Abstract
Despite the established efficacy of sorafenib in advanced hepatocellular carcinoma (HCC), a significant number of sorafenib-treated patients experience disease progression. Current guidelines recommend either best supportive care or clinical trial enrollment for this population. As such, there remains an unmet need for tolerable, life-prolonging strategies in the second-line setting. New information regarding the molecular pathogenesis of resistance to antiangiogenic therapy and positive post-progression experience with antiangiogenics in other tumor types has led to trials investigating the effect of continued use of sorafenib, alone or combined with other agents. Trials investigating the effect of switching from sorafenib to alternate antiangiogenic agents, phosphatidylinositol 3 kinase/AKT/mammalian target of rapamycin inhibitors, or cMet inhibitors are also underway. As these data emerge, clinicians may consider a new paradigm for managing advanced HCC. This article briefly reviews the mechanisms of disease resistance to antiangiogenic therapy as a vehicle for discussing clinical strategies to prolong survival in patients with advanced HCC that are currently employed at our institutions or are under investigation. Key ongoing trials investigating the use of molecularly targeted therapies in patients with progressive disease are also highlighted.
Introduction
Over the last 5 years, molecularly targeted therapy has become a mainstay in the treatment paradigm for advanced hepatocellular carcinoma (HCC) [Padzur, 2012; Zhu, 2012]. Sorafenib was the first and remains the only approved systemic therapy for advanced HCC. Approval was based on the results of two randomized, placebo-controlled phase III trials. The Sorafenib HCC Assessment Randomized Protocol (SHARP) trial, which enrolled 602 patients in 21 countries across Europe, North America, South America, and Australasia, demonstrated that sorafenib significantly prolonged overall survival (OS) compared with placebo (OS 10.7 versus 7.9 months, p < 0.001) [Llovet et al. 2008]. The Asia Pacific (AP) trial (N = 271) supported a similar survival advantage for sorafenib over placebo [OS 6.5 versus 4.2 months, hazard ratio (HR) 0.68; 95% confidence interval (CI) 0.50–0.93; p = 0.014] [Cheng et al. 2009]. The lower magnitude of sorafenib-related OS in the AP (versus SHARP) study population was likely due to the increased tumor burden in this heavily hepatitis B virus (HBV)-infected group. Grade 3/4 toxicities affecting more than 5% of patients in these trials included hand–foot skin reaction (SHARP = 8%, AP = 11%) and diarrhea (SHARP = 8%, AP = 6%).
In phase III trials examining the multikinase inhibitors sunitinib, brivanib, and linifanib individually, none of the agents demonstrated superiority to sorafenib in prolonging OS in the first-line setting [Cainap et al. 2013; Cheng et al. 2011; Johnson et al. 2012]. Sunitinib and linifanib inhibit members of the vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF) receptor families (see prescribing information for Sutent, Pfizer, Inc., New York, NY, USA), whereas brivanib inhibits VEGF and fibroblastic growth factor (FGF) signaling [Huynh et al. 2008]. Sorafenib inhibits several other kinases as well as VEGF and PDGF, including RAF kinase. This difference provides a rationale for additional studies examining combinations of sorafenib with sunitinib, brivanib, or linifanib to leverage the potential for additive or synergistic efficacy through inhibition of multiple pathways. Notably, the SEARCH (Sorafenib and Erlotinib, a Randomized Trial Protocol for the Treatment of Patients with Hepatocellular Carcinoma) trial (N = 720), which compared the efficacy of sorafenib plus the epidermal growth factor receptor (EGFR) inhibitor erlotinib versus sorafenib alone did not achieve its primary endpoint of a 33% improvement in OS (OS 9.5 versus 8.5 months, p = 0.204) [Zhu et al. 2012]. Trials evaluating first-line sorafenib alone or combined with other molecularly targeted agents, such as the dual c-Met/VEGFR-2 tyrosine kinase inhibitor (TKI) E7050 [ClinicalTrials.gov identifier: NCT01271504] are ongoing. The potential toxicities resulting from combining agents and the optimal dosing of combination regimens require further study.
Despite the established efficacy of sorafenib in advanced HCC, interim data from GIDEON (Global Investigation of Therapeutic Decisions in Hepatocellular Carcinoma and of Its Treatment with Sorafenib), a noninterventional study of patients with unresectable HCC and for whom the decision has been made to treat with sorafenib under real-life practice conditions, indicates that approximately one-third of patients discontinued sorafenib because of disease progression [Bronowicki et al. 2011]. Progression was determined radiographically using Response Evaluation Criteria in Solid Tumors (RECIST). It should be emphasized that treatment decisions for patients in this study were not protocol driven and were, therefore, diverse. In the SHARP study, the median time to radiologic progression was delayed in sorafenib-treated patients (5.5 versus 2.8 months, p < 0.001). However, 107 of 263 (40.6%) patients demonstrated radiologic progression by the cut-off date. These studies suggest that disease progression limits the impact of sorafenib on OS and that alternate strategies to manage advanced disease in the second-line setting need to be defined through clinical investigation.
