficiency was associated with an increased risk of liver cirrhosis (hazard ratio (HR), 1.30; 95% confidence interval (CI), 1.25–1.36), HCC (HR, 1.22; 95% CI, 1.08–1.37), and all-cause mortality (HR, 1.14; 95% CI, 1.13–1.16). Achieving sufficient vitamin D levels reduced the risk of all-cause mortality (HR, 0.93; 95% CI, 0.88–0.99) and aligned HCC outcomes (HR, 1.16; 95% CI, 0.68–2.00). However, it did not significantly reduce the risk of liver cirrhosis (HR, 2.05; 95% CI, 1.69–2.50). Dose–response analysis showed a U-shaped relationship for liver cirrhosis and HCC, with the lowest risks at 40–60 ng/mL.
Conclusion:
Serum vitamin D levels showed a nonlinear association with liver cirrhosis and HCC risk; deficiency independently increased the risks for cirrhosis, HCC, and mortality. Supplementation achieving sufficiency reduced mortality and normalized HCC risk but not cirrhosis risk, potentially reflecting limitations in reversing established disease. The lowest liver disease risk was associated with vitamin D levels of 40–60 ng/mL in this cohort, although causality and the clinical benefit of targeting this specific range require confirmation.
Plain language summary
Vitamin D is an important nutrient that our bodies need to stay healthy, but both too little and too much may be harmful. This study explored whether having low vitamin D levels increases the risk of serious liver diseases “such as liver cirrhosis and liver cancer” and the risk of death, and whether improving vitamin D levels can make a difference. Researchers analyzed the health records of millions of adults from various US hospitals between 2000 and 2024. They compared patients with low vitamin D levels to those with normal levels. In addition, they looked at a group of patients who had low vitamin D levels but later improved their levels through supplementation. The study found that people with low vitamin D levels had a higher risk of developing advanced liver diseases and were more likely to die compared to those with normal vitamin D levels. Interestingly, even after improving vitamin D levels, the risk of liver cirrhosis remained high, although the risk of death was reduced and liver cancer risk was similar to those with normal levels. The researchers also discovered that the relationship between vitamin D and liver health was not straightforward both very low and very high vitamin D levels were associated with greater risks, suggesting that an optimal balance is key. In simple terms, maintaining the right amount of vitamin D appears important for protecting liver health and reducing the risk of serious illness and death, although more research is needed to confirm the ideal levels and the benefits of aiming for them.
Keywords
Introduction
Vitamin D deficiency affects an estimated 30%–50% of the general population, representing a significant public health concern.1 –3 This fat-soluble vitamin plays a crucial role in maintaining skeletal health and regulating calcium and phosphorus metabolism. 4 Recent studies have linked vitamin D deficiency to an increased risk of chronic diseases, including cardiovascular disease, diabetes, and certain cancers.5,6
Liver cirrhosis and hepatocellular carcinoma (HCC) represent significant global health burdens. Cirrhosis, characterized by progressive liver fibrosis, is the 11th leading cause of death worldwide. 7 HCC, the most common primary liver cancer, ranks as the sixth most common cancer and fourth leading cause of cancer-related deaths globally. 8
Several observational studies have investigated the relationship between vitamin D deficiency and liver diseases. 9 For example, a cross-sectional study found that patients with non-alcoholic steatohepatitis had significantly lower vitamin D levels than healthy controls. 10 Another study reported that low vitamin D levels are associated with higher mortality rates in patients with liver cirrhosis. 11
The potential mechanisms underlying the association between vitamin D deficiency and the incidence of liver diseases have been examined in various experimental studies. Vitamin D is believed to exert anti-inflammatory, antifibrotic, and antiproliferative effects through the vitamin D receptor (VDR), which is expressed in liver cells, potentially modulating immune responses and cellular homeostasis.12 –14 Despite the growing evidence linking vitamin D deficiency to liver diseases, comprehensive studies assessing the impact of demographic factors such as age, sex, and race on this association are lacking. Furthermore, many existing studies have been limited by small sample sizes, single-center design, limiting generalizability, and insufficient adjustment for potential confounders, potentially biasing results.15,16 In addition, most studies focused on the association between vitamin D deficiency and liver disease severity. By contrast, the effect on disease incidence remains less explored, leaving uncertainty about its role in disease initiation. However, the clinical benefits of vitamin D supplementation remain controversial, as some studies report benefits, while others show no significant impact on long-term health outcomes. 17
Given these knowledge gaps and methodological limitations in prior research, the primary objectives of this study were to investigate the association between vitamin D deficiency and the risk of liver cirrhosis and HCC, to evaluate whether vitamin D supplementation can reduce these risks, and to explore how these risks vary according to demographic factors.
