Abstract
Background:
The success of anti-tumor necrosis factor (TNF) drug strategies in the treatment of inflammatory bowel disease (IBD) is altered by the development of anti-drug antibodies that reduce their efficacy. Studies have shown that the HLA-DQA1⋆05 allele increases the risk of immunogenicity to anti-TNF drugs approximately twofold.
Objective:
Analyze whether the presence of the HLA-DQA1⋆05 allele is associated with the development of immunogenicity and to evaluate the disease response to anti-TNF drugs (infliximab (IFX) and adalimumab (ADA)), according to the presence of this allele.
Design:
This is an observational retrospective cohort study, single center, to determine the impact of HLA-DQA1⋆05 on disease activity in patients with IBD at the Hospital Universitario Virgen Macarena.
Methods:
In total, 200 IBD patients were included: 109 treated with IFX and 91 with ADA. Data were collected using the computerized medical records from the DIRAYA program of the Servicio Andaluz de Salud. Response—defined as improvement—and remission—defined as the disappearance of symptoms and analytical/endoscopic signs—were assessed using activity indices (partial Mayo, Harvey–Bradshaw) in all patients. Anti-TNF drug levels were also determined, as well as the presence or absence of anti-IFX and anti-ADA antibodies. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement.
Results:
The HLA-DQA1⋆05 haplotype was present in 70 (35%) patients, including 39 (36%) treated with IFX and 31 (34%) with ADA. The risk of withdrawal, intensification, as well as antibody development, was higher in patients carrying the allele and on treatment with IFX or ADA.
Conclusion:
In our study, we demonstrated that there is an increased risk of immunogenicity in patients carrying the HLA-DQA1⋆05 genotype, which would support the idea of screening for this genetic variant before starting anti-TNF therapy, as its prevalence is high in the general population and increases the risk of treatment discontinuation due to loss of response.
Plain language summary
Anti-TNF drugs are commonly used to treat inflammatory bowel disease (IBD), but their effectiveness can be reduced if patients develop antibodies against them. This study investigated whether a specific gene, HLA-DQA1⋆05, increases the likelihood of forming these antibodies and affects the response to anti-TNF drugs, such as infliximab (IFX) and adalimumab (ADA).The study analyzed 200 IBD patients, including those treated with IFX and ADA. Researchers assessed drug levels, the presence of antibodies, and how well patients responded to the treatment. They found that 35% of the patients carried the HLA-DQA1⋆05 gene. Those with this gene had a higher risk of treatment issues, such as needing to stop or switch medications, and were more likely to develop antibodies.The findings suggest that screening for the HLA-DQA1⋆05 gene before starting anti-TNF therapy could be beneficial. Identifying patients with this gene might help predict who is at higher risk for treatment failure and allow for better management of their care.
Introduction
Despite the emergence of alternative therapeutic pathways, anti-tumor necrosis factor (TNF) alpha remains the mainstay of biological treatment in inflammatory bowel diseases (IBD) and is most often prescribed as a first-line therapy. Infliximab (IFX) and adalimumab (ADA) are both indicated in Crohn’s disease (CD) and ulcerative colitis (UC),1,2 and IFX has shown greater efficacy when initially given in combination with an immunosuppressant.3,4 However, many patients will experience primary failure or secondary loss of response, possibly through a mechanism of immunization. Early screening of patients at higher risk of anti-TNF failure would allow to elect the best personalized treatment according to each patient’s characteristics, but we lack reliable predictors. In this context, the PANTS study was conducted a few years ago to identify clinical and pharmacokinetic factors that could predict primary non-response to anti-TNF in CD patients. They highlighted that low drug concentrations and immunogenicity were associated with treatment failure, with a bidirectional negative relationship between them both, most likely via clearance of the drug, the risk being mitigated by combined therapy with a thiopurine or methotrexate. 5
A genome-wide association study was also performed on the PANTS study’s patients and first identified that the HLA-DQA1⋆05 variant could significantly increase the risk of immunogenicity toward IFX and ADA. 6 The human leucocyte antigen (HLA) complex plays a key role in immune tolerance. It is speculated that an aberrant response to bacteria mediated by the HLA genes occurs during IBD. Furthermore, the HLA complex has been implicated in the response to various medications. 7 Since 2020, few other publications, in the form of peer-reviewed articles or congress abstracts, have evaluated in IBD patients undergoing an anti-TNF course the role of HLA-DQA1⋆05 on immunogenicity or treatment failure, with contradictory conclusions.8–18 However, two meta-analyses agreed on its association with immunogenicity and secondary loss of response.19,20 In fact, authors have underlined the potential role of screening HLA-DQA1⋆05 carriage particularly in patients with contraindication to immunosuppressants, to guide decisions toward anti-TNF monotherapy or another treatment option. That could also be a helpful tool before the withdrawal of immunosuppressant in the context of a de-escalation.6,19 Nevertheless, no study has yet evaluated the existence of an association between HLA-DQA1⋆05 carriage and levels of serum anti-TNF concentrations.
