Abstract
Background and Aims:
Skin eruptions are prevalent among patients with inflammatory bowel diseases (IBD), often associated with therapies and frequently leading to dermatological consults and treatment interruptions. We aimed to assess the impact of joint shared decision-making in a multidisciplinary (MDT) IBD-DERMA clinic.
Methods:
This retrospective cohort study assessed a consecutive group of patients with IBD who were referred for consultation in an MDT clinic at a tertiary referral center in Israel.
Results:
Over 1 year, 118 patients were evaluated in the MDT-IBD-DERMA clinic: 68 (57.6%) males; age – 35.2 ± 13.5 years, disease duration – 7.1 (interquartile range: 3.7–13.9) years; Crohn’s disease – 94/118 (79.6%). Skin eruption induced by an anti–tumor necrosis factor (TNF) were the most common diagnoses [46/118 (39%)], including psoriasiform dermatitis (PD) – 31/46 (67.4%) and inflammatory alopecia (IA) – 15/46 (32.6%). Of these, 18 patients (39.1%) continued the anti-TNF agent concomitantly with a topical or systemic anti-inflammatory agent to control the eruption. The remaining 28 patients (60.9%) discontinued the anti-TNF, of whom 16/28 (57.1%) switched to ustekinumab. These strategies effectively treated the majority [38/46 (82.6%)] of patients. Continuation of the anti-TNF was possible in a significantly higher proportion of patients with PD: 12/31 (38.7%) than only one in the IA group, p = 0.035. There was a higher switch to ustekinumab among the IA 7/15 (46.6%) compared with the PD 7/31 (22.6%) group, P = .09. Following IBD-DERMA advised intervention, IBD deteriorated in 9/4 6(19.5%) patients, 5/9 on ustekinumab (PD versus IA, P = NS).
Conclusion:
Shared decision-making in an integrated IBD-DERMA clinic allowed successful control of skin eruptions while preserving control of the underlying IBD in more than 80% of cases. Patients with IA profited from a switch to ustekinumab.
Introduction
Inflammatory bowel diseases (IBD) are chronic conditions often requiring lifelong treatment(s). 1 Dermatological manifestations are a common complication of both IBD and its therapy.2,3 Since the approval of the first anti–tumor necrosis factor (TNF) agent in 1997 for Crohn’s disease (CD) and subsequently for ulcerative colitis (UC), reports of skin eruptions in patients on these therapies have increased.3,4 These paradoxical eruptions include psoriasis, psoriasiform dermatitis, and other inflammatory skin conditions. 5 The lack of evidence-based guidelines or consensus statements regarding the optimal approach to these complications may lead to fragmentation of care provided by various specialists, poor interprovider communication, and lapses in therapy. Furthermore, it frequently places the burden of care navigation and coordination on the patient.
There is an increasing awareness of the importance of comprehensive care among patients with IBD.6,7 Multidisciplinary team (MDT) clinics can pull together interdisciplinary providers around the IBD patients’ population and improve the quality of care, patient satisfaction, and possibly, outcomes.8 –11 However, outcome data from such an approach in the context of dermatological complications in IBD is lacking. Appreciating the MDT clinics’ advantages, during 2018, we founded an integrated IBD-dermatology (IBD-DERMA) clinic. The IBD-DERMA clinic operates with two experts that see and evaluate patients together: an IBD expert (HY) and a dermatologist (LP) with expertise in immunological skin disorders.
This study aimed to report our experience at the integrated IBD-DERMA clinic and the impact of joint shared decision-making for patients with skin eruptions induced by anti-TNF agents.
Methods
This retrospective cohort study was conducted at the Rabin Medical Center, a large tertiary referral center in Israel for IBD and dermatology. We describe a consecutive cohort of patients with IBD who developed a dermatological complication and were referred for consultation at the MDT IBD-DERMA clinic by either a gastroenterologist or a dermatologist. Demographic, clinical, and laboratory data were documented, including IBD-related therapy. The dermatological assessment was performed at the index and follow-up clinic visits. Skin histopathology was performed as needed.
To optimize patient experience and outcomes while decreasing health care expenses, we scheduled all patients with skin eruption to be seen jointly in the clinic (two physicians sitting in the room) by the same IBD specialist (HY) and dermatologist (LP). A global payment model was used, which covered the multidisciplinary clinic visit.
Patients with a skin eruption induced by an anti-TNF were assessed for IBD activity and the impact of anti-TNF therapy on IBD control. IBD activity at each visit was determined based on standard clinical, laboratory endoscopic, and imaging tools if available. Patients were graded for IBD activity based on the physician’s global assessment (PGA) according to the IBD expert assessment as either: quiescent (PGA: 0), mild (PGA: 1), moderate (PGA: 2), or severe (PGA: 3).