Although recent preclinical and clinical studies have increased our understanding of the mechanisms for resistance to antiangiogenic therapy, they offer limited guidance for effective management of disease progression [Li et al. 2010; Raza et al. 2010; Wenger et al. 2011]. In this review, we discuss key challenges in the management of HCC progression on sorafenib and review novel approaches to treatment of this patient population.
Disease progression: definitions and management
The management of patients treated with sorafenib for advanced HCC is complicated by several factors, including coexisting cirrhosis, evolving response criteria, and lack of predictive biomarkers. In the United States, nearly 90% of patients with HCC exhibit concomitant cirrhosis [Bartlett, 2008; Volk and Marrero, 2008]. Because the natural history of cirrhosis is known to significantly impact morbidity and mortality, it is often difficult to distinguish whether clinical deterioration arises from worsening liver function, treatment-related toxicity, or tumor progression. Studies have shown a poor correlation between clinical benefit provided by targeted agents such as sorafenib or by locoregional therapies and conventional methods of response assessment [Forner et al. 2009; Llovet et al. 2008]. Therefore, the RECIST assumption that tumor death results in a reduction in lesional dimensions may not apply to patients with heavily fibrotic/cirrhotic livers, making assessment of the clinical status of patients with HCC more difficult.
The SHARP study highlighted the insensitivity of RECIST in measuring treatment response. In that study, improved survival did not correlate with radiographic response rates. Although the disease control rate was 43%, objective response (partial response) was documented in only 2% of patients treated with sorafenib [Llovet et al. 2008]. This poor correlation between RECIST-assessed response and OS requires practitioners to rely primarily on OS to evaluate the efficacy of sorafenib and other targeted therapies. In clinical practice, this discordance could result in an early, unnecessary interruption in sorafenib treatment. It also emphasizes the need to develop more sensitive and accurate radiographic measures of tumor necrosis and tumor blood flow.
RECIST was amended in 2010 to account for the high level of vascularity associated with HCC and the observed discordance between apparent tumor death and reduction in lesion dimensions [Lencioni and Llovet, 2010]. The modified RECIST (mRECIST) assesses the target lesion(s) based on intratumor arterial enhancement by computed tomography or magnetic resonance imaging. This differentiation is of critical importance because the antiangiogenic activity of the multikinase inhibitor sorafenib is characterized by intralesional tumor necrosis and a temporary, but substantial, increase in tumor size [Horger et al. 2009]. Historically, patients treated with sorafenib for HCC typically achieved a best response of stable disease (SD) when assessed by RECIST, which may not reflect the full biologic effect of the agent [Cheng et al. 2009; Llovet et al. 2008]. In contrast, mRECIST may provide more meaningful data regarding survival benefit; in a small retrospective analysis of 53 patients treated with sorafenib for HCC, 42 with SD by RECIST were reclassified using mRECIST, with these results: 10 partial response (PR), 1 complete response (CR), 29 SD, and 2 progressive disease (PD) [Edeline et al. 2012]. Notably, while the median OS for the overall group was 11.2 months, the median OS in patients reclassified by mRECIST as objective responders, SD, or PD, was 17.1 months, 9.7 months, and 3.7 months respectively. Although these data suggest that broader use of mRECIST may provide a more precise assessment of treatment response, further study is needed to confirm these findings and validate this tool in HCC.
The role of serum biomarkers in predicting response and defining HCC progression continues to evolve. Historically, serum α-feto protein (AFP) has been widely used in HCC screening and diagnostic evaluations (e.g. CLIP staging), and to identify tumor progression in patients with an elevated baseline AFP [Bertino et al. 2012]. Several small studies have examined the role of AFP in evaluating response to antiangiogenic therapies in patients with HCC [Kuzuya et al. 2011; Yau et al. 2011]. Overall, these studies suggest that an early AFP decrease (>20% decline from baseline at 2–6 weeks) may be a surrogate marker for response and progression-free survival (PFS). Serial AFP measurements combined with radiographic assessment should be further evaluated to better understand their predictive utility.
