Abstract
Background:
Metabolic dysfunction-associated steatotic liver disease (MASLD) is more prevalent in patients with psoriasis compared to healthy individuals. Interleukin (IL)-17 and IL-23 inhibitors may have beneficial effects on MASLD by reducing systemic inflammation and improving metabolic parameters.
Objectives:
To assess the effect of IL-17 and IL-23 inhibitors on MASLD and liver fibrosis in patients with psoriasis.
Design:
We performed a systematic review that followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses guidelines.
Data sources and methods:
A literature search was conducted across four databases: MEDLINE, Embase, Web of Science, and Cochrane Central Register of Controlled Trials, from database inception to September 27, 2024. Observational studies and clinical trials that reported the presence of MASLD and/or liver fibrosis in patients with psoriasis/psoriatic arthritis treated with IL-17 or IL-23 inhibitors were included. The Newcastle Ottawa Scale (NOS) was used for risk of bias assessment in cohort studies, the Revised Cochrane Risk of Bias Tool (RoB2.0) in randomized controlled trials, and the Risk of Bias in non-randomized studies-of Interventions (ROBINS-I v.2) tool in non-randomized trials.
Results:
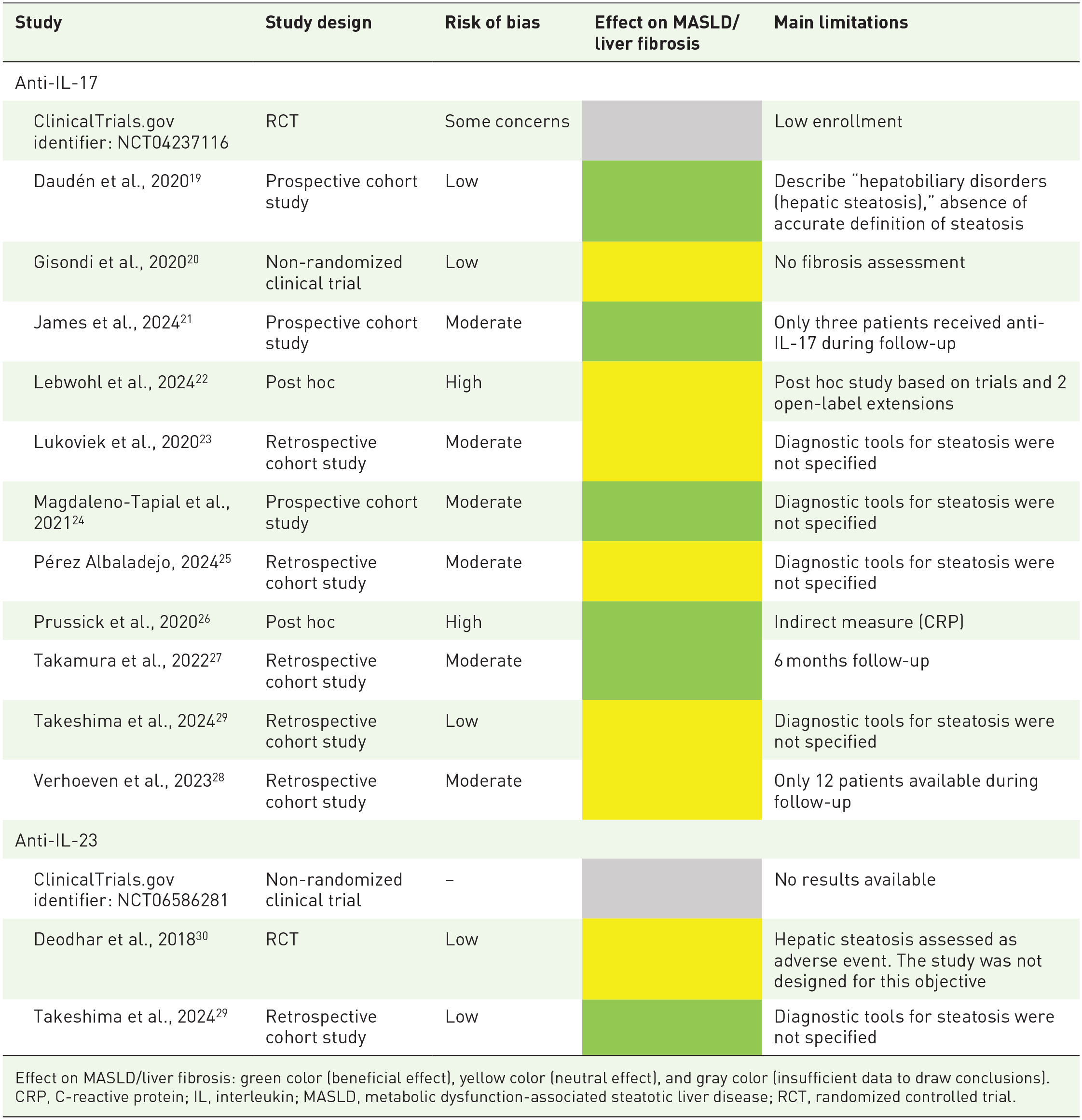
Fourteen studies were included: four clinical trials, five retrospective cohort studies, three prospective cohort studies, and two post hoc studies. Two cohort studies and one clinical trial showed a low risk of bias. Both post hoc studies had a high risk of bias. Eleven studies assessed the effect of IL-17 inhibitors on MASLD or liver fibrosis; six reported a neutral effect, while five demonstrated improvements in liver tests. Three studies evaluated IL-23 inhibitors; one showed neutral effects, another reported improvement in fibrosis-4 index (FIB-4) scores at 6 months, and the third was still in the recruitment phase.
