Abstract
Background:
Endoscopic submucosal dissection (ESD) is the first-line treatment for gastric mucosal lesions. The artificial ulcers caused by ESD can lead to bleeding, perforation, and other adverse events.
Objectives:
This study aimed to evaluate the efficacy of alternate mucosa–submucosa clip in preventing adverse events following ESD for gastric mucosal lesions.
Design:
Multicenter retrospective observational study.
Methods:
Data from 924 patients who underwent ESD for gastric mucosal lesions were retrospectively collected and categorized into completely closed and unclosed groups based on postoperative wound management. The incidence of adverse events and postoperative wound healing rate at 1 and 3 months were compared between the groups, and the factors related to delayed bleeding after ESD were analyzed.
Results:
The rate of delayed hemorrhage was lower in the completely closed group than in the unclosed group (0.8% vs 7.6%; p < 0.001), and subgroup analysis showed that this effect was consistent across all subgroups. The postoperative abdominal pain score was lower in the completely closed group than in the unclosed group (0 vs 2, p < 0.001). One month postoperatively, the wound healing rate was higher in the completely closed group (77.6% vs 55.1%; p < 0.001). Multivariable analysis showed that the presence of ulcers or scars on the lesion surface, and lesions located in the lower one-third of the stomach were risk factors for postoperative bleeding.
Conclusion:
Application of alternate mucosa–submucosa clip closing the wound of ESD reduced the risk of postoperative bleeding, alleviated postoperative abdominal pain, and promoted artificial wound healing.
Introduction
Endoscopic submucosal dissection (ESD) is the first-line treatment for early gastric cancer and precancerous lesions and has the advantages of faster recovery and preservation of the digestive tract.1–3 However, artificial ulcers caused by ESD can lead to bleeding, perforation, and other adverse events, among which bleeding is the most frequent. Moreover, severely delayed postoperative bleeding can lead to hemorrhagic shock.4,5 A Japanese study demonstrated that a mucosal defect of >3 cm after gastric ESD was a risk factor for delayed hemorrhage. 6 Hammad et al. reported that preventive clip closure of postoperative mucosal defects in colonic lesions reduced the rate of delayed bleeding by 1.8% in 463 patients. 7 However, few studies have focused on the use of clips to prevent delayed hemorrhage after ESD for gastric mucosal lesions. In clinical practice, mucosal defects <1 cm in diameter can be completely closed using clips, whereas mucosal defects >2 cm in diameter are difficult to close. Globally, experts have explored several methods such as endoscopic hand suturing (EHS), 8 O-ring blocking and ligation, 9 and “accordion” folding to close large postoperative gastric ESD wounds, 10 to minimize postoperative adverse events. Over the past decade, our center has applied the alternate mucosa–submucosa clip method for complete closure of artificial wounds after gastric ESD. In this study, we retrospectively collected data from patients with early gastric cancer and precancerous lesions who underwent ESD to evaluate the efficacy of the alternate mucosa–submucosa clip closure method in preventing adverse events after ESD.
Methods
Study design
This quality research project was constructed as a retrospective observational study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 11
Patients
This multicenter retrospective study enrolled 924 patients with gastric mucosal lesions who underwent ESD at 3 hospitals in China between January 2013 and December 2023. The study cohort included 562 patients from Zhongda Hospital Southeast University, 264 from the Second Affiliated Hospital Zhejiang University School of Medicine, and 98 from the Affiliated Changshu Hospital of Nantong University.
The inclusion criteria were: (1) Diagnosis of gastric mucosal lesions, including low-grade and high-grade intraepithelial neoplasia, and gastric carcinoma in situ, by gastroscopy before ESD and pathology after ESD; (2) single lesion with a specimen diameter of ⩾2 cm; and (3) availability of complete clinical, endoscopic, and pathological data. The exclusion criteria were: (1) Incomplete data; (2) presence of lymphovascular tumor embolus, lymph node metastasis, or distant metastasis; (3) serious bleeding tendency; (4) long- or short-term use of antiplatelet or anticoagulant drugs; and (5) wound following ESD sprayed with hemostatic powder, fibrin glue, and other medications. All patients underwent magnified gastroscopy and ultrasonic endoscopy to determine lesion depth before ESD. The patients were informed of the relevant risks of ESD and signed an operative consent form. The gastric ESD procedures were performed by expert endoscopists who had performed more than 500 cases of gastric lesions.
Based on the management of wounds after gastric ESD, the enrolled patients were divided into two groups: Completely closed (postoperative wound was completely closed using the alternate mucosa–submucosa clip method) and unclosed (postoperative wound was not closed using clips) groups.
