Abstract
Background:
Eicosanoid and related docosanoid polyunsaturated fatty acids (PUFAs) and their oxygenated derivatives have been proposed as noninvasive lipidomic biomarkers of nonalcoholic steatohepatitis (NASH). Therefore, we investigated associations between plasma eicosanoids and liver fibrosis to evaluate their utility in diagnosing and monitoring NASH-related fibrosis.
Methods:
Our analysis used baseline eicosanoid data from 427 patients with biopsy-confirmed nonalcoholic fatty liver disease (NAFLD), and longitudinal measurements along with liver fibrosis staging from 63 patients with NASH and stage 2/3 fibrosis followed for 24 weeks in a phase II trial.
Results:
At baseline, four eicosanoids were significantly associated with liver fibrosis stage: 11,12-DIHETE, tetranor 12-HETE, adrenic acid, and 14, 15-DIHETE. Over 24 weeks of follow up, a combination of changes in seven eicosanoids [5-HETE, 7,17-DHDPA, adrenic acid, arachidonic acid (AA), eicosapentaenoic acid (EPA), 16-HDOHE, and 9-HODE) had good diagnostic accuracy for the prediction of ⩾1 stage improvement in fibrosis (AUROC: 0.74; 95% CI: 0.62–0.87), and a combination of four eicosanoids (7,17-DHDPA, 14,15-DIHETRE, 9-HOTRE, and free adrenic acid) accurately predicted improvement in hepatic collagen content (AUROC: 0.72; 95% CI: 0.50–0.77).
Conclusion:
This study provides preliminary evidence that plasma eicosanoids may serve as noninvasive biomarkers of liver fibrosis and may predict liver fibrosis improvement in NASH.
Introduction
Nonalcoholic fatty liver disease (NAFLD) has become the most prevalent chronic liver disease worldwide.1,2 When unaccompanied by inflammation and cell damage, NAFLD is relatively benign and has a low risk of progression. However, patients with nonalcoholic steatohepatitis (NASH), the progressive form of NAFLD characterized by the presence of hepatic steatosis, lobular inflammation, and ballooning, are at high risk of liver fibrosis, cirrhosis, and hepatocellular carcinoma. 3 The presence of liver fibrosis represents the most important predictor of morbidity and mortality in patients with NASH.3–5
Although there are as yet no approved pharmacological therapies for NASH and NASH-related fibrosis, intensive research is underway and promising new therapies are in development. 6 Liver biopsy has long been considered the gold standard for the diagnosis of NASH and liver fibrosis, and is required in the setting of a clinical trial. However, the procedure is impractical for the staging of NAFLD and longitudinal follow up in routine clinical practice due to its cost and invasive nature. There is an urgent need for noninvasive, easy to perform, and widely available diagnostic alternatives for assessing the severity of fibrosis in the setting of NASH, as well as for monitoring treatment response and disease progression.
In a pilot study, we demonstrated that several eicosanoids and docosanoids, including arachidonic acid (AA), docosahexaenoic acid (DHA), and their oxygenated polyunsaturated fatty acid (PUFA) derivatives, generally referred to as “eicosanoids,” are potentially useful as biomarkers for the noninvasive diagnosis of NASH.7,8 Metabolome profiling has provided new insights into the molecular mechanisms of diseases, including NAFLD, suggesting potential innovative biomarkers for liver fibrosis. 9 Several studies have identified specific metabolomic signatures associated with disease stage in NAFLD.8–18 Among these metabolites, free PUFAs and their eicosanoid metabolites, as well as total eicosanoids, have been reported in patients with NAFLD.8,10,19,20 However, studies that have reported on total oxidized PUFAs,8,17 including esterified ones identified after base hydrolysis, are, as we have shown, unreliable because of dramatic decomposition during the experimental analysis. 21 Hepatic and circulating eicosanoid levels are reported to be altered in NASH patients,8,10,22 but data regarding the association of plasma eicosanoids with liver fibrosis and longitudinal association with a change in histological stage in patients with NAFLD are limited.
Using baseline data from 427 biopsy-proven NAFLD patients with available plasma eicosanoid assessment from phase II studies of simtuzumab, 23 and longitudinal data from 63 NASH patients with stage 2 or 3 fibrosis enrolled in a phase II study of selonsertib, 24 we investigated associations between plasma eicosanoids and fibrosis, and attempted to determine if longitudinal changes in plasma eicosanoids can predict changes in fibrosis over time.