Conclusion:
IL-17 and IL-23 inhibitors may provide beneficial effects on MASLD and liver fibrosis in patients with psoriasis. Larger, well-designed studies are needed to confirm these findings.
Trial registration:
PROSPERO CRD42024599350.
Introduction
The prevalence of metabolic dysfunction-associated steatotic liver disease (MASLD) has increased in recent years, reaching around 30% of the general population. This trend is parallel to the increase in the prevalence of type 2 diabetes mellitus (DM) and obesity, major drivers of its pathophysiology. 1 In fact, the prevalence of MASLD in patients with type 2 DM and obesity has increased above 50%.2,3 In addition, MASLD has important prognostic implications beyond liver disease, as it is an independent risk factor for cardiovascular morbidity and mortality. 4
In recent years, the terminology of the disease has changed to emphasize the influence of cardiometabolic factors on its pathophysiology, implying changes in diagnostic criteria. The classical term non-alcoholic fatty liver disease (NAFLD) was based on the presence of liver steatosis and the exclusion of other causes of hepatopathy. 5 Later, the term metabolic dysfunction-associated fatty liver disease (MAFLD) was developed in 2020 and required the presence of steatosis plus overweight/obesity, type 2 DM, or at least two metabolic risk abnormalities. 6 More recently, the term MASLD was developed in 2023 and required the presence of steatosis plus at least one cardiometabolic factor. 7
Around 40% of the global MASLD population is non-obese and almost a fifth is lean, and non-obese MASLD and lean MASLD occur in 12% and 5% of people, respectively. 8 These findings suggest that other factors, in addition to classic cardiometabolic factors, contribute to the pathophysiology of MASLD. Indeed, an association between MALSD/liver fibrosis and the systemic inflammatory burden of immune-mediated inflammatory diseases (IMID) such as inflammatory bowel disease, rheumatologic diseases, or dermatologic diseases has been described.9,10
A higher prevalence of MASLD in patients with psoriasis compared to healthy controls has been described in several studies, however, there is a lack of specific pharmacological treatment approved for MASLD. 11 Biological drugs for psoriasis, such as interleukin (IL)-17 and IL-23 inhibitors, could have beneficial effects on MASLD by decreasing the inflammatory response and metabolic syndrome. 12
Therefore, the aim of this systematic review was to assess the effect of IL-17 and IL-23 inhibitors on MASLD and liver fibrosis in patients with psoriasis.
Methodology
This systematic review was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 guidelines (PRISMA 2020). The protocol of this systematic review was pre-registered in PROSPERO (CRD42024599350). Due to the study design, institutional review board approval was not required.
Data sources and search strategy
A comprehensive literature search was carried out across multiple databases, including MEDLINE, Cochrane Central Register of Controlled Trials, Embase, and Web of Science, from database inception to September 27, 2024. In addition, clinicaltrials.gov was used to identify unpublished trials, and conference abstracts were reviewed to identify studies only published as abstracts. Finally, the references of eligible articles were reviewed to identify any potential studies that may have been overlooked in the electronic search.
Database searches were performed independently by two authors (J.G.F. and S.J.M.-D.) using the following search strategy: “Fatty Liver” OR “Non-alcoholic Fatty Liver Disease” OR NAFLD OR MASLD OR MAFLD OR “liver steatosis” OR “hepatic steatosis” OR “steatohepatitis” OR “metabolic associated fatty liver disease” OR “metabolic associated steatotic liver disease.” The Boolean operator “AND” was used to combine these terms with: anti IL17 OR anti IL23 OR guselkumab OR risankizumab OR tildrakizumab OR secukinumab OR ixekizumab OR bimekizumab OR brodalumab. No database filters, language, time, or other restrictions were applied. All references were imported to Rayyan. 13 Rayyan is an online tool designed for conducting systematic reviews and meta-analyses, allowing for the selection and review of references. This tool enables multiple researchers to review references simultaneously and independently, reducing selection bias. First, we manually reviewed all duplicate references detected by Rayyan, based on title, authors, and DOI. All authors independently conducted the selection process based on title, abstract, and full text while being blinded to each other’s decisions to minimize selection bias. Subsequently, full-text articles were reviewed again by all authors, and any discrepancies were discussed and resolved by consensus.
Selection criteria and definitions
The following inclusion criteria were applied: (1) studies assessing the effect of IL-17 or IL-23 inhibitors on liver steatosis and/or liver fibrosis, (2) studies conducted in patients with psoriasis and/or psoriatic arthritis; (3) original articles (case–control studies, cohort studies, clinical trials, post hoc), conference abstracts, editorial letters, or reports of interim results of clinical trials.