Methods
Data sources
The data source for this study was the TriNetX US Network, which includes de-identified electronic health records from a diverse population exceeding 116 million patients sourced from 64 healthcare organizations across the United States. 18 The dataset included demographic details; diagnostic information coded using the International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM); medication information coded using the Anatomical Therapeutic Chemical (ATC) classification system or RxNorm, a standardized nomenclature for clinical drugs developed by the US National Library of Medicine; and laboratory test results coded using Logical Observation Identifiers Names and Codes. To ensure data integrity, TriNetX employs a standardized framework that focuses on consistency, completeness, and validity. TriNetX adheres to the Health Insurance Portability and Accountability Act and the General Data Protection Regulation 19 providing a valuable repository of clinical data for healthcare and life sciences research.20,21
This study adhered to the Strengthening the Reporting of Observational Studies in Epidemiology initiative guidelines. 22 In June 2024, we collected and analyzed data using the American Collaborative Network, a subnetwork within TriNetX that includes 64 healthcare organizations. TriNetX is a global federated network that provides access to de-identified electronic health records from healthcare organizations worldwide, ensuring patient privacy and compliance with relevant data protection regulations.
Study design and study participants
This study evaluated the association between vitamin D deficiency and liver disease outcomes and investigated whether vitamin D supplementation influences these outcomes. The study period ranged from January 1, 2000, to May 30, 2024 (eTable 1 in Supplemental Material 1 for a summary of the study timeline and key definitions).
We initially identified patients with vitamin D levels between 20.00 and 80.00 ng/mL at any clinical visit during the study period (January 1, 2000, to May 30, 2024). Patients included in the initial cohort were identified based on meeting predefined criteria within the database (e.g., having qualifying vitamin D tests during the study period) rather than through consecutive or random sampling. Patients were classified as vitamin D deficient if their serum 25-hydroxyvitamin D [25(OH)D] levels ranged from 20.00 to 30.00 ng/mL, whereas vitamin D sufficiency was indicated by levels of 30.01–80.00 ng/mL. The threshold of >30 ng/mL for sufficiency aligns with the 2011 Endocrine Society’s clinical practice guideline, often applied in clinical settings for broader health benefits beyond the Institute of Medicine’s (IOM) ⩾20 ng/mL threshold, primarily based on bone health for the general population.23,24 Our study defined the deficient group as 20–30 ng/mL to specifically assess risks within this range, which is above severe deficiency thresholds (<20 ng/mL) but below the clinical sufficiency target. The upper limit of 80 ng/mL was chosen as a conservative operational threshold to exclude potentially excessive levels, informed by recent European expert consensus recommending avoidance of levels >60–80 ng/mL to reduce potential toxicity risk in certain populations. 25 For dose–response analysis, vitamin D levels were categorized into six groups (ng/mL): 20.00–25.00, 25.01–30.00, 30.01–40.00, 40.01–60.00, 60.01–70.00, and 70.01–80.00 ng/mL.
We then excluded patients younger than 20 years and those with a diagnosis of liver cirrhosis (ICD-10-CM code K74.60) or liver cancer (ICD-10-CM code C22.0) according to their outpatient or inpatient medical records. To allow a minimal induction time for disease development, a 1-year induction period was used to assess the occurrence of liver cirrhosis and HCC in exposed patients. 26
Exposed and non-exposed groups
We categorized the patients with vitamin D deficiency (exposed groups) into two groups based on their vitamin D status, as determined by serum 25(OH)D levels and vitamin D supplementation. The continuous deficiency group comprised patients with vitamin D deficiency for >1 year, as defined by a serum 25(OH)D level of 20.00–30.00 ng/mL continuously for 1 year without any record of sufficient levels (i.e., 30.01–80.00 ng/mL). The recovery group consisted of patients with an initial vitamin D deficiency who subsequently achieved vitamin D sufficiency within 1 year of starting vitamin D supplementation and maintained sufficient levels without reverting to a deficient status. The vitamin D sufficiency group (non-exposed group) comprised patients with serum 25(OH)D levels of 30.01–80 ng/mL without records of vitamin D deficiency.
Propensity score matching and covariates
We used propensity score matching based on patient characteristics to ensure comparability between the exposed and non-exposed groups. Matching was performed using TriNetX’s built-in tools to generate propensity scores and was performed on a 1:1 basis through greedy nearest-neighbor matching. We applied a caliper of 0.1 combined standard deviations (SDs) for variable matching between the exposed and non-exposed groups. The balance between the groups before and after matching was assessed using the standardized mean difference (SMD), with an SMD lower than 0.1 indicating well-matched groups.
