Abstract
Acute infectious diarrhea (AID) represents an important clinical entity both regarding morbidity and mortality rates, even in industrialized countries, and it leads to one of the major public health burdens, among gastroenterological diseases, with significant healthcare costs. Oral rehydration solution is the cornerstone of the therapy, but despite its proven efficacy in avoiding dehydration, it is still underused as it does not reduce the duration of diarrhea; hence, it is perceived as ineffective by caregivers. In this narrative review, we collected literature regarding the use of racecadotril, deeply discussing its role in the treatment of AID in both adults and children. Racecadotril has been studied in wide populations of patients, in many countries, and in different clinical settings. Its effectiveness in reducing the stool output and the duration of diarrhea has been proven, not only in the early phase of the disease. Racecadotril has been shown to increase the likelihood of home management of AID, to reduce hospitalizations and parenteral rehydration needs resulting in healthcare costs reduction. The current new formulations require only two-daily doses for adults and the pediatric syrup should simplify its use.
Plain language summary
Acute diarrhea is one of more frequent infectious diseases, with risk of dehydration if not adequately treated especially in children and the elderly, and this impacts on the healthcare costs beyond on the caregivers. Racecadotril, the first and only intestinal antisecretory drug, acting on the abnormal intestinal hypersecretion, decreases the loss of water and electrolytes from the gut, so reducing the dehydration risk, it increases the likelihood of home management of diarrhea. The efficacy of racecadotril has been demonstrated in all settings (inpatients, outpatients, and community-based), in patients of all ages (children, adults, and the elderly), in many countries both of high-income and low-middle-income. In addition, the safety of racecadotril has always been comparable to placebo and better than loperamide.
Introduction
Acute gastroenteritis (AGE) is a common clinical condition, causing 179 million illnesses annually. The estimated 30-day prevalence in the United States is 10.4% for AGE and 7.6% for diarrhea alone. The annual rates were 1.27 cases/person/year for AGE and 0.92 cases/person/year for AGE-induced diarrhea. 1 This condition is generally self-limiting but its symptoms such as diarrhea, dehydration, abdominal pain, and sometimes fever cause different levels of discomfort in affected patients. Viruses are the more frequent etiologic agents of AGE, compared with bacteria, parasites, and fungi. 2 For this reason, from this point forward, we will utilize the definition of acute infectious diarrhea (AID) to standardize the reading. In the pediatric population, AID may be more dangerous determining around 10% of pediatric deaths, mainly in low-income countries. Worldwide, 3–5 billion cases of AGE and nearly 2 million deaths have been registered each year in children under 5 years.3,4
AID is very frequent in children, and it should be treated at home or in outpatient settings. 5 Even the Italian Ministry of Health, in a report published in 2015 by Agenzia Nazionale per i Servizi Sanitari Regionali, stated that AID should not require hospitalization. Moreover, in the 2023 edition of the report, AID is listed among the diseases for which hospitalization should be avoided. 6
The World Health Organization’s (WHO) definition of acute diarrhea is a stool weight of more than 250 g per day, an abnormal stool frequency of at least three stools daily (for a maximum of 2 weeks), and consistency (soft or liquid). 7
Because of its self-limiting evolution, in otherwise healthy people, and due to the risk of dehydration, the milestone of therapeutic management of AID is represented by hydration, mainly using oral rehydration solution (ORS). Antisecretive drugs, drugs increasing intestinal transit time, or antimicrobics could be combined with this approach, to reduce diarrhea duration and/or to treat infections if a bacterial, fungal, or protozoal etiology is confirmed. Despite being useful in managing symptoms, compounds increasing intestinal transit time, such as loperamide, an opioid agonist, may be detrimental. In fact, they can favor the persistence of infectious agents in the intestinal lumen (in contrast with the Latin statement saying “Ubi pus, ibi evacuat”). Moreover, loperamide is contraindicated in children under 6 years and evidence under 12 years of age is limited. Furthermore, the abuse of loperamide in specific circumstances may determine paralytic ileum and toxic megacolon in children. On the other hand, antibiotics, by far the most frequent, are not useful in viral AID, and some of them might induce the selection and growth of aggressive strains such as, for example, Clostridium difficile. 8
Regarding other products frequently used in AID such as probiotics, smectite, and gelatin tannate, their exact mechanisms of action in humans in vivo are still not fully understood, so far. Therefore, a careful evaluation of the efficacy/safety balance suggests the use of drugs characterized by a safer mechanism of action in both adults and children. In this perspective, racecadotril, an enkephalinase inhibitor, could represent an interesting option. In this narrative review, we describe the available evidence on the efficacy and safety of racecadotril in adults and children. 9
Racecadotril: pharmacology
Racecadotril is a prodrug that, after oral administration, is converted into thiorphan by hydrolysis and it exerts its effects acting on enkephalinase, a peptidase located in the cell membrane, in different tissues, but mainly in the small intestine mucosa. This enzyme is involved in the metabolism of enkephalins, endogenous opioids that in physiologic conditions contribute to maintaining the secretion/absorption balance in the small intestine.10,11 In fact, enkephalines bind to δ opioid receptors of enterocytes, reducing the cyclic adenosine monophosphate (cAMP) synthesis. The final effect of this mechanism is a decrease in both water and electrolyte secretion in the gastrointestinal lumen 10 (see Figure 1).