Similar to evolving paradigms,12,13 for patients with a relatively mild skin eruption (involvement of less than 10% of body surface area except for functionally sensitive sites like face, palms and soles, skin folds, and genitals), who were considered to be dependent on the continuation of anti-TNF therapy for IBD control, we elected to spare the anti-TNF and to treat the eruption with either a topical anti-inflammatory agent (corticosteroidal ointments/intralesional injections, tacrolimus ointment, or phototherapy) or a systemic agent (oral steroids, methotrexate, acitretin, and apremilast) – continue anti-TNF strategy (i). For patients with severe skin eruptions, or if anti-TNF therapy was inadequate for IBD control (PGA ⩾ 2), we elected to discontinue the offending anti-TNF and based on individualized characteristics to add additional topical or systemic anti-inflammatory agent (s) or to switch to either a second anti-TNF or a switch out of class to ustekinumab – discontinue anti-TNF strategy (ii).
During follow-up visit(s), the assigned intervention’s impact on the dermatological condition and the IBD was defined and recorded.
Statistical analysis
Descriptive statistics for demographic and clinical characteristics include mean or median [interquartile range (IQR)] for continuous variables and frequency distributions for categorical data. The Student t test was used to compare continuous variables. Nonparametric distributed data were analyzed with the Mann–Whitney to compare continuous variables and chi-square tests or Fisher exact test to compare categorical variables.
Ethics
The local institutional review board approved the study protocol, waiving the requirement to obtain a signed informed consent form.
The reporting of this study conforms to the STROBE statement. 14
Results
Patients
Overall, 118 patients were evaluated and treated in the MDT-IBD-DERMA clinic over 1 year: 68 males (57.6%), age 35.2 ± 13.5 years with median disease duration of 7.1 years (IQR: 3.7–13.9). Ninety-four patients had CD (79.6% of the entire cohort), most with small bowel involvement (85.7%) and with either stricturing or penetrating (61.7%) disease, perianal involvement in 46.2%, reflecting a complicated disease course. Twenty-two patients had UC (18.6%), of whom 73.7% had extensive colitis. A quarter of the cohort had a history of IBD-related surgery, and 40.7% had at least one extraintestinal manifestation in addition to the dermatologic manifestation(s). Only 16.9% of patients had a history of dermatological conditions before the IBD diagnosis [mostly psoriasis (13%)]. Most patients (83.9%) were previously treated with a biological agent (78.8% with an anti-TNF) and 59% with an immunomodulator. Detailed data on patients’ characteristics are presented in Table 1.
Patients’ characteristics, N = 118.

IBD: inflammatory bowel diseases.
Dermatological diagnoses
The most common reason for patient referral was a skin eruption induced by an anti-TNF agent: 46/118 patients (39%). These skin eruptions manifested as either psoriasiform dermatitis (PD) involving the skin of body and extremities: diagnosed in 31/46 (67.4%) patients, or by scalp involvement with severe alopecia designated inflammatory alopecia (IA), diagnosed in 15/46 patients (32.6%). Representative examples of these eruptions are presented in Figure 1. PD was histologically characterized by PD with lymphohistiocytic infiltrate and eosinophils in the upper dermis and various degrees of interface changes and spongiosis and IA by similar findings combined with nonscarring alopecia. All other diagnoses encountered at our IBD-DERMA clinic are presented in Table 2. Aside from the anti-TNF-induced skin eruptions, most dermatological conditions did not require significant interventions that might have compromised IBD treatment and control.