As the current data remain scarce and still have been insufficient to provide clear guidance about HLA-DQA1⋆05 carriage in IBD patients undergoing treatment with an anti-TNF, we aimed to carry out additional work in our tertiary center, investigating its impact on immunogenicity and treatment following course (intensification or discontinuation). Moreover, we paid particular attention to its impact on serum drug concentration since, to our knowledge, no study has yet evaluated it during follow-up.
Methods
Study design
We conducted an observational, retrospective, single-center study, including adult outpatients of Sevilla’s University Hospital Virgen Macarena diagnosed with IBD, having received or currently receiving a first anti-TNF treatment with IFX or ADA, completing at least 2 years of follow-up. Data were collected using the computerized medical records from the DIRAYA program of the Servicio Andaluz de Salud. We reported demographic and clinical information such as gender, age, and smoking status, at the time of first anti-TNF initiation. History of IBD included disease localization and phenotype (CD), disease extension (UC), according to Montreal’s classification, existence of perineal disease (CD). We investigated the concomitant use of an immunosuppressant. Trough levels of IFX or ADA were also collected, with the therapeutic range defined as 3–7 µg/mL for IFX and 5–12 µg/mL for ADA, as well as anti-drug antibodies when the anti-TNF level was undetectable.
Genotypic and drug-level analysis
We performed a single screening of the HLA-DQA1⋆05 genotype. DNA was extracted using the MagCore Genomic DNA Whole Blood commercial kit (RBC Bioscience Corp., Taipei, Taiwan). The amplification and reverse hybridization were realized following the INNO-LiPA protocol for HLA-DQA1 alleles (Fujirebio Iberia, Barcelona, Spain).
Tests used to measure anti-drug levels and antibodies were performed using an enzyme-linked immunosorbent assay with Progenika kits (PROMONITOR®) (Grifols Diagnostic Solutions Inc., CA, UU). The sample used for both the measurement of anti-IFX and anti-ADA levels is serum. Sera can be stored or transported at refrigerator (RT) (20–25°C) for up to 2 days. Alternatively, sera can be stored at 2–8°C for up to 5 days; if serum will not be processed within 5 days, it should be frozen at −20°C or lower. If frozen sera are used, they have to be mixed thoroughly before analysis.
Study objectives
We aimed to analyze the impact of the HLA-DQA1٭05 haplotype on immunogenicity, anti-TNF trough levels, intensification or discontinuation of anti-TNF, and evolution of clinical scores. Immunogenicity was defined as at least one detectable amount of anti-drug antibodies ⩾10 AU/mL.
Statistical analysis
Statistical analysis was performed using IBM SPSS Statistics 25 for Windows (IBM Corporation, Armonk, New York, USA). Categorical variables were expressed as percentages, while continuous variables were presented as the median with an interquartile range. Comparison of qualitative variables was conducted employing the Chi-square test, while the comparison of quantitative variables was conducted employing either the Student’s t-test or the Wilcoxon rank sum test, based on the normality criteria. A two-sided p-value of 0.05 or less was considered indicative of statistical significance.
The fixed-effects Q statistic was used to perform a test of differential heterogeneity of the effect of combined therapy conditional on the HLA-DQA1⋆05. A survival analysis using Kaplan–Meier curves was conducted to estimate the time to anti-TNF discontinuation according to the presence of HLA-DQA1⋆05 as an independent variable, the log-rank test allowing to compare equality of distribution in both groups.