Racecadotril’s mechanism of action. In physiologic conditions, enkephalines reduce water secretion in the intestinal lumen and their metabolism is mediated by enkephalinases. In the presence of noxious stimuli, the water secretion may increase through the increase in second messengers like cAMP. In this condition, the inhibition of enkephalinases by racecadotril (converted into tiorphan) contributes to reducing diarrhea.
Differently from other products used in AID treatment, racecadotril, through a well-defined mechanism of action, reduces intestinal liquid loss due to the hypersecretion induced by intestinal infections, not impacting basal secretion. Therefore, it is directly addressed to the primary pathophysiological mechanism underlying AID.12–14
In adults, racecadotril is administered orally, according to the different formulations, at a total dosage of 300–350 mg, respectively, in three or two doses. In children, 1.5 mg/kg/dose is taken three times daily. The maximum period of treatment is 1 week.
Racecadotril’s pharmacokinetic properties are summarized in Table 1.
Racecadotril pharmacokinetics. 15

A new formulation of racecadotril 175 mg tablet showed shorter Tmax, greater bioavailability, and extended half-life in healthy volunteers, compared with 100 mg tablet, suggesting its use as a bis in die (b.i.d.) anti-diarrheal drug. 16
Clinical evidence
Racecadotril was approved in France in 1992, and it is now available in many countries in Europe, Latin America, Asia and Middle East, and Africa, even as an over-the-counter product, for the treatment of AID. The pivotal studies on the efficacy of racecadotril date back to the early 2000s and some of them measured the stool output, defined by the WHO as the most significant and reliable parameter to evaluate the effectiveness of anti-diarrheal drugs. 7 Since then, many studies confirmed racecadotril efficacy, both in adults and in children, in all clinical settings. Despite racecadotril being a drug with a good safety and efficacy profile (see the sections below), it is an underestimated therapeutic option. In real-world practice, loperamide or probiotics are the most prescribed compounds for the management of diarrhea. This narrative review aims to summarize the most important existing clinical evidence about the efficacy and safety of racecadotril in adults and children, showing that it has comparable efficacy with respect to loperamide, with a superior safety profile, especially in children.
