Abstract
Background:
Delayed post-polypectomy bleeding (DPPB) is the most common adverse event following colonic polypectomy, yet its management remains highly heterogeneous and lacks standardization. A considerable number of colonoscopies performed for DPPB may be unnecessary and do not result in hemostatic intervention.
Objectives:
To develop evidence-based statements to guide clinical decision-making in DPPB.
Design:
Multidisciplinary Delphi consensus statement.
Methods:
A panel of 29 experts in gastroenterology, hematology, radiology, and surgery was assembled. Through a systematic review of the literature and a modified Delphi process, consensus statements were developed through iterative rounds of anonymous voting. Statements were revised following anonymous voting and feedback at each round. Those achieving 80% agreement were accepted.
Results:
The expert panel reached a consensus on 36 statements, covering areas such as antithrombotic management, bowel preparation, colonoscopy indications, and therapeutic hemostatic modalities. Key recommendations include guidance for managing self-limited bleeding and risk stratification to reduce the rate of unnecessary colonoscopies, as well as recommendations for hemodynamically unstable patients who may require primary angioembolization. A practical clinical algorithm is proposed.
Conclusion:
This document provides a consensus-based framework for managing DPPB. These recommendations aim to improve patient outcomes and optimize healthcare resources while fostering a standardized approach to this common adverse event.
Plain language summary
Delayed post-polypectomy bleeding (DPPB) is the most common complication following the removal of polyps during a colonoscopy. Despite its frequency, there is no standardized approach for managing this issue. Many patients undergo colonoscopies that may not be necessary and do not lead to further treatment. This can result in wasted healthcare resources and unnecessary patient discomfort. To address this, a group of 29 experts in gastroenterology, hematology, radiology, and surgery reviewed the available scientific evidence. Using a structured process called the Delphi method, they developed 36 consensus statements to guide healthcare professionals. These statements provide clear recommendations for managing DPPB, such as when to perform a colonoscopy, how to handle antithrombotic medications (like blood thinners), and what treatments to use for severe bleeding. One key takeaway is that many cases of DPPB resolve on their own without the need for a repeat colonoscopy. For more severe cases, the guidance outlines the use of endoscopic treatments, radiological or surgical interventions if necessary. A practical algorithm is included to help doctors make decisions tailored to individual patients. These recommendations aim to improve patient outcomes, reduce unnecessary procedures, and optimize the use of healthcare resources. By fostering a consistent approach to managing DPPB, this consensus provides a valuable tool for healthcare professionals worldwide.
Introduction
Colonic polypectomy is a critical intervention for preventing colorectal cancer and has become the most common endoscopic procedure. 1 Delayed post-polypectomy bleeding (DPPB) is the most frequent adverse event (AE) of this technique. 2 Existing research primarily concentrates on assessing the incidence, risk factors, and efficacy of hemoclipping and other preventive methods.2 –5 However, there is a scarcity of studies exploring the management of DPPB.
Available data indicate that DPPB is often self-limited, suggesting an overutilization of colonoscopy.6–8 The decision-making process in these cases typically relies on the discretion of the treating physician or local clinical protocols. This accounts for the clinical management variability observed across different hospitals and the frequent occurrence of colonoscopies without clear benefit, ranging from 0% to 63%.2,6–12 Although repeating a colonoscopy may warrant endoscopic therapy in certain patients, many could benefit from a watchful waiting approach, as spontaneous resolution is relatively common.6–8
Current clinical guidelines from international scientific societies lack a comprehensive algorithm detailing when and for whom a colonoscopy should be performed. Reducing the number of ineffective colonoscopies would result in decreased costs, less consumption of healthcare resources, and avoidance of potential AEs associated with the procedure. Moreover, reducing colonoscopy frequency could improve the patient experience by avoiding the discomfort associated with the preparation and the procedure itself. In addition, the environmental impact of colonoscopy, which can reach up to 30 kg of CO2 per procedure, should not be underestimated. 13
An analysis of the literature highlights the necessity to generate clinical recommendations that optimize and standardize the clinical management of DPPB. Consequently, the mucosal resection and third-space group of the Spanish Society of Gastrointestinal Endoscopy (SEED) proposed the creation of a consensus statement to address this gap. This document aims to conduct a critical appraisal of the available evidence to generate a management algorithm based on the best available evidence for DPPB.
Methods
The project was promoted by the steering committee of the SEED group on mucosal resection and third-space endoscopy. The expert panel consisted of consultant gastroenterologists with a special focus on gastrointestinal endoscopy or clinical inpatient management, as well as hematologists, radiologists, and surgeons. The primary aim was to reach a consensus on a clinical management algorithm for delayed DPPB, regardless of the polyp removal technique used. The scope of the document did not include the prophylaxis of DPPB, given that contemporary clinical guidelines were readily available. 14
A Consensus Statement format was deemed appropriate given the expected limited body of evidence. In April 2023, the steering committee outlined the manuscript’s structure, established Task forces, and developed questions of interest in the PICO format (Population/Intervention/Comparisons/Outcomes). Systematic literature reviews were conducted in at least two databases. Task forces critically assessed the literature using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system through May 2024. In the absence of evidence or when GRADE was not applicable, recommendations were formulated as Good Practice Statements 15 or statements. Consensus among members was determined anonymously using a Delphi method. Statements were rated using a 5-point Likert scale on a web-based platform (Google Forms), with consensus defined as ⩾80% agreement (combining Agree and Strongly agree). The final manuscript received the SEED governing board’s endorsement. The manuscript is reported following the CREDES (Conducting and REporting of DElphi Studies) guideline. 16 Additional methodology information is provided in Supplemental Material.
Definitions
Delayed post-polypectomy bleeding is defined as any rectal bleeding occurring within 30 days after the completion of the index colonoscopy, requiring emergency room presentation, hospitalization, or reintervention (repeat endoscopy, angiography, or surgery).
(Strongly agree: 69.2%; agree: 30.8%)
Stigmata of recent bleeding is defined as the presence of any of the following findings at a polyp resection mucosal defect: active bleeding (either jet or oozing), a visible vessel, or an adherent clot that remains despite targeted jet irrigation.
(Strongly agree: 71.4%; agree: 25%)
Rebleeding is defined as (1) a new episode of hematochezia or rectorrhagia at least 48 hours after successful hemostatic therapy of the initial delayed post-polypectomy bleeding or after discharge; (2) a new drop in hemoglobin levels greater than 3 g/dL or additional transfusion requirements 48 hours after resolution of the baseline bleeding episode; (3) confirmation of stigmata of recent bleeding at the scar responsible for the initial delayed post-polypectomy bleeding on repeat endoscopy or angio-CT/angiography; and (4) any colonoscopy performed for suspicion of bleeding in research studies.
(Strongly agree: 50%; agree: 42.3%)
The definitions provided in this document are based on the available literature (see Supplemental Table 1) and agreed-upon consensus, prioritizing clinical relevance and applicability. They are intended to be used both in clinical practice and for research purposes in future studies.