Des-γ-carboxy prothrombin (DCP), an abnormal prothrombin whose level is increased in patients with HCC, may be used in screening and diagnosis. When measured in tandem with AFP, DCP may increase sensitivity and specificity for HCC detection [Semela and Dufour, 2004; Weitz and Liebman, 1993]. In the context of sorafenib treatment, Ueshima and colleagues performed a small retrospective study and found that time to progression (TTP) was significantly prolonged in patients whose serum DCP level increased at least twofold over baseline (p = 0.0296) in the first 2 weeks following treatment [Ueshima et al. 2011]. This increase may be transient and related to the hypoxic environment created by sorafenib. Thus, in patients treated with antiangiogenic therapies, transient increases in DCP may be indicative of response. However, the predictive utility of this marker may be limited, since sustained elevated levels have generally negative prognostic significance.
In the SHARP trial, the potential prognostic and predictive value of 10 candidate biomarkers was evaluated. Among the entire SHARP population, baseline concentrations of angiopoietin-2 (p = 0.001), VEGF (p = 0.015), AFP (p = 0.001), and alkaline phosphatase (p = 0.0003) were found to have independent prognostic value. Markers predictive of sorafenib treatment response were not identified, although nonsignificant trends were observed toward greater sorafenib benefit in patients with higher baseline s-c-KIT (p of interaction 0.081 for OS, 0.052 for PFS) or basic fibroblast growth factor (bFGF; p of interaction 0.078 for PFS), or low baseline hepatocyte growth factor (p of interaction 0.073 for OS, 0.396 for PFS) [Llovet et al. 2012a].
Mechanisms of resistance to antiangiogenic therapy
HCC growth depends on neovascularization. Antiangiogenic therapy prevents the initiation of neovascularization, thereby slowing tumor growth and inducing tumor regression [Folkman, 2004]. However, evidence suggests that compensatory pathways at the level of the tumor vasculature and microenvironment can promote resistance [Casanovas et al. 2005; Li et al. 2010; Mizukami et al. 2005; Raza et al. 2010; Wenger et al. 2011; Yu et al. 2002]. Multiple resistance mechanisms have been identified and are shown schematically in Figure 1. These mechanisms include reactive resistance, genetic alterations, overexpression of alternative signaling pathways, and vasculogenic mimicry. Following antiangiogenic exposure and a period of SD, murine models of pancreatic islet tumors have demonstrated a phenotypic resistance to anti-VEGF therapy, resulting in disease progression. It is believed that resistance may be ‘reactive’, that is, mediated by intratumoral hypoxia-induced upregulation of factors (e.g. FGF, ephrins, hypoxia-inducible factors 1 and 2, and interleukin 8) that restimulate angiogenesis via alternative pathways [Casanovas et al. 2005; Mizukami et al. 2005; Wenger et al. 2011; Yu et al. 2002].

Potential mechanisms of resistance to antiangiogenic drugs. Genetic and epigenetic alterations of tumor tissue may render cells less sensitive to hypoxia and consequently less sensitive to antiangiogenic agents. These mutations may allow the outgrowth of resistant cells after treatment. The hypoxic tumor environment generated by antiangiogenic treatment may also result in reactive resistance in which proliferative signaling and expression and secretion of proangiogenic factors are stimulated. Reactive resistance may lead to neovascularization, vascular mimicry, and enhanced cellular proliferation. Antiangiogenic treatment may also possibly select for cells with a higher propensity for invasiveness and metastases. FGF, fibroblast growth factor; IL-8, interleukin 8; PDGF, platelet-derived growth factor; VEGF, vascular endothelial growth factor.
Genetic alterations may impact resistance as well, as exemplified by the p53 tumor suppressor gene. Cancer cells, by definition, embody some degree of genetic instability. Mutations that lead to inactivation of the wild-type p53 tumor suppressor gene can reduce the susceptibility of cells to apoptosis in a hypoxic environment. Thus p53 mutant tumors may be less dependent on their vascular supply and therefore less responsive to antiangiogenic therapy [Yu et al. 2002]. Given that up to 58% of HCCs may harbor p53 mutations [Wu et al. 2007], exposure to antiangiogenic therapy may be associated with cellular overgrowth, which can ultimately foster therapeutic resistance.
Redundancy of antiangiogenic signaling pathways may also contribute to resistance. Inhibition of VEGF signaling can result in activation or upregulation of other proangiogenic signaling pathways involving members of the FGF, ephrin, and angiopoietin families. In addition, antiangiogenics may increase pericyte recruitment and neovascularization through several mechanisms, including upregulation of PDGF-β expression. The newly reorganized vasculature is less dependent on VEGF for growth and thus less susceptible to the effects of anti-VEGF therapy (reviewed by Raza and Wenger and colleagues) [Raza et al. 2010; Wenger et al. 2011].