Adults
Racecadotril efficacy in the management of AID has been documented in several clinical trials. In particular, Roge et al. compared racecadotril and loperamide at usual therapeutic doses in 69 patients with AID. The two drugs showed similar efficacy at 2 days. Nevertheless, in the racecadotril group, lower rates of abdominal distension and reactive constipation were observed. The authors observed that abdominal distension and abdominal pain were relieved sooner with racecadotril. The reason is probably related to the mechanisms of action of the two drugs: in fact, loperamide acts mainly in transit causing a pooling of fluid in the bowel lumen that may be responsible for distension and sometimes for bacterial colonization. The absence of antitransit activity by racecadotril accounts for its minor incidence of constipation and abdominal distension. 17
In a multicenter randomized, double-blind clinical trial, Vetel et al. evaluated 157 patients with AID treated with racecadotril ter in die (t.i.d.) or with loperamide 2 mg after each stool. In this study, the authors confirmed a similar efficacy for the two drugs in terms of stool number till recovery, associated signs and symptoms severity, and duration of diarrhea. However, in this study, the treatment with loperamide, instead of racecadotril, induced the development of constipation. 18
Some years later, Hamza et al. described the effects of racecadotril versus placebo in 70 patients with AID evaluating both stool weight and antisecretory activity. Racecadotril produced a significant decrease in stool weight (p = 0.025) and bowel movements number (p = 0.027) after 1 day of treatment. Abdominal distension prevalence was 5.6% with racecadotril versus 18.2% in the placebo group. Racecadotril and placebo safety profiles were comparable. It is noteworthy to observe that abdominal constipation incidence was significantly higher in the racecadotril group than in the placebo group (p = 0.02). In this study, stool weight appears to be a different clinical outcome and the study is limited by its missed assessment in the 24 h before treatment. Nevertheless, the two groups were homogeneous considering clinical symptoms. 19
In a multicenter single-blinded study in a wide population (945 outpatients) with AID, Prado documented that racecadotril reduced the duration of diarrhea (primary efficacy endpoint) and of abdominal pain duration, compared to loperamide. In the racecadotril group, constipation was less frequent than in loperamide-treated patients. Overall, 67 (14.2%) patients in the racecadotril group and 113 (23.9%) in the loperamide group (p = 0.001) experienced side effects. 20
Wang et al. performed a randomized single-blinded study aimed at comparing loperamide (2 mg b.i.d.) and racecadotril (100 mg t.i.d.) in 62 patients with AID. The authors documented that loperamide was faster in treating diarrhea versus racecadotril (13 h vs 19.5 h), but it induced the development of constipation. 21
In a randomized clinical trial, Gallelli et al. evaluated 61 elderly patients with AID and without signs of severe dehydration and bacterial infection. Patients were randomly allocated to racecadotril (100 mg t.i.d.) or loperamide 2 mg (followed by another tablet in case of unformed stool evidence, up to four tablets) arms. In the racecadotril group, stool normalization was faster compared to loperamide. Similarly, abdominal pain duration was lower, 14 h with racecadotril versus 28 h with loperamide. 22
In a systematic review and meta-analysis on 12 randomized controlled trials (RCTs) analyzing a total population of 2619 patients, Vetel et al. found that racecadotril treatment was associated with a 65% higher percentage of recovery compared to placebo (hazard ratio (HR) = 1.65 (1.38–1.97), p < 0.00001). Duration of diarrhea was similar to loperamide, but the number of patients developing constipation was significantly higher with the opioid receptors’ agonist (relative risk = 0.34 (0.22–0.51), p < 0.0001). 23 Coffin et al. obtained similar results in an Individual Patient Data (IPD) meta-analysis, comparing racecadotril to placebo. They showed a significantly higher recovery probability (HR = 1.8 (1.3–2.5), p < 0.001) and a 60% rate of responders in the racecadotril group. Despite these good results, the authors observed the differences in racecadotril and mainly loperamide doses from one study to another. A similar lack of homogeneity was found in the definition of constipation. 24