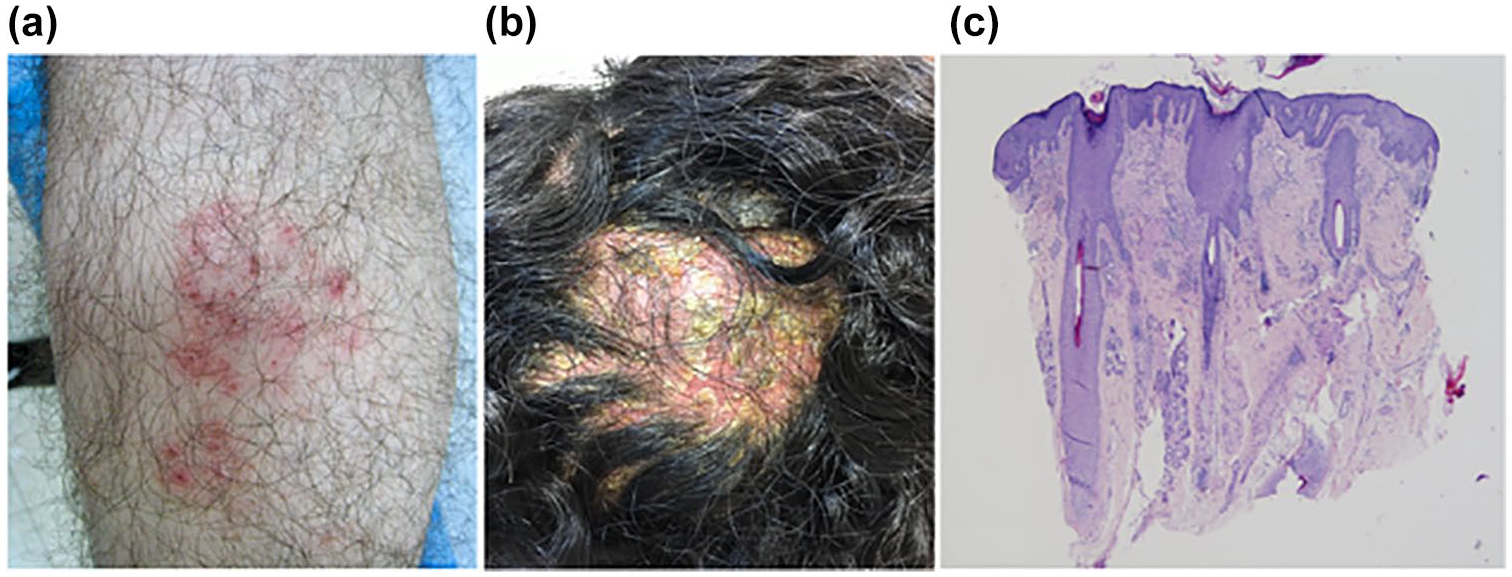
(a) Psoriasiform plaque on the shin of a 25-year-old man with Crohn’s disease. (b) Severe psoriasiform scalp inflammation with alopecia in a 29-year-old woman with Crohn’s disease. (c) Histopathology of the scalp lesion is shown in Figure 1(b), Psoriasiform epidermal hyperplasia, superficial dermal and peribulbar chronic inflammatory infiltrate, decreased number of sebaceous glands, miniaturization of hair follicles (H&E ×40).
Dermatological diagnoses, N = 118.

TNF: tumor necrosis factor.
Joint decision-making for skin eruptions induced by anti-TNFs
Characteristics of patients with skin eruptions induced by anti-TNFs are presented in Table 3. Like the entire cohort, most of this subgroup had CD (82.6%), most with a complicated phenotype (B2: 28.9%, B3: 26.4%, P: 44.7%); 14/46 patients were on infliximab, and 32/46 patients were on adalimumab. The mean duration of therapy with the offending anti-TNF until the first episode of the eruption was 37 ± 43 months, and there were no significant differences between infliximab and adalimumab (data not shown). Notably, at the index visit, 39.1% of this subgroup (18/46) had quiescent IBD (PGA-0), and 54% (25/46) active mild disease (PGA-1) while on anti-TNF therapy.
Characteristics of patients with anti-TNF induced skin eruption, n = 46.
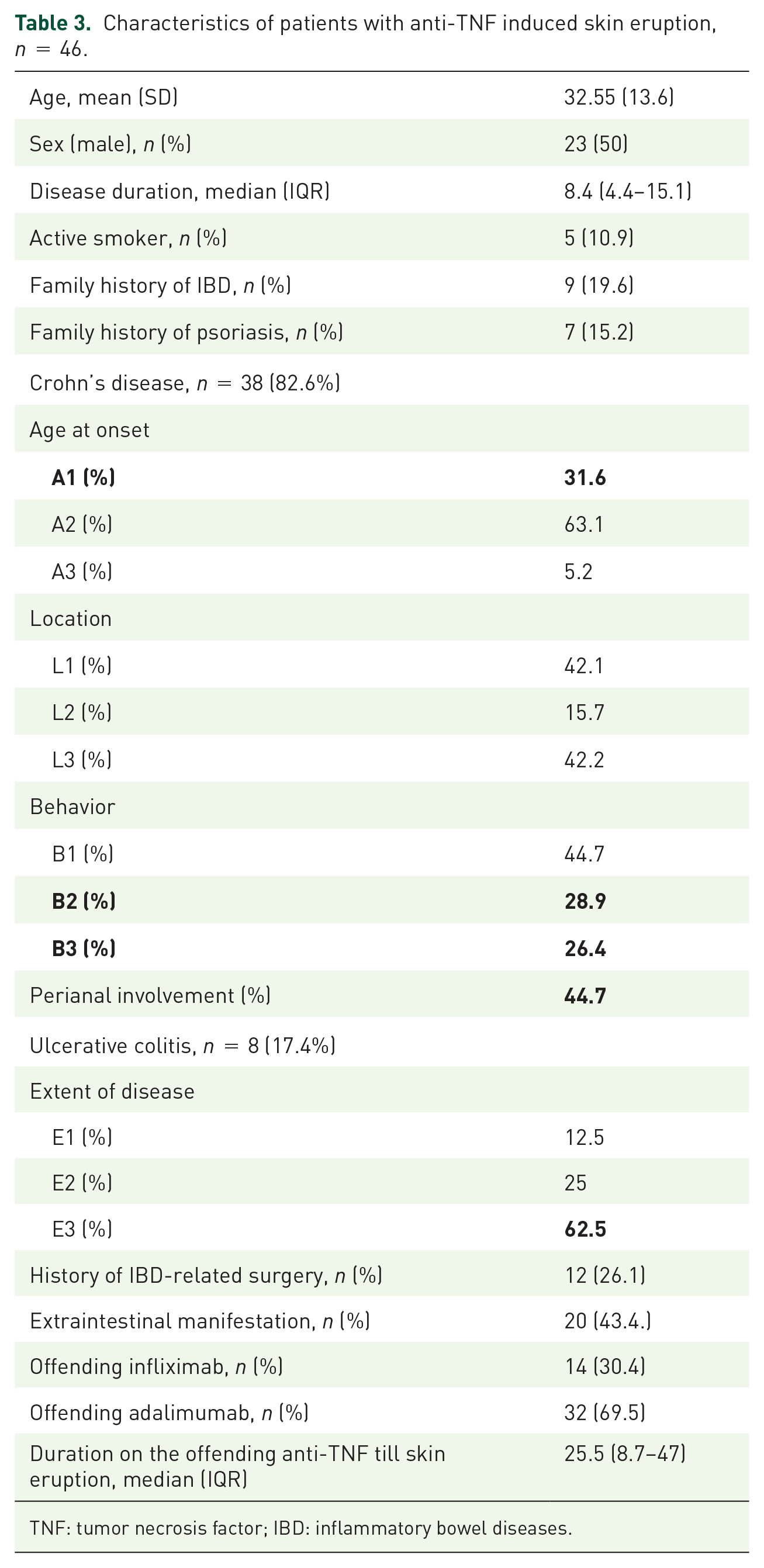
TNF: tumor necrosis factor; IBD: inflammatory bowel diseases.
As aforementioned, patients were treated according to one of two strategies: (1) 18/46 patients (39.1 %) continued the offending anti-TNF and concomitantly received either a topical or a systemic anti-inflammatory agent to control the skin eruption; (2) 28/46 patients (60.9%) discontinued the offending anti-TNF and were treated with or without a topical or systemic anti-inflammatory agent [16 patients (35%) were switched out of class]. An illustration of these elected strategies is depicted in Figure 2.