Furthermore, studies in melanoma have identified cells similar to embryonic precursors that can develop vasculogenic-like networks that ‘mimic’ embryonic vasculogenesis. In vivo, these networks may serve as vehicles for conducting fluid such as blood within the tumor. Vascular mimicry has also been observed in breast, lung, prostate, and ovarian carcinomas [Hendrix et al. 2003] as well as in HCC. One pilot study of patients with HCC (N = 20) who underwent orthotopic liver transplantation found that patients with explanted tumors in which vasculogenic mimicry was identified experienced more rapid post-transplant disease recurrence (p = 0.01) [Guzman et al. 2007].
In model systems, including in vivo models, heightened invasiveness and in some cases increased lymphatic and distant metastases have been observed after administration of inhibitors of the VEGF pathway or genetic ablation of the VEGF gene [Du et al. 2008; Ebos et al. 2009; Paez-Ribes et al. 2009]. These observations have led to speculation that, after an initial period of tumor suppression, antiangiogenic therapy may select for apoptosis-resistant and increasingly malignant cells [Bergers and Hanahan, 2008; Blagosklonny, 2004; Loges et al. 2010]. The clinical significance of this phenomenon remains to be determined. Several studies of patients with glioblastoma concluded that the proportion of distant recurrences does not differ between patients receiving bevacizumab and those receiving other regimens [Beal et al. 2011; Chamberlain, 2011; Platten et al. 2010; Pope et al. 2011; Shapiro et al. 2009]. Given that different tumor types may respond differently to antiangiogenic therapies, the question of whether antiangiogenic therapies select for a more aggressive malignant phenotype remains an area of active discussion and investigation.
Strategies to prolong overall survival: a potential paradigm shift?
Although resistance remains a challenge with long-term use of antiangiogenic treatment for cancer in general and HCC in particular, best practices for combating resistance—and thus halting disease progression—are lacking due to the paucity of data. Current guidelines from the National Comprehensive Cancer Network (NCCN), the American Association for the Study of Liver Diseases, and the European Society for Medical Oncology recommend supportive care or participation in a clinical trial for patients with HCC whose disease progresses on sorafenib treatment [Bruix and Sherman, 2011; NCCN, 2012; Verslype et al. 2012]. There is a clear need for effective treatment options in this patient population. In addition, the heterogeneity of affected patients (including differences in etiology, stage, and liver function) underscores the need for more personalized treatment approaches. Our increased understanding of the molecular mechanisms driving resistance to cytotoxic agents and the advent of targeted therapies have enabled new approaches, such as maintenance and add-on therapies, that represent a substantial evolution from more traditional treatment approaches in oncology. The following sections highlight treatment strategies currently under investigation as well as strategies used in our own clinical practice that could support consideration of new frameworks for second-line treatment of HCC in the near future.
Continuing sorafenib in the post-progression setting
Angiogenesis remains an important mechanism in disease progression. A key question is whether patients who progress continue to derive benefit from sorafenib treatment, either as a single agent or in combination with other treatments. Clinical trials in other tumor types with other antiangiogenics may shed some light on the potential effect of continuing sorafenib therapy. The Bevacizumab Regimens: Investigation of Treatment Effects and Safety (BRiTE) prospective observational cohort study (N = 642) found that patients with metastatic colorectal cancer (mCRC) who remained on bevacizumab after disease progression on a chemotherapy plus bevacizumab regimen exhibited significantly improved survival compared with patients who switched to a nonbevacizumab-containing regimen or no treatment at all (HR 0.46, p < 0.001) [Grothey et al. 2008]. Similar results were documented in a large randomized phase III intergroup study of patients with mCRC whose disease progressed on bevacizumab plus fluoropyrimidine-based chemotherapy [Arnold et al. 2012]. In that trial, patients who remained on bevacizumab as they were switched to a second fluoropyrimidine-based chemotherapy experienced significantly prolonged survival compared with patients treated with chemotherapy alone (11.2 versus 9.8 months; p = 0.0062). This study enabled a label extension for bevacizumab for use as a second-line treatment in patients with mCRC whose disease has progressed on a first-line bevacizumab-containing regimen (see prescribing information for Avastin, Genentech, Inc., South San Francisco, CA, USA).