Ethical statement
This study was approved by the research ethics committee of Sevilla’s university hospitals Virgen Macarena and Virgen del Rocio (internal reference 2379-N-20) in May 2022. The patients were included after providing informed written consent.
Results
Demographic characteristics and IBD history
We included 200 patients in our study. Table 1 illustrates their demographic characteristics and IBD history at the time of first anti-TNF initiation. They were a majority of men (61.5%) with a predominance of CD (60.5%). IFX was the most frequently used anti-TNF agent (54.5%) and almost all patients were receiving an immunosuppressant.
Demographic characteristics and IBD history.

CD, Crohn’s disease; HLA, human leucocyte antigen; IBD, inflammatory bowel disease; TNF, tumor necrosis factor; UC, ulcerative colitis.
Frequency of HLA-DQA1⋆05 carriage
The HLA-DQA1⋆05 haplotype was present in 70 (35%) patients, including 39 (36%) treated with IFX and 31 (34%) with ADA. Demographic characteristics and IBD history were not different in patients with or without the HLA-DQA1⋆05 haplotype (Table 1).
HLA-DQA1⋆05 carriage: impact on immunogenicity and serum anti-TNF concentration
Data about anti-drug antibodies and trough levels of anti-TNF are presented in Table 2. Forty-one (20.5%) patients had at least one detectable amount of anti-drug antibodies ⩾10 AU/mL during follow-up. Patients with HLA-DQA1⋆05 haplotype were significantly more likely to present anti-drug antibodies than patients without (35.7% vs 12.3%, p < 0.001). The difference remained statistically significant when analyzing separately patients treated with IFX (35.9% vs 12.9%, p = 0.006) and ADA (35.5% vs 11.7%, p = 0.009). Also, trough levels of anti-TNF were more frequently infratherapeutic in patients with HLA-DQA1⋆05 haplotype beyond 6 months of treatment with anti-TNF, reaching significance for the M6–M12 and after M24 periods. The difference remained significant for each anti-TNF in the M6–M12 period. Regarding the impact of immunosuppressive treatment, we did not find a different proportion of combined therapy in HLA-DQA1⋆05 carriers developing or not anti-drug antibodies, regardless of duration (Table 3). Also, we did not detect any significant heterogeneity in the effect of HLA-DQA1⋆05 on the immunogenicity rate in the presence or absence of concomitant immunosuppressant (HR 2.01 (1.57–2.58) vs HR 1.75 (1.37–2.22), phet = 0.14). The proportion of patients facing the apparition of anti-drug antibodies was significantly higher in patients having at least one infratherapeutic measure of the drug than patients having no infratherapeutic measure, whether for IFX (31.7% vs 8.3%, p = 0.0121) or ADA (40.0% vs 10.9%, p = 0.0053).
Immunogenicity and anti-TNF levels according to HLA-DQA1⋆05 carriage.

CD, Crohn’s disease; HLA, human leucocyte antigen; IBD, inflammatory bowel disease; TNF, tumor necrosis factor; UC, ulcerative colitis.
Combined therapy with immunosuppressants according to detection of anti-drug antibodies in HLA-DQA1⋆05 carriers.

ADA, adalimumab; HLA, human leucocyte antigen; IS, immunosuppressants; UC, ulcerative colitis.
HLA-DQA1⋆05 carriage and impact on treatment’s course: intensification, discontinuation
During follow-up, 102 (51%) patients experienced intensification of treatment, and 101 (50.8%) experienced discontinuation of treatment. Intensification occurred significantly more often in patients with HLA-DQA1⋆05 than in patients without (60.0% vs 46.2%, p = 0.042), as well as discontinuation (72.5% vs 39.2%, p < 0.001). The reason for discontinuation was most often loss of response in HLA-DQA1⋆05 carriers and non-carriers (78.0% and 68.6%, respectively), then intolerance (10.0% and 7.8%, respectively), without significant difference. When analyzing separately patients treated with IFX and ADA, we also found a significantly higher discontinuation rate in patients with HLA-DQA1⋆05 haplotype up to 24 months (64% vs 24.5%, p = 0.001, for IFX, and 52.2% vs 24.4%, p = 0.030, for ADA, respectively). Survival analysis for treatment persistence is illustrated in Figure 1 for both anti-TNF, IFX only, and ADA only.