The proportion of patients elected to continue the anti-TNF, to discontinue the anti-TNF, or to switch out of class.
In 16/28 patients (57.1%), a switch out of class to ustekinumab was recommended after discontinuation of anti-TNF. At the switch, 13/16 patients had active IBD despite anti-TNF therapy. The remaining three patients were recommended ustekinumab either because of severe IA or following a failure of methotrexate prescribed by the referring physician.
Notably, one patient with UC was switched in class to a second anti-TNF before the index visit. That switch failed and worsened the skin eruption; thus, we recommended discontinuation of the anti-TNF and a switch to methotrexate in combination with vedolizumab to control the colitis. None of the patients assessed by the IBD-DERMA team were switched in class; as for all our patients, we anticipated that ustekinumab would be significantly advantageous over a second anti-TNF.
Overall, 38/46 patients (82.6%) were considered effectively treated by these strategies, either with significant improvement or complete resolution of the skin eruption; PD – 26/31 patients (83.8%) versus IA – 12/15 patients (80%), p = NS. Detailed treatment strategies and outcomes are presented in Figure 3. Among patients with PD diagnosis, it was possible to continue the offending anti-TNF in 12/31(38.7%), while only 1/15 patients in the IA group (6.7%) profited from that strategy p = 0.035. This was reflected in a higher switch to ustekinumab among the IA 7/15 (46.6%) compared with the PD 7/31 (22.6%) group, p = 0.09. In patients diagnosed with PD, improvement/resolution was seen within a couple of weeks after introducing the treatment intervention. In cases of IA, complete hair regrowth was seen only several months after switching to ustekinumab. Details on patients with an effective intervention based on dermatological diagnosis are presented in Table 4.