Definitions of terms
In this study, delayed hemorrhage refers to bleeding that occurs within 24 h to 30 days after ESD and meets at least two of the following indicators: (a) Vomiting blood, black stools, dizziness, and other uncomfortable symptoms; (b) drop in hemoglobin >20 g/L; (c) drop in blood pressure >20 mmHg or increase in heart rate >20 beats/min; and (d) gastroscopy suggesting bleeding. Delayed perforation was defined as sudden postoperative symptoms of peritoneal irritation, postoperative abdominal plain films, or computed tomography (CT) suggesting the presence of free gas. Postoperative stenosis refers to a postoperative scar after ESD resulting in the narrowing of the digestive tract lumen. Postoperative abdominal pain was scored using a numeric rating scale ranging from 0 to 10 to indicate the degree of pain, with 0, 1–2, 3–5, and ⩾6 indicating no, mild, moderate, and severe pain, respectively. Patients selected a number from 0 to 10 to indicate their pain level according to their most genuine pain feelings, which were recorded by the nurse on the postoperative day. For patients with multiple pain scores, the highest score was selected. Wound healing rate was calculated by the percentage of ulcers that reached the healing stage endoscopically out of the total number of people that underwent gastroscopic follow-up.
Methodology
Data collection
Data related to lesion and endoscopic characteristics were collected, including endoscopic lesion characteristics, postoperative pathologic characteristics, and intraoperative indicators. According to the Japanese Gastric Cancer Association classification criteria, the stomach was vertically divided into the upper one-third (U), middle one-third (M), and lower one-third (L) regions. Endoscopic features included lesion morphology (elevated, flat, depressed) and the presence of ulcers or scars on the lesion surface.
ESD procedure
Main instruments
Olympus GIF-Q290J therapeutic gastroscope (Olympus Corp., Tokyo, Japan), Olympus transparent cap (D-201-11804), Alton injection needle (Alton Medical Instruments, Shanghai, China), Olympus KD-650L knife, Olympus FD-412LR hot biopsy forceps, sureclip (SDNROCC-D-26-195-C, SDNROCC-F-26-195-C; Nanjing Microtech, Nanjing, China), and Erbe VIO 300D high-frequency electrosurgical workstation (Erbe Elektromedizin, Tübingen, Germany).
Main steps
Before treatment, narrow-band light imaging and magnifying endoscopy were performed to determine the lesion boundary, and argon plasma coagulation was used to mark the point 0.5 cm from the edge of the lesion.
Submucosal injection of 1:10,000 epinephrine saline, containing methylene blue, was administered to sufficiently lift the lesion.
The mucosa surrounding the lesion was incised and progressively separated along the outer ring circumference of the marking point using a KD-650L knife. After complete lesion excision, the visible vessels in the artificial wound were routinely coagulated using hemostatic forceps.
Completely closed group
After prophylactic electrocoagulation of the artificial wound using hot hemostatic forceps, the wound was completely closed using an alternate mucosa–submucosa clip method. First, one side of the wound was apically anchored using a clip. Subsequently, the second clip closed one side of the mucosa and the postoperative wound musculature on one side to reduce the wound size. The third clip closed the contralateral mucosa and muscularis externa to further reduce the wound size. The above steps were repeated until the width of the wound was less than the maximum diameter of the clips after release. Finally, the wound was completely closed with a clip-to-mouth between the clips (Figure 1).
