Abstract
Objective:
This study was aimed at identifying
Method:
A facility-based cross-sectional study was conducted on 263 children aged below 14 years with diarrhea. A structured questionnaire was used to collect socio-demographic and clinical data after obtaining the necessary consent from their parents or caretakers. The culture and sensitivity tests were performed using the standard operating procedure of the microbiology laboratory.
Results:
Accordingly, 20/263 (7.6%), 95% confidence interval: 4.4%–11.4%
Conclusion:
High level of
Introduction
Diseases caused by enteric pathogens are of common public health concerns in many parts of the world including Ethiopia.1,2
They are species of particular concerns as they cause enteric fevers, food poisoning and gastroenteritis.
9
They are Gram-negative rods that commonly inhabit the intestinal tracts of humans and many animals.
10
It was estimated that 1.8 million cases of children died from diarrheal illness worldwide, a large proportion of which was attributed to infection by
Infections of
Materials and methods
Study area and period
The study was conducted in the Southern Nations, Nationalities and Peoples Region (SNNPR) at Hawassa Alamura Health Center from 1 April 2019 to 30 August 2019. Hawassa is the capital city of the SNNPR, located in the Southern part of Ethiopia, on the shores of Lake Hawassa which is one of the Great Rift Valley lakes situated around 270 km from Addis Ababa, the capital city of Ethiopia. The mean annual rainfall is about 950 mm, temperature about 20°C and humidity 70%–80%. The rainy season generally extends from June to October. The human population of Hawassa for 2015 was estimated at 351,469, with an annual growth rate of just over 4%. 20 Hawassa city has 7 sub-cities with 5 private, 1 general and 1 comprehensive specialized hospital and 10 health centers. Alamura Health Center is located in the Tabor sub-city and borderline between Fara and Hitata Kebele near Alamura Mountain.
Study design and population
A facility-based cross-sectional study design was conducted among children with diarrhea at Alamura Health Center. A convenient sampling technique was employed in which diarrheic pediatric patients below the age of 14 years were included. They were considered for the study only after obtaining the necessary consent from their parents or guardian and signing the document. The participants are excluded if their parents are not willing or refuse to sign. All diarrheic pediatric patients who visited Alamura Health Center for the diarrheal case of illness were the source of population.
Sample size determination
The sample size was calculated using a single population proportion formula: n = z2p (1−p)/d2, where n = sample size, z = confidence level at 95% (standard value of 1.96), M = margin of error at 5%, p = estimated prevalence of
Variable of study
The dependent variables were the presence of
Data collection
The socio-demographic and clinical data were collected after informing the parents/caregiver about the aim of the study. A face-to-face interview was conducted to collect the data with a structured questionnaire from parents or caretaker of the children who complained of diarrhea after they signed the consent and the child accepted the assent.
Laboratory diagnosis
The stool was collected using a screw cup container. The parents/caregiver was instructed to bring a fresh stool sample without any contamination before 30 min of collection. All stool specimens were placed into Carry Blair Transport Medium and transported to the Microbiology Laboratory of Hawassa University Comprehensive Specialized Hospital (HUCSH). The stool was inoculated on prepared culture media which is MacConkey, Xylose lysine deoxycholate (XLD) and selenite F-broth (Abtek, UK). The culture plates were incubated aerobically at 37°C for 24 h.