Epidemiology
The incidence of DPPB ranges from 0.2% to 7%, depending on the type of polyp, resection technique used, or the presence of risk factors such as active use of antithrombotics.2–4,9–11,17 Although specific data are scarce, reported transfusion rates vary widely from 5.5% to 75%, with a median hospitalization length of 3 days and a 30-day mortality rate of 0.18%.2,8,17–19 No information is currently available on the costs associated with DPPB. The rebleeding rate ranges from 7.6% to 14.9%, although predictive factors are still not well established.8–10,12 Details of the individual studies are provided in Supplemental Table 1.
Prognostic scales and grading severity
SEED recommends that clinical judgment is crucial in assessing patients with delayed post-polypectomy bleeding, with prognostic scales or tests serving as complementary tools rather than substitutes.
Good Practice Statement. (Strongly agree: 84.6%; agree: 15.4%)
In recent years, several prognostic scales have been developed for lower gastrointestinal bleeding (LGIB).20–24 Most studies are observational, aiming to identify key factors associated with LGIB prognosis. However, comprehensive validation of these prognostic indices remains incomplete, and none of them is primarily focused on DPPB.20–24
Among the various scales, the Oakland score has emerged as one of the most extensively studied. A recent meta-analysis highlighted its superior prognostic performance in predicting outcomes such as safe discharge, likelihood of major bleeding, and need for transfusion. 25 Importantly, the Oakland score has been particularly noted for its ability to identify patients suitable for early discharge with minimal risk of rebleeding. A score of 8 or lower in the Oakland score correlates with a 95% probability of safe discharge without additional complications, 21 and it has been cited in recent guidelines as a valid cutoff for clinical practice. 26 Similarly, the meta-analysis identified the Strate score as the most effective predictor of the need for hemostasis. 25
Despite the array of available scoring systems, current evidence does not support the exclusive endorsement of any single scale.26–28 The Oakland score, validated primarily in stable patients without active bleeding or anticoagulant therapy, appears most suited for predicting early discharge. However, its accuracy in patients presenting with DPPB remains unknown and may differ from other LGIB etiologies.
SEED suggests utilizing an endoscopy-focused classification (AGREE classification or ASGE Lexicon) for grading the severity of delayed post-polypectomy bleeding.
Weak recommendation, very low-quality evidence. (Strongly agree: 53.8%; agree: 34.6%)
The use of a standardized classification for grading the severity of AEs in endoscopy is essential for assessing procedure safety, allowing for direct comparisons, and represents a key element of high-quality colonoscopy. However, only a few classifications have been proposed, and none specifically for DPPB. These include the Clavien-Dindo Classification, developed in the surgical field in 2004, 29 the American Society for Gastrointestinal Endoscopy (ASGE) lexicon proposed in 2010, 30 and the AGREE (Adverse events in GastRointEstinal Endoscopy) classification introduced in 2021. 31 The AGREE classification and ASGE lexicon may be more appropriate for grading severity of DPPB, as they are specifically designed for endoscopic procedures and have been utilized in previous DPPB studies.7,8,32,33 We did not find any studies comparing the different classifications in the context of DPPB.
Initial management and blood transfusion
SEED recommends evaluating the patient’s general clinical condition and prioritizing resuscitation measures if necessary. The initial assessment should include a focused medical history, a physical examination (including an anorectal exam), and blood tests.
Good Practice Statement. (Strongly agree: 92.3%; agree: 7.7%)
There are no studies evaluating initial supportive measures in patients with DPPB. Recommendations are derived from expert consensus and clinical guidelines aimed at managing LGIB. Initially, the patient’s hemodynamic stability should be assessed, prioritizing resuscitation measures.26,34 It is recommended to secure two adequately sized peripheral veins and/or a central line. 35 Crystalloids are usually preferred over colloids for volume replacement and stabilization. 36 The initial patient evaluation should involve a focused medical history that details colonoscopy information (date, lesions removed, size, location, removal technique, symptom onset, and prophylactic scar therapy), relevant comorbidities, current pharmacological treatments (such as antiplatelets and anticoagulants), a rectal examination, and blood tests including a complete blood count and coagulation studies.26,34,37
There are no studies comparing out versus inpatient management in patients with DPPB. Most studies do not specify the criteria for patient admission or discharge. In a retrospective study of 548 patients with DPPB, 80.8% were admitted, but admission criteria were not detailed. 8 Another study of 69 DPPB patients found that 69.6% underwent therapeutic colonoscopy as inpatients, while the rest were managed as outpatients, with no details on observation time or discharge criteria. 10
Other authors advocate for admitting all patients.6,38 Burgess et al. propose an algorithm recommending 24 h of surveillance and considering discharge if there is no recurrence of bleeding, the patient remains hemodynamically stable, and there is no significant change in hemoglobin level during that time. 7 Van der Star et al. evaluated 42 patients and suggested discharge if bleeding stops spontaneously and does not recur during 24 h of observation. 9
In hemodynamically stable patients with significant cardiovascular comorbidity, red blood cell transfusion is recommended for hemoglobin levels ⩽8–9 g/dL with a post-transfusion target of 9–10 g/dL. In hemodynamically stable patients without cardiovascular comorbidity, the hemoglobin threshold for blood transfusion should be ⩽7 g/dL, with a target range of 7–9 g/dL.
Strong recommendation, very low-quality evidence. (Strongly agree: 69.2%; agree: 30.8%)
SEED recommends following severe bleeding protocols for blood transfusion in hemodynamically unstable patients, rather than relying solely on hemoglobin levels.