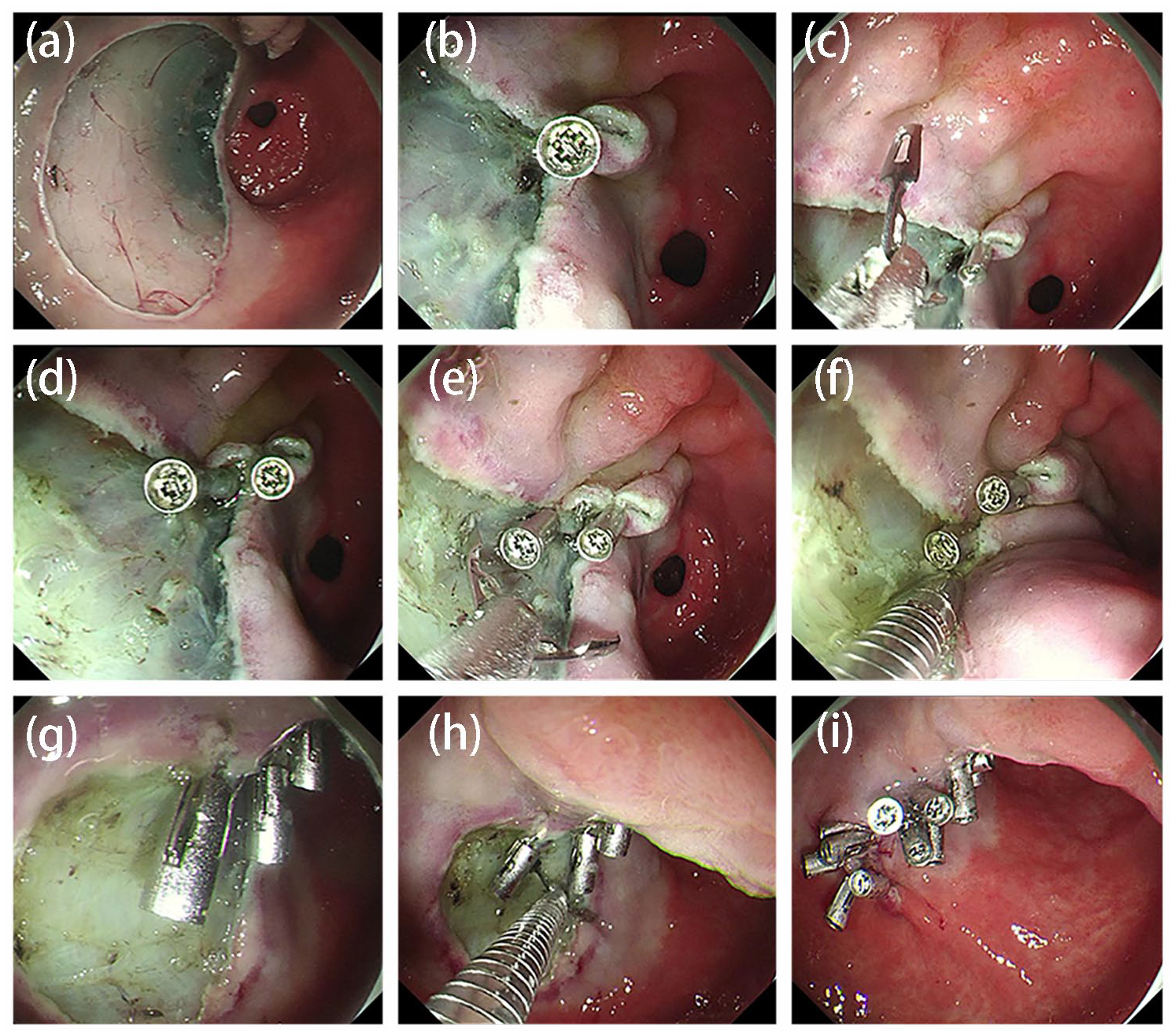
(a) Gastric ESD postoperative wound; (b) one side of the artificial wound is apically anchored using a clip; (c, d) the second clip closes one side of the mucosa and the postoperative wound musculature on one side to reduce the wound size; (e) the third clip closes the contralateral mucosa and muscularis externa to further reduce wound size; (f–i) the aforementioned steps are repeated until the width of the wound is less than the maximum diameter of the clips after release; finally, the wound is completely closed with a clip-to-mouth between the clips.
Unclosed group
After prophylactic electrocoagulation of the artificial wound using hot biopsy forceps, the wound was not treated with clips.
Postprocedural management
The patients were on bed rest; fasted for 24–48 h after ESD; routinely treated with acid-suppressing drugs. The patients were closely monitored for abdominal pain, blood vomiting, black stools, and other adverse events after ESD and were examined with abdominal radiography or CT, if necessary.
Outcome assessed
The primary outcome was the rate of delayed hemorrhage. The secondary outcomes were the rate of delayed perforation, stenosis, perforation, abdominal pain score, and 1- and 3-month wound healing rates afterward ESD.
Follow-up
Gastroscopy was performed at 1 and 3 months after ESD to observe the healing of the artificial wounds. The endoscopic staging of peptic ulcers was categorized into the active (A1, A2), healing (H1, H2), and scarring (S1, S2) stages.