Eberlin et al. conducted a multi-center, randomized, single-blinded study in 223 Chinese adult patients (aged 18–70) with AID. Patients received racecadotril (100 mg t.i.d.) or loperamide (2 mg t.i.d.) until recovery (defined as 12 h without bowel movements or two consecutive normal stool discharges) for a maximum of 3 days. Diarrhea, nausea, anal burning, and anorexia resolved with both drugs. However, the per-protocol analysis showed a better improvement with racecadotril. Adverse events were mild to moderate, and they were more common with loperamide than with racecadotril (17% vs 3.5%). Although coherent with the results of other studies, per-protocol analysis must always be considered a possible source of bias. 25
A systematic review by Fischbach et al. showed a racecadotril safety profile comparable to a placebo. Furthermore, compared with loperamide, racecadotril showed similar efficacy, but lower rates of constipation and abdominal discomfort. These authors mentioned even two studies where racecadotril and Saccharomyces boulardii safety profiles were similar but racecadotril was more efficacious. 26
Bouhnik et al. compared the efficacy of racecadotril 175 mg tablets b.i.d. with the one of racecadotril 100 mg t.i.d. in a randomized, controlled, double-blind, double-dummy study on 235 adults. The total number of diarrheic stools, from inclusion to recovery, was the main efficacy endpoint and an Associated Symptoms Index was calculated considering spontaneous abdominal pain, bloating, gurgling, nausea, vomiting, loss of appetite, fever, and asthenia severity as reported in the patient diary card. Racecadotril 175 mg b.i.d. showed to be more efficacious compared with racecadotril 100 mg t.i.d. (racecadotril 100 mg t.i.d. vs racecadotril 175 mg b.i.d. diarrheic stools total number (6 (3–9) vs 4 (2–6), p = 0.003), diarrhea duration (33 h (22–57) vs 24 h (8–45), p = 0.013), and Associated Symptoms Index (5.3 (2.7–8.5) vs 4.0 (1.8–5.6), p = 0.03)). Safety profiles of the two formulations were similar and no serious side effects were reported. 27
Coffin et al. performed an IPD meta-analysis aimed at comparing racecadotril 175 mg b.i.d. and racecadotril 100 mg t.i.d. efficacy in adults with AID. Two RCTs on 456 patients with similar endpoints were included. Racecadotril 175 mg b.i.d. induced a significantly higher reduction of stool number (−2.0 (−3.21, −0.78) p < 0.001) compared with racecadotril 100 mg t.i.d. Moreover, the last diarrheic stool was observed 25% earlier (p < 0.02). Finally, 10% more responders were observed in the group treated with the new formulation, with a Number Needed to Treat of 10. 16
Further details are presented in Table 2.
Adult clinical studies.

AEs, adverse events; HR, hazard ratio; ITT, intention-to-treat population; NDS, number of diarrheic stools; OR, odds ratio; PPP, per protocol population; RC, racecadotril; RR, relative risk; VAS, visual analog scale.
Children
The first studies evaluating the efficacy of racecadotril in children were made by Turck et al. and Salazar-Lindo et al. Turck et al. in their multicenter, parallel-group, double-blind, double-placebo, comparative study showed that racecadotril and loperamide, at the time used even in children, were equally effective in treating AID, but racecadotril had a better safety profile. 29
Salazar-Lindo et al. evaluated the differences in the 48-h stool output, the more reliable parameter to assess the anti-diarrheal drug’s efficacy, in 135 Peruvian hospitalized children with AID, comparing the efficacy of racecadotril combined with ORS to ORS plus placebo. Children treated with racecadotril experienced a 46% reduction of 48-h stool output (p < 0.001). In the same way, both median diarrhea duration and ORS volumes needed to rehydrate children were significantly lower in the racecadotril group, regardless of Rotavirus or not-Rotavirus etiology. No differences were found in the safety profiles of the two approaches. 30
With a similar study design, Cézard et al. compared the efficacy and tolerability of racecadotril as adjuvant therapy to ORS with ORS plus placebo in a randomized, double-blind, placebo-controlled study in 172 children hospitalized for AID in 13 French centers. Children receiving racecadotril had a significantly lower stool output in the first 24 and 48 h of treatment than the placebo group, up to 50%, and the tolerability did not differ between groups. 31
Besides these two trials, done in hospitalized patients to properly measure stool output, Cojocaru et al., in 166 French children with AID evaluated in a pediatric emergency department, confirmed a clear superiority, in decreasing stool number, in accelerating recovery from diarrhea and in reducing secondary medical visit need, of racecadotril added to ORS compared to ORS alone. 32
An open-label, non-controlled, prospective, multicenter, observational, and naturalistic study, done in clinical practice in Venezuela on a wide population, showed an evident efficacy of racecadotril associated with ORS in accelerating the diarrhea resolution in 3873 children in a real-world setting. In this study, the time-to-relief, defined as the time from the first racecadotril dose to the last watery bowel movement, observed in this study was less than 24 h, with a good perception, both by doctors and by parents, of racecadotril performance as childhood acute watery diarrhea treatment. 33