Materials and methods
Patients and study design
Baseline assessment
We performed a cross-sectional analysis of baseline data from 427 participants who were screened for phase II studies of simtuzumab 23 : GS-US-321-0105 [ClinicalTrials.gov identifier: NCT01672866] (84 patients enrolled and 217 screen-failed patients); and GS-US-384-1497 [ClinicalTrials.gov identifier: NCT02466516] (72 enrolled and 54 screen-failed patients) with baseline plasma eicosanoid assessment and liver biopsy. The designs of these studies have been described previously.23,24
Briefly, adult patients 18–65 years of age with body mass index (BMI) of at least 18 kg/m2 were eligible for enrollment in the two studies, and were screened at 111 centers in the United States, Canada, and Europe from November 2012 to October 2016. To be eligible for study GS-US-321-0105, patients were required to have chronic liver disease from NASH (defined histologically as macrovesicular steatosis involving >5% of hepatocytes with associated lobular inflammation) and fibrosis stage 3 or 4 (bridging fibrosis) based on a modified Ishak classification. Eligibility for study GS-US-384-1497 is described in the longitudinal assessment section below. All patients had to have aspartate aminotransferase (AST) and alanine aminotransferase (ALT) values no higher than 10 times the upper limit of normal, and serum creatinine level lower than 2.0 mg/dl based on central laboratory values.
Patients with any of the following conditions were excluded from the two studies: any history of hepatic decompensation, including ascites, hepatic encephalopathy, or variceal hemorrhage; weight-reduction surgery in the prior 5 years; infection with the hepatitis B or C virus; alcohol consumption greater than 21 ounces/week for men or greater than 14 ounces/week for women; positive screen result for illegal drug use; or clinically significant cardiac disease. In addition, patients in the cirrhosis study were excluded if they had a Child–Pugh–Turcotte score higher than 7, a Model for End-Stage Liver Disease score higher than 12, or a history of solid organ transplantation.
The studies were approved by the institutional review board or independent ethics committee at each participating sites available at ClinicalTrials.gov [ClinicalTrials.gov identifiers: NCT01672866 and NCT02466516] and conducted in compliance with the Declaration of Helsinki, Good Clinical Practice guidelines, and local regulatory requirements. Written informed consent was obtained from all patients prior to study initiation.
Longitudinal assessment
We also analyzed longitudinal data – plasma eicosanoid, liver biopsy, and hepatic collagen content – at baseline and week 24 from 63 NASH patients with stage 2 or 3 fibrosis enrolled in a phase II study of selonsertib (GS-US-384-1497 [ClinicalTrials.gov identifier: NCT02466516]). The study design and procedures have been described previously. 24 Briefly, adult patients 18–70 years of age were enrolled at 23 sites in the United States and Canada from 8 June 2015 to 31 March 2016. To be eligible, patients were required to have a liver biopsy within 3 months of screening consistent with a diagnosis of NASH and stage 2 or stage 3 fibrosis according to the NASH Clinical Research Network (CRN) histologic scoring system. All patients had a NAFLD activity score (NAS) of 5 or higher, with a score of at least 1 point for each of the three components (steatosis, hepatocellular ballooning, and lobular inflammation). All patients had at least three of the following features of the metabolic syndrome: abdominal obesity, hypertension, elevated fasting glucose, elevated levels of serum triglycerides, or low levels of high-density lipoprotein cholesterol. Patients were randomly assigned in a 2:2:1:1:1 ratio to receive 24 weeks of treatment with 6 or 18 mg of selonsertib, 6 or 18 mg of selonsertib with 125 mg of simtuzumab, or 125 mg of simtuzumab alone. Selonsertib was administered orally once daily and simtuzumab was administered as weekly subcutaneous injections. After 24 weeks of treatment, 32% of the participants included in this study had at least a 1-stage reduction in fibrosis, 52% had no change in fibrosis stage, and 16% had an increase of at least 1-stage in fibrosis. In addition, 38% of the participants had at least 20% reduction in hepatic collagen content, 22% no significant change in hepatic collagen content, and 40% had an increase of at least 20% in hepatic collagen content. Treatment groups were combined for this analysis.
Histology
Biopsy samples of all participants included in each baseline and longitudinal study were read centrally by a single experienced pathologist who was blinded to treatment assignment, but not to biopsy sequence. Histologic assessments included the adequacy of the biopsy specimen, confirmation of the diagnosis, fibrosis staged according to a modified Ishak classification (GS-US-321-0105) and the NASH CRN system for both studies (GS-US-321-0105 and GS-US-384-1497). Biopsy specimens were graded according to the NAS, a standardized grading system for steatosis (on a scale of 0–3), lobular inflammation (on a scale of 0–3), and hepatocellular ballooning (on a scale of 0–2), with higher scores indicating increasing disease activity. Computer-assisted morphometry was also used to quantify hepatic collagen and fat content using picrosirius red-stained liver sections, as well as a smooth muscle actin (a-SMA) expression. 25
Outcomes
Baseline assessment
The primary outcome was the individual stage of fibrosis; the secondary outcome was the presence of advanced fibrosis (stage 3 or 4 according to the NASH CRN system).
Longitudinal assessment
The primary outcome was improvement in liver fibrosis, defined as a ⩾1-stage reduction of fibrosis. 24 The secondary outcome was improvement in hepatic collagen content as defined by a relative reduction of at least 20% from baseline to week 24.
