Abstract
Inflammatory bowel diseases (IBD) and psoriasis are chronic inflammatory conditions belonging to the heterogeneous group of immune-mediated inflammatory diseases (IMIDs). A significant bidirectional link between these two entities has been observed, conditioning an increased risk of IBD in patients with psoriasis and vice-versa. Biological therapies used for IBD may lead to the occurrence of psoriasis as a “paradoxical reaction.” The objective of this study is to analyze the current evidence on the association between psoriasis and IBD, particularly finding case reports of the appearance or aggravation of psoriasis under therapy with interleukin-12/23 (IL-12/23) and IL-23 inhibitors. We conducted comprehensive research to identify studies examining the association between psoriasis and IBD and to find case presentations that reported the appearance or aggravation of psoriasis under biologic therapy with IL-12/23 and IL-23 inhibitors up to March 2024. Clinical trials for IL-12/23 and IL-23 inhibitors in IBD were analyzed to find cases of paradoxical psoriasis as registered adverse events. The sources of evidence are PubMed and ClinicalTrials.gov. For each included case report, data on patient characteristics concerning their age, sex, and comorbidities were selected. Moreover, information regarding the indication for biologic therapy, time to onset of paradoxical psoriasis after starting treatment, clinical presentation, and management of the paradoxical psoriasis was extracted. We found 10 reported cases of ustekinumab-induced new-onset or worsening psoriasis and one reported case of paradoxical psoriasis induced by risankizumab in the literature. Four cases of paradoxical psoriasis have been also registered in clinical trials involving ustekinumab treatment in IBD. Psoriasis can constitute a rare paradoxical adverse event of ustekinumab treatment, but further studies are needed to better clarify the cytokine imbalance that leads to this phenomenon induced by inhibition of IL-12/23 and IL-23. Still, few real-world data exist to draw any conclusions, but risankizumab may positively treat psoriasis induced by ustekinumab.
Case report
We present a case report of a 30-year-old female patient with Crohn’s disease (CD) with a personal history of tonsillectomy and a familiar history of colon rectal cancer (maternal grandfather). She has been affected by an inflammatory form of CD localized to the terminal ileum with concomitant peri-anal disease since January 2010. In her medical history, the patient has experienced multiple disease flare-ups treated first with cycles of steroids and successively with adalimumab from January 2013 to February 2018, when she experienced a clinical flare confirmed by ileocolonoscopy and bowel ultrasound. She then started ustekinumab in March 2018, with a classical regimen with intravenous induction and then 90 mg subcutaneously every 8 weeks that led to and maintained clinical, endoscopic, and ultrasonographic remission. In July 2022, the patient experienced a new disease flare, characterized by a Harvey–Bradshaw index (HBI) score of 8, a fecal calprotectin level of 457 mcg/g, and ultrasound findings indicating thickening of the terminal ileum over a 15 cm length, with a maximum wall depth of 5.5 mm. Following the significant and prolonged remission achieved with ustekinumab, a new reinduction phase was initiated with a single intravenous administration of ustekinumab at a dosage of 6 mg/kg, followed by maintenance therapy of 90 mg every 8 weeks. In October 2022, clinical remission (HBI < 3) was assessed with a fecal calprotectin level of less than 150 mcg/g, with normal routine blood tests. She reported, a few weeks before, the onset of a cutaneous lesion on the lower third of the legs. Physical examination revealed well-defined, erythematous, scaly plaques suspicious of psoriasis (Figure 1). A following punch biopsy of the skin lesions showed histological findings consistent with the diagnosis of psoriasis (Figure 2). After a multidisciplinary discussion with the dermatology department, it was decided to switch the biologic therapy from ustekinumab to risankizumab with the following induction schedule: 150 mg at week 0 and week 4 and then every 12 weeks. After 6 months of risankizumab therapy, there was almost complete resolution of the psoriatic lesions and the CD remained in clinical remission (the patient reported 1–2 bowel movements per day without smooth evacuation or abdominal mass), biomarker remission (CRP <0.3 mg/dL and fecal calprotectin <150 mcg/g), and with a maximum wall thickness of 4.2 mm in the ileum. In April 2024, risankizumab was adjusted to gastroenterological dosage, that is 360 mg every 8 weeks.
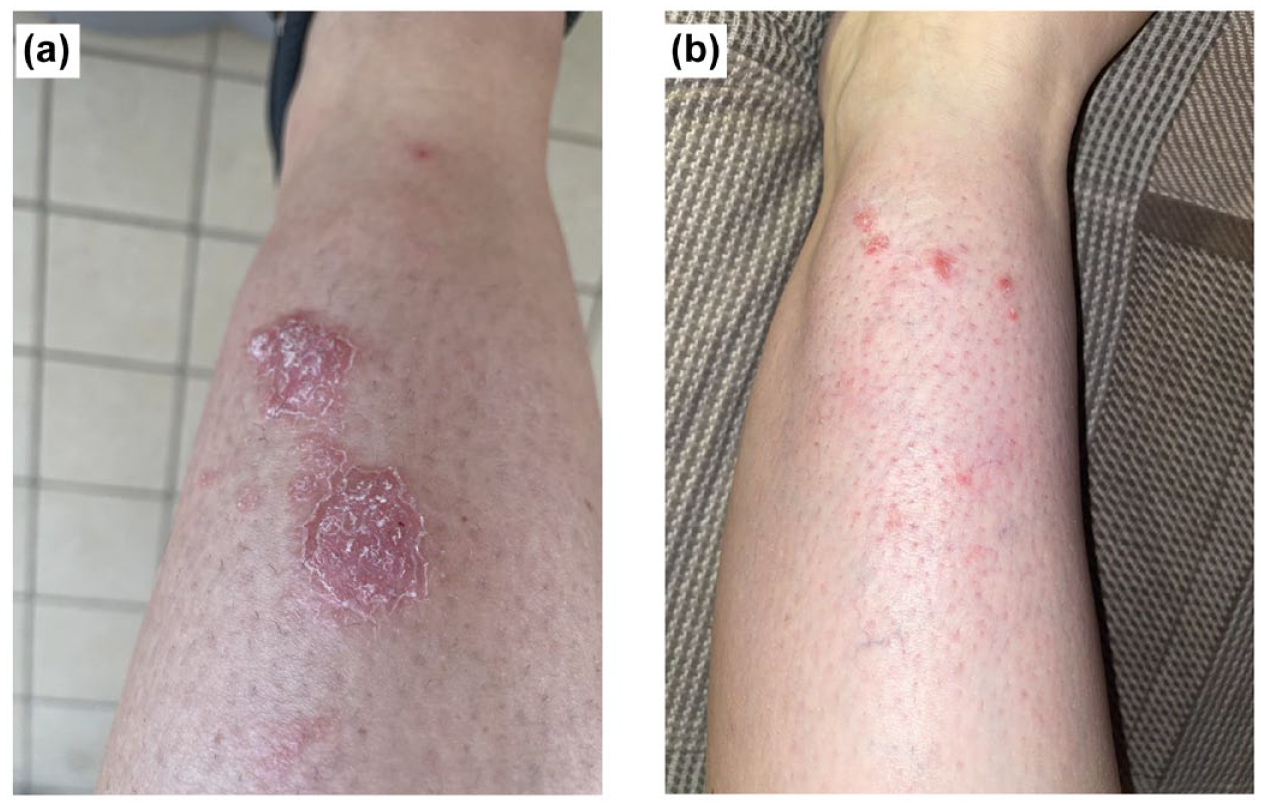
Psoriasis of the left leg. (a) Onset during therapy with ustekinumab. (b) Resolution during therapy with risankizumab.