Statistical analyses
SPSS (version 27.0; IBM, Armonk, NY, USA) software, was used for statistical analyses. Measured data are expressed as means ± standard deviations for normal distributions and were compared using independent samples t test for comparisons between the two groups. Skewed distribution quantitative data are expressed as median (interquartile range (IQR)) and assessed using the Mann–Whitney U test for comparisons between the two groups. Categorical data are expressed as frequencies (%) and were compared between the groups using the Pearson χ2 test, corrected χ2 test, or Fisher’s exact probability method. Stratified analysis was analyzed by logistic regression analysis. Risk factors for postoperative bleeding after gastric ESD were analyzed using logistic regression analysis. p < 0.05 was considered statistically significant.
Results
Patient characteristics
A total of 924 patients fulfilled the inclusion criteria, including 479 patients in the completely closed group and 445 patients in the unclosed group. Most patients were male (69.3%) in the completely closed group with a median age of 65 (IQR, 57–72) years. In the unclosed group, there were 317 male patients (71.2%) with a median age of 66 (IQR, 58–71) years. The smoking history, alcohol consumption, and comorbidities of the two groups are presented in Table 1.
Baseline characteristics of patients.

Characterization of endoscopic lesions and postoperative pathological features
In the completely closed group, the lesions were mainly located in the lower one-third of the stomach (39.9%), with predominantly elevated lesions (45.1%). Ulcers or scars were present on the lesion surface in 84 (17.5%) patients. However, the lesions in the unclosed group were located primarily in the lower one-third of the stomach (164 (36.9%) patients), and the distribution of lesion location significantly differed between the two groups (p = 0.031). The size of the specimens in the completely closed group was similar to that in the unclosed group (38 (30–48) and 40 (30–50) mm, respectively; p = 0.514). The postoperative pathological types in the completely closed group were low-grade dysplasia in 214 (44.7%) patients, high-grade dysplasia in 94 (19.6%), and gastric carcinoma in 171 (35.7%), with the lesion mainly infiltrating the epithelial layer (57.0%). No significant differences were evident between the groups in terms of lesion morphology (p = 0.504), ulcers or scars present on the lesion surface (p = 0.407), pathological type (p = 0.058), depth of infiltration (p > 0.05), and the horizontal positivity rates (p = 0.943; Table 2).
Comparison of lesion characteristics and postoperative pathological features between groups.