Survival analysis of time to discontinuation of treatment according to the presence or absence of HLA-DQA1⋆05 haplotype for both anti-TNF (a), infliximab only (b), and adalimumab only (c).
Multivariate analysis
A multivariate analysis was performed (up to 24 months), with the studied variables: the development of immunogenicity, drug withdrawal, and the possibility of drug intensification, considering potential confounding factors (age, gender, smoking status, type of IBD, and presence or absence of the disease and presence or not of the allele studied). The results were that the presence of the allele increases the risk of developing immunogenicity by up to sevenfold (1943 immunogenicity (1943–25,929), a higher probability of drug withdrawal (HR = 2943–25,929), and withdrawal (HR = 2.732 (1.608–4.640), p < 0.001) and a higher probability of having to intensify treatment, specifically, 2.156 times (1.334–3.485). In the latter case, the greatest intensification would occur in patients with CD (Tables 4–6). Age, as can be seen, also seems to be related, although it would not be of clinical significance as the HLA allele is a DNA variation and therefore not modifiable by the age of the patients.
Cox regression for immunogenicity (treatment time up to 24 months).
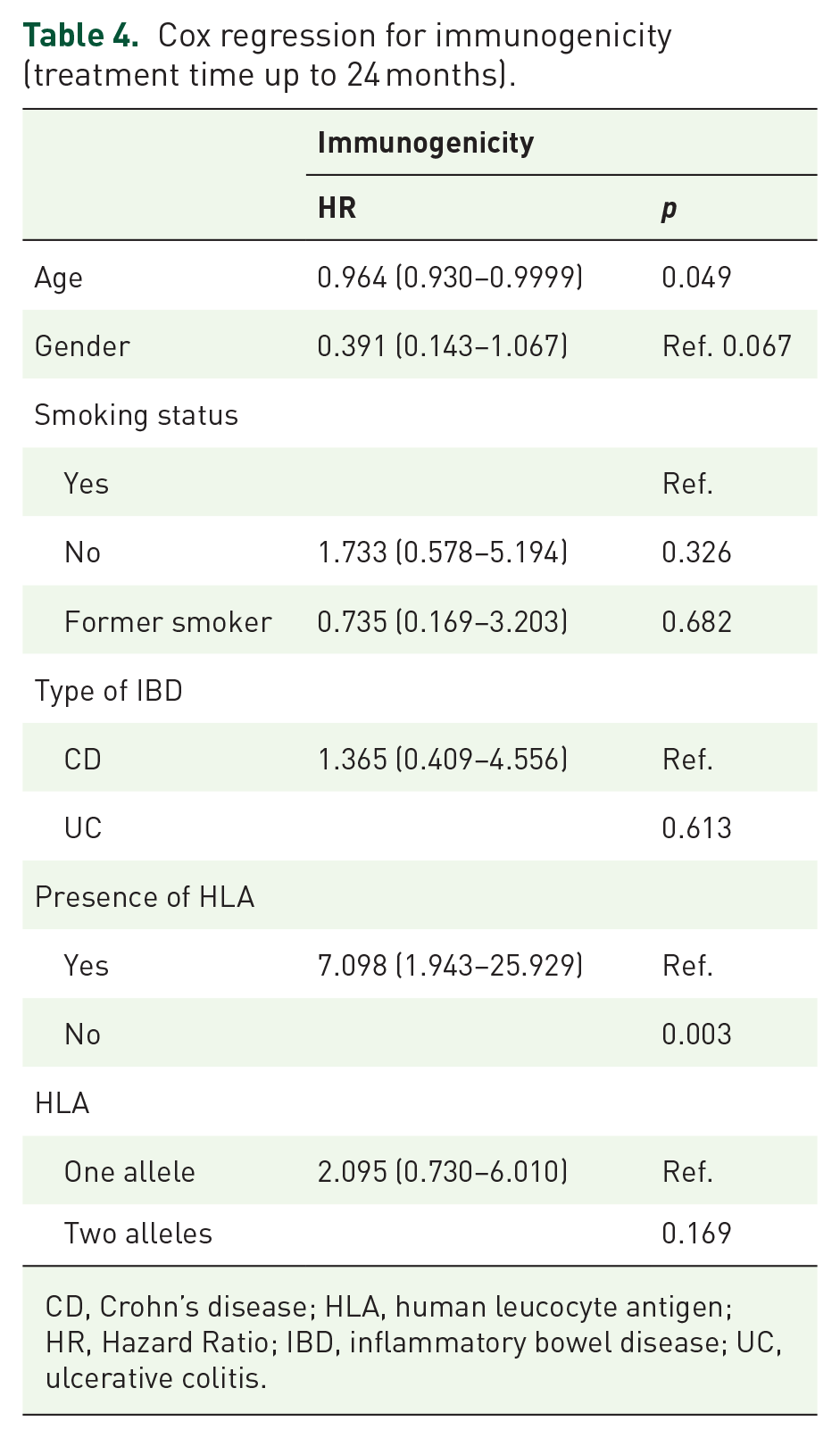
CD, Crohn’s disease; HLA, human leucocyte antigen; HR, Hazard Ratio; IBD, inflammatory bowel disease; UC, ulcerative colitis.