The baseline characteristics of the patients included various demographic, lifestyle, socioeconomic, medication, comorbidity, and laboratory test parameters, all assessed prior to the index date. The age at index was recorded as the age of the patients in years on the date of diagnosis. Patients were categorized according to sex (male or female) and race (White, Black/African American, or Asian). Lifestyle characteristics encompassed nicotine dependence, as indicated by ICD-10 Code F17 and body mass index.
Socioeconomic status was assessed using the proportion of patients with identified socioeconomic health hazards (ICD-10 codes Z55–Z65), including issues related to education and literacy, employment and unemployment, and housing and economic circumstances. Medication use was documented for corticosteroids (ATC code: R01AD), aspirin (RxNorm code: 1191), and metformin (RxNorm code: 6809).
Comorbidities related to vitamin D deficiency and liver diseases were recorded, including hypertension diseases (ICD-10 codes I10–I1A), chronic lower respiratory diseases (ICD-10 codes J40–J4A), type 2 diabetes mellitus (ICD-10 code E11), chronic kidney disease (CKD; ICD-10 code N18), non-alcoholic fatty liver disease (ICD-10 code K76.0), acute myocardial infarction (ICD-10 code I21), viral hepatitis (ICD-10 codes B15–B19), alcoholic liver disease (ICD-10 code K70), portal hypertension (ICD-10 code K76.6), and autoimmune hepatitis (ICD-10 code K75.4).
Laboratory tests conducted to assess liver health and diagnose liver diseases included measurements of alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase, bilirubin, albumin, gamma-glutamyl transferase, and alpha-1-fetoprotein.
Index date and follow-up period
The index date for each group was clearly defined to reduce immortal time bias. For the continuous deficiency group, it was the first blood test confirming the deficiency. For the recovery group, it was the start date of supplementation within 1 year of diagnosis. For the sufficiency group, it was the first test confirming sufficient levels.
A 1-year washout period excluded patients diagnosed with liver cirrhosis or HCC within the first year after the index date, ensuring outcomes occurred post-exposure.
Outcomes
The target outcomes examined in this study included cirrhosis (ICD-10-CM codes K74.60), liver cancer (ICD-10-CM code C22.0), and all-cause mortality.
Statistical analysis
Descriptive statistics were calculated for all the baseline characteristics of the study cohort. Continuous variables are presented as mean (SD), and categorical variables are presented as frequencies (%). Survival probabilities for each adverse outcome were estimated using Kaplan–Meier analysis. Log-rank test results were used to analyze the differences. Cox proportional hazard regression models were used to calculate hazard ratios (HRs) and 95% confidence intervals (CIs) for the associations between vitamin D status and the risk of liver cirrhosis, HCC, and all-cause mortality after adjusting for potential confounders. The analysis tracked patients based on their index dates. Patients were censored at the date of their last follow-up or at the end of the study period, whichever occurred first, if they had not experienced an outcome of interest. Censoring accounted for patients who were lost to follow-up and those with incomplete data. The proportional hazard assumption was tested for each model. If the assumption was violated, stratified Cox models based on different time intervals were used.
To address the competing risks, we incorporated death as a competing event. We combined death with the target outcomes and conducted a Cox regression analysis to estimate the associations between vitamin D status and the risk of liver cirrhosis, HCC, and all-cause mortality. 27
All statistical analyses were performed using TriNetX. The charts for this analysis were generated using R programming in RStudio version 4.4.1 (The R Foundation for Statistical Computing, Vienna, Austria).
Results
Characteristics of the study subjects
This study initially included 3,067,155 patients. After propensity score matching, 1,204,760 patients with continuous vitamin D deficiency and an equal number of patients with vitamin D sufficiency were enrolled. The selection process is illustrated in Figure 1.

Flowchart of stratification of the study population from the TriNetX US Database based on vitamin D levels.
eTables 2 and 3 in Supplemental Material 1 present the baseline characteristics of the study participants before and after propensity score matching. Prior to matching, significant differences were observed between the exposed and non-exposed groups across various demographic, lifestyle, socioeconomic, comorbidity, and medication-use factors. After matching, these differences were largely attenuated, with SMDs lower than 0.1 for all variables, indicating an adequate balance between the matched groups.