Eicosanoids assessment
Lipid extraction
Serum samples for lipidomic profiling were obtained within 90 days of the liver biopsy. All serum samples were stored at −80°C, thawed once, and immediately used for free fatty acid and eicosanoid isolation as described previously. 8 Briefly, 50 μl plasma was spiked with a cocktail of 26 deuterated internal standards (individually purchased from Cayman Chemicals, Ann Arbor, MI, USA) and brought to a volume of 1 ml with 10% methanol. The samples were then purified by solid phase extraction on Strata-X columns (Phenomenex, Torrance, CA, USA), using an activation procedure consisting of consecutive washes with 3 ml of 100% methanol followed by 3 ml of water. The eicosanoids were then eluted with 1 ml of 100% methanol, and the eluent was dried under vacuum, dissolved in 50 μl of buffer A [consisting of water-acetonitril-acetic acid, 60:40:0.02 (v/v/v)], and immediately used for analysis as follows: For free fatty acids analysis, 50 µl of serum was spiked with deuterated fatty acid standards, and the free fatty acids were isolated by selective extraction with methanol and isooctane. The extracted fatty acids were derivatized and analyzed by gas chromatography and MS, as described in detail elsewhere. 26
Reverse-phase LC/MS
Eicosanoids in plasma were analyzed and quantified by LC/MS/MS as previously described.8,26 Briefly, eicosanoids were separated by reverse-phase chromatography using a 1.7 µM 2.1 × 100 mm BEH Shield Column (Waters, Milford, MA, USA) and an Acquity UPLC system (Waters). The column was equilibrated with buffer A, and 5 μl of sample was injected via the autosampler. Samples were eluted with a step gradient starting with 100% buffer A for 1 min, then to 50% buffer B (consisting of 50% acetonitril, 50% isopropanol, and 0.02% acetic acid) over a period of 3 min, and then to 100% buffer B over a period of 1 min. The LC was interfaced with an IonDrive Turbo V ion source, and mass spectral analysis was performed on a triple quadrupole AB SCIEX 6500 QTrap mass spectrometer (AB SCIEX, Framingham, MA, USA). Eicosanoids were measured using electrospray ionization in negative ion mode and multiple reaction monitoring (MRM) using the most abundant and specific precursor ion/product ion transitions to build an acquisition method capable of detecting 158 analytes and 26 internal standards. The ionspray voltage was set at −4500 V at a temperature of 550°C. Collisional activation of the eicosanoid precursor ions was achieved with nitrogen as the collision gas with the declustering potential, entrance potential, and collision energy optimized for each metabolite. Eicosanoids were identified by matching their MRM signal and chromatographic retention time with those of pure identical standards.
Quantitation of lipids
Eicosanoids were quantitated by the stable isotope dilution method. Briefly, identical amounts of deuterated internal standards were added to each sample and to all the primary standards used to generate standard curves. To calculate the amount of eicosanoids and free fatty acids in a sample, ratios of peak areas between endogenous metabolite and matching deuterated internal standards were calculated. Ratios were converted to absolute amounts by linear regression analysis of standard curves generated under identical conditions.
Statistical analysis
Data preparation
Out of the 161 eicosanoids measured in the LipoNexus platform, 34 were selected for further analyses based on: (1) 75% samples were ⩾LLOQ (31 metabolites), and (2) previous evidence for relevance in NASH based upon the pilot study (three metabolites). 8
Data analysis
Baseline analysis of the association of individual plasma eicosanoids with fibrosis stage was assessed using Jonckheere-Terpstra (ordinal stage categories) trend tests. The longitudinal analysis of the association of individual markers with binary response (e.g. improvement in fibrosis stage) was assessed using Wilcoxon tests. The performance of individual markers for the detection of liver fibrosis or improvement in fibrosis or hepatic collagen content was assessed using area under the receiver operating characteristics curve (AUROC) based on the whole dataset and 5-fold cross-validation repeated 100 times. An ad hoc approach was performed to combine markers for monitoring improvement responses. Specifically, plasma eicosanoids were considered if their AUROC (based on the full dataset) or Wilcoxon p value for change from baseline to week 24 from univariate analyses passed certain thresholds [e.g. AUROC ⩾0.65 or Wilcoxon p ⩽ 0.1, and ⩾75% of samples were above limit of quantitation (ALOQ)]. Additionally, a systematic two-step approach was considered for monitoring improvements in fibrosis. The first step was to identify the most informative markers (including baseline and change from baseline) based on consistent signals across multiple methods including logistic regression, random forests, generalized, unbiased, interaction detection and estimation (GUIDE), and regularized regression. A weighted score based on the importance assessed by each method was then formed to rank the markers. In the second step, logistic ridge regression (including baseline and change from baseline as predictors) was used to establish the multi-marker classification algorithm based on the selected markers, and its performance assessed using AUROC. All statistical analyses were performed using R and Graphpad Prism software.
Results
Baseline associations between eicosanoids and fibrosis stage
Analysis population
Data from 427 patients with baseline histological data and plasma eicosanoid assessment were used in this analysis. Participants had a mean age of 52 years and BMI of 34 kg/m2. The distribution of fibrosis stages was F0: 7%, F1: 22%, F2: 24%, F3: 34%, and F4: 12%. Detailed baseline demographic and clinical characteristics of the participants are provided in Table 1.
Baseline characteristics of the study population.