Bacterial identification
The colonies were examined morphologically for size, shape and ability to ferment lactose. Those bacterial colonies with non-lactose fermenting characteristics with H2S for
Antibiotics susceptibility testing
A pure colony of isolated bacteria was mixed with normal saline to make a 0.5 McFarland standard suspension for susceptibility testing and then swabbed on Mueller Hinton agar. The susceptibility pattern of the isolates was determined for ciprofloxacin (CIP; 5 μg), augmentin (AUG; 30 μg), gentamicin (GEN; 10 μg), chloramphenicol (CAF; 30 μg), co-cotrimoxazole (COT; 25 µg), tetracycline (TAT; 30 µg), ampicillin (AMP; 10 µg), ceftriaxone (CRO; 30 µg), cefuroxime (CRX; 30 µg) and ceftazidime (CAZ; 30 µg). After incubation for 24 h at 37°C, the diameter of each zone of inhibition was measured with a ruler in millimeters. The results were then interpreted according to Clinical and Laboratory Standards Institute (CLSI) guidelines antimicrobial susceptibility breaking points 2018 and recorded as sensitive (S), intermediate (I) or resistant (R). 23
Quality control
A pre-test was conducted on 5% of the questionnaire before conducting the study. The validity and completeness of the data were verified daily. Sterility of culture media and biochemical tests were checked by overnight incubation of un-inoculated media from each batch of preparation. Standard strains of
Data analysis
Data were entered into Statistical Package for the Social Sciences (SPSS) version 20 and were analyzed to make inferences on the frequency of occurrence of enteric pathogens associated with diarrhea and to show bacterial resistance pattern to locally prescribed antibiotic substances. Descriptive statistics were performed to get the frequency of dependent and independent variables. Binary logistic regression analysis was conducted to identify real predictor of
Ethical consideration
The study was conducted after obtaining formal permission from the Southern Nations Nationality and People Regional Health Office, Hawassa City Administration Health Office, Alamura Health Center Manager and Laboratory Head. The patients were included in the study only when the parents or caretakers of the patients sign the consent letter. The culture and antimicrobial susceptibility results were communicated to the concerned bodies in the health center within 72 h.
Results
Socio-demographic characteristics of the study subjects
A total of 263 diarrheic pediatric patients from Alamura Health Center were enrolled for the study with a mean and standard deviation of age 6.8 ± 3.7 years. The frequency and percentage of pediatrics age range enrolled for the study were 0–4, 88 (33.5%); 5–9, 103 (39.2%); and 10–14, 72 (27.4%). An almost equal ratio of male to female was enrolled for the study (130:133). Regarding the residence, most of the study subjects 155 (58.9%) were from an urban area and 108 (41.1%) patients were from a rural area. Concerning the educational status of the mothers of pediatric patients, most of them were educated (81%) ranging from reading and writing to university graduate level and the rest 19% were illiterates. The marital status of their mothers: 178 (67.7%) were married, 43 (16.3%) divorced and 41 (15.6%) widowed. The mean and standard deviation of the family size was 5.6 ± 1.9 persons. The average income of the family was 3743.3 ± 2568.1 Ethiopian birr. Most of the study participants have a large family size with a relatively low income of <1500 birr per month and from this number, the diarrhea positive was 12 (57.1%; Table 2).