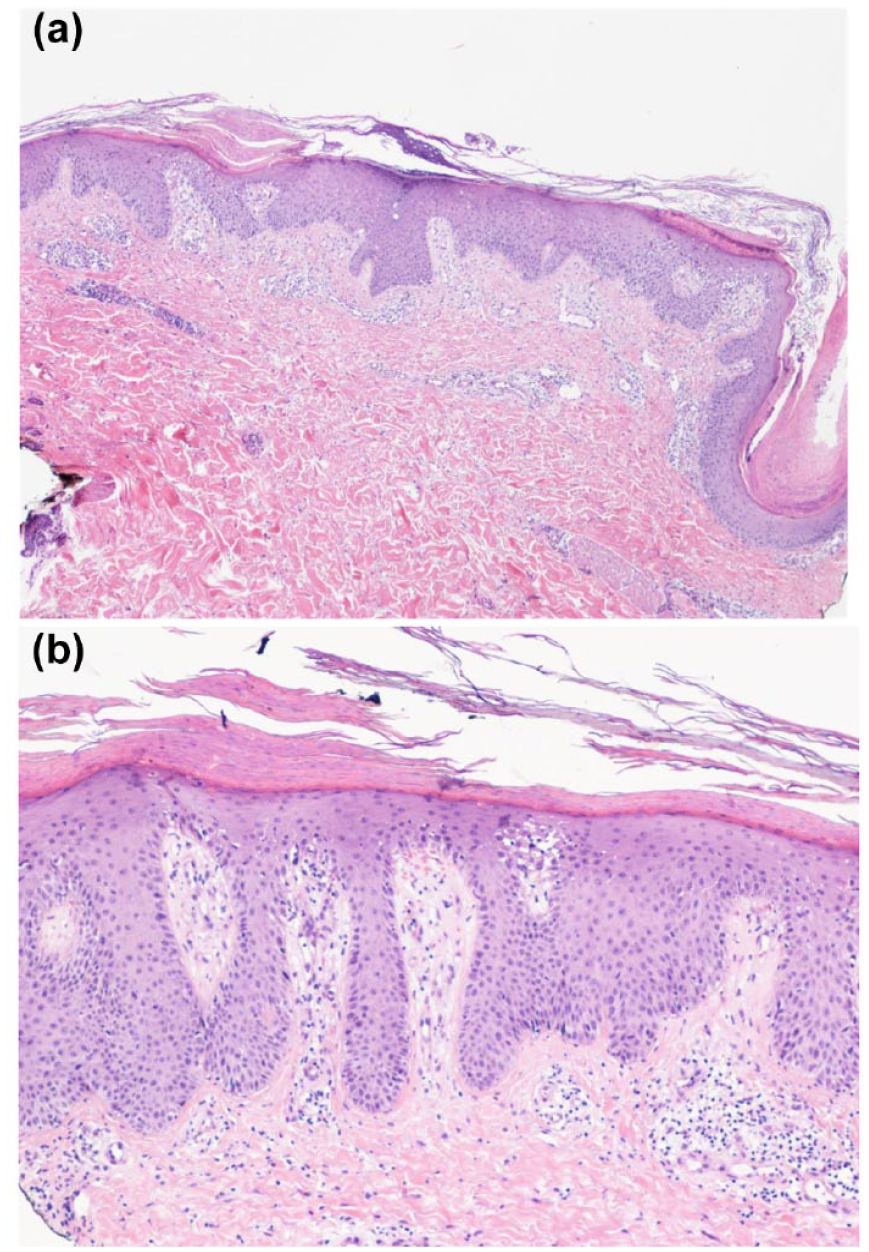
Histopathologic findings at punch biopsy of the leg. (a) Small magnification (EE 4×). The epidermis shows moderate acanthosis and plurifocal spongiosis. Mild orthokeratosis is noted along the entire surface, and plurifocal parakeratosis is also seen at the hair infundibulum. Small and scattered intracorneal accumulations of polymorphonucleates in the context of parakeratosis. In the dermis, the presence of mixed superficial inflammatory infiltrate and capillary ectasia is predominantly in the papillary dermis. (b) Moderate magnification (EE 10×), we note psoriasiform and more regular epidermal hyperplasia, focal spongiosis, and parakeratotic scale on the surface. The inflammatory infiltrate is shown to consist of elements of chronic inflammation, small lymphocytes, and histiocytes, mixed with polymorphonucleates, with predominantly superficial pericapillary arrangement. No obvious eosinophilic granulocytic component is noted. Small extravasation of emazias. Minimal surface edema in association with capillary ectasia in the papillary dermis. Conclusion (for both): the histologic picture is consistent with the clinical hypothesis of psoriasis.
In this patient with CD was the new onset of psoriasis caused (or facilitated) by ustekinumab or it would have happened anyway? This review aims to tentatively respond to this question after a careful revision of the current evidence about the association between psoriasis, inflammatory bowel disease (IBD), and biologic therapies targeting interleukin-12/23 (IL-12/23) and IL-23.