Cox regression for drug withdrawal (treatment time up to 24 months).

CD, Crohn’s disease; HLA, human leucocyte antigen; IBD, inflammatory bowel disease; UC, ulcerative colitis; HR, Hazard Ratio.
Cox regression for intensification (treatment time up to 24 months).

CD, Crohn’s disease; HLA, human leucocyte antigen; IBD, inflammatory bowel disease; UC, ulcerative colitis; HR, Hazard Ratio.
Discussion
Since its first description from the PANTS study, the HLA-DQA1⋆05 haplotype has been a candidate of interest to improve the management of anti-TNF in IBD and therefore go further toward the personalized care of patients. However, some recent results failed to report an association of this variant with anti-drug antibody formation11,12 or drug persistence.11,17,18 Our study brings new insight into this controversial topic.
We reported in our cohort a 35% of HLA-DQA1⋆05 carriers, which is consistent with all literature data of 31%–46% carriage in European or North American countries.6,10–18 In such patients, we demonstrated a higher rate of anti-drug antibodies, for both IFX and ADA, alongside more frequent infratherapeutic anti-TNF levels. The concomitant immunosuppressive treatment did not reduce the immunogenicity rate in the presence of the HLA-DQA1⋆05 haplotype. Anti-TNF intensification and discontinuation occurred more often in the HLA-DQA1⋆05 carriers, without difference between patients treated with IFX or ADA. We did not identify any tolerance issue in HLA-DQA1⋆05 patients, as the proportion of discontinuation for intolerance was comparable in the two subgroups.
Regarding the impact of HLA-DQA1⋆05 on anti-TNF treatment’s course, our results are in line with the literature, as a rate twice as high of loss of response is generally assumed, based on the two available meta-analyses.19,20 There are no previous data about clinical scores and biochemical markers, only one study to our knowledge investigated the rate of clinical remission that was similar in carriers and non-carriers. 18
We confirmed a positive role of HLA-DQA1⋆05 on the development of immunogenicity, specifically seven times more risk. This value is higher than that found in other studies, such as Sazonovs (almost twice the risk) or, more recently published, in Solitano et al. (1.75 times more). However, we found that the apparition of antidrug antibodies was associated with more frequent infratherapeutic levels of anti-TNF and that infratherapeutic levels of anti-TNF occurred preferentially in HLA-DQA1⋆05 carriers. Thus, the mechanism of HLA-DQA1⋆05 carriage’s effect on immunogenicity is unclear. Kennedy et al. 5 already highlighted in the PANTS study a bidirectional negative association of immunogenicity and low drug concentrations, as detection of anti-drug antibodies was an independent risk factor of further low anti-TNF level, and conversely low anti-TNF level was the major independent risk factor for further development of immunogenicity. In the same way, Dubinsky et al. 21 have demonstrated that a dashboard-driven strategy to optimize IFX to achieve target drug concentrations in IBD patients was able to lead to a lower rate of anti-drug antibody formation and a longer persistence of anti-TNF. In fact, a new concept of “optimized monotherapy” rather than combination therapy is emerging since several authors concluded that proactive drug therapeutic monitoring (pTDM) enabled to reach higher IFX concentrations with subsequent better drug durability, in combination with azathioprine appearing to improve anti-TNF efficacy only through increasing pharmacokinetic features of IFX.22–24 So, unlike us, other authors have mostly described that the use of immunosuppressants in HLA-DQA1⋆05 carriers could reduce immunogenicity,6,8,10,12 but they did not investigate the serum drug concentrations. On another hand, two recent studies evaluated the influence of the HLA-DQA1⋆05 variant in patients with IBD and anti-TNF treatment with pTDM. They reported a low rate of immunogenicity that was associated with infratherapeutic drug levels and failure to achieve drug target concentration, but not with HLA-DQA1⋆05.11,18 It, therefore, seems that, including in HLA-DQA1⋆05 carriers, pTDM improves anti-TNF concentrations and thus corrects the negative influence of this haplotype.