Studies were excluded in case of: (1) studies not including IL-17 or IL-23 inhibitors or results for these drugs were not reported separately; (2) studies reporting liver damage different from liver steatosis or liver fibrosis; (3) studies not including patients with psoriasis/psoriatic arthritis or results for psoriasis/psoriatic arthritis were not reported separately; (4) case reports, reviews, meta-analysis, or guidelines; (5) non-human studies; (6) in vitro studies.
Data extraction, outcomes, and analysis
Each included study was critically reviewed by all authors and discrepancies were discussed and resolved through consensus. We extracted the following variables: study location, study design, publication type, characteristics of psoriasis/arthropathy, previous systemic treatments for psoriasis, age of participants, gender, body mass index (BMI), metabolic comorbidities, years since psoriasis diagnosis, characteristics of study groups (intervention/control, exposed/non-exposed cohort), sample size, diagnosis of fatty liver and liver fibrosis, effect on fatty liver and/or liver fibrosis, and conclusions/other comments of interest.
Considering the update of terminology and diagnostic criteria in recent years, the terms NAFLD, MAFLD, or MASLD were used in the extracted data following the terminology used in the original article. In the rest of the review, the term MASLD was used as it is the most recent unless we wanted to emphasize the differences between the three terms.
NAFLD was defined as the presence of liver steatosis after excluding significant alcohol consumption and secondary causes of fat accumulation in the liver. 5 MALFD was defined as the presence of liver steatosis and one of the three following conditions: overweight/obesity (BMI ⩾25 kg/m2 in Caucasians or ⩾23 kg/m2 in Asians), type 2 DM, or at least two metabolic risk abnormalities in case of lean/normal weight patients. The metabolic risk abnormalities were: waist circumference ⩾102/88 cm in Caucasian men and women or ⩾90/80 cm in Asian men and women, blood pressure ⩾130/85 mmHg or specific drug treatment, plasma triglycerides ⩾150 mg/dL or specific drug treatment, plasma HDL-cholesterol <40 mg/dL for men and <50 mg/dL for women or specific drug treatment, prediabetes (HbA1c 5.7%–6.4%, fasting glucose 100–125 mg/dL or 2-h post-load glucose 140–199 mg/dL), homeostasis model assessment of insulin resistance score ⩾2.5 and plasma high sensitivity C-reactive protein >2 mg/L. 6 MASLD was defined as the presence of liver steatosis plus at least one cardiometabolic risk: BMI ⩾25 kg/m2 in Caucasians (⩾23 kg/m2 in Asians) or waist circumference >94/80 cm in men and women, fasting serum glucose ⩾100 mg/dL or 2-h post-load glucose levels ⩾140 mg/dL or HbA1c ⩾5.7% or type 2 diabetes or treatment for type 2 diabetes, blood pressure ⩾130/85 mmHg or specific treatment, plasma triglycerides ⩾150 mg/dL or lipid-lowering treatment, and plasma HDL-cholesterol ⩽40 mg/dL in men or ⩽50 mg/dL in women or lipid-lowering treatment. If additional drivers of steatosis were identified, it was consistent with a combination etiology called MetALD or ALD in case of alcohol. 7
The CASPAR (Classification Criteria for Psoriatic Arthritis) criteria were used to define psoriatic arthritis. 14 To meet the criteria, a patient must have inflammatory articular disease (joint, spine, or entheseal inflammation) and score ⩾3 points (Table 1).
CASPAR criteria.

CASPAR, Classification Criteria for Psoriatic Arthritis; PsA, psoriatic arthritis.
Risk of bias assessment
The risk of bias assessment was independently performed by two authors (S.J.M.-D. and J.G.F.), and disagreements were resolved by consensus involving two additional authors (L.P.-T. and M.A.M.). The Newcastle-Ottawa Scale (NOS) was used to evaluate the quality of cohort studies. 15 NOS for cohort studies contains eight items: four items for the selection of exposed/non-exposed group (representativeness of the exposed cohort, selection of the non-exposed cohort, ascertainment of exposure, and ensuring that the outcome of interest was not present at the start of the study), one item assessing the comparability of the groups, and three items for outcome evaluation (assessment of outcome, length of follow-up period, and adequacy of follow-up). A maximum of one star can be assigned to each item except for comparability, which can be assigned two stars (maximum total score of nine stars). Studies achieving at least seven stars were considered to have low risk of bias.
The Risk-of-bias visualization (robvis) is an online tool that was used to represent risk of bias of trials. 16 The RoB 2 scale is composed of five domains: Domain 1 (Risk of bias from the randomization process), Domain 2 (Risk of bias due to deviations from the intended interventions), Domain 3 (Risk of bias due to missing outcome data), Domain 4 (Risk of bias in measurement of the outcome), and Domain 5 (Risk of bias in selection of the reported result). 17 An overall low risk of bias was determined if the study had a low risk of bias for all domains. This scale was also used to evaluate the risk of bias of post hoc studies, as there is no specific scale for this type of study and they were mainly based on randomized controlled trial (RCT).
The Risk of Bias in non-randomized studies-of Interventions (ROBINS-I v.2) tool was used to evaluate the quality of non-randomized studies included in this review.16,18 This tool is composed of seven domains: Domain 1 (Bias due to confounding), Domain 2 (Bias due to selection of participants), Domain 3 (Bias in classification of interventions), Domain 4 (Bias due to deviations from intended interventions), Domain 5 (Bias due to missing data), Domain 6 (Bias in measurement of outcomes), and Domain 7 (Bias in selection of the reported result). Studies were considered to have an overall low risk of bias if they were judged to be at low risk of bias across all seven domains.