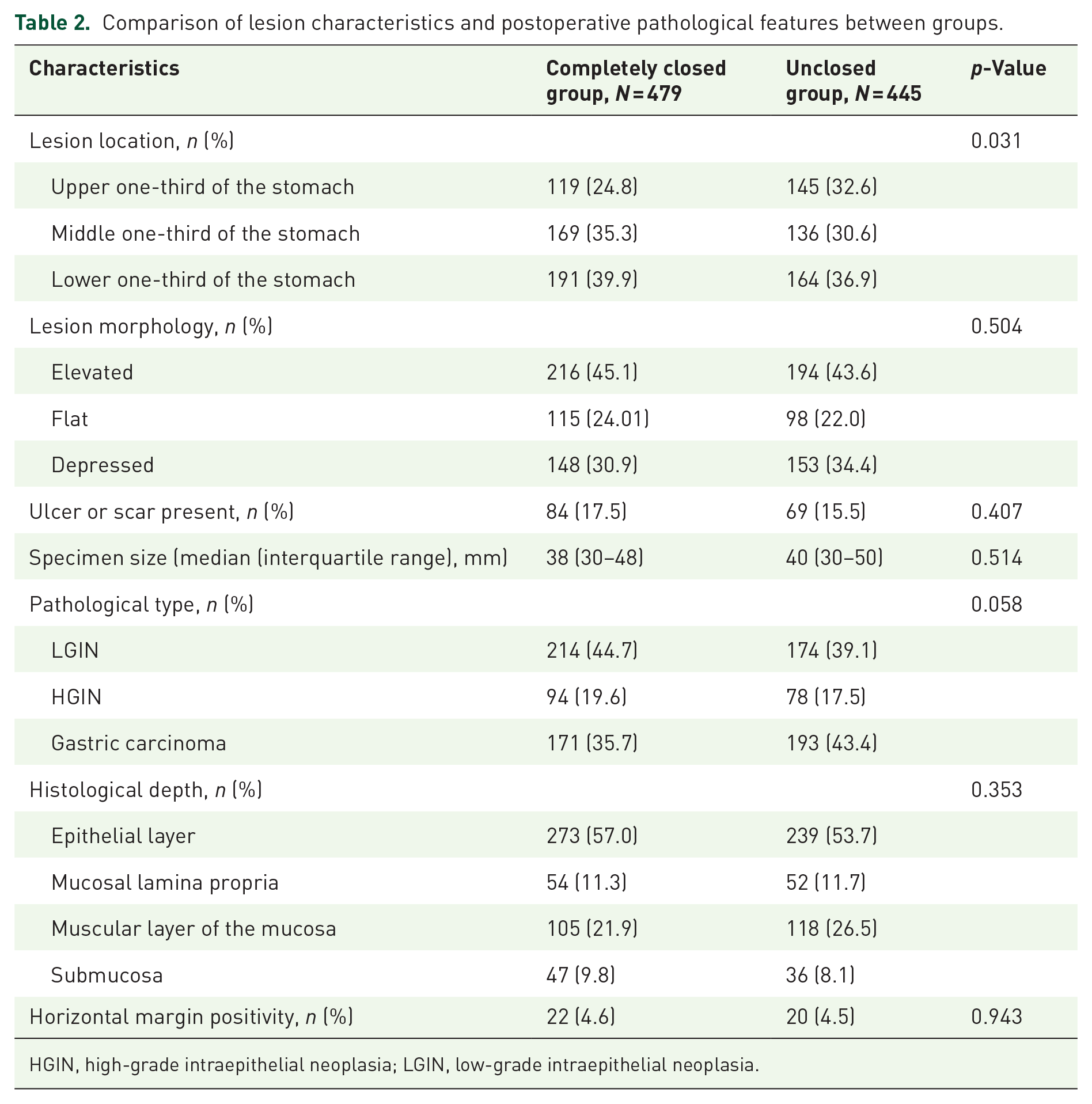
HGIN, high-grade intraepithelial neoplasia; LGIN, low-grade intraepithelial neoplasia.
Gastric ESD procedure analysis
The en bloc resection rates in the completely closed and unclosed groups were 96% and 97.1%, respectively. The median procedure times for the completely closed and unclosed groups were similar (60 (40–75) and 50 (40–77.5) min, respectively; p = 0.593). There were no significant differences in en bloc excision rate (p = 0.385), and muscular injury rate between the two groups (p = 0.539; Table 3).
Comparison of gastric endoscopic submucosal dissection procedure and wound closure between groups.

Postoperative adverse events and follow-up
All patients successfully underwent ESD, however, patients in the unclosed group had significantly longer hospital stays than those in the completely closed group (8 (7–10) and 8 (7–11) days, respectively; p = 0.018). In the completely closed group, 4 (0.8%) patients had delayed postoperative bleeding, which was considerably lower than that in the unclosed group (34 (7.6%) patients; p < 0.001). No intra- or postoperative perforations were observed in either group. Postoperative stenosis occurred in 2 (0.4%) patients in the completely closed group and 4 (0.9%) patients in the unclosed group, with no significant difference between the groups (p = 0.617). The postoperative abdominal pain scores were significantly lower in the completely closed group than in the unclosed group (0 (0–1) and 2 (2–3) days, respectively; p < 0.001; Table 4).
Comparison of postoperative adverse events and follow-up between groups.

ESD, endoscopic submucosal dissection.
One month after ESD, 232 (48.3%) patients in the completely closed group underwent gastroscopy, of whom 180 (77.6%) had ulcers completely healed to the white scar stage, with a significantly higher healing rate than that in the unclosed group (51.1%; p < 0.001). Gastroscopy was repeated 3 months after ESD, and all patients in both groups achieved full wound healing.
Subgroup analysis of postoperative bleeding after ESD for gastric mucosal lesions with different wound closure strategies
Figure 2 summarizes the effect of different wound management strategies after gastric ESD on delayed postoperative bleeding. No significant interaction was observed between the different wound management strategies in all subgroups including sex, age, history of diabetes mellitus, history of chronic renal failure, history of cardiovascular disease, muscle injury, presence of ulcers or scars, lesion location, and specimen size (p for interaction > 0.05). The benefit of complete wound closure compared to wound non-closure was consistent across all subgroups.