Good Practice Statement. (Strongly agree: 60.7%; agree: 39.3%)
No specific recommendations regarding transfusion for DPPB have been identified. A post hoc study in patients with LGIB 39 concluded that, although there were no statistically significant differences between restrictive strategies (Hb threshold <7 mg/dL) and liberal strategies (transfusion threshold <9 mg/dL), a trend toward reduced rebleeding and severe events was observed with restrictive strategies. The recommendation for restrictive transfusion strategies is primarily extrapolated from evidence in upper gastrointestinal bleeding. 40 Patients with significant cardiovascular comorbidity or advanced age may benefit from a less restrictive transfusion threshold. Given the lack of specific studies on DPPB, our recommendations are in line with current guidelines on LGIB.26,34,37
For patients with hemorrhagic shock, a different approach is warranted, involving volume resuscitation and blood component transfusion according to severe bleeding protocols. Platelet transfusion and fibrinogen replacement should be guided by laboratory parameters.41–44
Management of anti-thrombotics
The SEED proposal for antithrombotic management is summarized in Figures 1 and 2. Our recommendations for the management of anti-thrombotics have been adapted and adhere to current LGIB guidelines, given the lack of specific evidence for DPPB.26,34,37
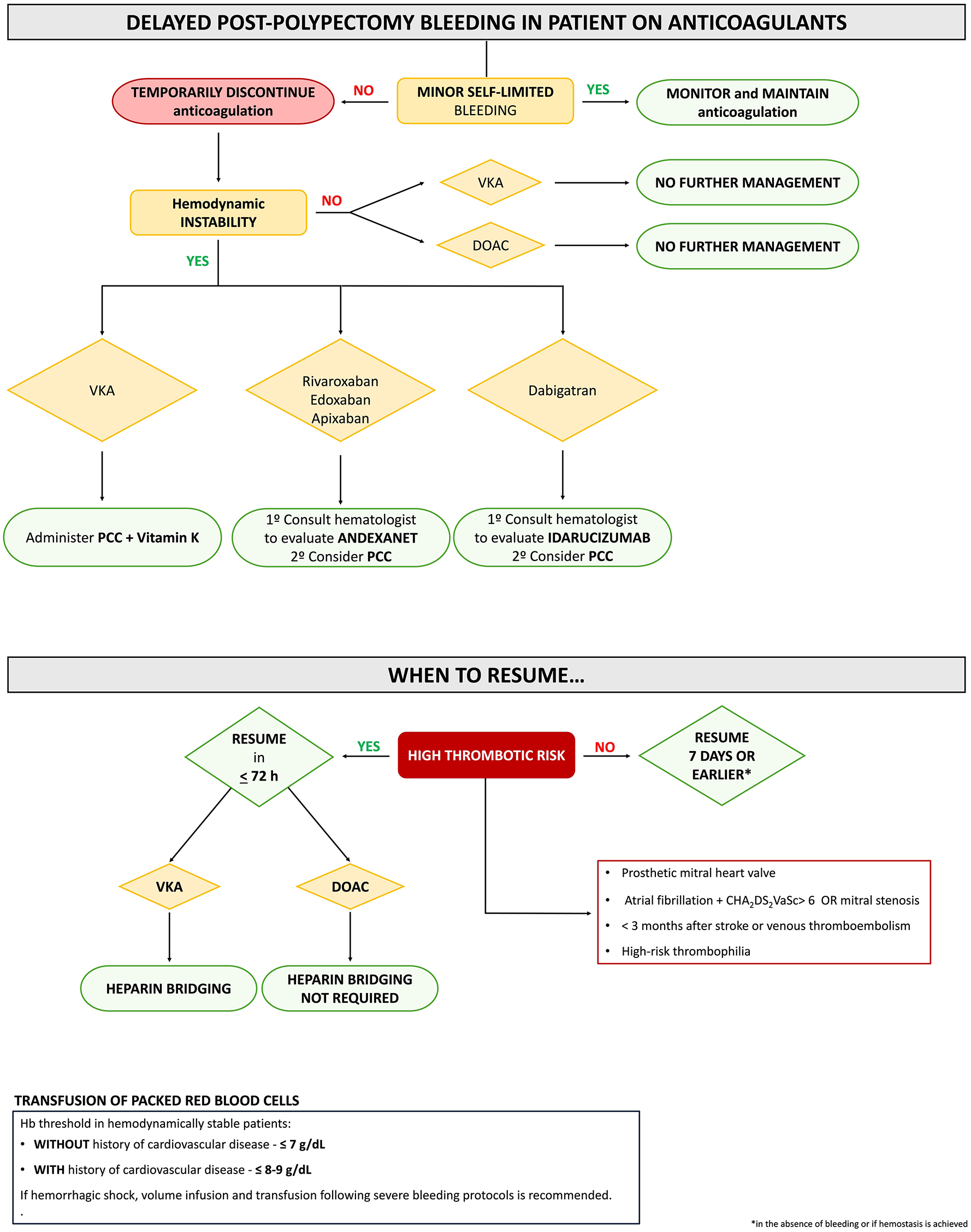
Delayed post-polypectomy bleeding in patients on anticoagulants.
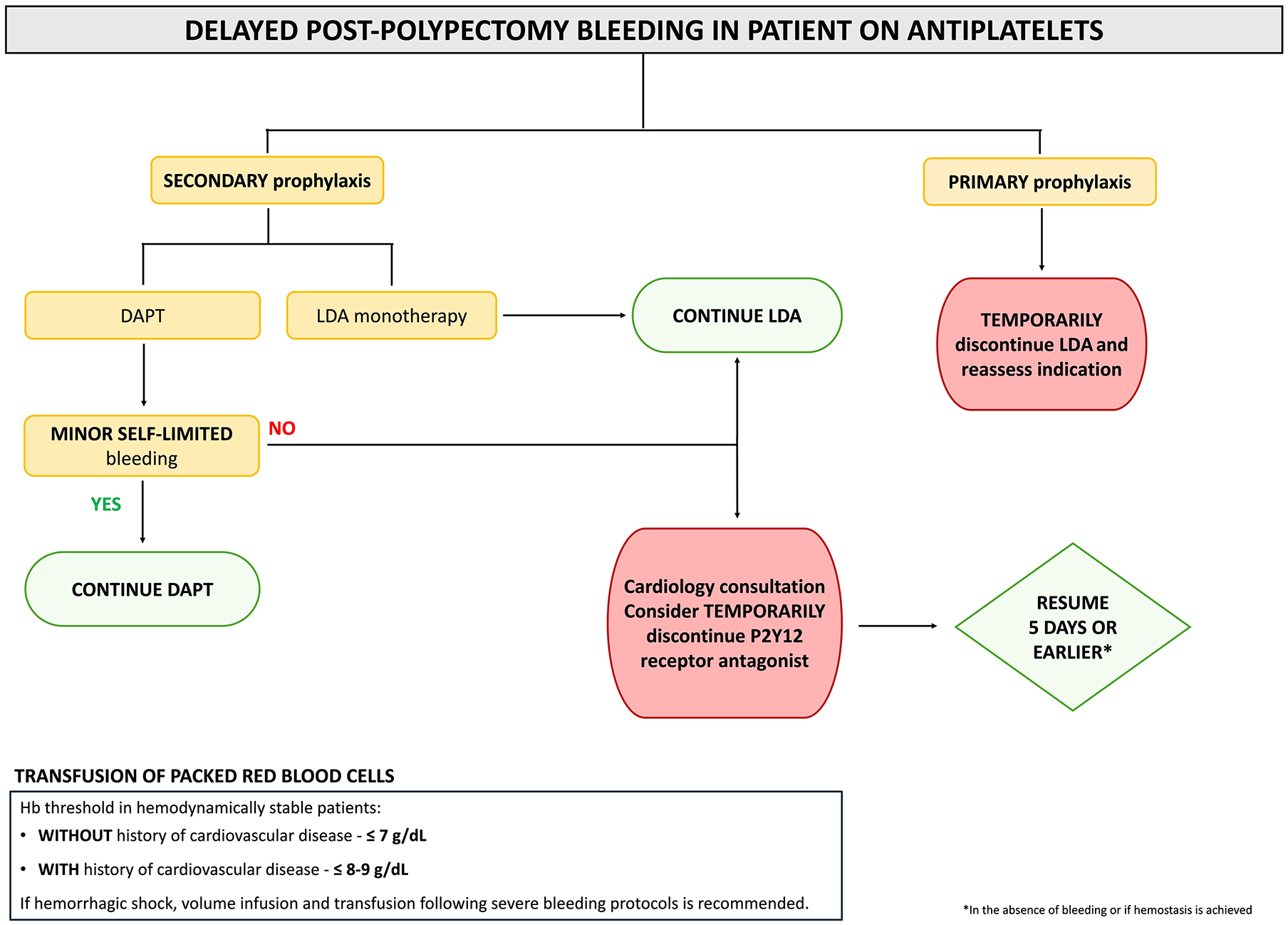
Delayed post-polypectomy bleeding in patients on antiplatelets.