Results
A total of 244 records were identified from databases (PubMed 39, Embase 173, Web of Science 24, Cochrane 6, ClinicalTrials.gov 2). After an initial electronic search and subsequent manual identification of duplicate records, 54 records were excluded, leaving 190 records for screening. During the screening by title and abstract, 160 references were removed and 30 full-text studies were assessed. After applying inclusion and exclusion criteria, 13 studies identified through databases were included in the systematic review (ClinicalTrials.gov identifier: NCT04237116).19–29 The most frequent reasons for exclusion were: non-human studies (n = 7), no treatment with IL-17 or IL-23 inhibitors (n = 6), and no MASLD population (n = 2). One additional record was identified during the review of references cited in eligible articles. In conclusion, a total of 14 studies were incorporated into this systematic review. The study selection flow diagram can be found in Figure 1. Agreement between investigators regarding study eligibility was excellent (Κ = 0.813).

Flowchart of study selection.
Study characteristics
Five records (36%) corresponded to retrospective cohort studies23,25,27–29 and three (21%) were prospective cohort studies.19,21,24 In addition, four (29%) clinical trials were included. Among these trials, one (7.1%) was a double-blind, placebo-controlled randomized trial, 30 one (7.1%) was an open-label and non-randomized trial, 20 one (7.1%) was discontinued prior to publication but had available results (ClinicalTrials.gov identifier: NCT04237116), and one (7.1%) was in the recruitment phase (ClinicalTrials.gov identifier: NCT06586281). Finally, two (14%) post hoc studies were included.22,26 Prussick et al. 26 used data from two identically designed, randomized, double-blind psoriasis trials; and Lebwohl et al. 22 included data from five phase III/IIIb trials conducted over 2 years, including their open-label extensions.
All studies were carried out between 2018 and 2024. Eight (57%) of them were performed in Europe (ClinicalTrials.gov identifier: NCT04237116),19–21,23–25,28 another two (14%) in Japan27,29, and one (7.1%) in the USA (ClinicalTrials.gov identifier: NCT06586281), while three studies (21%) used data from multiple countries.22,26,30 The main characteristics of the studies are detailed in Table 2.
Main characteristics of the 14 trials and observational studies included in the systematic review.

AHA/NHLB, American Heart Association/National Heart, Lung, and Blood Institute; ALP, alkaline phosphatase; ALT, alanine aminotransferase; APRI, AST to platelet ratio index; AST, aspartate aminotransferase; BMI, body mass index; BSA, body surface area affected; CAP, controlled attenuation parameter; CI, confidence interval; CRP, C-reactive protein; DAPSA, Disease Activity index for Psoriatic Arthritis; DM, diabetes mellitus; ELF, enhanced liver fibrosis score; FIB-4, fibrosis-4 index; HBV, hepatitis B virus; HCV, hepatitis C virus; HSI, hepatic steatosis index; IL, interleukin; LAP, lipid accumulation product; MAFLD, metabolic-associated fatty liver disease; MASH, metabolic dysfunction-associated steatohepatitis; MASLD, metabolic-associated steatotic liver disease; MRI-PDFF, magnetic resonance imaging derived proton density fat fraction; MS, metabolic syndrome; NA, not available; NAFLD, non-alcoholic fatty liver disease; NCE ATP III, National Cholesterol Education Programme Adult Treatment Panel III; NFS, NAFLD fibrosis score; NS, non-statistically significant; PASI, Psoriasis Area and Severity Index; PsA, psoriatic arthritis; PsO, psoriasis; PUVA, psoralen and ultraviolet A; RCT, randomized controlled trial; s.c., subcutaneous; TE, transient elastography; TNF-α, tumoral necrosis factor-α; UK, United Kingdom; ULN, upper limit of normal; US, ultrasound; USA, United States of America; UVB, ultraviolet B.
Regarding the assessment of MASLD diagnosis, two (14%) studies used imaging techniques (ClinicalTrials.gov identifier: NCT04237116), 21 one (7.1%) study used controlled attenuation parameter (ClinicalTrials.gov identifier: NCT06586281), one (7.1%) study used non-invasive serological parameters, 20 two (14%) studies described a diagnostic exclusion approach,22,25 one (7.1%) relied on MAFLD criteria, 27 one (7.1%) used NAFLD criteria, 28 one (7.1%) study described “hepatobiliary disorders (hepatic steatosis)” 19 , and five (36%) studies mentioned MASLD but did not provide sufficient diagnostic criteria.23,24,26,29,30
Additionally, data on liver fibrosis were not available in five (36%) studies (ClinicalTrials.gov identifiers: NCT04237116, NCT06586281).19,20,30 Eight (57%) studies used non-invasive serological biomarkers,21–23,25–29, and one (7.1%) used non-invasive serological biomarkers combined with transient elastography (TE). 24
Participants characteristics
The sample size in the observational studies included in our review ranged, ranged from 10 participants 24 to 2845. 19
Regarding clinical trials, Deodhar et al. 30 reported a sample size of 149 participants (100 patients treated with guselkumab and 49 assigned to placebo). Similarly, Gisondi et al. 20 included 130 participants (66 treated with secukinumab and 64 treated with methotrexate). The ClinicalTrials.gov identifier: NCT04237116 had low enrollment rate with seven patients who received secukinumab and three patients receiving placebo, and it was prematurely terminated due to this reason.