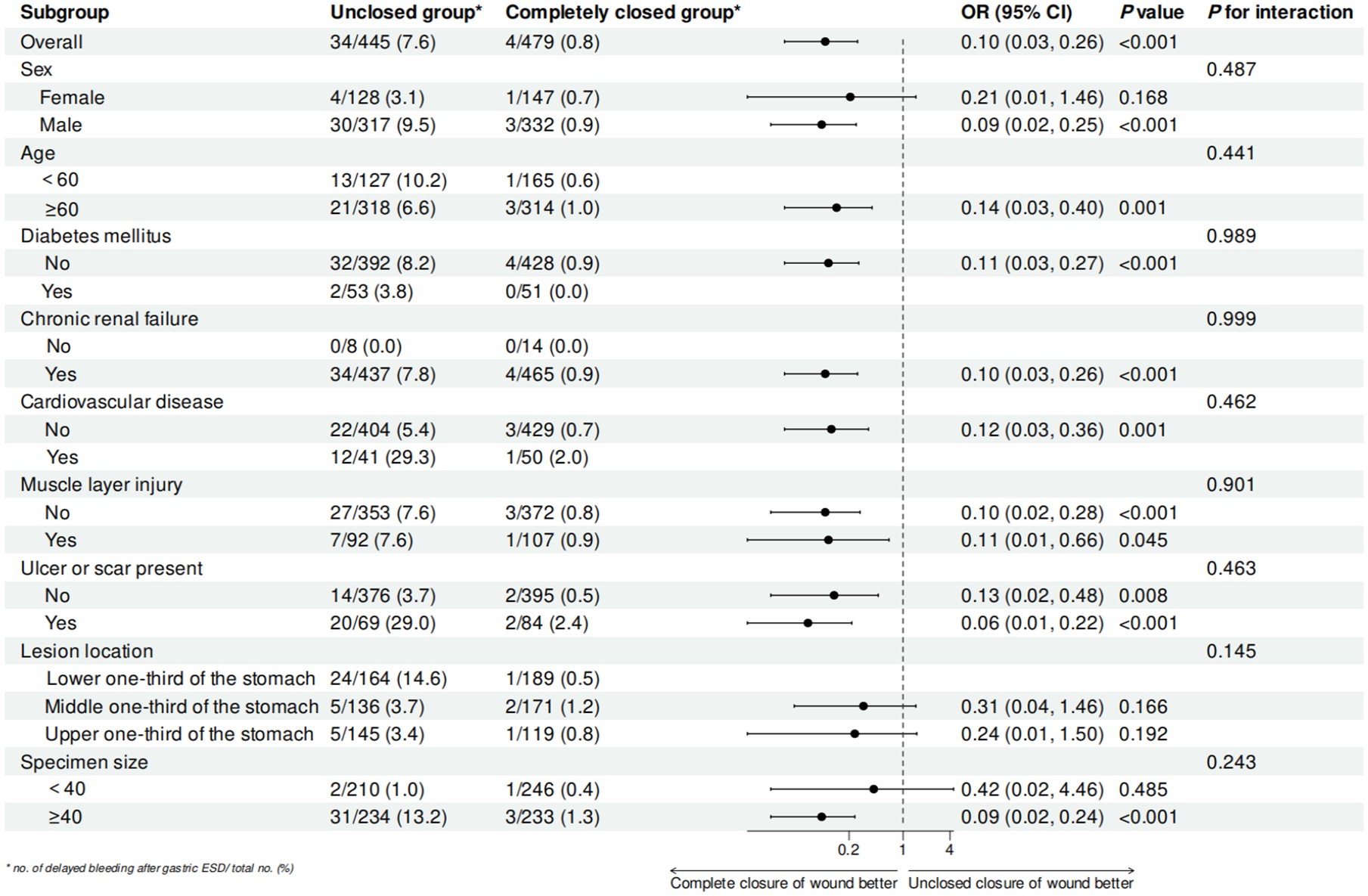
Subgroup analysis of postoperative bleeding after ESD for gastric mucosal lesions with different management of wound closure. Odds ratios and 95% confidence intervals were estimated with the use of logistic regression models, stratified according to sex, age, history of diabetes mellitus, history of chronic renal failure, history of cardiovascular disease, muscle injury, presence of ulcers or scars, lesion location, and specimen size.
Risk factors of delayed bleeding after ESD for gastric mucosal lesions
Univariable and multivariable logistic regression analysis of delayed postoperative bleeding after ESD for gastric mucosal lesions was performed in the two groups, and the presence of ulcers or scars on the lesion surface (p < 0.001; odds ratio (OR) = 6.91; 95% confidence interval (CI), 2.7–15.42), lesions located in the lower one-third of the stomach (p = 0.002; OR = 3.32; 95% CI, 0.05–10.93) in this study were identified as risk factors for postoperative bleeding (Tables 5 and 6).
Univariable analysis for delayed bleeding in patients after ESD for gastric mucosal lesions.