Introduction
IBDs and psoriasis are chronic inflammatory conditions characterized by a lifelong relapsing-remitting course.1,2 Both entities belong to the heterogeneous group of immune-mediated inflammatory diseases (IMIDs),2,3 consisting of apparently unrelated conditions involving common multifactorial etiology and inflammatory pathways. 3 In particular, this group of illnesses is characterized by an uncontrolled abnormal immune response which subsequently leads to increased production of inflammatory cytokines causing tissue damage and maintaining the chronic process. 2
IBD includes ulcerative colitis (UC) and CD, chronic inflammatory disorders of the gastrointestinal tract, often associated with concomitant IMIDs. 4 Skin diseases are among the most common IMIDs observed in patients with IBD, 4 with evidence for a significant association between IBD and psoriasis. 5 Several studies reported a significant bidirectional association between these two entities, conditioning an increased risk of IBD in patients with psoriasis and vice versa.4,5 This observed link has been hypothesized to be driven by shared genetic, microbiological, immunological, and environmental factors.2–4
Biological agents revolutionized the treatment of IMIDs by targeting specific immune modulation. Distinct IMIDs differentiate for synthesis and function of different inflammatory cytokines, thus justifying the different responsiveness to corticosteroids, immunosuppressors, biologics, and small molecules. 2 The widening use of biological agents across rheumatologic, dermatologic, and gastrointestinal diseases highlighted a new class of adverse events, the so-called paradoxical reactions. 6 The skin is frequently involved in these inflammatory immune-mediated reactions and cutaneous paradoxical manifestations became a specific class effect of targeted biological agents. Among these, paradoxical psoriasiform reactions have been increasingly recognized since the widespread use of biological agents. 6 Particularly, paradoxical psoriatic reactions induced by tumor necrosis factor (TNF)-inhibitors are now well known,6,7 first described in rheumatologic patients and subsequently across other indications, such as IBDs. 4
Similar cases of paradoxical reactions have also been reported following the long-term use of new anti-interleukin biologic agents, including IL-12/23. 6 Less is known about the possible paradoxical cutaneous effects of IL-23 inhibitors, which are currently used with success in IBD, psoriasis, and psoriatic arthritis. 8 On the contrary, another class of biologic therapy, namely anti-IL-17, currently used for psoriasis, may induce IBD or worsen its clinical course. 9
The co-occurrence of two or more IMIDs in the same patient is frequent, and IMID patients are generally more likely than the general population to have another IMID, also related to specific treatments.6,7,10 Sometimes, it might even be difficult to differentiate concomitant IMIDs from paradoxical reactions occurring among patients with immune-mediated pathologies following the use of biologics. 2 A retrospective observational study is ongoing (PROTECT-IMID, ClinicalTrials.gov number NCT05696106), aiming at assessing the risk of an incident second IMID in patients treated with biologics (including IL-23 inhibitors), small molecules, thiopurines, or methotrexate for a first IMID (IBD, inflammatory rheumatologic diseases, or psoriasis).
This review aims to analyze the current evidence on the peculiar association between psoriasis and IBD, being psoriasis either a concomitant IMID frequently observed in IBD patients or even a possible cutaneous paradoxical adverse event triggered by biologics used to treat IBD.
Materials and methods
We searched in the PubMed database until March 2024 to identify studies examining the association between psoriasis and IBD and to find case presentations that reported the appearance or aggravation of psoriasis in the context of biologic therapy with IL-12/23 and IL-23 inhibitors. The following terms were used: “Inflammatory Bowel Diseases,” “Crohn’s Disease,” “Ulcerative Colitis,” “Psoriasis,” “IMID,” “Paradoxical psoriasis,” “Paradoxical psoriasiform reactions,” “IL-12/23 inhibitors,” “IL-23 inhibitors,” and “IL-23 blockers.” References of original articles and relevant reviews were screened to find additional publications. The search was limited to articles published in English (Supplemental material).
One independent reviewer extracted data from the included studies using a standardized data extraction form, according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) recommendations. 11 For each included case report, data on patient characteristics concerning their age, sex, and comorbidities were selected. Moreover, information regarding the indication for biologic therapy, time to onset of paradoxical psoriasis after starting treatment, clinical presentation, and management of the paradoxical psoriasis was extracted.
In addition, clinical trials for IL-12/23 and IL-23 inhibitors in CD and UC were searched and analyzed in the ClinicalTrials.gov database to find cases of paradoxical psoriasis as registered adverse events.
We conducted a narrative synthesis of the findings from the included databases, analyzing the current evidence on the association between psoriasis and IBD, and particularly finding case reports of appearance or aggravation of psoriasis under biologic therapy with IL-12/23 and IL-23 inhibitors, thus identifying areas for future research.
IBD and psoriasis
Epidemiology
IBD and psoriasis are both chronic relapsing disabling IMIDs showing a wide spectrum of manifestations among affected individuals.2,3
The phenotypes of IBD traditionally involve UC, characterized by mucosal colonic inflammation continuously and with different extensions from the rectum, 12 and CD, which causes transmural inflammation discontinuously and theoretically affects any part of the gastrointestinal tract, even if terminal ileum and colon are the most frequent involved sites. 13
On the other hand, psoriasis is a chronic papulosquamous skin disease involving several phenotypes, with chronic plaque or psoriasis vulgaris being its most common form (90% of all cases).14,15 Psoriasis vulgaris is characterized by well-demarcated, erythematous plaques covered by silvery lamellar scales, 15 anatomically symmetric and mainly localized in extensor sides of the knees and elbows, in the lumbosacral region, and in the scalp. 14 Furthermore, psoriasis consists of other less common forms including guttate (2% of all cases), erythrodermic (2%–3%), and pustular psoriasis.14,16 Erythrodermic is a severe form of psoriasis and may be potentially life-threatening 14 since it impairs the thermoregulatory capacity of the skin 16 ; any form of psoriasis, during a flare, may become erythrodermic. 15 Other forms of psoriasis are “inverse or flexural” (involving skin folds and intertriginous areas), palmoplantar, sebopsoriasis, and nail psoriasis.14,15
The connection between IBD and psoriasis was first reported in 1968. 17 It has been estimated that psoriasis is present in 3.6% of patients with CD in 2.8% of patients with UC and up to 6.7% of IBD patients treated with anti-TNF agents.3,5,18 On the contrary, it has been observed that the prevalence of IBD is higher in patients with psoriasis than in age- and sex-matched controls (odds ratio (OR) 2.49 for CD and 1.64 for UC). 19 We have to underline that variations in prevalence data of psoriasis and IBD as well as of their association have been reported in different age groups and different geographical regions and nationalities.5,20
Other interesting relationships have been reported in this peculiar setting. For instance, the risk of developing IBD in patients with psoriasis appears to be higher in females and young people, 21 and a mild form of psoriasis, instead of the plaque type, appears to be more frequently associated with IBD. 22 Moreover, the clinical course of the intestinal disease appears to be worsened in patients with CD and psoriasis than in those with CD only 23 while the severity of psoriasis appears to be associated with a higher risk of developing IBD. 5
Pathogenesis
The most widely accepted pathogenic model predicts that both IBD and psoriasis are the consequence of an aberrant immune-inflammatory response arising in genetically predisposed individuals triggered by several environmental and host factors. 1 It follows that possible explanations for the identified association between these two entities include shared genetic background abnormalities, combined immune dysfunction, and mutual environmental and host factors. 24 The similarities in pathogenic mechanisms are further emphasized by the efficacy of the same treatment in both diseases, and in particular biological therapy targeting TNF-α, IL-12/23, and IL-23. 25
Genetic associations
Genetic association studies identified various genome areas that are associated with both psoriasis and IBDs.1,5,26 Here, we briefly discuss those genetic loci that have shown well-established associations for both diseases.