Overall, there was a higher proportion of men compared to women in nine (64%) studies (ClinicalTrials.gov identifier: NCT04237116),19–21,23,25,27,29,30, and only two (14%) studies had a higher proportion of women.24,28 In addition, two (14%) studies did not provide information regarding the gender of the participants22,26 and one (7.1%) study did not report available results (ClinicalTrials.gov identifier: NCT06586281). None of the studies targeted a specific age group.
Regarding the clinical type of psoriasis, plaque psoriasis was the most frequent type found in all the studies reporting this information.19,22,23,27,29 Regarding the severity of the disease, the average PASI score of patients at the start of 6 (43%) studies was over 10 (severe psoriasis; ClinicalTrials.gov identifier: NCT04237116),19,20,22,23,30 while in 3 studies, it was under 10: Takamura et al., 27 Magdaleno-Tapial et al., 24 and Pérez Albaladejo et al. 25
Risk of bias assessment
The summary of NOS scores for risk of bias assessment of cohort studies is shown in Table 3, the Cochrane risk of bias tool scores for RCT v.2 in Figure 2, and the Risk of Bias in Non-randomized Studies-of Interventions v.2 in Figure 3.
Risk of bias assessment of the eight cohort studies included in the systematic review according to the NOS.

Single star: one point. Double stars: two points. A maximum of one star can be assigned to each item except for comparability, which can be assigned two stars (maximum total score of nine stars).
NOS, Newcastle Ottawa Scale.

Risk of bias assessment of the two randomized controlled trials included in the systematic review according to the RoB 2.0 for randomized trials.

ROBINS-I tools (Version 2).
Our analysis showed that two cohort studies (25%) had a NOS total score ⩾7 points,19,29 whereas six studies (75%) had 5 or 6 points.21,23–25,27,28 A weakness found in the studies included in this systematic review was that five of them (63%) lacked an unexposed cohort.23–25,27,28 However, all studies adequately represented the exposed cohort, focusing on patients with moderate-to-severe psoriasis treated with IL-23 or IL-17 inhibitors.
Two randomized clinical trials (ClinicalTrials.gov identifier: NCT04237116) 30 and two post hoc studies22,26 were included. One trial was judged to have a low risk of bias across the five domains 30 whereas the other trial showed some concerns regarding adherence to the intervention (ClinicalTrials.gov identifier: NCT04237116). In contrast, both post hoc studies22,26 had a high risk of bias due to the selection of reported results.
Finally, the open-label, non-randomized clinical trial showed a low risk of bias for all domains of ROBINS-I v.2. 20
Prevalence of MASLD and liver fibrosis
The prevalence of MASLD was reported in three studies21,27,28 and ranged from 30% to 83%. Most studies compared continuous values of non-invasive fibrosis biomarkers during follow-up21–25,28,29 while Lukoviek et al. 23 also reported the prevalence of patients with fibrosis-4 index (FIB-4) >1.3 (13%) at baseline, Takamura et al. 27 the prevalence of FIB-4 >1.3 (21%) and NAFLD Fibrosis Score (NFS) >−1.455 (25%), and Takeshima et al. 29 the prevalence of FIB-4 >1.3 at baseline (38% for secukinumab, 28% for ixekizumab, 33% for brodalumab, 22% for guselkumab, and 20% for guselkumab).
Impact of treatment with IL-17 inhibitors on MASLD and liver fibrosis
On the one hand, 6 (50%) of 12 studies showed a neutral effect of IL-17 inhibitors on liver outcomes.20,22,23,25,28,29 Gisondi et al. 20 in an open-label, non-randomized clinical trial, concluded that treatment with secukinumab for 12 months had a neutral effect on metabolic parameters as well as the hepatic steatosis index (HSI) and the lipid accumulation product (LAP). Similarly, Lebwohl et al. 22 in a post hoc analysis including five clinical trials, found that treatment with bimekizumab for 2 years did not increase FIB-4 and APRI scores. In addition, four retrospective cohort studies found no differences on FIB-4 after treatment with IL-17 inhibitors.23,25,28,29 Moreover, Takeshima et al. 29 observed no change in FIB-4 after treatment with IL-17 inhibitors in patients with FIB-4 >1.3 at baseline. Although Lukoviek et al. 23 observed an improvement after treatment with secukinumab in six patients with FIB-4 >1.3 at baseline, the authors concluded that secukinumab seemed to have no influence on NAFLD.