Although these studies suggest that targeted therapies, particularly antiangiogenic agents, may provide therapeutic benefit in the setting of tumor progression, it is not clear how generalizable this concept is. For example, in a cohort of 19 patients with glioblastoma treated with bevacizumab plus irinotecan after progression on bevacizumab monotherapy, none of the patients responded and the median PFS was 30 days [Kreisl et al. 2009]. These results underscore the fact that continued administration of targeted agents at disease progression may only be applicable to specific combinations of tumors and agents.
Two clinical studies revealed a potential beneficial effect with continual sorafenib in advanced HCC post progression. The first study examined the feasibility of using an increased dose of sorafenib after disease progression in patients with advanced HCC. In this trial, 101 patients whose disease had progressed on the standard sorafenib regimen (400 mg twice a day) were randomized to receive either an increased dose (600 mg twice a day) or best supportive care (BSC). Higher-dose sorafenib was associated with significantly longer TTP (median 4.0 versus 2.0 months; p = 0.04), incremental but nonsignificant improvement in PFS (3.9 versus 2.6 months; p = 0.07), but similar OS (6.0 versus 6.1 months; p = 0.14) [Pressiani et al. 2011]. The lack of effect on OS may reflect the fact that increasing the dose of the same inhibitor may not address the emergence of redundant pathways or reactive resistance. The second study retrospectively investigated 43 patients with HCC whose disease progressed during sorafenib therapy and who, based on clinician assessments of liver function, tumor stage, and economic status, received BSC (A), sorafenib (B), sorafenib plus locoregional treatment (C), or an alternate therapy without sorafenib (locoregional treatment or brivanib) (D) [Woo et al. 2012]. Median survival times following disease progression were 80 days, 131 days, 302 days, and 813 days in groups A, B, C, and D respectively. OS in groups B, C, and D was significantly improved compared with group A (p = 0.001), but the differences among groups B, C, and D were not statistically significant (possibly because the number of patients in each treatment group was small). Although the data in this study would seem to support the notion that sorafenib continued to provide benefit over BSC, the survival statistics are almost certainly impacted by selection bias, making it difficult to fully evaluate the differential impact of the treatment regimens. Sorafenib was considered to be tolerable, but investigators suggested caution in patients with Child-Pugh B liver function.
In contrast to the notion of continuing sorafenib in the post-progression setting, preclinical and clinical studies examined the role of temporary treatment interruption in maximizing sorafenib efficacy in progressive HCC. In a murine model of HCC, tumor cells that acquired resistance to sorafenib during continuous dosing were removed and propagated in the absence of sorafenib for at least 2 weeks. When these cells were then implanted into new hosts, sorafenib again profoundly retarded tumor growth, indicating that the tumor cells had regained sensitivity to sorafenib [Tang et al. 2010]. The investigators propose that treatment interruptions may slow or potentially reverse the development of molecular resistance mechanisms and thus confer a prolonged therapeutic effect after reintroduction of the agent. In a single case study of a 54-year-old man with advanced HCC with lung metastases, sorafenib was discontinued due to progressive disease and treatment with an investigational oral cytotoxic agent TS-1 (tegafur, gimeracil, and oteracil; Taiho Pharmaceutical Co. Ltd., Tokyo, Japan) was initiated [Okuwaki et al. 2012]. The cytotoxic agent was stopped after one cycle due to progressive disease and sorafenib was reinstituted at the patient’s request. Eight months after the reintroduction of sorafenib, the patient experienced PR; at the time the case report was published, the patient was continuing to receive sorafenib at a reduced dose (400 mg/day) [Okuwaki et al. 2012].
Sorafenib in combination with agents affecting alternative disease pathways
Disease progression during sorafenib treatment may be associated with the emergence of alternative proliferative signaling cascades that could serve as potential targets for blockade. If the basis of tumor progression is an activated alternative pathway, using a combination treatment to inhibit the additional pathway could be beneficial as long as the cascade inhibited by sorafenib remains suppressed.
Combination treatments using other targeted inhibitors have shown promise in treating HCC and other tumor types. A small phase II study (N = 59) examined combined treatment with bevacizumab and the EGFR inhibitor erlotinib for advanced HCC; median PFS was 7.2 months and median OS was 13.7 months [Kaseb et al. 2012]. Although this was a single-arm study, the median OS compared favorably with that observed in the sorafenib arms of the SHARP and AP trials (OS 10.7 and 6.5 months respectively). A multi-institution, randomized trial comparing first-line sorafenib with the bevacizumab–erlotinib combination is ongoing [ClinicalTrials.gov identifier: NCT00881751]. In melanoma, resistance to therapy with BRAF kinase inhibitors is associated with reactivation of the mitogen-activated protein kinase pathway. In a phase I/II trial (N = 247) combination therapy with a selective BRAF and a selective MEK inhibitor resulted in improved PFS in patients with metastatic melanoma [Flaherty et al. 2012].