A retrospective Italian study compared the efficacy of racecadotril associated with ORS (Group 1) versus ORS alone (group 2), in children hospitalized with AID, assessing the need to switch from oral to parenteral rehydration and the probability of early discharge. The percentages of children requiring parenteral rehydration, due to diarrhea persistency, were 26.9% and 42.9% in groups 1 and 2, respectively. Consistently, 65.4% of group 1 children were discharged earlier, compared to 40% of group 2, with a statistically significant difference between groups (p < 0.05). 34
An open-label, randomized study done in India analyzed the efficacy of racecadotril plus intravenous rehydration therapy, compared to intravenous rehydration therapy alone, in children with watery non-bloody diarrhea. Children using racecadotril had more than 50% reduction in stool frequency and a higher probability of switching to oral rehydration therapy. 35
Another Indian study confirmed that racecadotril significantly decreased the frequency of daily stools, the duration of diarrhea, the recovery time, and the ORS volume administered, compared to placebo, in hospitalized children with AID. 36
An Individual-Patient-Data meta-analysis performed in 2011 concluded that racecadotril had a clinically relevant effect in reducing diarrhea duration and stool output, irrespective of dehydration level, etiologic agent, or age of infected children, both in inpatient and outpatient settings, and both in high-income and middle-/low-income countries. 37
In 2015, a network meta-analysis done in Mexico showed racecadotril therapeutic advantage as an adjuvant therapy of rehydration, compared to smectite, several kinds of probiotics, zinc, and placebo, in the management of pediatric AID. 38
Gordon and Akobeng in their systematic review of seven RCTs evaluated the efficacy and safety of racecadotril compared with placebo or other interventions for the treatment of acute diarrhea in children. Although the authors identified several biases such as selection, performance, attrition, and reporting type, they concluded that by adding racecadotril to ORS, the length of symptoms as well as the stool number and volume can be reduced. Therefore, the use of racecadotril appears safe and well-tolerated in children with AID. 39
Moreover, a more recent meta-analysis, including 58 trials performed in nine countries, from China to Spain and France, from Ecuador to Kenya, India, Peru, Guatemala, and Egypt, found that racecadotril, compared to other anti-diarrheal treatments, such as smectite, probiotics, loperamide, antiviral drugs and antibiotics, or placebo, reduced diarrhea duration and stools number, showing a better global effectiveness, already on the second day of therapy. Based on this evidence of efficacy, the authors concluded that, compared to other treatments, racecadotril is preferable in AID, having also a safety profile similar to a placebo and better than loperamide. 40
Compared to this large amount of evidence showing racecadotril effectiveness, some authors did not confirm the same therapeutic outcomes. However, analyzing these studies in detail, we can note some relevant methodological biases, such as a high number of dropped-out patients and the use of an Indian copy of the molecule not validated by the company that held the drug patent. Therefore, this drug was not equivalent to the original molecule hence this may have impacted treatment efficacy. Furthermore, the authors did not consider additional critical co-infections causing osmotic diarrhea such as Salmonella, Shigella, and Campylobacter beyond HIV infection) that could have influenced the final outcome, as the authors themselves highlighted.41,42
In the same way, a recent Cochrane review reported a modest benefit of racecadotril under a low-very low certainty of evidence, concluding that more data are needed to improve the certainty of evidence in racecadotril’s efficacy in diarrhea. 43
Lastly, in a case report by Tran et al., racecadotril was reported to be efficacious in reducing both the stool number and the need for total parenteral nutrition in a child affected by chronic diarrhea due to microvillous inclusion disease, after the failure of loperamide. Although racecadotril is not indicated in this disease and it is only a single case, it is interesting to note that watery diarrhea reappeared after the withdrawal of racecadotril and improved again after its reintroduction. 44
Further details are presented in Table 3.
Pediatric clinical studies.