Antiplatelet discontinuation
SEED recommends that aspirin for secondary prophylaxis should not be withheld. It may be temporarily withheld only in cases of severe ongoing bleeding and/or hemodynamic instability.
Strong recommendation, very low-quality evidence. (Strongly agree: 61.5%; agree: 38.5%)
SEED recommends withholding aspirin for primary prophylaxis during the acute phase of DPPB.
Strong recommendation, very low-quality evidence. (Strongly agree: 61.5%; agree: 38.5%)
SEED recommends against discontinuing dual-antiplatelet therapy (aspirin and a P2Y12 receptor antagonist) in patients with delayed post-polypectomy bleeding. In cases of severe ongoing bleeding and/or hemodynamic instability, discontinuing the P2Y12 receptor antagonist while continuing aspirin may be considered. This approach should not be undertaken without prior consultation with a cardiologist.
Strong recommendation, very low-quality evidence. (Strongly agree: 61.5%; agree: 34.6%)
In 2021, the European Society of Gastrointestinal Endoscopy (ESGE) recommended withholding low-dose aspirin (LDA) during LGIB if taken for primary prophylaxis. However, the maintenance of LDA was advocated in secondary prevention indications. Routine discontinuation of dual-antiplatelet therapy (DAPT) was not recommended. LDA should be maintained, and the P2Y12 receptor antagonist can be either continued or temporarily withheld, depending on the severity of bleeding and ischemic risk. 26
Evidence regarding the management of antiplatelet therapy in LGIB is limited, and evidence is even more limited in the context of DPPB, with no specific studies conducted to date. A cohort study reported a higher 1-year all-cause mortality (hazard ratio 2.16, 95% confidence interval (CI): 1.39–3.35) among 548 patients taking LDA for secondary prevention if it was interrupted following an episode of gastrointestinal bleeding (16% LGIB). 45 This result was in line with previous observational data. 46 While continuing antiplatelets for secondary prophylaxis may be associated with a relatively higher risk of rebleeding,46–49 this was not consistently detected in all studies.37,50,51 The potential beneficial impact on ischemic risk46,47,49 and mortality45–47 suggests that continuation is the preferred approach. Therefore, we recommend refraining from routinely withholding LDA when indicated for secondary prevention.
This recommendation does not apply to LDA for primary prevention, as current guidelines restrict its use to a very limited population and discourage its use in those with an increased bleeding risk. 52 Consequently, we recommend withholding LDA in primary prevention patients presenting with DPPB and reassessing its indication.
Evidence regarding the management of DPPB patients on DAPT (LDA and a P2Y12 receptor antagonist) is limited. Most of these patients have undergone coronary intervention and stent placement within the last 6–12 months, leading to a significantly high ischemic risk. A multidisciplinary approach involving cardiology is essential, as discontinuing DAPT after acute coronary syndrome and stent placement is linked to an increased risk of ischemic events and mortality. 53 Cardiology guidelines currently accept a shorter period of DAPT (from 1 to 3–6 months) in patients with high bleeding risk. Therefore, the timing of the DPPB episode relative to the prior stent placement is crucial in determining whether to withdraw the P2Y12 receptor antagonist. 54 Data from a randomized controlled trial (RCT), conducted in a limited number of patients with upper gastrointestinal bleeding, suggest that continuation of LDA in DAPT patients is associated with a reduced incidence of ischemic events in the short term. 55 Therefore, since most DPPB cases are self-limited and have lower mortality and rebleeding rates compared to other LGIB etiologies, 8 we recommend against the routine discontinuation of DAPT, particularly within the first 1–3 months following acute coronary syndrome. In severe cases with associated hemodynamic instability, temporary interruption of the P2Y12 receptor antagonist while maintaining LDA may be considered.26,34,37
Antiplatelet resumption
SEED suggests that low-dose aspirin and P2Y12 receptor antagonists should be resumed within 5 days or earlier in the absence of bleeding or if hemostasis is achieved.
Weak recommendation, very low-quality evidence. (Strongly agree: 57.7%; agree: 38.5%)
No data exist on the optimal time to resume antiplatelet agents in the setting of DPPB. A multicenter prospective analysis on the short-term outcomes of withholding antithrombotic therapy revealed higher in-hospital rebleeding rates in LGIB patients on antiplatelet agents. 47 While most rebleeding episodes occurred within 5 days of admission, they were comparable to cases where therapy was uninterrupted or resumed within 5 days. Also, a retrospective cohort study (162 with LGIB) found no significant difference in rebleeding rates when antiplatelet therapy was resumed within 7 days. 56 Another study demonstrated that restarting antiplatelets within 7 days significantly reduced the risk of major adverse cardiovascular and cerebrovascular events, with a higher net clinical benefit, and no significant increase in rebleeding risk. 49 Although not specifically for LGIB, this study defined a cutoff of 9 days to resume therapy for net clinical benefit in patients with gastrointestinal bleeding related to P2Y12 receptor antagonists or aspirin therapy.
Interrupting P2Y12 receptor antagonists after coronary stent implantation is consistently linked with a heightened risk of major cardiovascular events.57,58 Therefore, it is imperative to resume P2Y12 therapy as promptly as feasible following any interruption. Uninterrupted aspirin therapy, with P2Y12 receptor antagonists withheld for no more than 5–7 days, should be reasonable in severe LGIB cases, similar to upper gastrointestinal bleeding. 59
Antiplatelet reversal
SEED suggests against platelet transfusions to reverse antiplatelet therapy in patients with delayed post-polypectomy bleeding.
Weak recommendation, very low-quality evidence. (Strongly agree: 61.5%; agree: 30.8%)
Only two relevant studies have been identified from 2013. The first, a case–control study involving patients admitted with gastrointestinal hemorrhage without thrombocytopenia who were on antiplatelet agents showed a significant increase in mortality, along with non-significant increases in recurrent bleeding and thrombotic events associated with platelet transfusion. 60 An RCT involving 190 patients with intracerebral hemorrhage, who were assigned to receive either platelet transfusion or standard care, reported a higher rate of death in the platelet transfusion group (odds ratio (OR) 2.05, 95% CI: 1.18–3.56). 61 Moreover, this study noted a substantial, albeit not statistically significant, increase in thrombotic events in the platelet transfusion group. 62
Anticoagulation withdrawal
SEED recommends temporarily stopping anticoagulant drugs in cases of delayed post-polypectomy bleeding. However, in selected cases of mild bleeding (Oakland scale score ⩽8), continuation of anticoagulation may be considered, provided there are no new signs of bleeding, and the patient remains clinically stable.