-1p31.3: it harbors the IL23R gene, which encodes a subunit of the IL-23 receptor and subsequently influences the binding capacity of IL-23. 24 IL-23 is essential for the differentiation and activation of Th17 lymphocytes, which produce IL-17, an interleukin that has been found to contribute to the pathogenesis of both psoriasis and IBD.27–30
-5q31: implicates genes IL-4, IL-5, and IL-13, which encode respective interleukins acting as inflammatory mediators. 26 In this locus, variants for both psoriasis and IBD have been found.31–33 Genetic locus 5q33.1 represents an established psoriasis and CD common risk locus,26,34 being involved in IL-23/Th17 signaling.
-6p21: it implicates both CDKAL1 and HLA-Cw6 genes. 26 Remarkably, HLA-Cw6 represents a major disease allele at PSORS1, 35 which is the main psoriasis susceptibility locus. 34 On the other side, it has been suggested a general role for the chromosome 6 locus in IBD. 36
-16q: it involves the NOD2/CARD15 gene, whose variants have been identified as well-established genetic risk factors for CD. 37 A meta-analysis by Zhu et al. 38 did not find a direct association between psoriasis and common polymorphisms of NOD2/CARD15 but emphasized the importance of the protein encoded by this gene in the pathogenesis of psoriasis.
-19p13: it corresponds to psoriasis susceptibility locus 6 (PSORS6) 39 and IBD6. 34
-20q13: it involves ZNF313 gene for psoriasis 40 and TNFRSF6B for IBD. 41
Common immune dysregulation
In both IBD and psoriasis, there exists a genetic concordance alongside overlapping immune activation mechanisms 42 (Figure 3).
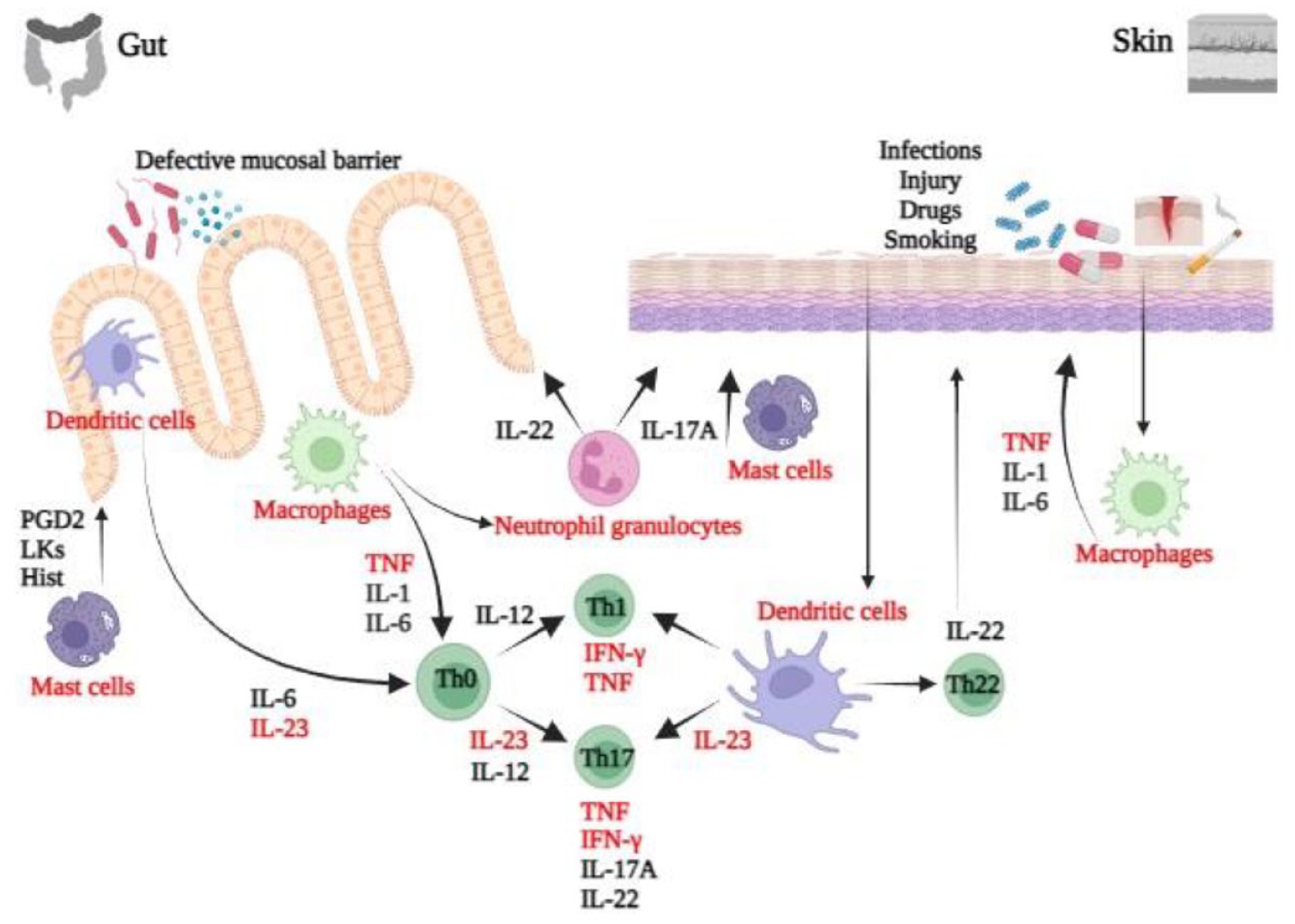
IBD and psoriasis common pathogenesis. Gut epithelium (left) and skin epithelium (right). Shared inflammatory pathways are marked in red. Both innate immunity and adaptive immunity play a key role in triggering and maintaining both skin and gut chronic inflammation. 43 Particularly, the IL-23/Th17 cells axis and its interaction with keratinocytes and intestinal mucosa cells are crucial for the pathogenesis of both psoriasis and IBDs.