Main outcomes
The mean durations of follow-up in the vitamin D deficiency and vitamin D sufficiency groups were 4.5 (3.8) and 4.1 (3.6) years, respectively. The majority of patients (98%) started vitamin D testing and follow-up between 2010 and 2024, whereas approximately 20,000 patients initiated follow-up between 2000 and 2010. Cirrhosis was significantly more frequent in the vitamin D deficiency group (5714 cases vs 3901 cases), resulting in an adjusted HR of 1.30 (95% CI = 1.25–1.36). HCC was also more common in the vitamin D deficiency group (659 cases vs 477 cases), yielding an HR of 1.22 (95% CI = 1.08–1.37). Moreover, the vitamin D deficiency group displayed a higher frequency of all-cause mortality (61,167 deaths vs 48.951 deaths), corresponding to an HR of 1.14 (95% CI = 1.13–1.16; Table 1).
Comparison of outcome risks between continuous deficiency, deficiency recovery, and their respective vitamin D sufficiency groups*.
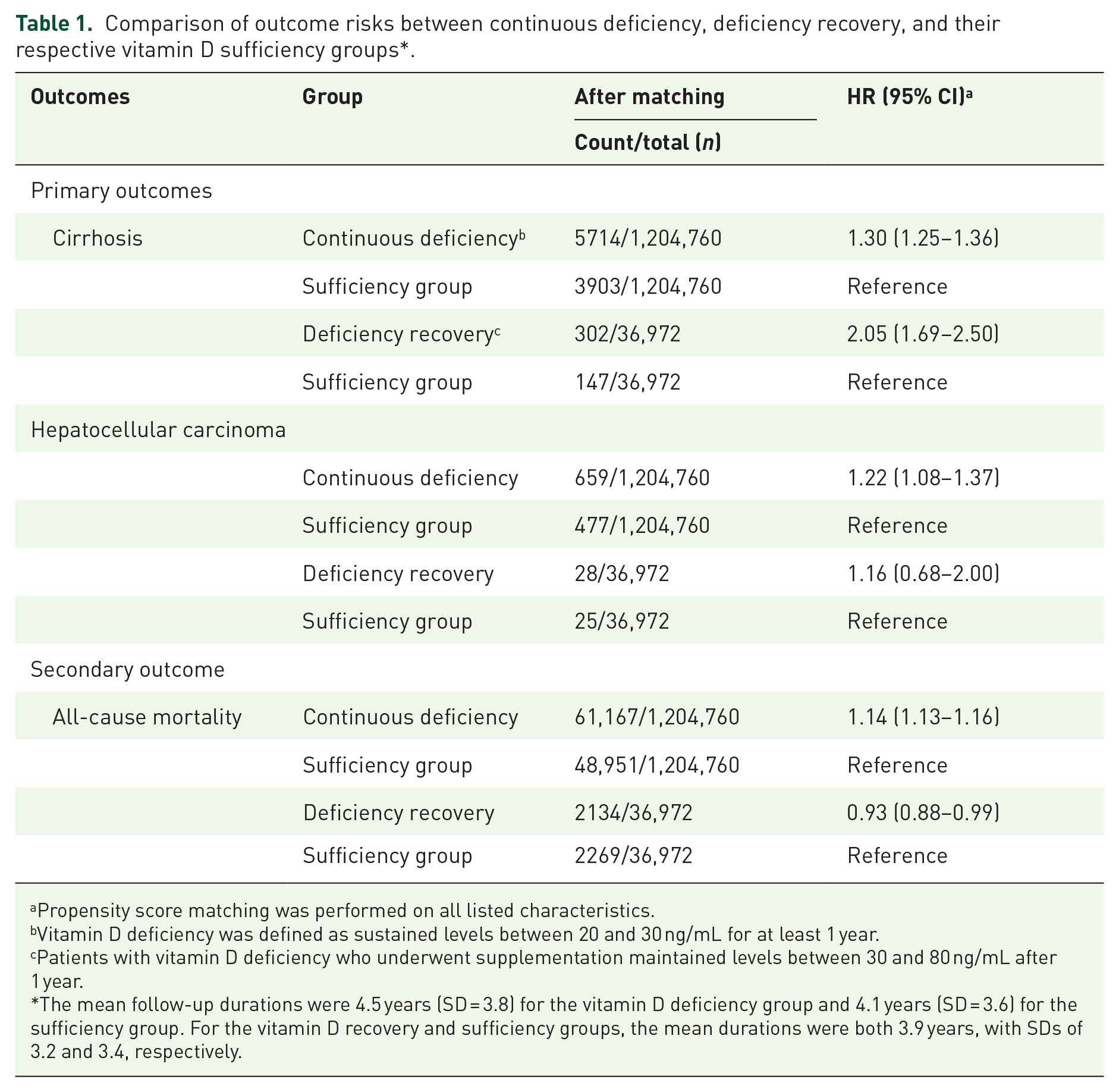
Propensity score matching was performed on all listed characteristics.