On the other hand, 5 (42%) of 12 studies showed a beneficial effect of IL-17 inhibitors on liver outcomes.19,21,24,26,27 Daudén et al. 19 conducted a prospective cohort study that evaluated the long-term safety of various systemic drugs for psoriasis through the BIOBADERM registry. This was a Spanish national multicenter registry that collected data on adverse medication effects and observed that patients treated with secukinumab had a lower risk of hepatic steatosis compared to those receiving methotrexate (adjusted incidence ratio 0.1, 95% confidence interval (CI) (0–0.5); p < 0.01). James et al., 21 in a prospective cohort, reported an improvement in magnetic resonance imaging (MRI) derived proton density fat fraction after 6 months of treatment with secukinumab, although no differences were observed in serological markers. However, Takamura et al. 27 observed an improvement in serological indices NFS and FIB-4 after 6 months of treatment in their retrospective cohort.
In addition, Magdaleno-Tapial et al., 24 in another prospective cohort study where patients were treated with secukinumab for at least 2 years, observed an improvement in liver stiffness measured by TE, regardless of weight, BMI, and analytical values. Prussick et al. 26 published a post hoc study including two identically designed RCT, where a decrease in C-reactive protein was found in patients with elevated aspartate aminotransferase (AST)/alanine aminotransferase (ALT) ratio (>1.4) who were treated with brodalumab (16.2 vs 2.8 mg/L, p = 0.013).
Finally, one (8.3%) study (ClinicalTrials.gov identifier: NCT04237116) had a very low enrollment so we could not draw solid results or conclusions. This was a terminated RCT that assessed the effect of secukinumab versus placebo on ALT levels in patients with psoriasis. Only 10 patients completed follow-up (ClinicalTrials.gov identifier: NCT04237116). ALT levels improved at week 12 (40.3 ± 12) compared to baseline (60.5 ± 35.7) in patients treated with secukinumab.
Impact of treatment with IL-23 inhibitors on MASLD and liver fibrosis
Only one study was specifically designed to assess the effect of IL-23 inhibitors on liver outcomes. This retrospective cohort study observed a significant improvement in FIB-4 after 6 months of treatment with IL-23 inhibitors (guselkumab and risankizumab). 29 In addition, the RCT published by Deodhar et al. 30 reported the presence of hepatic steatosis as potential adverse event of IL-23 inhibitors, with a low prevalence 0–3% in both IL-23 inhibitors and placebo groups. The NCT06586281 was in the recruiting status so there were no results available (ClinicalTrials.gov identifier: NCT06586281).
Results on the impact of IL-17 and IL-23 inhibitors on MASLD and liver fibrosis in patients with psoriasis are summarized in Table 4.
Summary of the effects of IL-17 and IL-23 inhibitors on MASLD and liver fibrosis in patients with psoriasis.
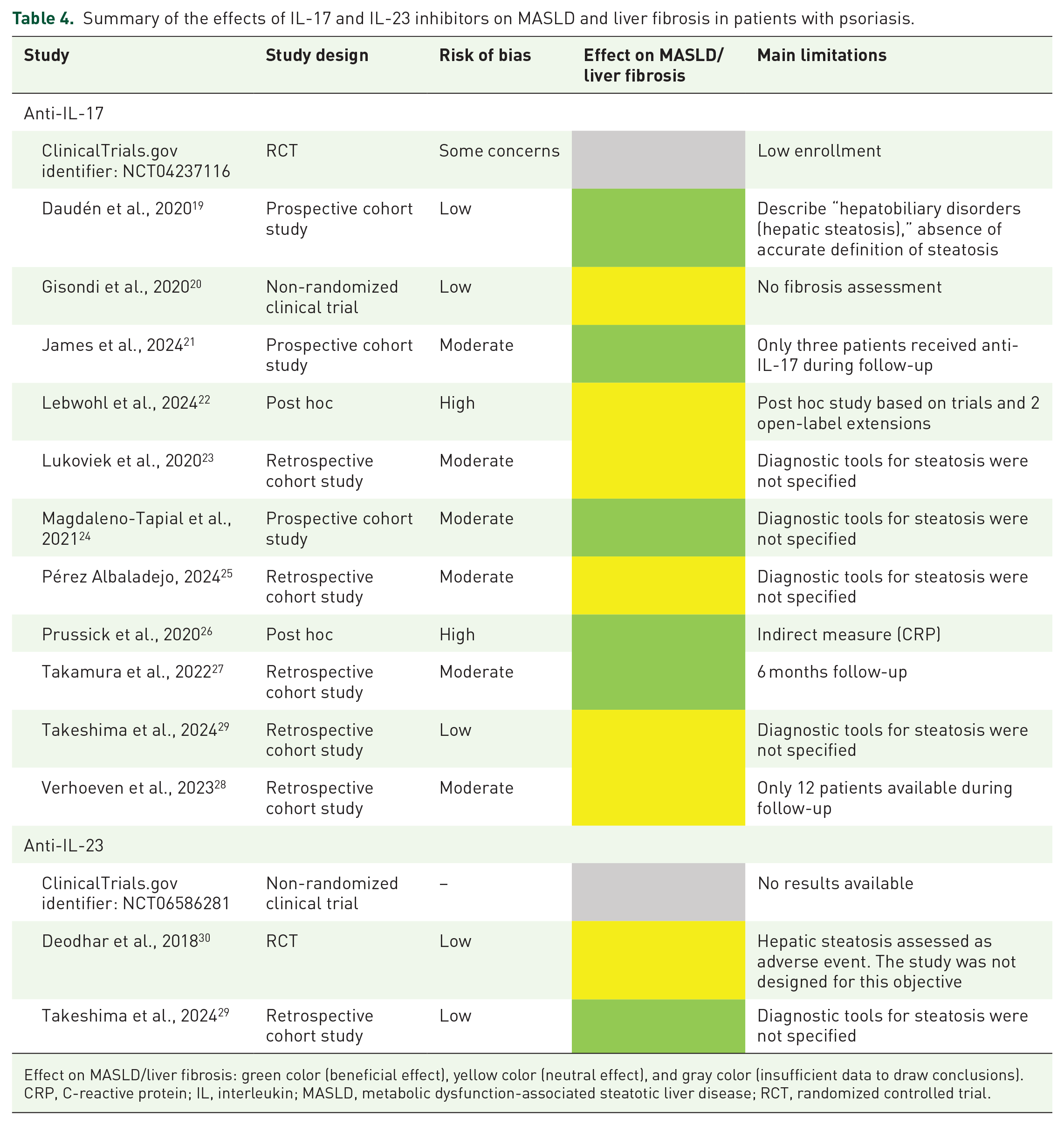
Effect on MASLD/liver fibrosis: green color (beneficial effect), yellow color (neutral effect), and gray color (insufficient data to draw conclusions).