Combination treatment with histone deacetylase (HDAC) inhibitors represents another intriguing possibility. Imbalances in HDAC activity may contribute to inappropriate gene activation that occurs in HCC [Coradini and Speranza, 2005]. A number of HDAC inhibitors, including vorinostat, which is used in the treatment of cutaneous T-cell lymphoma, have either been approved or are in development for the treatment of a variety of tumor types. Resminostat is an oral pan-HDAC inhibitor currently under investigation in patients with advanced HCC whose disease has progressed on sorafenib. In an open-label, two-arm phase I/II study, preliminary data in 15 evaluable patients showed PFS at 12 weeks in 67% of patients treated with resminostat plus sorafenib and 33% of patients treated with resminostat alone [Bitzer et al. 2012a, 2012b]. Second-line combination treatment with doxorubicin has also been explored. In a retrospective study of 14 patients treated with doxorubicin combined with sorafenib after disease progression on sorafenib, 36% achieved SD lasting more than 4 months; median PFS was 3.4 months (95% CI 2.30–8.36) and the median OS was 10.1 months (95% CI 4.34–11.45) [Abou-Alfa et al. 2012]. However, 57% of patients experienced grade 3/4 toxicity, including neutropenia, febrile neutropenia, thrombocytopenia, gastrointestinal effects, and mucositis. The authors concluded that comparative assessment of the combination versus single -agent sorafenib and BSC is needed to confirm a synergistic relationship between the agents and to define the relative benefit of the combination therapy.
Switching to an alternative agent after disease progression on sorafenib
As described above, redundancy of angiogenic pathways and upregulation of alternative pathways can contribute to antiangiogenic resistance. Switching from sorafenib to another targeted therapy at disease progression represents another potential treatment strategy for the refractory or resistant tumor. A number of targeted agents are currently under investigation in the second-line setting in advanced HCC (Table 1). It will be interesting not only to determine which among these may have clinical benefit, but also whether sequential treatment post sorafenib is preferable to combined treatment.
Clinical trials of targeted agents for second-line treatment of hepatocellular carcinoma.

CR, complete response; DB, double blind; FGF, fibroblast growth factor; FGFR, fibroblast growth factor receptor; mTOR, mammalian target of rapamycin; OL, open label; OS, overall survival; PD, progressive disease; PDGFR, platelet-derived growth factor receptor; POC, proof of concept; PR, partial response; RR, response rate; SD, stable disease, TTP, time to progression; VEGF, vascular endothelial growth factor; VEGFR, vascular endothelial growth factor receptor.
How we treat patients with advanced hepatocellular carcinoma with disease progression post sorafenib
Dr Goldenberg: In my practice, patients who demonstrate PD or intolerance to sorafenib must be reevaluated both clinically and radiographically. If a substantial decrease in performance status (PS) and marked progression of tumor burden is observed, I offer supportive care. If the patient’s PS and hepatic function remain adequate and there is evidence of locoregional progression within the liver or extrahepatic disease, I will consider treatment with additional targeted therapies if deemed potentially tolerable. It is generally accepted that patients who demonstrate progressive systemic disease are typically not candidates for locoregional intervention.
Based on the modest effect of single-agent bevacizumab in HCC, the encouraging first-line efficacy of the combination of erlotinib and bevacizumab [Kaseb et al. 2012], and the concept that withdrawal of sorafenib may unmask the activity of an inhibited cascade, we employed a combination treatment with sorafenib and erlotinib alternating every other day, plus bevacizumab every 2 weeks in 15 patients (Figure 2). This low-dose, alternating targeted regimen was designed to minimize high-grade skin toxicity. As the patients were projected to have limited survival after sorafenib progression, the extended time to further progression after multidrug treatment was encouraging.