Vitamin D deficiency was defined as sustained levels between 20 and 30 ng/mL for at least 1 year.
Patients with vitamin D deficiency who underwent supplementation maintained levels between 30 and 80 ng/mL after 1 year.
The mean follow-up durations were 4.5 years (SD = 3.8) for the vitamin D deficiency group and 4.1 years (SD = 3.6) for the sufficiency group. For the vitamin D recovery and sufficiency groups, the mean durations were both 3.9 years, with SDs of 3.2 and 3.4, respectively.
Further analysis assessed patient outcomes after achieving sufficient vitamin D levels following 1 year of supplementation. As presented in Table 1, the risk of cirrhosis remained elevated in treated patients, with an adjusted HR of 2.05 (95% CI = 1.69–2.50). In addition, the risk of HCC did not differ significantly between the supplementation group and the sufficient group (adjusted HR = 1.16, 95% CI = 0.68–2.00), while a noticeable decrease in all-cause mortality was observed (adjusted HR = 0.93, 95% CI = 0.88–0.99). These results suggest that while normalization of vitamin D levels is associated with reduced mortality rates (Figure 2), it does not appear to substantially alter the incidence of liver cirrhosis or HCC.
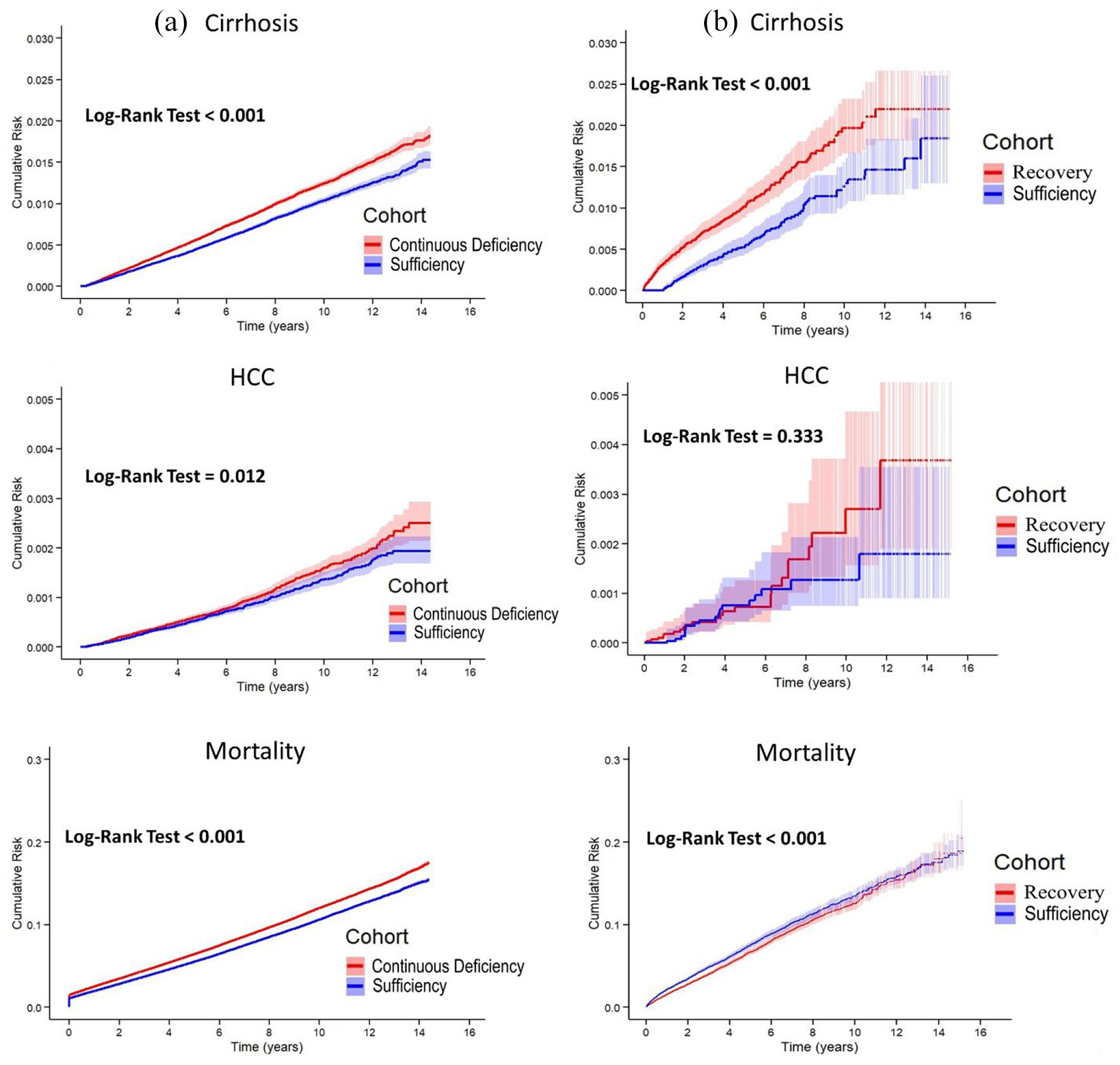
Cumulative risk of liver cirrhosis, hepatocellular carcinoma, and all-cause mortality by vitamin D status: (a) continuous deficiency versus vitamin D sufficiency and (b) recovery from deficiency versus vitamin D sufficiency.