Median and IQR values and n (%) are provided, unless otherwise noted.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; GGT, gamma-glutamyl transferase; HDL, high-density lipoprotein; IQR, interquartile range; LDL, low-density lipoprotein; NAFLD, nonalcoholic fatty liver disease; NAS, NAFLD activity score; NASH, nonalcoholic steatohepatitis; T2D, type 2 diabetes.
Plasma eicosanoids are associated with fibrosis stage
We identified four plasma eicosanoids significantly associated with fibrosis stage: 11, 12-DIHETE (p = 0.0094), tetranor 12-HETE (p = 0.0135), adrenic acid (p = 0.0328), and 14,15-DIHETE (p = 0.0481; Figure 1). In addition, three plasma eicosanoids were significantly associated with the presence of advanced fibrosis (stage 3 and 4): 7,17-DHDPA (p = 0.01), 11,12-DIHETRE (p = 0.03), and DHK-PGD2 (p = 0.03; Table 2, Supplemental Table S1). Four plasma eicosanoids were significantly associated with cirrhosis (stage 4 fibrosis): 8-HETE (p = 0.0103), 11-HETE (p = 0.0212), adrenic acid (p = 0.0259), and 15-HETRE (p = 0.0299; Table 3). However, there was no significant correlation between the plasma eicosanoids associated with stage of fibrosis 3 or 4 and grade of lobular inflammation Supplemental Table S2.

Baseline association of plasma eicosanoids with fibrosis stages. Plasma concentration of the four eicosanoids significantly associated with liver fibrosis stages are depicted as whisker plots across liver fibrosis stages: (A) 11,12-DIHETE; (B) tetranor 12-HETE; (C) adrenic acid; (D) 14,15-DIHETE. p values were determined using the Jonckheere test.
Eicosanoids that are significantly altered in NASH subjects with advanced fibrosis (F3/4).

Eicosanoids that are significantly higher in NASH subjects with cirrhosis (F4).

Multi-variable adjusted p value for age, sex, BMI, and presence of T2D.
BMI, body mass index; NASH, nonalcoholic steatohepatitis; T2D, type 2 diabetes.
Longitudinal associations between changes of eicosanoids and liver fibrosis improvement
Analysis population
Of the 72 patients with biopsy-proven NASH and stage 2 or 3 fibrosis who were randomized and treated in the phase II trial of selonsertib, 63 with evaluable eicosanoid assessment and liver biopsy at baseline and week 24 were included in the analysis. Detailed baseline characteristics are provided in Table 4.
Baseline characteristics of the longitudinal study population.

Median and interquartile range (IQR) values and n (%) are provided, unless otherwise noted.
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; GGT, gamma-glutamyl transferase; NAFLD, nonalcoholic fatty liver disease; NAS, NAFLD activity score; T2D, type 2 diabetes.
Changes in plasma eicosanoid are associated with liver fibrosis improvement
We identified seven eicosanoids (5-HETE, 7, 17-DHDPA, adrenic acid, arachidonic acid, EPA, 16-HDOHE, and 9-HODE) as the most informative markers using a weighted score for the prediction of ⩾1-stage improvement of fibrosis. The median relative changes of these seven eicosanoids stratified by liver fibrosis changes (fibrosis improvement, no change and worsening) are shown in Figure 2A. Baseline and longitudinal change values of these seven eicosanoids were evaluated as predictors for fibrosis improvement at week 24. The AUROC of the individual eicosanoids ranged between 0.52 and 0.67 and the combination of the seven eicosanoids yielded a good diagnostic performance for the prediction of liver fibrosis improvement [AUROC 0.74; 95% confidence interval (CI): 0.62–0.87; Table 5 and Supplemental Table S3].