CRP, C-reactive protein; IL, interleukin; MASLD, metabolic dysfunction-associated steatotic liver disease; RCT, randomized controlled trial.
Discussion
Given the relationship between psoriasis and MASLD, the shared pathophysiological mechanisms, and the lack of specific treatment for MASLD, we conducted a systematic review to assess the effects of IL-17 and IL-23 inhibitors on MASLD and liver fibrosis in patients with psoriasis. We found that IL-17 and IL-23 inhibitors may have a beneficial effect on MASLD, consistent with its pathogenesis.
Despite the heterogeneity in the terminology used for liver disease by different studies, a high correlation between all terms has been described in literature, especially between NAFLD and MASLD. 31 In addition, a complete overlap between patients with NAFLD and MASLD in terms of metabolic comorbidities and degree of liver injury has been recently demonstrated in other series including IMID. 32
MASLD is the leading cause of liver disease and fibrosis in individuals with risk factors, affecting 25%–30% of the general population and occurring 1.5–3 times more frequently in patients with psoriasis, with a prevalence of up to 65% in this group.1,33 The close relationship between MASLD and psoriasis is attributed to shared systemic inflammation, leading Mantovani et al. 34 to propose the “liver-skin axis.” In this axis, T cells from psoriasis lesions release pro-inflammatory cytokines such as TNF-α, IL-6, and IL-17, which enter systemic circulation and contribute to the pathogenesis of MASLD. Conversely, hepatic macrophages may exacerbate psoriasis lesions by producing similar pro-inflammatory molecules. 34 This association is particularly evident in severe cases of psoriasis, regardless of other risk factors. 33
Th17 cells and IL-17 play a key role in the pathophysiology of both conditions, linking the progression of MASLD to steatohepatitis and liver fibrosis in psoriasis. 35 Studies in animal models have shown that IL-17 neutralization improves hepatic steatosis, suggesting a potential therapeutic strategy targeting this shared mechanism.36,37 IL-23 promotes the differentiation of Th17 cells and the production of IL-17. 38 In addition, IL-23 is responsible for subclinical inflammation in psoriasis patients without visible skin lesions. This has led some studies to investigate its effect on patients with MASLD.29,30
Regarding human studies, this systematic review included 14 studies that assessed the impact of IL-17 and/or IL-23 inhibitors in the development or progression of MASLD and liver fibrosis in patients with psoriatic disease. Studies that assessed the effects of IL-17 inhibitors on MASLD and/or liver fibrosis showed either a neutral effect on liver status or a significant improvement. None of the studies reported a detrimental effect of IL-17 inhibitors in psoriasis patients with MASLD or liver fibrosis.
In the BIOBADERM registry, it was noted that patients treated with secukinumab had a lower risk of hepatic steatosis compared to methotrexate. 19 However, this study does not define the methods or diagnostic criteria for hepatic steatosis, nor does it reflect the effects of secukinumab in patients with an established diagnosis of MASLD or liver fibrosis. It would be highly beneficial for national multicenter registries like this to provide more detailed data to allow a better assessment of MASLD and liver fibrosis in psoriatic patients in future publications.
Lebwohl et al. 22 conducted a post hoc analysis of five clinical trials evaluating the effects of bimekizumab on serological indices of liver fibrosis, finding no significant differences after 2 years of treatment. This study calculated the FIB-4 and APRI serological indices at baseline and after 2 years of treatment. No statistically significant differences were found, but in the groups at higher risk of liver fibrosis (FIB-4 >1.3 and APRI >0.5), a numerical improvement in both indices was observed. Similarly, Gisondi et al. 20 examined the effects of secukinumab over 1 year on HSI and the LAP. No differences were observed in LAP, whereas an improvement in HSI was noted after 1 year of treatment with secukinumab, although not statistically significant.