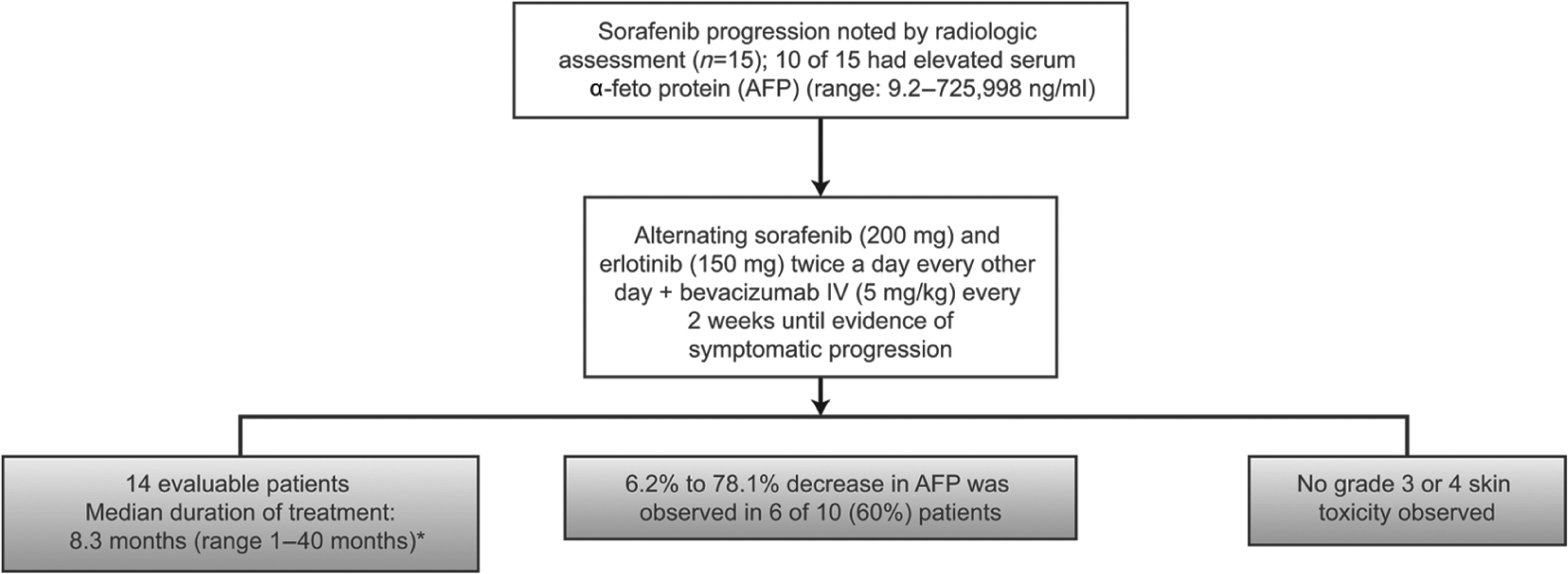
Treatment schema and outcomes in patients receiving sorafenib-based combination therapy following disease progression on sorafenib (data courtesy of Dr A. Goldenberg).
Dr He: Of 89 patients treated with sorafenib in our institution between 2009 and 2011, 32 patients continued sorafenib for a median period of 12 weeks (range 2–39 weeks) after radiologic assessment showed disease progression. Sorafenib was discontinued after symptomatic progression. A total of 33 patients received additional systemic treatments after progressing on sorafenib, and some of these patients received more than one line of treatment. Fifteen patients received second-line treatment on a clinical trial, and 18 patients received additional systemic treatment off clinical protocol due to lack of available clinical trials at the time of progression. Treatment schema for all patients and their responses are outlined in Figure 3.
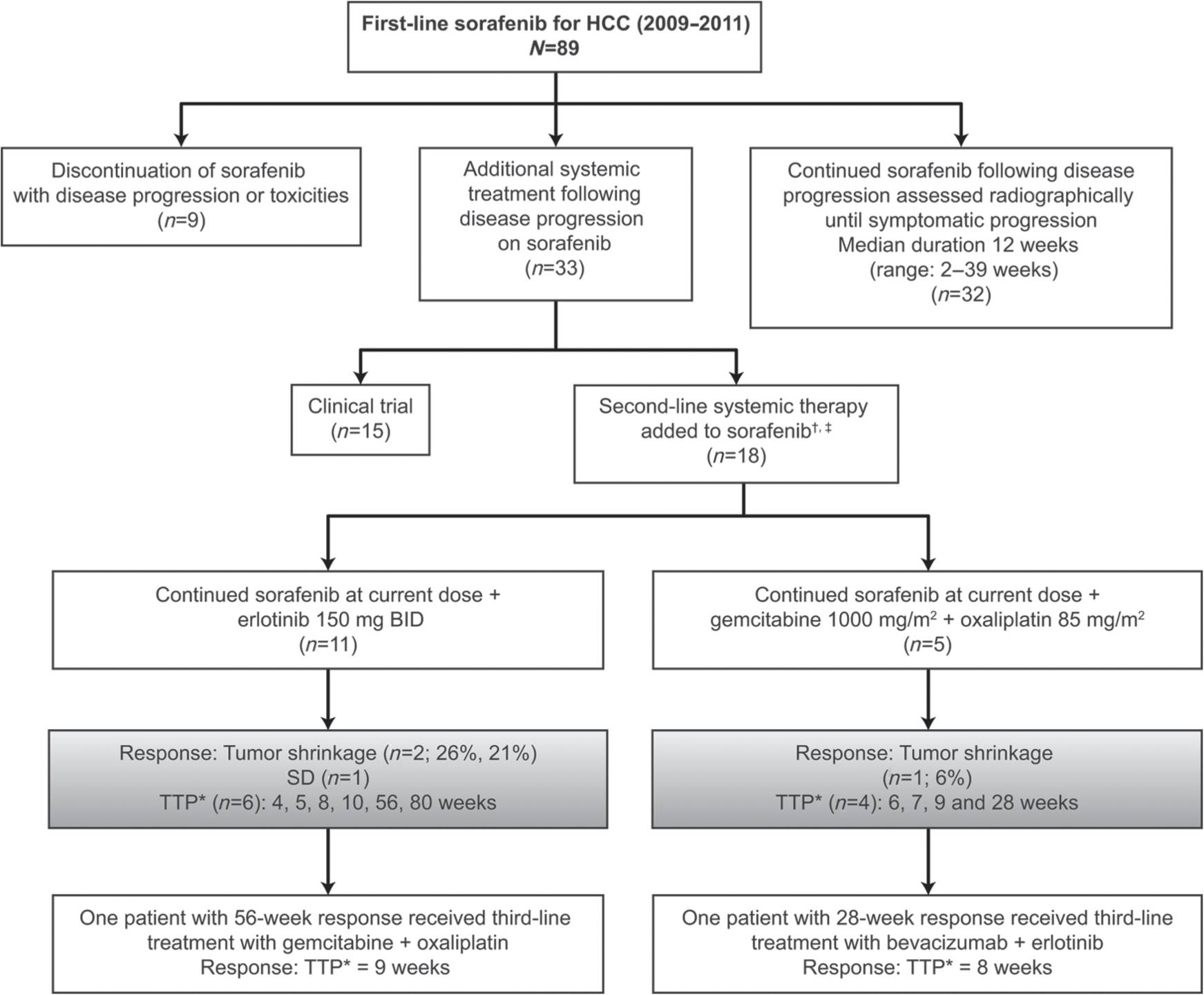
Treatment schema and response to sorafenib-based combination therapy following progression on sorafenib (data courtesy of Dr R. He).
Prolonged disease control in the subset of patients who received combination treatments cannot be explained by a slow-growing cancer, since patients progressed rapidly on the subsequent line of treatment. This observation suggests that sorafenib-based combinations may produce clinical benefit in certain patients. Unfortunately, no tumor samples from these patients are available to enable investigation of biomarkers that may predict responsiveness.
Future directions and conclusion
Sorafenib has served as the standard of care for patients with advanced HCC for the past 5 years, based on two pivotal trials supporting prolonged survival over BSC as well as other head-to-head studies failing to report an OS advantage of treatment with other agents over sorafenib. However, due to inherent