The magnitude of Shigella and Salmonella
The overall magnitude of
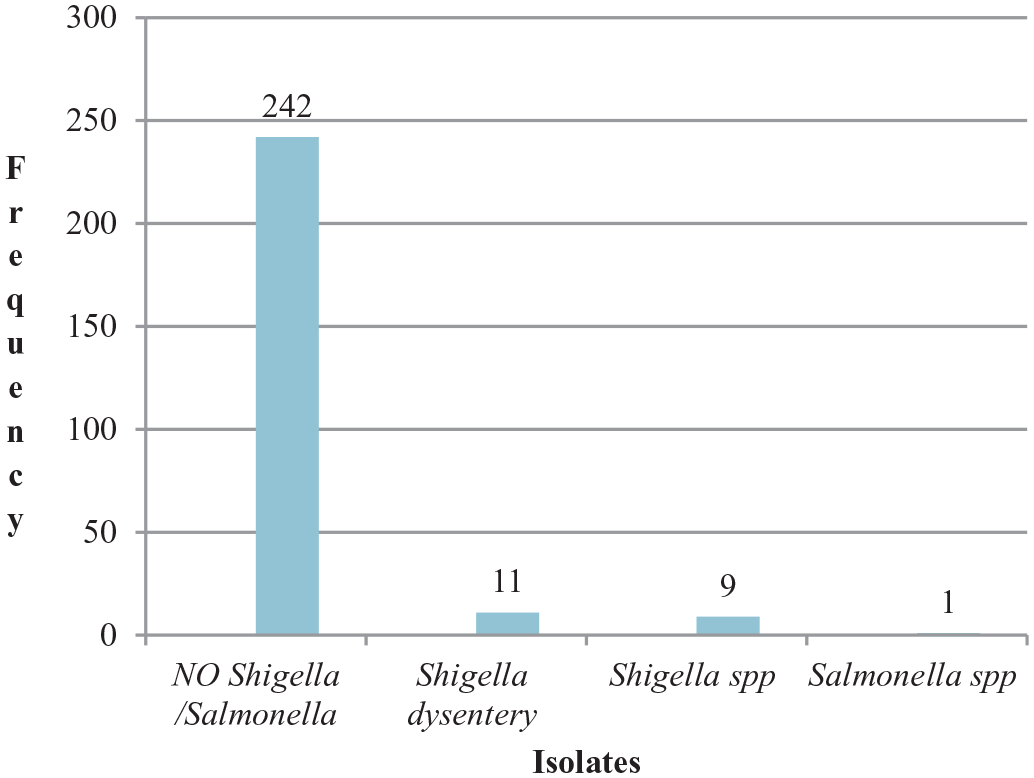
Magnitude of
Salmonella typhi
A single
Other Shigella species
Shigella dysenteriae
Antimicrobial susceptibility profile of

S: sensitive; I: intermediate; R: resistant; AMP: ampicillin; COT: co-cotrimoxazole; CIP: ciprofloxacin; CRO: ceftriaxone; CAZ: ceftazidime; GEN: gentamicin; CAF: chloramphenicol; CRX: cefuroxime; AUG: augmentin; TAT: tetracycline.
Associated risk factors
Among the study participants, 162 (61.6%) patients showed a history of diarrhea; of these, 17 (81.0%) were positive for current infection. Of all diarrheic children, the type of diarrhea was watery for 111 (42.2%), mucoid for 103 (39.2%) and bloody for 49 (18.6%). Children with mucoid diarrhea were more 18 (85.7%) as compared to the rest of the patients. Most of the children, 170 (64.6%), had diarrhea once a day and most of the bacteria, 11 (52.4%), was isolated from these patients. Most of the study subjects used pipe water, 159 (60.5%), for drinking, and the children infected in these categories were more than 17 (81.0%).
Regarding hand-wash, 221 (84.0%) affected children practiced hand-washing after defecation and the rest 42 (16.0%) wash their hands only for some time. Among the 16% of patients who wash their hands only for some time, nearly 95.2% of the patients were affected. Most of the food taken by the children before the illness was cooked food, 82 (31.2%), even if the bacterial infection was dominantly isolated from children who ate food at night, 8 (38.1%). Most of the children enrolled in the study were those who stored their food in closed containers, 223 (84.8%); lack of hand-wash before and after meal, 178 (67.7%); wash food containers, 157 (59.7%); well-nourished, 238 (90.5%); vaccinated, 202 (76.8%); and had animal contact 137 (52.1%). Correspondingly, most of the bacteria were isolated from those who stored food in an open container, 16 (76.2%); lack of hand-wash after or before meal, 15 (71.4%); washing of food container for some time, 17 (81.0%); well-nourished, 18 (85.7%); vaccinated, 14 (66.7%); and had animal contacts, 13 (61.9%) (Table 2).
Bivariate analysis of socio-demographic characteristics and clinical data of diarrheic pediatric patients in the Alamura Health Center, Southern Ethiopia, 2019.

COR: crude odds ratio; CI: confidence interval; AOR: adjusted odds ratio; ETB: Ethiopian Birr.
The bivariate analyses indicate that family with monthly income >1500 (crude odds ratio (COR) = 2.250, 95% CI: 0.86–5.902, p = 0.099), educational status of mother who can read and write (COR = 5.170, 95% CI: 0.62–43.05, p = 0.129), previous history of diarrhea (COR = 0.35, 95% CI: 0.115–0.078, p = 0.067), watery diarrheal type (COR = 11.69, 95% CI: 0.988–138.44, p = 0.051), mucoid (COR = 16.75, 95% CI: 2.130–131.67, p = 0.007) were the candidate variables for multivariable analysis.