Primarily, abnormal permeability of the skin and intestinal barriers allows antigens, including those from altered microbiota, to interact with pattern recognition receptors on dendritic cells and pro-inflammatory M1 macrophages, triggering immune responses.5,26 Cells of the innate immune system, such as dendritic cells, M1 macrophages, mast cells, and natural killer cells, are implicated in both conditions, exhibiting increased activity and pro-inflammatory cytokine production.44–50 Furthermore, the adaptive immune system, including T-helper cells (Th1, Th17), regulatory T-cells (T-regs), and cytokines like TNF-α, IL-1, IL-12/23, and IL-6, plays a significant role in both skin and gut inflammation. 42 The IL-23/Th17 axis is particularly crucial in both psoriasis and IBD pathogenesis, with increased Th17 cell proportions and cytokine production observed in affected tissues.26,51 IL-23, pivotal in the Th17 pathway, promotes inflammation, angiogenesis, and Th17 cell proliferation and survival, with elevated levels detected in both conditions.36,52–56
Environmental factors and microbiota
The progressive increase in the incidence of IMID is probably a consequence of the progressive globalization and acquisition of Western habits, diet, and lifestyle in new geographic areas. 57 Environmental factors are thought to influence the development of IMID by triggering abnormal immune responses (both innate and cell mediated).
Due to the relatively low concordance rates in monozygotic twins, the role of environmental determinants appears to be substantial. 3 In particular, a relevant part seems to be played by hygiene, higher socio-economic status, vitamin D exposure, cigarette smoking, drugs (antibiotics, biologics), infections, stress, trauma with different roles, and different evidence for each IMID.58,59
It is known that smoking habit is associated with an increased risk of developing CD 60 and psoriasis. 61 To note, smoking appears to modify the risk of IBD in patients with psoriasis, 21 enhance the risk for inflammatory chronic skin diseases, including psoriasis, in patients with IBD. 62
Microbiota dysfunction has been implicated as a contributing factor in the pathogenesis of both IBD and psoriasis. 24 The gut microbiota influences the local immune system but also impacts the systemic immune system. 63 The concept of a “gut–skin axis” has been proposed to elucidate the connection between alterations in the gut microbiota, abnormal intestinal permeability, and disrupted immune balance, all of which may contribute to inflammation in both the gut and the skin. 1
A study conducted by Eppinga et al. 63 in 2016 observed an IBD-like reduction in Faecalibacterium prausnitzii and an increase in Escherichia coli among psoriasis patients. Faecalibacterium prausnitzii serves as an energy source for colonocytes and possesses anti-inflammatory properties that modulate the balance between T effector cells (such as Th1, Th2, and Th17) and T-regs.63,64 Alterations in commensal gut bacteria levels may lead to dysregulation of T-cell populations, instigating systemic inflammation in both the skin and the gut. 65 In addition, research by Tan et al. 66 in 2018 showed that Akkermansia muciniphila, a bacterium notably diminished within the gut microbiota of patients with IBD, is also reduced in patients with psoriasis. 67
IBD and psoriasis during therapy with anti-IL-12/23 and anti-IL-23 antibodies
In IBD patients, paradoxical psoriasis induced by therapeutic TNF-α inhibition is a well-described phenomenon.68–70 Over the last two decades, cases of paradoxical psoriasis have also been reported following the administration of other biologic agents, including IL-12/23 inhibitor ustekinumab and IL-23 inhibitors, first in dermatological patients and subsequently in IBD ones. In this review, we performed a literature review and an analysis of clinical trials of this class of biological agents in IBD to find cases of paradoxical psoriasis induced by both ustekinumab and IL-23 inhibitors, aiming to raise awareness of this rare paradoxical phenomenon among gastroenterologists.
Anti-IL-12/23 antibodies
Ustekinumab is a fully human IgG1κ monoclonal antibody that inhibits the biological activity of IL-12 and IL-23 through their common p40 subunit. 71 It was first licensed for psoriasis and psoriatic arthritis, and it received approval for use in CD in 2016 and UC in 2019. 52
We found 10 reported cases of potentially ustekinumab-induced new-onset or worsening psoriasis in the literature (Table 1). The latency period of these reactions ranged from 2 days to 3 years following first administration.
Reported cases of ustekinumab-induced psoriasis in the literature.

ADA, Adalimumab; CD, Crohn’s disease; FU, follow up; IFX, Infliximab; UST, ustekinumab.
In particular, six cases72–77 reported a paradoxical flare of psoriasis in patients with underlying psoriasis after ustekinumab treatment for dermatological indications. Five of these cases72–75,77 featured the appearance of pustular psoriasis in patients with chronic plaque psoriasis after starting IL-12/23 inhibitor therapy. This presentation with altered morphology from plaque-type to pustular-type psoriasis and the time course of the disease about the drug made a drug-induced flare of psoriasis more likely. Only in one case, 76 the morphology was that of plaque-type psoriasis flare without a pustular component.
Four cases of ustekinumab-induced paradoxical psoriasis concerned gastrointestinal indication (CD) for the IL-12/23 inhibitor use.78–81 In two of these cases, the morphology was that of pustular psoriasis,78,80 while in one case 79 paradoxical psoriasis developed with an inverse presentation; the fourth case involves a biopsy-confirmed psoriasiform rash in a patient undergoing ustekinumab treatment. 81 Benzaquen et al. 78 described the case of a palmoplantar pustular psoriasis which developed after 3 weeks from ustekinumab induction for a refractory CD, with complete recovery of cutaneous manifestations 2 weeks after cessation of treatment and therapeutic swap to golimumab. Darwin et al. 79 reported the first case of paradoxical psoriasis with an inverse presentation: this case concerns a 56-year-old woman with a history of adalimumab-induced palmoplantar pustular psoriasis who developed a flare of inverse psoriasis after 15 months of treatment with ustekinumab, which did not require ustekinumab discontinuation but only topical therapy. In 2021, Barahimi et al. 80 described a case of reproducible paradoxical psoriasiform rash after ustekinumab administration, histologically compatible with subcorneal pustular dermatosis and subsequently indistinguishable from pustular psoriasis 82 in a patient without a previous history of psoriasis. The chronology between cutaneous manifestation and drug administration, including resolution between doses and with cessation of the drug, as well as subsequent recurrence with reintroduction of the drug, suggested a drug-induced phenomenon. Given the improvement in CD, ustekinumab was continued and the rash was managed with topical corticosteroids. Recently, Klimko et al. 81 reported the case of a 39-year-old female patient with a long-standing colonic CD who developed a psoriasiform rash 3 years after ustekinumab therapy onset. Histologic findings of the skin biopsy revealed some typical psoriasis signs such as epidermal hyperplasia, parakeratosis, Munro’s microabscesses, inflammatory infiltrate in the upper dermis, thinned granular cell layer of the epidermis, and dilated dermal capillaries. Ustekinumab treatment was continued and a complete resolution of the lesions was obtained with topical steroid therapy.