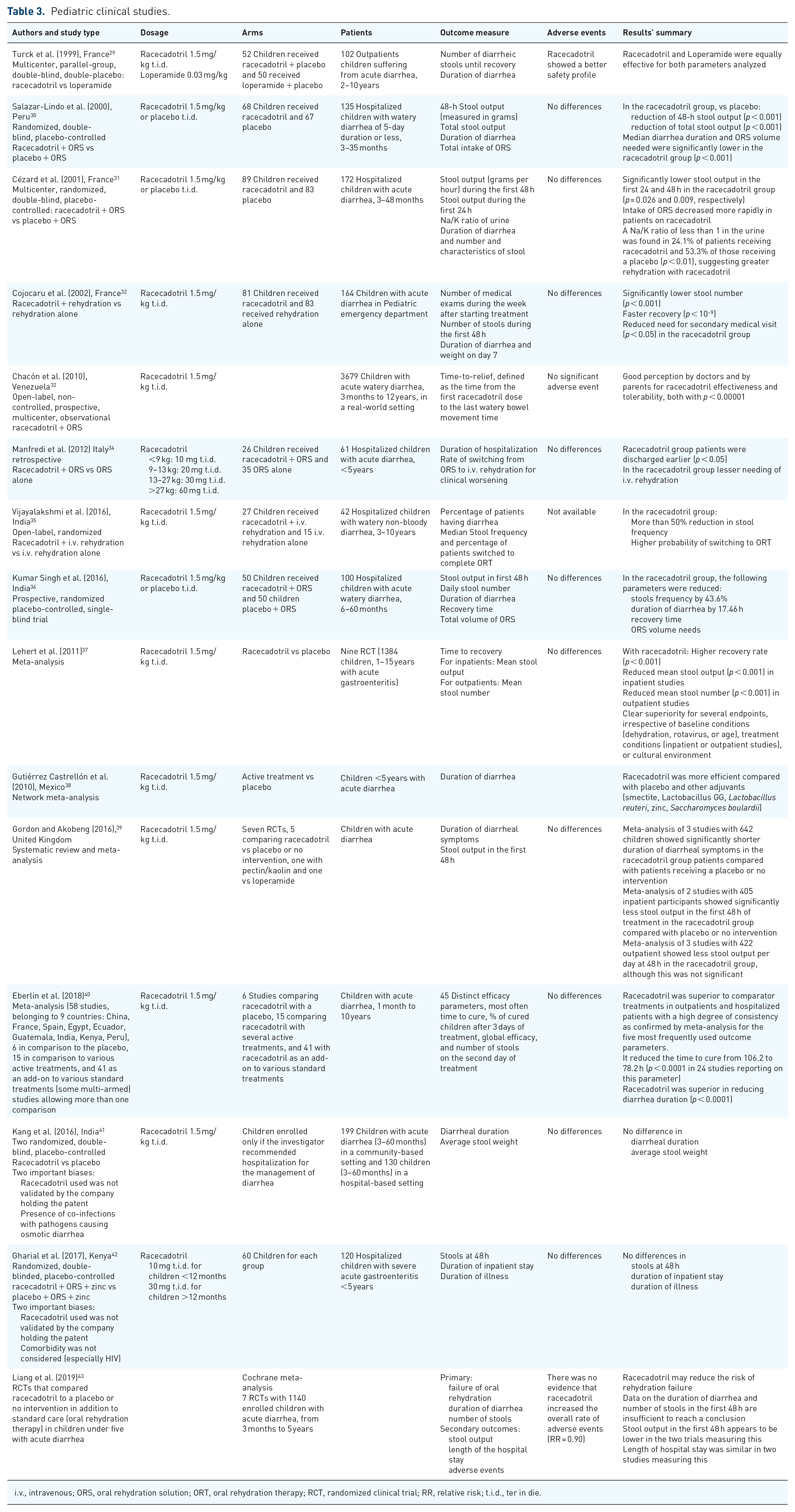
i.v., intravenous; ORS, oral rehydration solution; ORT, oral rehydration therapy; RCT, randomized clinical trial; RR, relative risk; t.i.d., ter in die.