Similarly, those who used pipe water source (COR = 2.993, 95% CI: 0.978–9.16, p = 0.055), wash hands of their child after toilet for some time (COR = 200, 95% CI: 25.602–1562.348, p = 0.000), store food in open containers (COR = 29.1, 95% CI: 9.78–86.37, p = 0.000) washing habit of food containers for some time (COR = 7.306, 95% CI: 2.38–22.4, p = 0.001) were candidate variables for multivariable analysis with p value of ⩽0.25 (Table 2).
However, in multivariate analysis, after adjustment, those who had a habit of washing the hands of children after toilet use for some time (adjusted odds ratio (AOR) = 235.1, 95% CI: 20.9–2643.3, p = 0.000) and store cooked food in an open container (AOR = 36.44, 95% CI: 5.82–228.06, p = 0.000) showed a statistically significant association of
Discussion
The overall magnitude of
In this study, 7.6% (4.6%–11.0%) of
A single
Our study revealed that the highest rates of antibiotic resistance of
Studies have shown that
The clinical variables showed that there are high rates of infection associated with mucoid diarrhea with a history of diarrhea, no malnutrition. However, none of these variables were statistically associated. Contrasting to our finding, the type of diarrhea with watery consistency from Bahir Dar was with high rates and statistically associated.
33
Similarly, a study from Ambo showed that mucoid diarrhea was with higher rates of infection.
35
Our study also showed that those who have taken vaccination were highly affected, which is in disagreement with the study reported from Ambo, which spells that children who were not vaccinated were at higher risk and significantly associated.
35
Host factors associated with malnutrition, such as a compromised immune system, environmental enteric dysfunction and enteric microbiome may predispose malnourished children to be more severe to disease infection.68–71 Children with malnutrition may also be more likely to live in households of low socioeconomic status where poor access to clean water,72,73 sanitation and hygiene may expose them to greater fecal microbial loads and a higher risk of pathogens associated with mortality such as
Behavioral factors such as the source of water from the pipeline, washing of hands after defecation for some time, consumption of food before illness, storing of food in open containers for later use, not washing hands before and after a meal and cleaning of cooking containers for some time constitute associate factors for the infection. However, the multivariate analysis showed that those who had a habit of washing hands for some time as compared to those who practice hand-washing always were at risk of infection. This can be justified that regular hand-washing using detergent is important for the prevention of
Limitation of study
Our study does not indicate the total magnitude of
It does not identify bacteria at the species level due to a lack of anti-sera in the local market.
Conclusion
Our study indicated that there was a high rate of Shigellosis and incidence of single
Supplemental Material
sj-pdf-1-smo-10.1177_20503121211009729 – Supplemental material for Magnitude, risk factors and antimicrobial susceptibility pattern of Shigella and Salmonella , among children with diarrhea in Southern Ethiopia: A Cross-sectional Study
Supplemental material, sj-pdf-1-smo-10.1177_20503121211009729 for Magnitude, risk factors and antimicrobial susceptibility pattern of
Footnotes
Acknowledgements
We would like to acknowledge Hawassa University, SNNPR Health Bureau, Alamura Health Center and Hawassa University Comprehensive Specialized Hospital for their co-operation and study participants at large.
Author contributions
M.H., T.A., B.T., E.M., Z.B. equally conceived the idea, developed the proposal, collected the data, performed the analysis and prepared the manuscript; T.A. and Z.B. has made a final edition of the document. All authors have read and approved the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from *Hawassa University College of Medicine and Health Science Institutional Review Board (IRB/094/11)*.
Ethical clearance
The study was conducted after obtaining formal permission from the Southern Nation Nationality and People Regional Health Office, Hawassa City Administration Health Office, Alamura Health Center Manager and Laboratory Head. The patients were included in the study only when their parents or caretakers agree to the investigation and sign the consent. Culture results and antimicrobial susceptibility results were communicated to the concerned bodies in the health center within 72 h.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported for data collection by Hawassa University.
Informed consent
Written informed consent was obtained from all subjects before the study.
Availability of data and material
All the data supporting the findings can be obtained from the corresponding author.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