Causality assessment in all cases was plausible for ustekinumab in induction of paradoxical psoriasis due to a compatible temporal relation according to time to onset and recovery after cessation of treatment,72,73,75,77,78,80 negative assessment of an alternative etiology (nor bacterial or viral infection, nor other treatment taken by the patient), and in some cases78,80,81 nor previous history of psoriasis. As largely mentioned, psoriasis could also be a concomitant IMID of CD, but these characteristics of presentation suggested a paradoxical drug-induced etiology.
Furthermore, we reviewed adverse event reports in clinical trials investigating ustekinumab as a treatment for IBDs. Data from the IM-UNITI trial 83 and long-term extension 84 evaluating the long-term (5 years) efficacy, safety, and immunogenicity of ustekinumab in patients with CD reported one case of pustular psoriasis. This case developed in the study arm including 354 patients (1/354, 0.28%) who were treated with ustekinumab 90 mg subcutaneous q8w in the long-term extension study (week 44–272). No other cases of psoriasis were recorded in a phase IIa trial, 85 CERTIFI phase IIb trial, 86 and two phase III trials (UNITI-I and UNITI-2) evaluating the safety and efficacy of ustekinumab in patients with moderately to severely active CD. 87 In addition, the phase III POWER study (ClinicalTrials.gov number NCT03782376) evaluated the efficacy and safety of a re-induction dose of ustekinumab in CD patients with secondary loss of response to ustekinumab maintenance therapy. Up to week 36, one case of psoriasis has been registered in the ustekinumab reinduction group (1/108, 0.93%), while no cases have been described in the continuous q8w subcutaneous maintenance group. Other possible cases have been reported in the SEAVUE study, 88 which is another phase IIIb study comparing the efficacy and safety of ustekinumab to adalimumab in CD patients. Safety analysis has been conducted up to 81 weeks and demonstrated one case of psoriasis (registered as a “serious adverse event”) in the adalimumab-treated group of patients (1/195, 0.51%) while no “serious adverse” cases of psoriasis have been registered in the ustekinumab-treated group. However, between other (not including serious) adverse events, three cases of psoriasis have been reported in the adalimumab arm (3/195, 1.54%) and two cases (2/191, 1.05%) in the ustekinumab arm.
Conversely, no cases of psoriasis have been reported up to week 104 in the STARDUST study, 89 which compared treat to target versus routine care maintenance strategies in CD patients treated with ustekinumab. No reported case of psoriasis also derived from the UNIFI trial 90 and long-term extension 91 evaluating the efficacy and safety of ustekinumab up to week 220 of maintenance therapy in UC.
Post-marketing surveillance for CD patients treated with ustekinumab under real-world clinical practice is also being evaluated by an observational prospective study (STELARA CD PMS, ClinicalTrials.gov number NCT03942120) whose results are still not available.
Anti-IL-23 antibodies
During the last few years, there has been notable interest in developing IL-23 selective therapies offering all the benefits of anti-IL-23p40 therapies with less “off-target” effect derived from the inhibition of IL-12 signaling. 52
Specific IL-23p19 inhibitors have been developed and already approved for the treatment of psoriasis (risankizumab, guselkumab, and tildrakizumab) and psoriatic arthritis (risankizumab and guselkumab). Furthermore, risankizumab and mirikizumab have recently been approved, respectively, for moderate-to-severely active CD and for the treatment of moderate-to-severe UC in adult patients. 92 Guselkumab is currently in phase III of clinical trials for the treatment of both CD and UC.
Selective IL-23p19 inhibitors were associated with substantially greater efficacy in psoriasis treatment compared with IL-12/23p40 blockers. 93 Similarly, a matching-adjusted indirect comparison was carried out using data from risankizumab trials and published aggregated data from ustekinumab trials for CD. Higher rates of clinical and endoscopic outcomes during induction for risankizumab compared to ustekinumab have been demonstrated. 94 Furthermore, SEQUENCE trial 95 directly compared the efficacy and safety of risankizumab versus ustekinumab in patients with moderate to severe CD and prior anti-TNF failure. Results have been recently presented and demonstrated higher rates of clinical remission and response at weeks 24 and 48 (significantly higher for week 48 CDAI clinical remission) with risankizumab compared to ustekinumab.
Similarly, phase III VIVID-1 study 96 showed mirikizumab numerically superior rates of endoscopic response, endoscopic remission, and corticosteroid-free CDAI clinical remission compared to ustekinumab. Mirikizumab was nominally statistically superior to ustekinumab in achieving combined CDAI clinical remission and endoscopic response at week 52.
Preliminary indicative comparison data between guselkumab and ustekinumab derived from the GALAXI-1 trial, which included a ustekinumab group as a reference group only: active comparison of guselkumab with ustekinumab will be evaluated in larger phase III trials. 97
We here report data about the occurrence of psoriasis in patients with IBD treated with risankizumab, mirikizumab, and guselkumab, that is the anti-IL-23 antibodies currently licensed or studied in patients with UC and CD (Figure 4). In Table 2 and Figure 5, we report the main features of all anti-IL-23 antibodies considered for the treatment of psoriasis and IBD.

Flowchart illustrating the screening process for relevant studies reporting cases of paradoxical psoriasis induced by IL-12/23 and IL-23 inhibitors.
Summary of anti-IL-12/23 and anti-IL-23 antibodies..

CD, Crohn’s disease; FAP, familial adenomatous polyposis; GVHD, graft-versus-host disease; IL, interleukin; IV, intravenous; N/A, not available; PsA, psoriatic arthritis; PsO, plaque psoriasis; q4w, every 4 weeks; q8w, every 8 weeks; q12w, every 12 weeks; SC, subcutaneous; UC, ulcerative colitis.

IL-23 inhibitors molecular structure and differences. Mirikizumab is a humanized IgG4 variant monoclonal antibody; guselkumab is a fully human IgG1 monoclonal antibody with a native Fc region, while risankizumab is a humanized IgG1 antibody with a mutated Fc region. Differences between anti-IL-23 therapeutic antibodies may be related to unique molecular characteristics: binding of these therapeutic antibodies to CD64 is particularly interesting since CD64+ IL-23-producing myeloid cells are increased in the inflamed colon in IBD. 98
Risankizumab
Risankizumab is a humanized IgG1 monoclonal antibody that selectively inhibits IL-23 by targeting its p19 subunit and it is currently the only IL-23p19 inhibitor approved for the treatment of moderate-to-severe CD. It was first approved for the treatment of moderate-to-severe plaque psoriasis and psoriatic arthritis.