genetic alterations in HCC tumor cells and signaling and epigenetic changes promoted by inhibition of angiogenesis, resistance to sorafenib may be inevitable. By exploring measures of disease progression, models of resistance, and alternative approaches to therapeutic management, this review highlights select methods that may potentially move clinicians closer to improving outcomes for patients with advanced HCC. Wider adoption of mRECIST may allow for a more accurate assessment of disease progression and facilitate meaningful comparisons of new regimens in patients with advanced HCC. Future investigations may further define the potential role of biomarkers such as AFP or DCP, or identify additional biomarkers that may play a role in predicting and monitoring therapy in clinical practice. Novel dosing strategies for sorafenib require additional study to determine their benefit in reducing resistance. While current guidelines call for BSC or clinical trial enrollment for patients whose disease progresses on sorafenib, our experience indicates that continuation of sorafenib with additional targeted therapy may provide the best chance for delaying disease progression. Based on our current understanding of the mechanisms of drug resistance, clinical trials are actively examining new approaches for reversing or circumventing sorafenib resistance. As we continue to develop greater insight into the pathogenesis of disease progression, perhaps a new paradigm for rational sequencing of therapies will emerge to provide clinically meaningful advances in survival and other outcomes for patients with advanced HCC.
Footnotes
Acknowledgements
The authors acknowledge the medical writing and editorial support provided by Percolation Communications LLC.
Funding
The authors acknowledge the funding by Onyx Pharmaceuticals, Inc. and Bayer HealthCare.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