Safety
Based on this evidence, we can state that racecadotril has a very good safety and tolerability profile, is not different from placebo, and clearly better than loperamide. Post-marketing data cumulate knowledge from about 71 million adults and 65 million infants and children. Overall, there were only 530 (0.00075%) and 265 (0.00041%) individual case safety reports, respectively. 45
The most common side effect of racecadotril is headache (2/96 patients in the clinical trial by Baumer et al.). 28 Furthermore, cutaneous/subcutaneous (including erythema, edema, urticaria, and itching have been described) and gastrointestinal signs and symptoms have been reported. It is not futile to repeat that generally in all studies both in adults and in children adverse events incidence is not significantly different from placebo.11,46 Nevertheless, racecadotril is not indicated in antibiotic-induced diarrhea and chronic diarrhea due to a lack of clinical studies in these indications. Moreover, there are little data available on patients with severe renal or hepatic insufficiency and the drug must be administered according to the indications of the summary of product characteristics (SmPC). In patients with prolonged vomiting, bioavailability may be reduced. Patients with hereditary metabolic deficiencies, like galactose intolerance, Lapp-lactase deficit, or glucose-galactose malabsorption, must avoid racecadotril. 11
The risk of drug interaction (DDI) is very low even if it has been reported an increased risk of angioedema in patients using ACE inhibitors. The mechanism beside this side effect is the increase of bradykinin the degradation of which is mediated by both ACE and a neutral endopeptidase inhibited by racecadotril. Therefore, patients taking ACE inhibitors should be warned in advance that in case of diarrhea, the concomitant use of racecadotril could increase the risk of developing angioedema. 47 Despite this interaction risk being reported in racecadotril’s SmPC, its evidence is limited to case reports. 48
Discussion
Oral rehydration by gluco-electrolyte solutions represents the cornerstone of diarrhea management, but because it does not reduce the duration of diarrhea, the need for an effective anti-diarrheal drug is a matter of fact. Being viral etiology (mainly by Norovirus, Rotavirus, and Adenovirus), the cause by far more frequent of AID, antibiotic treatment should be limited to a small percentage of cases where the presence of bacterial pathogen strains (Shigella, Salmonella, Campylobacter, Escherichia coli, Vibrio cholerae) is confirmed and sensitivity of the strain to the antibiotic assessed. Protozoa and fungi-induced AID cases that require specific therapies are even less frequent. 49 On the other hand, treatments aimed at reducing the severity and duration of diarrhea and associated symptoms, not impacted by rehydration, could be administered, to decrease the burden of AID.
For many years, probiotics have been considered a useful option, especially to restore the activity of healthy intestinal flora, stimulate the immune system, and compete with pathogens for binding sites on epithelial cells. However, the evidence about this therapeutic option is still limited and it seems to be less efficacious compared to antidiarrhoeic drugs in adults. Moreover, recent studies in children stated that probiotics are ineffective in treating AID50,51 and the last position paper of the European Society of Pediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) 52 considers probiotics as ineffective or with a very low efficacy in children with AID. Moreover, a recent AGA technical review stated that probiotics are not useful or not effective in most gastrointestinal diseases in adults. 53
In this context, the use of some antidiarrheal drugs needs to be well understood since their use may certainly reduce the intensity of the symptoms but may be also associated with possible complications. In particular, loperamide has shown several safety concerns due to the risk of toxic megacolon, necrotizing enterocolitis, syncope, QT alterations, ventricular arrhythmias, and cardiac arrest.54,55 In children, antidiarrheal drugs are also associated with hemolytic uremic syndrome. Despite these observations, the real problem seems related to the abuse of these compounds. In fact, Osugi et al. 56 showed, in their retrospective study on 10,246 patients, 204 of whom received antidiarrheal drugs, that the administration of these compounds is not related to the increase in return visits to hospital accesses. Their work suggests that antidiarrheal drugs, if not abused, have more benefits than risks.