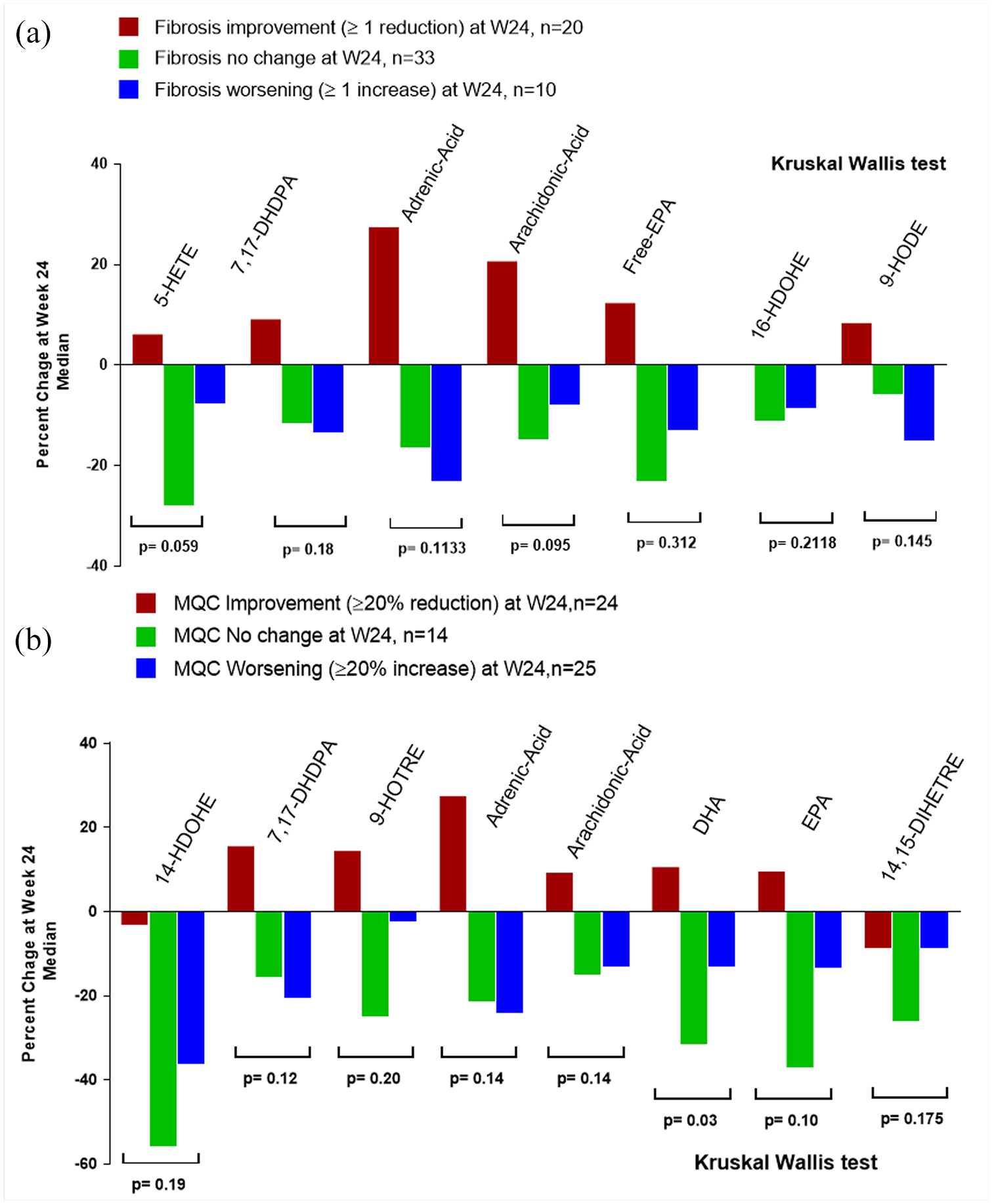
Plasma eicosanoid changes are associated with changes in liver fibrosis and hepatic collagen content. Median changes in plasma eicosanoids from baseline to week 24 of the most informative biomarkers. (A) Stratified by liver fibrosis changes: red bar participants with improvement of liver fibrosis ⩾1 stage (n = 20), green bar participants with no change in liver fibrosis (n = 33), and blue bar participant with liver fibrosis worsening ⩾1 stage (n = 10). (B) Stratified by MQC changes as assessed by % picrosirius red stained area: red bar participants with improvement of MQC ⩾20% (n = 24), green bar participants with no change in MQC (n = 14), and blue bar participant with MQC worsening ⩾20% (n = 25). p value was determined using the Kruskal–Wallis test.
Performance of an eicosanoid panel for the prediction of liver fibrosis improvement.
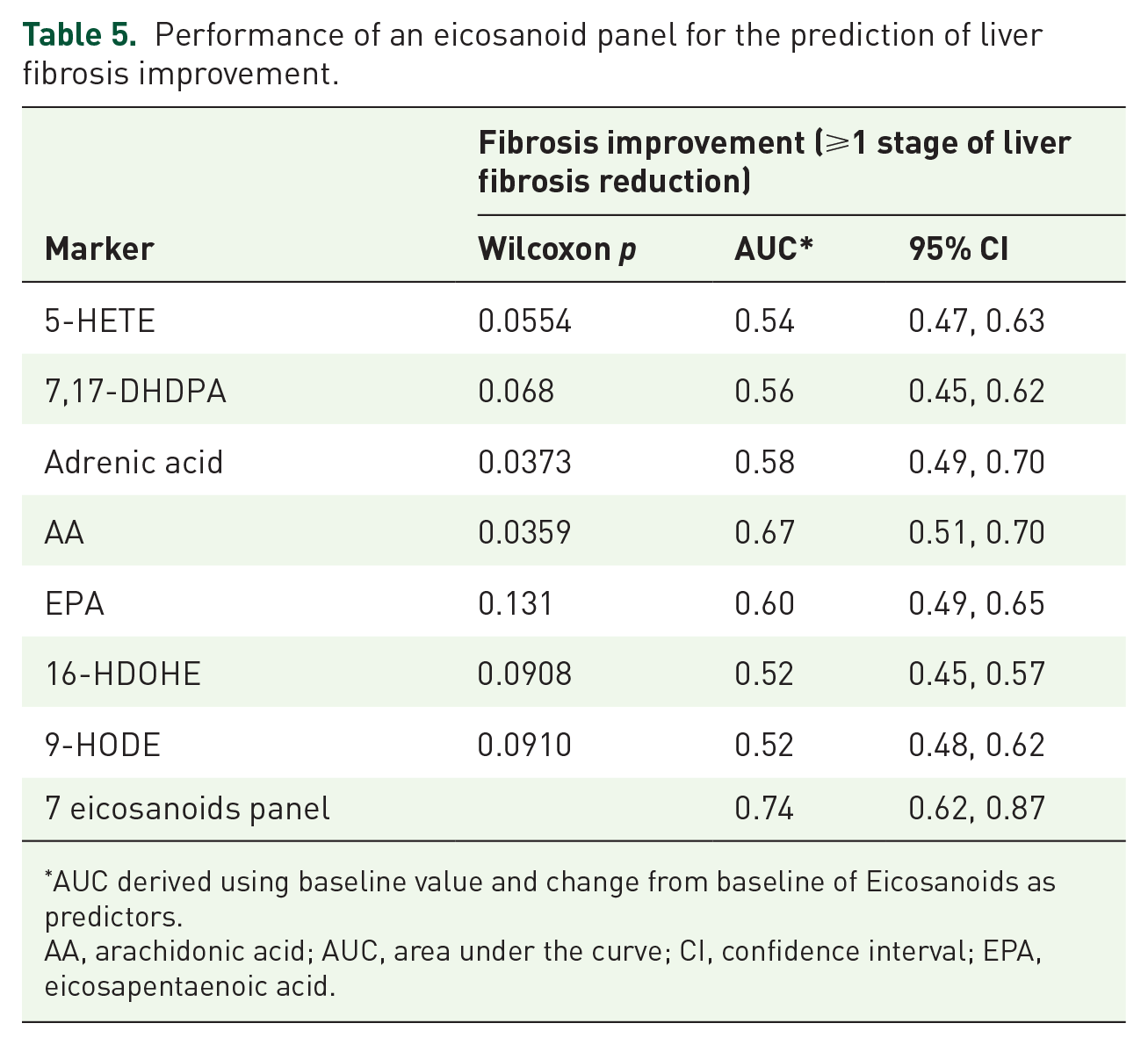
AUC derived using baseline value and change from baseline of Eicosanoids as predictors.
AA, arachidonic acid; AUC, area under the curve; CI, confidence interval; EPA, eicosapentaenoic acid.
Changes in plasma eicosanoids are associated with improvement in hepatic collagen content
We also examined associations between changes in plasma eicosanoids and hepatic collagen content. We identified eight eicosanoids: 14-HDOHE, 7,17-DHDPA, 9-HOTRE, adrenic acid, AA, DHA, eicosapentaenoic acid (EPA), and 14,15-DIHETRE as the most informative markers using a weighted score for the prediction of hepatic collagen content improvement (⩾20% relative reduction from baseline to week 24). The median relative changes of these seven eicosanoids stratified by hepatic collagen content changes (improvement, no change, and worsening) are shown in Figure 2B. Baseline and longitudinal changes of these eicosanoids were evaluated as predictors of hepatic collagen content improvement at week 24. The AUROCs of the top four individual eicosanoids ranged from 0.49 to 0.69 and the combination of these four eicosanoids yielded a good diagnostic performance for the prediction of hepatic collagen content improvement with an AUROC of 0.72 (95% CI: 0.50–0.77; Table 6).