Good Practice Statement. (Strongly agree: 53.8%; agree: 38.5%)
None of the reviewed articles offered conclusive evidence on the benefits or harms of discontinuing anticoagulation in the context of DPPB.56,63–68 The main guidelines offer recommendations on this matter with a weak or very weak level of evidence.26,34,37,69 In general, they recommend suspending anticoagulation during the bleeding episode but suggest considering its continuation in cases of mild LGIB (Oakland score ⩽8). In any case, it seems reasonable to suspend anticoagulation until clinical stability is confirmed and bleeding has ceased.
Anticoagulation reversal
SEED suggests reversing anticoagulation only in patients experiencing ongoing severe bleeding and/or hemodynamic instability.
Weak recommendation, low-quality evidence. (Strongly agree: 73.1%; agree: 23.1%)
In patients with ongoing severe bleeding or hemodynamic instability, SEED recommends withholding vitamin K antagonists and administering intravenous vitamin K and prothrombin complex concentrate (PCC), or fresh frozen plasma if PCC is not available.
Strong recommendation, low-quality evidence. (Strongly agree: 67.9%; agree: 28.6%)
SEED recommends consulting a local hematologist before using reversal agents in patients on direct oral anticoagulants who are experiencing ongoing delayed post-polypectomy bleeding and hemodynamic instability.
Strong recommendation, low-quality evidence. (Strongly agree: 60.7%; agree: 35.7%)
Anticoagulation reversal should be reserved for patients with ongoing severe bleeding and/or hemodynamic instability, as it may increase the risk of thromboembolic events. 70 Since the publication of the British Society of Gastroenterology, American College of Gastroenterology, and ESGE guidelines,26,34,37,71 no relevant studies specifically focused on patients with DPPB and vitamin K antagonists (VKAs) have been published to justify statements differing from other sources on LGIB.
Regarding the reversal of direct oral anticoagulants (DOACs), four observational studies have been conducted from 2019 to the present.72–75 Three studies assessed the effectiveness of Idarucizumab in patients on dabigatran therapy,72–74 two of which involved patients with gastrointestinal bleeding.73,74 The third study did not specify whether gastrointestinal bleeding was included. 72 Finally, one sub-analysis of the ANNEXA-IV cohort evaluated the yield of Andexanet alpha in patients with gastrointestinal bleeding. 75 All studies were observational, lacked a control group, and did not specify if bleeding was a result of prior polypectomy. Therefore, these studies do not provide sufficient evidence to support or refute the recommendation of Idarucizumab or Andexanet alfa. Although no specific evidence supports hematology consultation in this scenario, we recommend it as a precautionary measure due to the high cost, potential risks, and the limited familiarity that other specialists may have with reversal agents.
Anticoagulation resumption
SEED recommends resuming anticoagulants (vitamin K antagonists and direct oral anticoagulants) following delayed post-polypectomy bleeding in patients with an indication for anticoagulation.
Strong recommendation, low-quality evidence. (Strongly agree: 82.1%; agree: 17.9%)
SEED suggests resuming anticoagulants following delayed post-polypectomy bleeding on day 7 or earlier. In patients with high thrombotic risk, anticoagulation should preferably be restarted within 72 hours, provided hemostasis has been achieved.
Weak recommendation, very low-quality evidence. (Strongly agree: 57.7%; agree: 34.6%)
Overall, current recommendations suggest resumption within 7 days of the bleeding event, given the overall benefit in reducing thromboembolic complications.34,37,71 According to the ESGE, it is reasonable to restart warfarin and DOACs from day 7 onwards in patients at low thrombotic risk after interruption. In those with high thrombotic risk, heparin bridging is recommended, preferably within 72 h. 26
The benefits of resuming anticoagulation after hospitalization for LGIB are well established. In a large cohort study of patients experiencing diverticular hemorrhage, discontinuation of anticoagulation was associated with an increased risk of cardiovascular events, while no significant association was found with rebleeding. 51 Similarly, in a cohort of patients with atrial fibrillation, resumption of VKAs or DOACs following LGIB was linked to a lower hazard of all-cause mortality and a comparable risk of recurrent gastrointestinal bleeding compared to those who did not resume anticoagulation. 76 An analysis of patients from the UK audit of LGIB on anti-thrombotics revealed that rebleeding rates in the DOAC (20.0%) and warfarin groups (23.1%) were similar to the unexposed group (19.4%). The use of DOACs or warfarin was not associated with either in-hospital rebleeding or hospital readmission due to further bleeding. 56 Moreover, an analysis of 150 patients with LGIB while on anticoagulants found that resumption of anticoagulation was not associated with recurrent bleeding. When the cohort was expanded to include all patients with GIB, resumption of anticoagulation was linked to a reduced risk of ischemic events during follow-up and decreased mortality. 47
On the other hand, a meta-analysis of observational studies demonstrated that resuming anticoagulation after hospitalization for GIB is associated with a reduction in thromboembolic events and mortality, though it may potentially increase the risk of recurrent GIB. 77 Another cohort study found that resumption of anticoagulant therapy was associated with a lower risk of thromboembolism but a higher risk of bleeding, regardless of the timing. 78 The risk of recurrent bleeding was particularly higher in patients with previous bleeding, lower glomerular filtration rate, and those with an index major bleeding event.
Bowel preparation
SEED suggests the use of oral preparation over enemas or no preparation in the case of suspected delayed post-polypectomy bleeding originating in the right, transverse, or descending colon.
Weak recommendation, low-quality evidence. (Strongly agree: 57.1%; agree: 32.1%)
SEED suggests that rectosigmoidoscopy may be performed without oral preparation, preferably with enemas, in delayed post-polypectomy bleeding from rectosigmoid lesions.
Weak recommendation; very low-quality evidence. (Strongly agree: 39.3%; agree: 53.9%)
The absence of RCTs and meta-analyses limits the comparison of colon preparation methods for DPPB. Current guidelines support oral preparation for LGIB, 26 which is associated with substantial improvement in the cecal intubation rate.79,80 Recent data suggest that lower-volume preparation (2 l) may improve tolerance without compromising the detection of the bleeding site or cecal intubation.81–83 Without prior preparation, the risk of intestinal perforation is increased. 80 Comparative studies report that polyethylene glycol (PEG) preparation has a superior diagnostic rate of bleeding focus (97.1% vs 84%, p = 0.008) and fewer repeat colonoscopies (18.8% vs 44.0%, p < 0.001) compared to enemas. 84
The rationale for omitting bowel preparation is based on the fact that colonoscopy is performed shortly after polypectomy in distal locations, and the laxative effect of the presence of blood may sufficiently clean the colon. 10 Single-center studies explore the role of colonoscopy without preparation in LGIB.10,79,85,86 Indeed, data exist for patients with DPPB who did not receive preparation before colonoscopy. 87 The cecal intubation rate was 74% in patients prepared with PEG compared to 41% in patients prepared with enema or without preparation, although therapeutic intervention was possible in 15 out of 16 patients with DPPB. 87 Another retrospective analysis included 69 patients with DPPB from polyps <1 cm and no oral preparation. Colonoscopy was successfully performed in all patients with no AEs, and hemostasis was achieved in 90% of patients. 10 The absence of oral preparation has also been proposed for patients admitted to the intensive care unit or elderly patients to avoid associated discomfort.79,86 Only in cases of single or low-number distal polypectomies (rectum or sigmoid), preparation with enema or without preparation may be considered.10,84,87
SEED recommends using a preparation based on polyethylene glycol (PEG) for colonoscopy following delayed post-polypectomy bleeding. Both high (4 liters) or low volume (2 liters or 1 liter) PEG-based regimens are acceptable.