We reviewed the literature and found one case report of paradoxical psoriasis induced by risankizumab described by McFeely et al. 100 It involved a patient with dermatological indication (plaque psoriasis) for IL-23p19 inhibitor use. The patient was already on adalimumab for uncontrolled plaque psoriasis and for this reason was switched to risankizumab, subsequently developing pustular psoriasis. Interestingly, the patient has never had a history of pustular psoriasis in the 22 years that she had been diagnosed with psoriasis. We did not find cases of paradoxical psoriasis induced by risankizumab in patients using this drug for active CD, probably due to its recent release for gastrointestinal indication.
We also analyzed results for adverse events in clinical trials investigating risankizumab in CD and we did not find reported cases of paradoxical psoriasis. Particularly, no cases have been reported in a phase II trial 101 nor the respective long-term extension. 102 Likewise, results from two phase III induction trials ADVANCE and MOTIVATE for risankizumab in CD patients 103 did not report cases of paradoxical psoriasis. Data from the phase III maintenance study FORTIFY trial (ClinicalTrials.gov number NCT03105102) 104 did not show new safety risks, but detailed reports on adverse events are not available yet on ClinicalTrials.gov. Safety data will also be available from another ongoing phase III clinical trial (ClinicalTrials.gov number NCT06063967) which aims at assessing the safety and effectiveness of risankizumab subcutaneous induction treatment in adult CD patients. Other important information about safety will also be evaluated by an observational study (APPRISE, ClinicalTrials.gov number NCT05841537) evaluating real-world clinical effectiveness and safety of risanzkizumab in CD. More safety data might also emerge from ongoing phase III trials assessing risankizumab in UC (ClinicalTrials.gov numbers NCT03398148 and NCT03398135).
Mirikizumab
Mirikizumab is a humanized IgG4 variant monoclonal antibody selectively targeting the IL-23 p19 subunit and it is currently in advanced development for IBD treatment.
No cases of paradoxical psoriasis have been reported in the phase II trial evaluating the efficacy and safety of mirikizumab in UC.105,106 Mirikizumab showed efficacy as a treatment of moderately to severely active UC in phase III LUCENT-1 and LUCENT-2 clinical trials in which no cases of paradoxical psoriasis have been reported (up to week 40). 107 A phase III clinical trial (LUCENT-3, ClinicalTrials.gov number NCT03519945) is still ongoing evaluating the long-term efficacy and safety of mirikizumab in UC.
Safety concerns will also be evaluated in LUCENT-URGE (ClinicalTrials.gov number NCT05767021), a phase IIIb single-arm study that investigates bowel urgency and other outcome measures in UC patients treated with mirikizumab up to 36 weeks.
Mirikizumab is also being evaluated for CD indication. Phase II SERENITY trial evaluated its efficacy and safety in patients with active CD 108 and did not report cases of psoriasis until 208 weeks. Phase III VIVID-1 trial (ClinicalTrials.gov number NCT03926130) and its open-label extension VIVID-2 (ClinicalTrials.gov number NCT04232553) for mirikizumab in CD are still ongoing.96,109
Guselkumab
Guselkumab is a fully human IgG1-lambda that inhibits p19 IL-23 and it is already approved for the treatment of plaque psoriasis and psoriatic arthritis.
A phase II (GALAXI-1) and phase III (GALAXI 2 and GALAXI 3) clinical trials (ClinicalTrials.gov number NCT03466411) have been designed to evaluate the efficacy and safety of guselkumab in adult patients with moderate-to-severe CD. Their safety results are still not available. GRAVITI clinical trial (ClinicalTrials.gov number NCT05197049) is also active, evaluating the efficacy and safety of subcutaneous guselkumab in participants with CD up to week 109. Furthermore, another phase III, open-label, multicenter study (ClinicalTrials.gov number NCT04397263) is still ongoing and aims to evaluate the safety of guselkumab in CD patients. Guselkumab is also being assessed in the treatment of patients with active fistulizing, perianal CD in a phase III clinical trial (FUZION CD, ClinicalTrials.gov number NCT05347095): safety data will be collected and the total duration of the study for participants will be up to 118 weeks.
In UC, the efficacy and safety of guselkumab induction and maintenance have been designed to be evaluated in QUASAR phase IIb/III trials. Phase IIb safety results were consistent with the known safety profile of this drug in plaque psoriasis and psoriatic arthritis, 110 but detailed data on adverse events are still not available and future results from maintenance therapy will find out possible adverse paradoxical reactions.
Another phase III multicenter study (ASTRO, ClinicalTrials.gov number NCT05528510) is still ongoing and aims to evaluate the efficacy and safety of Guselkumab subcutaneous induction therapy in patients with UC with an overall study duration up to 112 weeks.
Furthermore, the VEGA clinical trial evaluated the efficacy and safety of the combination therapy guselkumab + golimumab in patients affected by UC: no paradoxical skin reactions have been reported up to week 50. 111
Discussion
Psoriasis and IBD, including CD and UC, belong to the heterogeneous group of IMID. A significant link between these two entities is strongly suggested by epidemiological data showing the presence of either psoriasis or IBD associated with significantly increased odds of having the other disease as well. 5 The observed link between psoriasis and IBDs is also supported by shared genetic abnormalities, and overlapping pathophysiological patterns involving both innate and adaptive immunity and environmental factors.26,34 However, the link between these two illnesses is currently far from clear, and many times it might be difficult for clinicians to precisely define it. In particular, psoriasis can be associated with IBD as an independent concomitant IMID or constituting a paradoxical adverse event of biological therapy.
The similarities in the pathogenesis of the two illnesses are also reflected in the overlapping of their therapeutic approaches, particularly represented by biological TNF-α inhibition, IL-12/23 inhibitors, and IL-23 blockers. 26 Another aspect of the association between IBD and psoriasis is represented by the possibility of paradoxical cutaneous adverse events caused by biological agents used for both diseases. Paradoxical psoriasis induced by TNF inhibitors is nowadays a well-known phenomenon, but less is known about the possible similar effect of IL-12/23 inhibitor ustekinumab and IL-23p19 inhibitors. The pathophysiology of these paradoxical manifestations has not yet been clearly understood, but it might involve a cytokine-vicious circle in genetically predisposed patients. 72 In particular, increased IFN-α levels may derive from decreased IL-23 and Th17 cell-induced TNF-α levels. 75 IFN-α may then promote T-cell activation, 73 favoring their homing to the skin. 78 IFN-α also stimulates and activates T cells to produce TNF-α and IL-17, sustaining inflammatory mechanisms for paradoxical psoriasis lesions. 78
Other few cases of psoriasis associated with the use of ustekinumab are present in the literature: six cases reported a flare of psoriasis in patients who were using ustekinumab for dermatological indications, and four case reports involved patients treated for their CD. This uncommon adverse event manifested from 2 days to 3 years following the first administration of the drug as either a new-onset or exacerbation of pre-existing psoriasis. Flares of psoriasis after ustekinumab started were characterized in quite all cases (5/6) by altered morphology from plaque-type to pustular-type psoriasis, thus suggesting a drug-induced etiology. Other common features suggesting a paradoxical drug-induced etiology were as follows: the temporal relationship between drug administration and psoriasis development, the prompt resolution after cessation of treatment73,77,78 and subsequent recurrence with reintroduction of the drug,73,75,76,80 the negative assessment of an alternative etiology, and in some cases78,80,81 nor previous history of psoriasis. In six cases (6/10), paradoxical psoriasis required ustekinumab discontinuation, while three patients (3/10) continued the drug while psoriasis was well controlled with topical therapy. In one case (1/10), an initial discontinuation of the drug was followed by an attempt of re-induction: paradoxical psoriasis reappeared but was controlled by topical steroid therapy.