Performance of an eicosanoid panel for the prediction of hepatic collagen improvement.
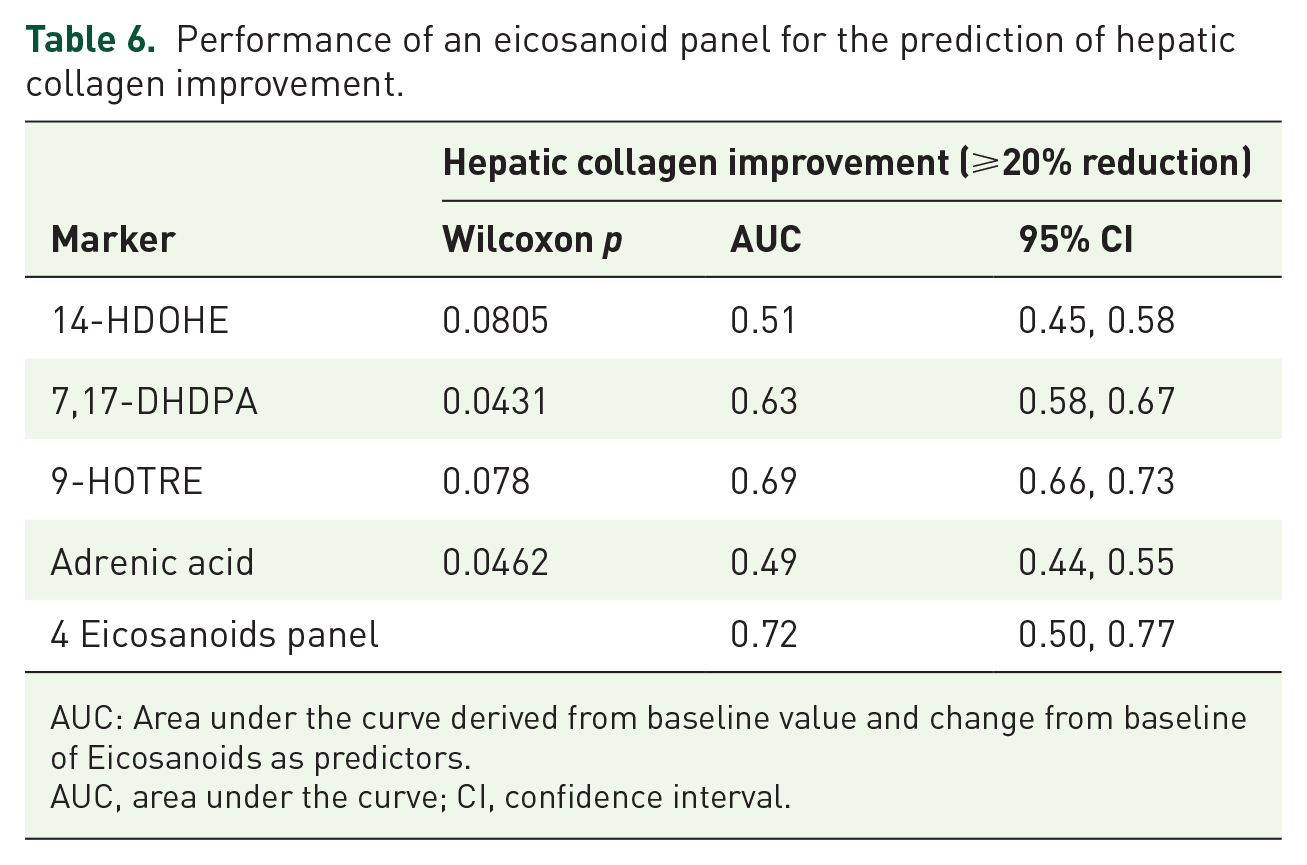
AUC: Area under the curve derived from baseline value and change from baseline of Eicosanoids as predictors.
AUC, area under the curve; CI, confidence interval.
We performed sensitivity analysis and there were no significant differences between the change in plasma eicosanoids and the treatment arm in the study.
Discussion
Main findings
Using a large cohort of patients with biopsy-proven NAFLD across the spectrum of fibrosis severity, we have identified four plasma eicosanoids significantly associated with fibrosis stage. Furthermore, using data from a multicenter phase II trial of selonsertib in patients with NASH and stage 2 or 3 fibrosis, we showed that changes in individual plasma eicosanoids were associated with improvement in both histological liver fibrosis stage and hepatic collagen content. The combination of these individual eicosanoids yielded a good diagnostic performance for the prediction of liver fibrosis improvement and for the prediction of hepatic collagen improvement. This proof-of-concept study demonstrates that plasma eicosanoids can serve as noninvasive biomarkers of liver fibrosis and improvement of liver fibrosis in patients with NAFLD. However, further studies are needed to confirm the diagnostic performance of the combination of plasma eicosanoids for the detection of liver fibrosis improvement in NAFLD. These novel data have potentially important implications for the noninvasive assessment of treatment response in NAFLD, which remains a major unmet need in the field.
Findings in the context of the published literature
Several studies have previously reported alterations of plasma eicosanoids associated with the presence of NASH that are less likely to be found in patients with NAFL or in healthy controls.8,10,22 Our study builds on these previous data, and further assesses associations between plasma eicosanoids and liver fibrosis in NAFLD. Three of the four plasma eicosanoids associated with liver fibrosis in this study are consistent with our previous pilot study, which demonstrated that 11,12-DIHETE, tetranor 12-HETE, and 14,15-DIHETE were associated with