Strong recommendation, low-quality evidence. (Strongly agree: 53.8%; agree: 38.5%)
Preparations based on PEG 4 l, and more recently PEG 2 l or PEG 1 l, have been tested according to dosage and timing of the usual preparation intake.81,88 An RCT in patients with LGIB found that low-volume bowel preparation showed the same efficacy and tolerability with better satisfaction compared with high volume, 88 but no direct comparisons of different PEG volumes exist for DPPB patients. Attention should be paid to avoid broncho-aspiration, especially in high-risk situations such as low consciousness levels. We do not recommend administering oral laxatives rapidly, as this increases the risk of AEs. Placement of a nasogastric tube may improve tolerance in patients with limited oral intake and at low risk of aspiration. 26 No evidence exists for sodium picosulfate/magnesium citrate preparations in DPPB.
Colonoscopy indication
SEED suggests against the routine use of colonoscopy for all patients with delayed post-polypectomy bleeding.
Weak recommendation, very low-quality evidence. (Strongly agree: 65.4%; agree: 34.6%)
SEED suggests performing risk stratification to identify patients with delayed post-polypectomy bleeding who are most likely to benefit from a colonoscopy.
Weak recommendation, very low-quality evidence. (Strongly agree: 57.7%; agree: 42.3%)
In hemodynamically stable patients, SEED suggests an initial observation period (6–24 hours) to assess spontaneous bleeding cessation.
Weak recommendation, very low-quality evidence. (Strongly agree: 57.7%; agree: 42.3%)
SEED suggests performing a colonoscopy in cases of hourly rectal bleeding or when 3 or more risk factors for active bleeding and/or therapeutic intervention are present (hemodynamic instability at admission, hemoglobin drop ⩾2 g/dL, need for blood transfusion, active antithrombotic use, American Society of Anesthesiologists score ⩾ III)
Weak recommendation, very low-quality evidence. (Strongly agree: 42.3%; agree: 50%)
SEED suggests performing a colonoscopy in patients with persistent bleeding 24 hours after onset, regardless of the presence of risk factors for active bleeding and/or therapeutic intervention.
Weak recommendation, very low-quality evidence. (Strongly agree: 61.5%; agree: 34.6%)
The decision to perform an invasive procedure is typically based on clinical variables at admission and the physician’s expertise. In the systematic literature search conducted for this consensus statement, we did not identify any meta-analysis or RCT evaluating the benefit of colonoscopy in patients with DPBB. Descriptive studies examining the role of colonoscopy are summarized in Supplemental Material. The main finding of these studies, primarily retrospective, is that the rate of repeat colonoscopy ranges from 28% to 100%, although no therapeutic intervention is performed in 0%–63% of cases. Among patients who did not receive an endoscopy and those who underwent colonoscopy without hemostatic intervention, 3%–12% and 0%–20% experienced rebleeding, fewer than 10% were readmitted, and none died during the 30-day follow-up. Therefore, the evidence suggests that a substantial number of patients can be managed without intervention with a low risk of adverse outcomes.2,7–10,12,18,84,89–92
A consensus algorithm based on the available evidence and expert opinion for the management of DPPB is provided in Figure 3. For hemodynamically unstable patients following initial resuscitation, a computed tomography angiography (CTA) scan is preferred, with radiologic embolization if active bleeding is detected. If no active bleeding is identified, a colonoscopy is recommended.

Proposed management of delayed post-polypectomy bleeding.
For hemodynamically stable patients, given that bleeding often ceases spontaneously and a significant proportion of patients with DPPB can be managed without a colonoscopy, the spontaneous cessation of bleeding is considered the primary clinical criterion for decision-making. To develop the algorithm, we reviewed the literature to identify factors associated with active bleeding and/or therapeutic intervention (Table 1). Relevant predictors were selected based on their consistency (presence in at least two studies), reproducibility, low interobserver variability, solid pathophysiological basis, and high magnitude of effect (OR > 1.5–2.0). The expert panel determined that hourly rectal bleeding is the most significant criterion for guiding decisions due to its indication of high-volume bleeding. The definition of hourly hematochezia is not well established in the literature.7,9 By consensus, the panel agreed that it should be defined as persistent hematochezia occurring at least once per hour for a period of at least 4 h, which clinically indicates significant bleeding. In addition, hemodynamic instability at admission, a hemoglobin drop of ⩾2 g/dL, need for blood transfusion, antithrombotic use, and American Society of Anesthesiologists score ⩾ III were deemed relevant predictors for the need for colonoscopy.
Predictors of active bleeding, need for therapeutic intervention, or moderate/severe bleeding.

ASA, American Society of Anesthesiologists; CI, confidence interval; DPPB, delayed post-polypectomy bleeding; Hb, hemoglobin; OR, odds ratio; p, p value; RR, risk ratio.
To date, only two logistic predictive models have been developed. The first model, originating from Australia, was not subjected to internal validation, and no information regarding its calibration or discrimination properties was provided. 7 The second model, developed in Spain, underwent internal validation through bootstrapping; however, it has yet to undergo external validation. 8 This model provides estimates of the probability of active bleeding. In this study, patients exhibiting three or more risk factors had a predicted probability of >25% of active bleeding, which justified the panel’s recommendation for colonoscopy even in the absence of hourly hematochezia, as this subgroup is likely to benefit from the procedure. For hemodynamically stable patients without a high probability of requiring endoscopic hemostatic intervention (i.e., <3 risk factors), and given the uncertain calibration of the model in real settings, a 24-h timeframe to assess bleeding cessation appears reasonable. 9 If bleeding persists or recurs, a colonoscopy is recommended.
Colonoscopy timing
SEED suggests that for patients requiring colonoscopy, it should be performed at some point during their hospital stay, preferably during the first available non-urgent time slot. The timing of the colonoscopy should be adapted to available resources and the patient’s clinical condition.
Weak recommendation, very low-quality evidence. (Strongly agree: 39.3%; agree: 53.6%)
There is no high-quality evidence demonstrating the benefit of early colonoscopy (within the first 24 h after admission). 26 We did not find any study showing a difference in clinical outcomes based on the timing of colonoscopy in patients with DPPB. Therefore, the most logical advice seems to follow established recommendations for LGIB. According to current guidelines, patients with major LGIB should undergo a colonoscopy at some point during their hospital stay.26,34 Subsequent published cohorts are consistent with these recommendations.93,94
Hemostatic therapy
Indication for hemostatic treatment
SEED suggests applying an endoscopic hemostatic treatment when there is active bleeding at the post-polypectomy ulcer. Further evidence is needed to clarify the prognostic value and to establish the management of other endoscopic stigmata of recent bleeding.