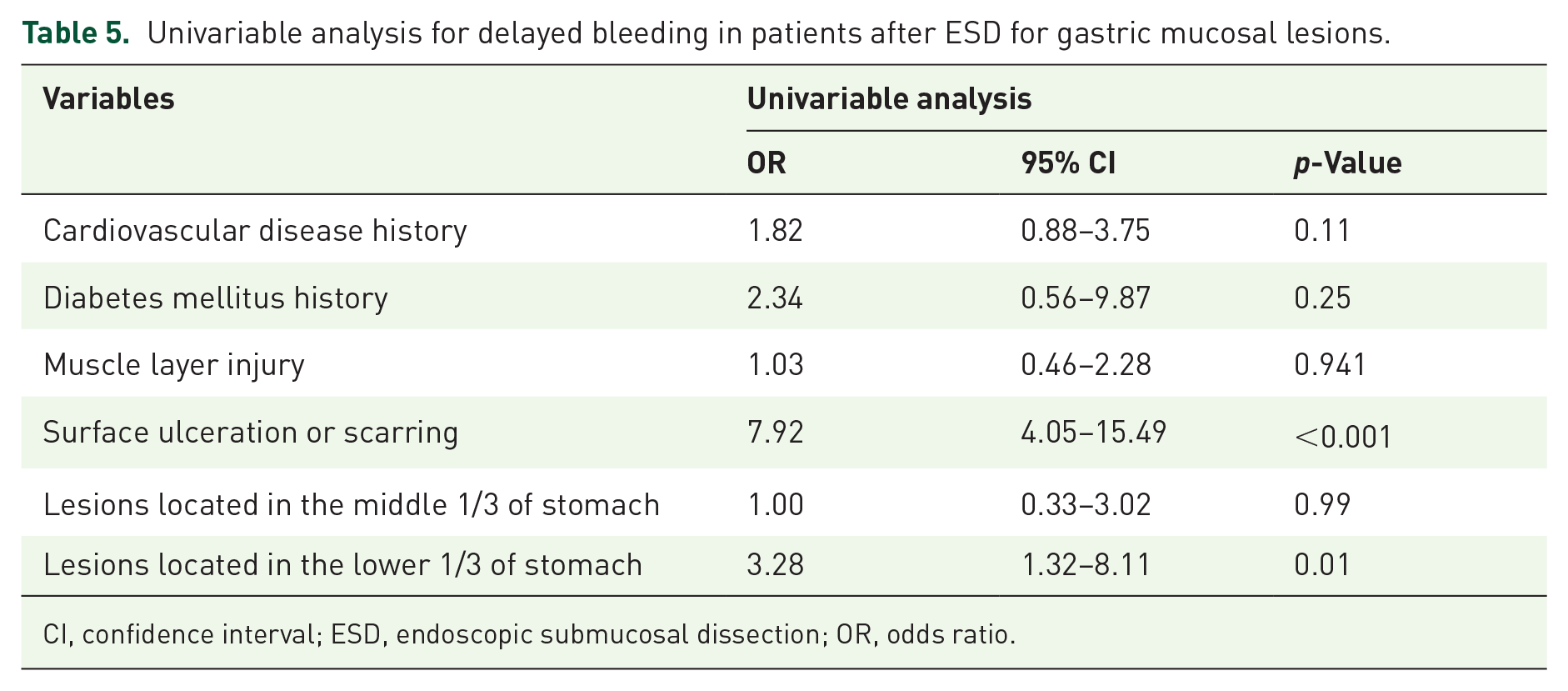
CI, confidence interval; ESD, endoscopic submucosal dissection; OR, odds ratio.
Multivariable analysis for delayed bleeding in patients after ESD for gastric mucosal lesions.

Model 1: no adjusted variables. Model 2: the factors of diabetes mellitus history, and cardiovascular disease history were adjusted. Model 3: the factors of diabetes mellitus history, cardiovascular disease history, muscle layer injury, lesions located in the middle 1/3 of the stomach, and specimen size were adjusted were adjusted.
CI, confidence interval; ESD, endoscopic submucosal dissection; OR, odds ratio.
Discussion
This multicenter retrospective study reports a new technique to close the artificial wound after gastric ESD, which was first proposed internationally and performed in our center in 2013. The results demonstrate that the alternate mucosa–submucosa clip closure technique could reduce postoperative adverse events including delayed bleeding, and postoperative abdominal pain.