NASH. 8 These data suggest that eicosanoids may be involved in a common pathway leading to both NASH and liver fibrosis in patients with NAFLD. Indeed, alterations in hepatic eicosanoid levels have been reported in humans; these alterations in proinflammatory eicosanoids may induce hepatic oxidative damage in NASH. 22 However, further studies are needed to determine whether these novel biomarkers are directly involved in hepatic fibrogenesis in NASH and to identify the precise pathway(s) involved.
This is the first study to assess associations between longitudinal changes in plasma eicosanoids and changes in histological stage of fibrosis or hepatic collagen content. The association between eicosanoid levels and fibrosis does not necessarily mean that eicosanoids have a causal role in the development of NASH and liver fibrosis; changes in eicosanoid levels may in some cases simply reflect alterations in pathways associated with disease activity. However, adrenic acid, AA, and DHA, which were identified among the most informative biomarkers to predict improvement of both liver fibrosis and hepatic collagen content, have been shown to be involved in the pathogenesis of NAFLD. AA is the precursor of eicosanoids through three main pathways including cytochrome P450s, cyclooxygenases, and lipoxygenases.8,19 Recently, Hall et al. have reported an increased release of AA from the phospholipid membrane and production of eicosanoid species in the liver of NASH that promote inflammation and cell injury. 22 In addition, an increase in plasma adrenic acid levels has been reported in children with hepatic steatosis, 27 and adrenic acid accumulation contributes to disease progression in NAFLD in mice. 28 Finally, alterations in plasma DHA concentrations have been previously reported in patients with NASH,8,10 and studies suggest that dietary supplementation of DHA may be effective at lowering liver fat content in NAFLD. 29 Hence, changes in combination ‘panels’ of these eicosanoids could be a more direct measure of disease activity, and may be better biomarkers of histological improvement in NAFLD patients than classic indirect markers of liver fibrosis such as ALT, AST, and platelet count.