Sensitivity analysis
We conducted a separate analysis focusing on patients with any history of vitamin D deficiency without the requirement of a 1-year follow-up. The results were consistent with the main findings, revealing similar risks of liver cirrhosis, HCC, and all-cause mortality as observed in the continuous vitamin D deficiency group. This differentiation highlights the influence of both short-term and prolonged vitamin D deficiencies on the risk of adverse outcomes (eTable 4 in Supplemental Material 1). To evaluate the impact of different confounding factors, we performed multiple analyses with varying levels of adjustment (eTable 5 in Supplemental Material 1). Starting from the crude model and progressively adding demographic factors, socioeconomic status, lifestyle factors, medications, and comorbidities, the associations remained robust across all models, though the magnitude of the associations varied slightly. The final fully adjusted model (Model 6) demonstrated consistent results with our main findings. To ensure the robustness of the associations, appropriate adjustments were made to address the violation of the proportional hazard assumption in the Cox regression model. In addition, we examined the effects of different follow-up durations (eTable 6 in Supplemental Material 1). The association of vitamin D deficiency with liver cirrhosis, HCC, and all-cause mortality remained significant across both short-term (3 years) and long-term (15 years) follow-up, with only minor variations in HRs noted. To account for the potential competing risks of death, we performed a competing risk analysis (eTable 7 in Supplemental Material 1). These results were consistent with our primary findings, highlighting the significant associations between vitamin D deficiency and increased risks of liver cirrhosis, HCC, and all-cause mortality.
In addition, we conducted sensitivity analyses to evaluate the effects of vitamin D supplementation (eTable 8 in Supplemental Material 1). These findings demonstrated that even after vitamin D supplementation and achievement of normal levels, patients with initial vitamin D deficiency still had an increased risk of liver cirrhosis and HCC. Notably, the risk of all-cause mortality was significantly improved compared with that in the non-exposed group, suggesting that vitamin D supplementation has a positive effect on overall mortality.
External validation
To assess the generalizability of our findings, we performed external validation using data from the TriNetX Global Collaborative Network, which included patient data from 77 global healthcare organizations (eTable 9 in Supplemental Material 1). This analysis involved a large cohort of 1,339,529 matched pairs of patients with and without vitamin D deficiency. The results of this external validation were highly consistent with our primary analysis conducted using the TriNetX US Collaborative Network, suggesting that the observed associations between vitamin D deficiency and increased risks of liver cirrhosis, HCC, and all-cause mortality are generalizable across different healthcare settings and patient populations internationally.
Subgroup analysis
We expanded our subgroup analyses by including demographic characteristics, such as age, sex, and race, to further investigate the association between vitamin D deficiency and the risk of liver cirrhosis, HCC, and all-cause mortality (Figure 3). The risk of liver cirrhosis was stratified by the presence of comorbidities including non-alcoholic fatty liver disease, viral hepatitis, diabetes mellitus, chronic liver disease, and CKD. These findings indicate that a continuous vitamin D deficiency is generally associated with an elevated risk of liver cirrhosis across these subgroups.

Risk of liver cirrhosis, HCC, and mortality stratified by demographic and clinical factors in continuous vitamin D deficiency and sufficiency groups.
Similarly, the risk of HCC was stratified according to the comorbidities. The risk of HCC was notably elevated among different demographic subgroups, particularly among patients with CKD, women, white patients, and those aged 60 years. Finally, the association between persistent vitamin D deficiency and all-cause mortality was stratified using the same set of comorbidities. Subgroup analysis generally indicated that vitamin D deficiency was associated with increased risks of liver diseases and mortality, with this trend displaying a degree of consistency across most demographic and comorbidity subgroups.