Among antidiarrheal drugs loperamide, racecadotril, and bismuth salicylate, much less used, are the possible options. 49
Racecadotril showed better tolerability compared to loperamide, being less frequently associated with constipation and other side effects. Constipation related to loperamide treatment may also lead to severe complications like bacterial retention, which is harmful because AID could be induced by toxin-producing bacteria. Furthermore, loperamide has a certain potential of DDI being metabolized by CYP2C8 and CYP3A4 enzyme isoforms of cytochrome p450 (CYP450). Extrusion by P-glycoprotein (P-gp) limits loperamide central nervous system (CNS) side effects but the presence of P-gp inhibitors may eventually promote loperamide access to CNS. Despite the possible risks related to these mechanisms, loperamide is a drug very easy to access even as an over-the-counter product, and it is probably the most frequent therapeutic solution chosen by many adult patients. 27
Racecadotril offers a safe option both in adults and in children after the 3 months of age, whereas loperamide is recommended only for subjects ⩾12 years old. 11
Many trials, systematic reviews, and meta-analyses in children agreed showing that adding racecadotril to ORS reduces stool output and diarrhea duration in both inpatient and outpatient settings. Based on this evidence, racecadotril has been recommended for the treatment of AID in children by many international guidelines, from the ones by the World Gastroenterology Organization 57 and by ESPGHAN/ESPID (European Society for Pediatric Infectious Diseases), 58 the panels’ guidelines panel from Spain and Latin America, 41 and the recommendations by the German Society for Pediatric Gastroenterology and Nutrition. 59 In addition, the Italian Ministry of Health, in its webpage regarding the treatment of traveler’s diarrhea, recommends the use of racecadotril. 60
Furthermore, we should not forget that AID generates significant direct and indirect costs. Parents of children affected by AID reported high distress levels during the diarrhea episode and a feeling of exhaustion and helplessness. 61 An Italian study showed that the average cost of each access to the emergency department for AID is about €141.00, and the average cost for each hospitalization ranges between €1478.00 and €2000.00. On the other hand, the cost of AID home management was estimated at €18.20 for each patient. 62 Furthermore, parents of children affected by acute diarrhea lose on average 4 days of work or school for each AID episode.
In this regard, several studies showed an important advantage of racecadotril. Rautenberg et al. observed that the acute watery diarrhea management cost changed significantly in economic terms when racecadotril was added to ORS in children under 5 years. The results of the combination therapy were a total cost reduction of £379.00 (about €440.00) in favor of racecadotril and a total incremental quality-adjusted life years gain in favor of racecadotril of +0.0008. However, they stress the main cost determinants. Reconsultation rate and therefore access to primary care (not including dehydration) is the primary cost factor and needs to be evaluated through diarrhea duration. In fact, in this study, the outcome is related to second accesses. Racecadotril, resolving symptoms in 48 h, will reduce this cost. The second factor is access to secondary care with a consequential increased risk of nosocomial infections. The reduction of the duration of diarrhea may impact also those cost generators. Lastly, the impact on parents is relevant since absence from work is the natural consequence of a child’s illness. 63
Similar racecadotril cost-effectiveness advantages have been demonstrated in both high-income and low/middle-income countries.63–66 Racecadotril advantages are summarized in Figure 2.

Racecadotril’s advantages. As discussed in the text, (1) the action on enkepalinases and therefore on secretion only avoids transit inhibition; (2) less severe side effects if compared to loperamide, especially toxic megacolon, paralytic ileum, and cardiac side effects; (3) pharmacoeconomic analysis showed a benefit in terms of costs; (4) the high level of safety makes racecadotril is the best option in children; and (5) clinical trial showed similar or superior efficacy to loperamide.
Robert Edelman, in 1985, defined that the ideal characteristics of any compound for treating AID should be those:
to inhibit fluid secretion/stimulate fluid absorption by intestinal mucosa;
to have a rapid onset of action;
to limit constipation effects;
not to interfere with recovery of local bowel functions;
to minimize CNS effects;
to have low abuse potential;
to have low costs. 67
Effectively, by the evidence of literature and this narrative review, racecadotril fulfills all these criteria.
Conclusion
Racecadotril, an enkephalinases inhibitor seems to be an effective and safe option in AID associated with a very low number of DDI. Its efficacy is superior to probiotics and, in children, it improves rehydration efficacy and increases the probability to manage successfully at home the episodes of AID. Racecadotril effectiveness has been confirmed in both children and adults with AID, in inpatient and outpatient settings, in several clinical trials, meta-analyses, and systematic reviews done all over the world, from high-income to middle- and low-income countries. Furthermore, a favorable racecadotril cost/effectiveness balance should encourage its use in clinical practice. The actual availability of an adult formulation requiring only two-daily doses and of a pediatric syrup should simplify its use.