Weak recommendation, very low-quality evidence. (Strongly agree: 38.5%; agree: 53.8%)
No studies have been conducted to evaluate the benefit of applying a hemostatic endoscopic treatment according to the stigmata found at the post-polypectomy ulcer. Some authors advocate treating only active bleeding,6,95,96 while others propose managing additional bleeding stigmata (visible vessel, adhered clot) to reduce the rebleeding rates, similar to Forrest’s classification.7–10,97 Most articles published are retrospective, with several methodological limitations.
The largest study conducted to date evaluates a cohort of 548 patients with DPPB. Among 394 patients managed by colonoscopy, the bleeding point was located in 87.3% of cases: 74 (21.5%) presented with active bleeding, 161 (46.8%) with a visible vessel-adherent clot, and 109 (31.7%) with fibrin-hematin. Endoscopic hemostatic therapy was performed in all lesions with active bleeding, most scars with a visible vessel-adherent clot (97.5%), and half of the ulcers with fibrin-hematin. Despite this management, 47 patients rebled within the first 30 days with no differences observed in the rebleeding rate stratified by ulcer stigmata. Rebleeding rates were lower in patients not receiving a colonoscopy (or radiologic/surgical intervention) and also in those who were not treated during re-colonoscopy. 8
In another retrospective study (N = 69), colonoscopy was performed in 100% of patients, and all but three received endoscopic treatment whether they had active bleeding, visible vessel/adhered clot, or hematin. Greater rebleeding rates were found in patients presenting active bleeding or visible vessel/clot compared with hematin. 10
Another multicentric study analyzed 42 cases of DPPB. A second colonoscopy was performed in almost three-quarters of patients and hemostatic therapy was applied in 100% of patients with active bleeding (n = 6), in 80% with stigmata of recent bleeding (n = 16), while individuals with clean ulcer base or not visualized did not receive any therapy. Rebleeding rates were similar among patients receiving endoscopic hemostatic therapy and those left untreated, without significant differences between groups with and without active bleeding. 9
Ultimately, the ulcer’s assessment and the decision to apply a hemostatic approach rely on the endoscopist. Moreover, the interpretation of the culprit ulcer is operator-dependent. In addition, visible vessels and adherent clots are considered into the same category, and visible vessels can be part of a normal post-polypectomy mucosal defect, which complicates the establishment of a stigmata-based therapeutical decision. Applying an endoscopic treatment when there is active bleeding seems logical, but further evidence is needed to clarify the prognostic value and management of visible vessels, adherent clots, pigmented spots, or clean bases.
First-line and rescue hemostatic treatment
SEED recommends the use of mechanical therapy as first-line treatment in patients with delayed post-polypectomy bleeding, either as mono- or combined therapy. Thermal and/or injection therapy may also be used, depending on the endoscopist’s preference and the devices available.
Strong recommendation, low-quality evidence. (Strongly agree: 69.2%; agree: 30.8%)
SEED suggests the use of hemostatic powders, mechanical, thermal, and/or injection therapy as rescue treatment, either as mono- or combined therapy, regardless of the approach used during the previous colonoscopy.
Weak recommendation, low-quality evidence. (Strongly agree: 57.7%; agree: 38.5%)
The current scientific evidence evaluating endoscopic treatments for use as first-line or rescue therapy is insufficient to rank these treatments from an evidence-based approach.
In terms of first-line endoscopic treatment in patients with DPPB, the most used devices are through-the-scope clips (TTSCs), which have resulted in very high bleeding arrest rates (between 93% and 100%) and acceptable rebleeding rates (up to 12%).9,11,92,97–108 Although deploying a TTSCs in a post-polypectomy scar can be challenging due to induration, compared to an ulcer immediately after polypectomy, this device is well known and widely available. The largest cohort of patients treated with TTSC alone (N = 185) describes an initial hemostasis rate of 100% and an incidence of rebleeding of 10%. 12 Thermal therapy includes both contact modalities (e.g., heater probe, hemostatic forceps, snare-tip coagulation) and non-contact modalities (e.g., argon plasma coagulation). It has been described as an effective monotherapy but carries a risk of transmural injury, so caution is warranted. Sclerosants with or without adrenaline, as well as hemostatic powders, could also be used as first-line treatments.11,92,106,109,110
For combined therapy, sclerosis, thermal therapy, and TTSCs have been used together with good results.9,97,100,111–113 In the largest DPPB cohort, monotherapy and combined therapy were used in similar proportions, with the most common combination being sclerotherapy and TTSCs. 8
Rescue treatment modalities have been scarcely reported. TTSCs and over-the-scope clips have shown very high hemostatic rates, with the latter option covering a wider area.11,99,112 Thermal therapy, hemostatic powders, and band ligation of pedunculated polyps are also viable alternatives for rebleeding.97,109,111 Each of these therapeutic options can be employed regardless of the approach taken during the previous colonoscopy. Currently, there is no published evidence supporting the use of hemostatic gels in the management of DPPB, further research is needed before incorporating these agents into clinical practice.
Radiology
Diagnosis
SEED suggests performing CT-angiography in patients with delayed post-polypectomy bleeding experiencing hemodynamic instability or when colonoscopy has failed to control the bleeding, before radiological or surgical treatment.
Weak recommendation, very low-quality evidence. (Strongly agree: 57.7%; agree: 30.8%)
SEED recommends against the use of other radiological studies (e.g., conventional angiography or red blood cell scintigraphy) due to their limited accuracy in identifying the bleeding origin and their low availability.
Strong recommendation, very low-quality evidence. (Strongly agree: 57.7%; agree: 30.8%)
No studies evaluating the diagnostic accuracy of CTA in the detection of bleeding in patients after an index polypectomy have been conducted. Extrapolating data from other sources of LGIB, CTA has a sensitivity and specificity of 90% and 92%, respectively. 114
Unlike LGIB secondary to diverticulosis or vascular malformations, where identifying the bleeding origin can be challenging, the location of the DPPB is usually determined by previous colonoscopy. Nevertheless, CT-angiography provides information on the distinction between arterial and venous bleeding and plays a role in the evaluation of the vascular supply prior to selective arterial embolization. Thus, the cecum and ascending and transverse colon are part of the territory of the superior mesenteric artery, while the splenic flexure, descending and sigmoid colon, and rectum correspond to the inferior mesenteric artery. This scheme is generalized, and depending on the patient, there may be variants that make it more complex to establish the vascular supply of the scar. 115 Thus, CTA is useful prior to angiographic embolization when colonoscopy has failed due to massive bleeding, poor bowel preparation, or in hemodynamically unstable patients.2,116
Although conventional angiography could also show the vascular anatomy, it is an invasive procedure requiring higher doses of radiological contrast, is not widely available, and has less sensitivity than CTA. 117 Therefore, angiography is usually performed prior to embolization and after a positive CTA result. Red blood cell scintigraphy has no role in DPPB due to its low availability and greater complexity compared to CTA.