Analysis of different vitamin D levels
Figure 4 illustrates the relationship between serum 25-hydroxyvitamin D levels and the risks of liver cirrhosis, HCC, and all-cause mortality, revealing a U-shaped association for liver disease outcomes. The lowest risks for liver cirrhosis and HCC were observed at 40–60 ng/mL, whereas both lower levels (<30 ng/mL) and higher levels (>70 ng/mL) were associated with increased risk. However, for all-cause mortality, the risk was primarily elevated at lower vitamin D levels (<30 ng/mL), while higher levels (>70 ng/mL) did not show a significant increase. These findings suggest that both vitamin D deficiency and excess may adversely affect liver-related outcomes, whereas mortality risk appears to be mainly driven by deficiency.

Association between 25-hydroxyvitamin D levels and risks of liver cirrhosis, HCC, and all-cause mortality. This figure illustrates the nonlinear association between 25-hydroxyvitamin D levels and HRs for liver cirrhosis (a), HCC (b), and all-cause mortality (c). The solid line represents the estimated HR, while the dashed lines indicate the 95% confidence intervals. The black dots represent spline knots used in the restricted cubic spline regression model.
Discussion
Main findings
This study identified a significant association between vitamin D deficiency and increased risks of liver cirrhosis, HCC, and all-cause mortality, independent of potential confounders. While vitamin D supplementation was linked to a reduced risk of all-cause mortality and HCC, it did not significantly affect liver cirrhosis risk, suggesting that timing and disease progression may influence intervention effectiveness. Notably, the U-shaped relationship observed for liver cirrhosis and HCC underscores the potential risks of both deficiency and excessive vitamin D levels, while all-cause mortality appears primarily driven by low vitamin D levels rather than excess. These findings highlight the importance of maintaining optimal vitamin D levels within a moderate range, rather than focusing solely on correcting deficiency.
Subgroup analyses also suggested potential variations in risk across populations, with heightened risks noted particularly in patients with CKD and older adults in Figure 3, potentially reflecting differences in vitamin D metabolism or underlying health status, although further investigation is needed.28,29
Limited effectiveness in advanced disease and paradoxical findings
The mechanisms underlying the effect of vitamin D on liver health are multifaceted. Vitamin D modulates immune responses, reduces inflammation, and mitigates fibrosis through its interaction with VDRs expressed in various liver cells.12,30 –32 However, the relationship between vitamin D and liver diseases is more complex than initially understood and is potentially influenced by factors such as disease stage, genetic background, and comorbid conditions.
The broader systemic effects of vitamin D, particularly in cancer research, have been explored extensively. For example, in digestive tract cancers, vitamin D modulates immune responses by interacting with PD-L1, leading to improved survival outcomes in specific subgroups. 33 This evidence suggests that the benefits of vitamin D might be more pronounced in systemic conditions than in organ-specific pathologies, providing a potential explanation for the discrepancy observed in our study. While vitamin D supplementation has been associated with reduced cancer-specific and overall mortality,34,35 its impact on liver-specific outcomes appears to be limited, as observed in our analysis.
At the cellular level, vitamin D can activate autophagy through both the genomic and non-genomic pathways, potentially maintaining cellular homeostasis by inhibiting oxidative stress and apoptosis.36 –38 This mechanism could partly explain the observed survival benefits in our study, as vitamin D may confer protection against systemic stressors and comorbid conditions rather than directly impacting liver-specific pathogenesis.
Regarding liver cirrhosis, our findings partially align with those of previous studies demonstrating an association between vitamin D deficiency and cirrhosis.39,40 However, the lack of a protective effect from supplementation may suggest that the role of vitamin D is more relevant to early-stage fibrosis rather than established cirrhosis, as indicated by several studies reporting minimal effects of supplementation on hepatic function in advanced cirrhosis.41,42
Interpreting the increased cirrhosis risk (HR 2.05) in the recovery group requires caution, as this paradoxical finding persisted across follow-up durations (eTable 7 in Supplemental Material 1). Potential explanations include residual reverse causality (despite a 1-year washout period designed to mitigate it), supplementation being potentially ineffective against pre-existing irreversible liver damage, selection bias or confounding related to which patients received and responded to supplementation, and limitations in available data regarding the supplementation details (dose, adherence). Therefore, this finding likely reflects complex underlying patient factors and inherent observational study limitations rather than indicating a harmful effect or causation by vitamin D supplementation itself.