Furthermore, we reviewed results from clinical trials investigating ustekinumab as a treatment for IBDs and found four cases of psoriasis registered as adverse events, but little information is available regarding the features of these cases.
Focusing also on the new IL-23p19 inhibitors, only one case has been reported of risankizumab-induced flare of psoriasis in a patient using risankizumab for dermatological indication. No other cases of paradoxical psoriasis were registered in available clinical trial data for risankizumab, guselkumab, and mirikizumab in IBD patients.
The small number of case reports included in the review represents its main limitation. However, as far as we know, it summarizes the current literature and results from clinical trials on this topic in gastroenterology. Second, the included reports differed in their methodologic descriptions, making it challenging to comprehensive summary.
The starting point of this review was to define whether psoriasis occurred in our patient with CD treated with ustekinumab should be considered as an onset of a concomitant IMID or a paradoxical effect of therapy. The presentation some weeks after intravenous re-induction with ustekinumab could lead to considering the paradoxical reaction as the more likely explanation in our patient. This interpretation appears to be coherent with the other cases of psoriasis that occurred during therapy with ustekinumab. On the other hand, a definitive answer about this issue cannot be provided as well as other definitive answers about the real role of the several pathogenic mechanisms linking IBD and psoriasis. At this point, from a practical point of view, we can apply the principle of Occam’s razor and then accept the simplest explanation that requires the fewest assumptions. In our case, the simplest explanation is that IBD and psoriasis are strictly associated and can coexist in the same patient, as clearly demonstrated by several epidemiological studies.3,5,18,19,21–23 From this fact, efforts should be made to raise awareness about the fact that psoriasis is frequently associated with IBD and that therapies used for IBD may lead to the occurrence of psoriasis. All patients with IBD should be asked and examined for skin manifestations by gastroenterologists112,113: cutaneous manifestations should be promptly recognized and early referred to dermatologists to be correctly diagnosed.42,112,113 Indeed, an early diagnosis is crucial for reducing the progressively deleterious course of both IBD and psoriasis. Therefore, gastroenterologists should be able to recognize the signs and symptoms suggestive of psoriasis (requiring dermatological evaluations) and be familiar with the onset of psoriasis as a side effect of therapies used to treat IBD. A dermatologist is considered a relevant figure in the management of patients with IBD and IMIDs involving skin are among the more frequently taken into account by gastroenterologists evaluating patients with IBD. 114 However, the interaction between gastroenterologist and dermatologist for the management of concomitant IMIDs has not been yet formally structured as already done for other clinical settings, such as rheumatology. 115
In parallel, further studies are needed to better clarify the pathophysiology of the cytokine imbalance which leads to the paradoxical phenomenon of psoriasis induced by inhibition of IL-12/23 and IL-23. This will also help to identify patients at risk of this event and to address personalized and safer therapy. Cutaneous paradoxical reactions can be clinically relevant and disabling for patients and it follows that a specific specialistic and adequate clinical management is mandatory. Of note, the severity of skin eruptions should be defined considering not only the extent of the psoriatic eruption but also its impact on the patient’s quality of life, highlighting the consequence of paradoxical psoriasis on the patient’s health. 116 A multidisciplinary approach allows for better management of various IMIDs and improves patients’ health-related quality of life. 117
In patients with IBD and a personal or familial history of psoriasis, the use of anti-IL-12/23 and anti-IL-23 should be preferred to anti-TNF agents. The concomitant presence of IBD and psoriasis increases morbidity and healthcare use and further worsens the quality of life of patients making their care more complex and clinical decision-making more challenging. This will be more relevant in the next few years due to the worldwide increasing prevalence of IMIDs, even in younger subjects.
Finally, we can affirm that (a) de novo onset of psoriasis and worsening of already present psoriasis may be a rare paradoxical effect of ustekinumab, an anti-IL-12/23 antibody; (b) too few real-world data exist to draw any conclusions about a similar situation with anti-IL-23 antibodies; and (c) risankizumab may positively treat psoriasis induced by ustekinumab.
Conclusion
Nowadays, a rising number of advanced therapies are effective both for IBD and psoriasis, such as anti-TNF agents and anti-IL-12/23. It has been described, with different evidence levels, that these therapies may cause the paradoxical new onset of psoriasis or worsening of an already present psoriasis requiring additional therapies.
Several anti-IL-23 antibodies will soon be available for IBD. Studies will need to be conducted to compare the single agents to better assess their efficacy and their safety profile, including the possibility of inducing paradoxical psoriasis. Moreover, the identification of risk factors associated with efficacy and safety will help us in the direction of personalized medicine. In particular, randomized controlled trials should be performed in the peculiar settings of concomitant IBD and psoriasis.
In addition, Scientific Societies of Gastroenterology and Dermatology should identify those signs and symptoms that require an early, proper, and rapid mutual referral as well as provide recommendations for the shared management of patients with IBD and concomitant (or paradoxical) psoriasis.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241299564 – Supplemental material for Psoriasis and inflammatory bowel disease: concomitant IMID or paradoxical therapeutic effect? A scoping review on anti-IL-12/23 and anti-IL-23 antibodies
Supplemental material, sj-docx-1-tag-10.1177_17562848241299564 for Psoriasis and inflammatory bowel disease: concomitant IMID or paradoxical therapeutic effect? A scoping review on anti-IL-12/23 and anti-IL-23 antibodies by Cristina Bezzio, Carolina Aliai Micol Cavalli, Gianluca Franchellucci, Arianna Dal Buono, Roberto Gabbiadini, Davide Scalvini, Sofia Manara, Alessandra Narcisi, Alessandro Armuzzi and Simone Saibeni in Therapeutic Advances in Gastroenterology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