Treatment
SEED suggests that angiographic embolization could be of choice in patients with delayed post-polypectomy bleeding either with hemodynamic instability or when colonoscopy has failed to control the bleeding, provided that active bleeding is confirmed by computed tomography angiography.
Weak recommendation, very low-quality evidence. (Strongly agree: 53.8%; agree: 38.5%)
SEED suggests that angiographic embolization should be elected ahead of surgical treatment in patients with delayed post-polypectomy bleeding either with persistent hemodynamic instability after initial resuscitation or when colonoscopy has failed to control the bleeding
Weak recommendation, very low-quality evidence. (Strongly agree: 46.4%; agree: 53.6%)
The evidence evaluating angiographic embolization to control DPPB is limited, with no direct comparisons between radiological intervention and endoscopy or surgery. Considering its greater complexity with the associated risk of colonic ischemia around 1%–4%,118,119 this technique should be reserved for those patients presenting hemodynamic instability or when colonoscopy has failed. Morbidity and mortality are likely higher in patients with LGIB undergoing surgery, 37 making angiographic embolization preferable to surgery in these cases.
According to the largest cohort of individuals presenting with DPPB, 2.6% of patients required primary angiographic embolization. 8 However, this approach can also be used as rescue therapy in patients experiencing rebleeding.2,7,8 The technical success rate regarding embolization in patients with DPPB is nearly 100% and the clinical success ranges from 93% to 100%, without major AEs.2,7,8,120–123 Nonetheless, its success is related to the time elapsed between CTA and arterial embolization, and <90 min has been highly associated with detection of extravasation in patients with LGIB. 124
Endovascular interventions to control bleeding traditionally included intra-arterial vasopressin infusion and mechanical occlusion of the culprit arterial vessel; however, the first option has been replaced by the second one, due to its higher complexity. 125 There are different materials to perform embolization (gelatin sponges, cyanoacrylate glue, ethylene or polyvinyl alcohol, and microcoils), and the choice will rely on professional experience and local availability. During arterial embolization, it is essential to determine if extravasation is present to reduce the risk of subsequent ischemia, which is related to the inability to achieve super-selective embolization.116,118,119,126 In addition, despite the absence of extravasated contrast, embolization could be conducted based on CTA findings considering the risk of AEs and the patient’s context. Accordingly, TTSCs placed during the previous endoscopy could help with identifying the bleeding location and the vessel supply, even if no extravasation is observed.
Surgery
SEED suggests that surgery should only be undertaken in patients with refractory delayed post-polypectomy bleeding when endoscopic or radiological treatment has failed, and always based on local expertise and case-by-case assessment.
Strong recommendation, very low-quality evidence. (Strongly agree: 84.6%; agree: 15.4%)
The proportion of patients experiencing DPPB requiring surgery varies widely, ranging from 0.2% to 10%.2,7,8,18,127–134 This variability in incidence can be attributed to heterogeneity in study designs, differences in the definition of DPPB (where immediate and delayed bleeding are not always distinguished), and timeframes ranging from 14 to 30 days post-procedure.
Up to now, neither RCTs nor non-randomized interventional studies have directly assessed the effectiveness of surgery as a therapeutic option compared to radiological, endoscopic, or conservative approaches. Proposing an RCT to compare the efficacy of surgery with more conservative therapeutic approaches seems illogical and ethically questionable. Given the widespread availability of less invasive alternatives like endoscopic and endovascular radiological interventions and the higher mortality associated with surgery, it is rational to reserve surgery for cases where less aggressive options have failed.
Regarding the surgical approach, segmental colectomy is the most commonly used technique, due to its lower morbidity and mortality compared to extended colectomy. 135 However, in patients experiencing massive bleeding with multiple post-polypectomy ulcers in whom identifying the responsible one proves challenging after imaging tests or even after an on-table colonoscopy in the operating room, subtotal colectomy seems to be a reasonable option. 130 Considering recent surgical advances of the last few years, laparoscopy emerges as the preferred surgical approach over laparotomy, contingent upon individual experience and the patient’s medical status. On an alternative note, transanal surgery is a safe and effective option for managing rectal bleeding. 2 This more conservative surgical approach could even be an alternative to endoscopy in rectal ulcers, although it depends on local expertise. Finally, surgery may be warranted in managing AEs arising from the endoscopic and interventional radiological treatment of DPPB, such as endoscopic perforation or ischemia after radiological embolization.18,131
Conclusion
This consensus statement provides an evidence-based framework for managing DPPB, addressing a critical gap in current guidelines. The recommendations emphasize individualized care, optimizing healthcare resources, and reducing unnecessary colonoscopies. Given that many cases of DPPB are self-limited, a risk-stratified approach can minimize unnecessary procedures while ensuring timely intervention for high-risk patients.
However, these recommendations rely largely on expert consensus due to limited high-quality evidence, and the proposed algorithm has yet to undergo external validation, which may affect its generalizability. In addition, a formal cost analysis is lacking, though reducing unnecessary colonoscopies could improve resource utilization. To validate these recommendations, we are conducting the COLOHEM-II study, a prospective evaluation of the proposed algorithm. Future research should focus on validating risk stratification models, comparing hemostatic therapies, and integrating cost-effectiveness analyses. Despite these limitations, this consensus represents a key step toward standardizing DPPB management, providing a structured and practical guide for clinicians.
Supplemental Material
sj-docx-1-tag-10.1177_17562848251329145 – Supplemental material for Delphi consensus statement for the management of delayed post-polypectomy bleeding
Supplemental material, sj-docx-1-tag-10.1177_17562848251329145 for Delphi consensus statement for the management of delayed post-polypectomy bleeding by Enrique Rodríguez de Santiago, Sandra Pérez de la Iglesia, Diego de Frutos, José Carlos Marín-Gabriel, Carolina Mangas-SanJuan, Raúl Honrubia López, Hugo Uchima, Marta Aicart-Ramos, Miguel Ángel Rodríguez Gandía, Eduardo Valdivielso Cortázar, Felipe Ramos Zabala, Marco Antonio Álvarez, Marina Solano Sánchez, Jesús Manuel González Santiago, Eduardo Albéniz, Gonzalo Hijos-Mallada, Nerea Castro Quismondo, Miguel Fraile-López, David Martínez Ares, Javier Tejedor-Tejada, Luis Hernández, Joan B. Gornals, Sergi Quintana-Carbo, Juan Ocaña, João A. Cunha Neves, Juan Martínez Martínez, María López-Cerón Pinilla, Carlos Dolz Abadía and María Pellisé in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
None.
Authorship statement
Guarantor of the article: Enrique Rodríguez de Santiago assumes responsibility as the guarantor of the article and takes full accountability for the integrity of the work as a whole, from its inception to the final published version.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