Overall, our results support the generally admitted idea of screening for the HLA-DQA1⋆05 variant as a predictive factor before anti-TNF initiation, since its prevalence is high in the general population and it increases the risk of treatment discontinuation for loss of response. However, it cannot be considered sufficient in itself to decide whether to use an anti-TNF or another biologic, combination therapy or a monotherapy, as many other elements related to patient and IBD features need to be taken into account. As Solitano et al. 19 pointed out the positive predictive value of developing anti-drug antibodies in patients carrying HLA-DQA1⋆05 is rather low (30%), whereas its high negative predictive value of 80% should reassure us about prescribing anti-TNF monotherapy in a non-carrier patient. In light of our results and the literature data, we hypothesize that a pTDM should be proposed in patients detected with this variant if an anti-TNF is needed rather than another treatment class (Figure 2). However, considering the other biologics, the HLA-DQA1⋆05 variant does not seem to influence ustekinumab and vedolizumab efficacy25,26 while there are no data in anti-JAK or anti-IL23 patients.

Proposed decision tree in place of HLA-DQA1⋆05 screening.
The principal strength of our study was the analysis of serum anti-TNF concentrations, which helped us formulate a new proposal on the role of HLA-DQA1⋆05 screening. The main limitation was its retrospective design which resulted notably in heterogeneous anti-TNF levels sampling time. In addition, our definition of immunogenicity of at least one detectable amount of anti-drug antibodies ⩾10 AU/mL, with a low threshold, without considering transient antibodies and neutralizing versus non-neutralizing antibodies, may have overestimated the development of immunogenicity. At last, the results cannot easily be extrapolated to other populations outside Europe or North America, as the frequency of the HLA-DQA1⋆05 variant is not the same, but this can apply to all other published studies until now.
Conclusion
To conclude, our study emphasizes that the HLA-DQA1⋆05 haplotype is associated with more frequent immunogenicity and infratherapeutic levels of the drug in IBD patients undergoing treatment with IFX or ADA, resulting in higher intensification and discontinuation rates. Subsequently, in the era of personalized medicine and in the context of various options with advanced therapies rising, genetic screening could be of great utility to physicians before the initiation of a biologic to help decision-making.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241278145 – Supplemental material for Carriage of the HLA-DQA1⋆05 haplotype is associated with a higher risk of infratherapeutic drug concentration and higher immunogenicity in patients undergoing treatment with anti-TNF for inflammatory bowel disease
Supplemental material, sj-docx-1-tag-10.1177_17562848241278145 for Carriage of the HLA-DQA1⋆05 haplotype is associated with a higher risk of infratherapeutic drug concentration and higher immunogenicity in patients undergoing treatment with anti-TNF for inflammatory bowel disease by Pilar Navajas Hernández, Samer Mouhtar el Halabi, Ana Caridad González Parra, Teresa Valdés Delgado, Belén Maldonado Pérez, Luisa Castro Laria, Cloé Charpentier and Federico Argüelles-Arias in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
To Antonia Sáez for her willingness and great contribution in carrying out the statistical analyses. To the Biochemistry Department of our Hospital, especially Dr Concepción González, for all her support from the beginning of this project. To our Hospital Pharmacy Service, for always helping us in all the studies we undertake with drugs for inflammatory bowel disease.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