Strengths and limitations
There are several notable strengths of this study, including the use of well-characterized cohorts from clinical trials with paired liver biopsies centrally read by a single, experienced hepatopathologist. In addition, the large sample size of the cohort for the baseline assessment (n = 427) including all stages of liver fibrosis enabled us to study associations between plasma eicosanoids and stages of fibrosis, which was not possible in the previous seminal studies in patients with NASH due to limited sample size.8,10 We acknowledge that, due to the study design, baseline plasma samples were not collected on the same day as the baseline liver biopsy. Moreover, the multicenter study design allowed the inclusion of participants with various ethnic origins, different environmental exposures, or dietary behavior, which limits the risk of bias and increases the likelihood of generalizations from the study. Among the seven eicosanoids that predict longitudinal changes in fibrosis stage, adrenic acid and 7,17-DHDPA are associated with liver fibrosis and advanced fibrosis, respectively. The lack of complete overlap between the eicosanoids associated with stage of fibrosis in the baseline assessment and eicosanoids associated with longitudinal changes could be due to the specific population included in the longitudinal study. Contrary to the baseline assessment, which included a whole spectrum of stage of fibrosis (F0–F4), the longitudinal assessment included only patients with F2 and F3 and an NAS ⩾5. Hence, the current study may not have captured the markers associated with a decrease of cirrhosis or improvement from a lower stage of disease activity. Moreover, potential confounding factors such as drug effects, improvement in inflammation or lipid metabolism, and changes in BMI or insulin resistance could not have been assessed in this proof-of-concept study and further studies are needed to confirm the validity of the seven eicosanoids panel in independent population with various stages of NAFLD. However, the eicosanoids associated with higher level of liver fibrosis stage F3 and F4 were not significantly correlated with the grade of lobular inflammation, which suggests that the association observed between these plasma eicosanoids and liver fibrosis may not be driven by inflammatory liver damage. Finally, the quantitative assessment using deuterated standards and LC/MS/MS provides a comprehensive profiling of plasma eicosanoids and free fatty acids.8,21 Our study also has limitations. We also acknowledge that statistical adjustment for multiple comparison were not performed in this exploratory study and further study is needed to confirm the biological importance of the identified eicosanoids. Even though the combination of plasma eicosanoids predicts improvement in liver fibrosis, plasma eicosanoids are not liver specific. Therefore, change in plasma eiconsanoids may reflect change in PUFA metabolism in other organs related to the improvement in liver fibrosis. The causal role of eicosanoids in the development of liver fibrosis has not been established, and further longitudinal studies are needed.
Implications for future study
In this proof of concept study using well-characterized cohorts, we demonstrate that plasma eicosanoids are associated with liver fibrosis in NAFLD, and that changes in plasma eicosanoids can predict liver fibrosis improvement. Further validation in independent cohorts with larger sample sizes to confirm the diagnostic performance of a combination of plasma eicosanoids to predict histological improvement in NAFLD is warranted.
Supplemental Material
Supplemental_Data – Supplemental material for Plasma eicosanoids as noninvasive biomarkers of liver fibrosis in patients with nonalcoholic steatohepatitis
Supplemental material, Supplemental_Data for Plasma eicosanoids as noninvasive biomarkers of liver fibrosis in patients with nonalcoholic steatohepatitis by Cyrielle Caussy, Jen-Chieh Chuang, Andrew Billin, Tao Hu, Ya Wang, G. Mani Subramanian, C. Stephen Djedjos, Robert P. Myers, Edward A. Dennis and Rohit Loomba in Therapeutic Advances in Gastroenterology
Footnotes
Author contributions
Cyrielle Caussy: Formal analysis, writing – original draft, writing – review and editing.
Jen-Chieh Chuang: Data curation, formal analysis, visualization, writing – original draft, writing – review and editing.
Tao Hu and Ya Wang: Data curation, formal analysis, visualization.
G. Mani Subramanian, C. Stephen Djedjos, Robert Myers, and Andrew Billin: Conceptualization, formal analysis, funding acquisition, methodology, writing – review and editing.
Edward A. Dennis and Rohit Loomba: Conceptualization, formal analysis, funding acquisition, methodology, supervision, writing – original draft, writing – review and editing.
Conflict of interest statement
EAD and RL are co-founders, consultants, and hold equity in LipoNexus Inc. CC is a consultant to LipoNexus, Inc. The remaining authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by LipoNexus, Inc. and Gilead Sciences, Inc.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