Previous studies have shown that the rate of delayed hemorrhage after gastric ESD ranges from 1.8% to 15.6%.12–14 The role of clips in preventing postoperative bleeding following ESD for gastric mucosal lesions has rarely been reported. Our study revealed that the postoperative bleeding rate decreased to 0.5% by the alternate mucosa–submucosa clip closure technique, suggesting that wound closure has advantages, which is consistent with previous findings. A prospective observational study employed the endoscopic ligated O-ring closure technique, which combines an O-ring with an endoscopic variceal ligation device to prevent submucosal pouch formation by immobilizing the muscularis propria in patients with gastric ESD on long-term antithrombotic therapy, significantly reduced the rate of delayed hemorrhage after gastric ESD in this population. 9 In contrast to our findings, another prospective study used a detachable snare to close post-ESD ulcers and examined its effect on postoperative bleeding rates. In that study, there was no significant difference in delayed bleeding rates between the closed and unclosed groups, 15 which may be due to the fact that the wound was effectively closed in only 60% of the cases in this study.
Nevertheless, it is difficult to directly close defects >2 cm using conventional clips due to the thick gastric mucosa. Ikenoyama et al. reported the successful closure of a 10 cm postoperative gastric ESD wound using a floss-assisted mucosal–submucosal clip and the wound remained completely closed on the third postoperative day of gastroscopic follow-up. 10 Over the past 10 years, our center has widely used the alternate mucosa–submucosa clip closure technique for postoperative gastric ESD defects. In our study, there are still three cases of postoperative bleeding in the complete group, and the reason was high-risk behaviors, such as weight-bearing and strenuous activities after discharge from the hospital, leading to premature dislodgement of the clips that triggered delayed hemorrhage. The subgroup analysis of our study showed that the advantage of complete closure of the wound to reduce the delayed bleeding rate was consistent across subgroups, suggesting that the alternate mucosa–submucosa clip closure technique is suitable for different locations and sizes of lesions in the stomach.
Currently, studies on the relief of postoperative pain after gastric ESD are limited to drug-related studies, wherein the causes of postoperative pain include ulcers and electrical burns. Lee et al. reported that intravenous dexamethasone (0.15 mg/kg) was effective in reducing postoperative abdominal pain after ESD for early gastric cancer. 16 Another study showed that the use of a fentanyl patch could reduce immediate postoperative pain on the second day after ESD. 17 Conversely, in our study, closing the postoperative ulcers significantly reduced the abdominal pain score, and all patients in the complete closure group had no or mild pain without further need for analgesic medication. The reduction in pain in the completely closed group was attributed to the fact that the exposed muscle layers were protected from direct stimulation by acid and bile after the complete closure of the artificial wound.
Furthermore, in terms of postoperative wound healing, a randomized controlled trial demonstrated that routine closure with a detachable snare and clips after gastric ESD decreased the percentage of open ulcerated wounds from 43% to 18% after 8 weeks. 15 Akimoto et al. reported that EHS promoted the healing of artificial ulcers after gastric ESD in a living porcine model and that sutured mucosal defects were completely covered by regenerating epithelium within 14 days of suturing. 18 In our study, we found that after complete closure of the wound using the alternate mucosa–submucosa clip method, the complete healing rate of the wound was as high as 80% at 1 month postoperatively, which was higher than that in the unclosed group, consistent with the findings of previous studies.
Additionally, the multivariable analysis in this study showed that the presence of ulcers or scars on the lesion surface was a risk factor for bleeding after ESD, possibly because ulcers or scars lead to unclear layers of lesion peeling, insufficient hemostatic electrocoagulation, and therefore an increased risk of bleeding. This finding is consistent with the results of two previous studies.19,20 However, the presence of scars and ulcers did not influence postprocedural bleeding in some other studies.21–23 Multivariable analysis also revealed that lesions located in the lower one-third of the stomach were a risk factor, and the reasons for this result may be as follows: (1) The artificial wound formed in this location after ESD is exposed to high concentrations of gastric acid and stimulated by refluxed bile, which makes artificial wounds in this area more susceptible to postoperative hemorrhage, and (2) during ESD procedures, wounds in this area are less frequently subjected to routine prophylactic coagulation, thus increasing the rate of postoperative bleeding.
Limitations
Our study had some limitations. First, this was a retrospective study, and the management of post-ESD wounds was not randomized. Second, we did not confirm whether the wound remained completely closed after gastric ESD by second-look endoscopy. Therefore, further validation is required in future multicenter, prospective, and large-sample clinical studies.
Conclusion
In conclusion, complete closure of post-ESD defects by alternate mucosa–submucosa clip could reduce the risk of postoperative bleeding, alleviate patients’ postoperative abdominal pain, and shorten the healing time of the artificial wounds. In addition, this technique requires no additional equipment and is simple to perform, making it a safe and effective defect closure technique that should be promoted.