U-shaped association with liver disease
Our study identified a U-shaped association between vitamin D levels and the risk of liver cirrhosis and HCC (Figure 4), with the lowest risk observed in the 40–60 ng/mL range. While this optimal range requires further validation, recent literature provides context for its potential physiological significance. Several reviews suggest this range might be “preferred” or “ideal” for maximizing overall health benefits, potentially including non-skeletal effects like immune function.43,44 Mechanistically, optimal immune function, which plays a crucial role in liver disease pathogenesis (e.g., inflammation, HCC surveillance), has been suggested by some reviews to be better supported within this range, 43 potentially providing a relevant physiological rationale for our findings. However, it is crucial to emphasize that a universal consensus on this specific optimal range is lacking, particularly for liver disease outcomes, and the evidence for non-skeletal benefits remains under active investigation. Therefore, our observation primarily highlights a potential nonlinear association specific to liver outcomes in our cohort and should be interpreted cautiously pending further mechanistic studies and external validation.
Regarding specific outcomes after achieving sufficiency, the finding that HCC risk was comparable to controls aligns with some studies suggesting vitamin D’s protective effects.45,46 Nonetheless, supplementation benefits in this context remain debated, potentially influenced by factors like VDR polymorphisms affecting HCC susceptibility 47 and limitations inherent in previous underpowered or inconsistent clinical trials on vitamin D in liver disease. 48
Moreover, the U-shape observed for liver outcomes differs from the generally nonlinear (risk decreasing then plateauing) association reported between vitamin D and all-cause mortality in recent reviews, 28 implying distinct effects on different health outcomes.
Our observation that lower vitamin D levels are associated with increased all-cause mortality receives additional support from recent studies. For example, Miwa et al. also recently reported that vitamin D deficiency (<20 ng/mL) was significantly associated with higher mortality in a cohort of patients with established cirrhosis, 49 reinforcing the potential prognostic significance of vitamin D status during the progression of liver disease.
Study limitations
Our study had several limitations. Although extensive, the TriNetX database may contain inconsistencies due to variations in data entry across healthcare institutions. Missing data, particularly in the long-term follow-up, could have affected our analysis of disease progression. Key variables, such as lifestyle factors and socioeconomic status, might not have been comprehensively recorded, potentially limiting our ability to control for all relevant confounders.
Sample representativeness is a concern, as TriNetX data may not fully reflect underrepresented populations, potentially limiting the generalizability of our findings. The follow-up duration might have been insufficient to capture long-term outcomes, especially for slow-progressing conditions, such as cirrhosis and HCC. Moreover, we could not assess details of the supplementation regimens (e.g., specific dose, formulation, precise duration) or patient medication adherence, which may have influenced the interpretation of the effects of vitamin D supplementation.
Despite using propensity score matching and a large sample size, residual confounding factors cannot be ruled out. As this was an observational study, we could not infer causality between vitamin D deficiency and liver outcomes. However, we implemented several measures to enhance the credibility of our findings, including sensitivity analysis, competing risk analysis, and external validation. These efforts aimed to mitigate some inherent limitations of observational studies and to strengthen the robustness of our results.
Conclusion
Vitamin D levels exhibit a nonlinear association with liver disease risk, with both deficiency and excess linked to increased risk. Achieving sufficient levels was associated with lower mortality and normalized HCC risk but did not reduce cirrhosis risk, potentially reflecting limitations in reversing established disease. The lowest liver disease risk was associated with vitamin D levels of 40–60 ng/mL in this cohort, although causality and the clinical benefit of targeting this specific range require confirmation. These findings suggest that early screening and maintaining optimal vitamin D levels may be beneficial for high-risk individuals, though further research is needed to establish definitive clinical recommendations.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251338669 – Supplemental material for Nonlinear associations of serum vitamin D levels with advanced liver disease and mortality: a US Cohort Study
Supplemental material, sj-docx-1-tag-10.1177_17562848251338669 for Nonlinear associations of serum vitamin D levels with advanced liver disease and mortality: a US Cohort Study by Yuan-Tsung Tseng, Chun-Hsiang Wang, Jung-Der Wang, Kow-Tong Chen and Chung-Yi Li in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848251338669 – Supplemental material for Nonlinear associations of serum vitamin D levels with advanced liver disease and mortality: a US Cohort Study
Supplemental material, sj-docx-2-tag-10.1177_17562848251338669 for Nonlinear associations of serum vitamin D levels with advanced liver disease and mortality: a US Cohort Study by Yuan-Tsung Tseng, Chun-Hsiang Wang, Jung-Der Wang, Kow-Tong Chen and Chung-Yi Li in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
Ms. Tseng and Prof. Li had full access to all of the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. Dr. Wang contributed equally to this work.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