Lukoviek et al., 23 in a retrospective cohort study on secukinumab, found a reduction in FIB-4 levels in patients with elevated indices following treatment. Of the 68 study participants, 9 showed a FIB-4 >1.3, and 6 of them improved after switching to secukinumab, although the follow-up period was not specified. Takamura et al., 27 in a retrospective cohort study, observed improvements in the NFS and FIB-4 indices after 6 months of treatment with ixekizumab and secukinumab. Similar findings were reported by Prussick et al. 26 in their post hoc analysis: patients with elevated AST/ALT indices treated with brodalumab and ustekinumab (anti-IL-12 and IL-23) showed significant improvement after 48 weeks compared to those treated with ustekinumab alone. Patients with an AST/ALT ratio >1.4 were analyzed, and a reduction in C-reactive protein was observed at 48 weeks, but not in FIB-4. This suggests that anti-IL-17 could reduce liver inflammation, even if this is not reflected in transaminase levels. However, C-reactive protein is not a specific marker for the liver. James et al. 21 also performed a retrospective cohort study on patients treated with secukinumab. While they found no differences in serological markers of hepatic steatosis, they observed significant improvement in MASLD using imaging tests (MRI). From our point of view, a possible explanation for these mixed findings could be differences in study design, treatment duration, and patient populations.
Regarding the effects of IL-23 inhibitors, fewer studies are available in the literature. Deodhar et al. 30 conducted a RCT where guselkumab was compared against placebo and reported cases of hepatic steatosis at 52 weeks as “adverse events”; however, the study was not specifically designed for this purpose and extracted data should be cautiously interpreted. Takeshima et al. 29 demonstrated in their retrospective cohort study a significant improvement in patients with elevated FIB-4 after 6 months of treatment with guselkumab or risankizumab, whereas no significant differences were observed in patients treated with anti-IL-17 therapies. However, it would be very useful to know the progression of non-invasive biomarkers in the long term.
A possible explanation for the beneficial effects of anti-IL-23 therapies is their role in Th17/Treg cell regulation, reducing differentiation into Th17 cells and thereby lowering IL-17 levels. 38 However, these results should be interpreted with caution, as the study conducted by Takeshima et al. 29 observed that secukinumab and ixekizumab were more frequently used in patients with PsA compared to other biologics. This suggests another possible explanation: PsA patients exhibit greater systemic inflammation compared to patients with plaque psoriasis. Furthermore, this is a retrospective cohort study. More prospective studies are needed to provide stronger scientific evidence. Currently, there is an open-label, non-randomized clinical trial recruiting patients to evaluate the effect of guselkumab on MASLD in psoriasis patients.
The variability in the diagnostic methods used for MASLD and liver fibrosis is one of the limitations of this review. In some studies, the diagnosis is mentioned without a clear definition, while others rely on specific criteria, confirming steatosis or fibrosis with indirect serological markers (such as the HSI, FIB-4) or imaging modalities such as liver elastography or MRI. A similar heterogeneity is evident in the evaluation of treatment response, where some studies assess the effect by measuring liver enzymes or C-reactive protein levels, and others by observing changes in imaging tests. This variability limits the generalizability of our results and complicates direct comparisons across the studies.
Despite the limitations, these studies support the hypothesis that early application of biological treatments may modify the disease course. 39 These therapies targeting TNF-α, IL-17, IL-12, and IL-23 could reduce inflammation, prevent the emergence of resident memory T cells, and ultimately decrease the comorbidity and mortality associated with psoriatic disease. 40
The studies included in this systematic review varied in their methodological quality, most of them were observational, and some of them had a moderate to high risk of bias due to potential confounding factors. In addition, two studies were post hoc so liver assessment was not primarily defined as an endpoint. The short follow-up duration of the studies (a maximum of 2 years) and the limited sample size during follow-up was another limitation of the included studies. For conditions such as liver fibrosis or MASLD, where disease progression could be slow, the follow-up time should ideally continue for several years. Additionally, several studies lacked baseline data on liver disease severity or did not include an unexposed control cohort, which restricts the interpretability of the findings. Only one completed RCT was included, which was deemed to have a low risk of bias and showed results regarding the impact of IL-23 inhibition. 30 However, this RCT did not assess the potential improvement in liver fibrosis, the main long-term prognostic factor of liver disease.
The variability in the terminology used for liver disease and diagnostic criteria for MASLD or NAFLD across the included studies also represented a challenge in synthesizing the results. Some studies relied on imaging techniques like liver TE 24 or MRI, 21 while most of them used indirect indices such as FIB-4, NFS, APRI, or even CRP, which may not be as accurate in early-stage fibrosis detection. This lack of standardization in diagnostic approaches may account for some of the discrepancies observed between studies.
This is, to our knowledge, the most recent systematic review on this topic. While other reviews assessed the role of previously approved systemic treatments for psoriasis, this review specifically focused on the effect of novel therapies such as IL-17 and IL-23 inhibitors. Our review included a relatively large number of studies, considering that these drugs have recently been approved. In addition, our results were biologically plausible. However, new prospective and specifically designed studies are needed in the future to evaluate the effect of these drugs on MASLD in patients with psoriasis.
Conclusion
This systematic review, which includes the latest and highest-quality studies to date, highlights the potential beneficial effect of IL-17 and IL-23 inhibitors on MASLD and liver fibrosis in patients with psoriasis. However, further high-quality prospective studies are needed to confirm these findings.