Detailed treatment strategies and outcomes for patients with skin eruptions induced by anti-TNFs.
Effective strategies for anti-TNF induced skin eruption among patients based on dermatological diagnosis, n = 38.
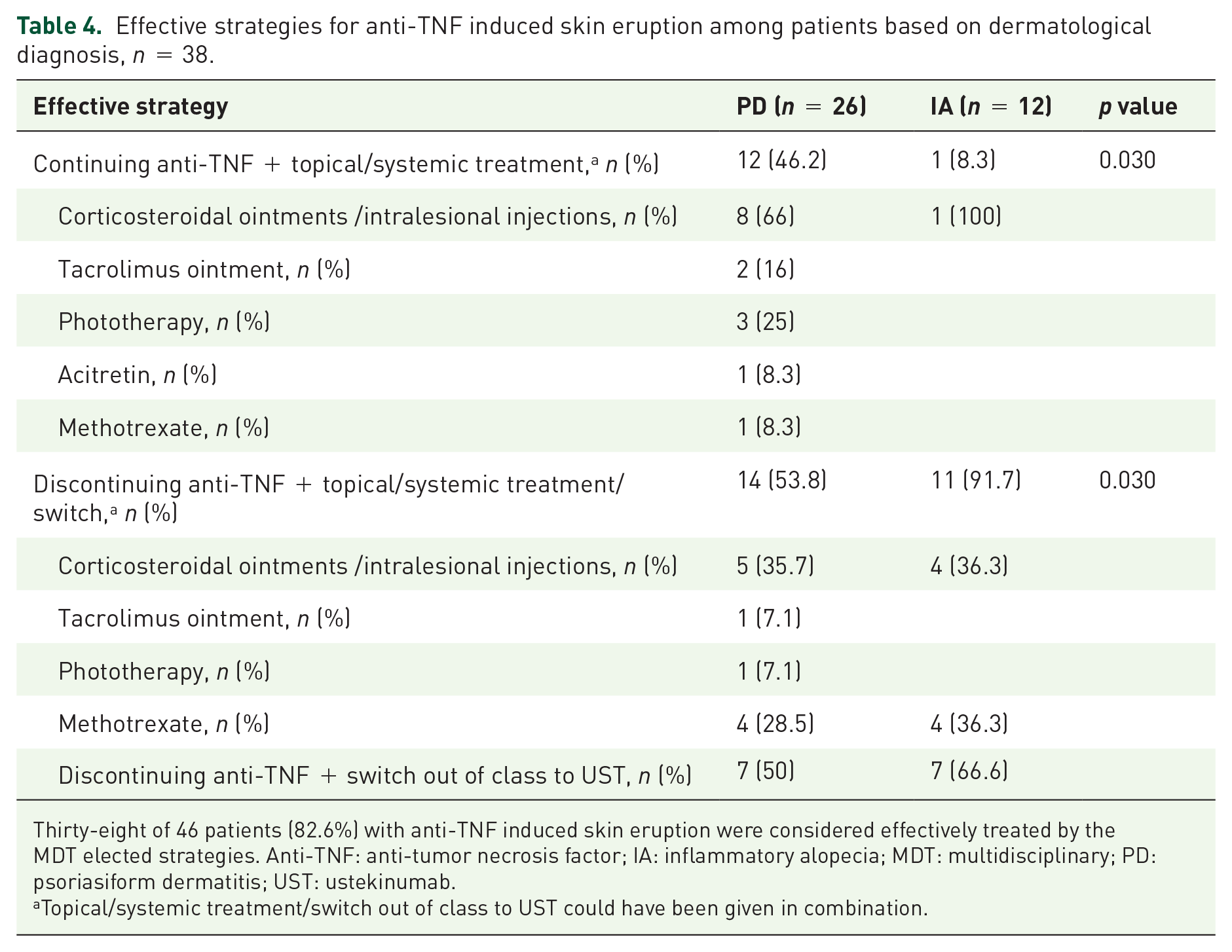
Thirty-eight of 46 patients (82.6%) with anti-TNF induced skin eruption were considered effectively treated by the MDT elected strategies. Anti-TNF: anti-tumor necrosis factor; IA: inflammatory alopecia; MDT: multidisciplinary; PD: psoriasiform dermatitis; UST: ustekinumab.
Topical/systemic treatment/switch out of class to UST could have been given in combination.
Finally, following these interventions, almost 90% of patients were either in sustained remission [21/46 patients (45.6%)] or maintained mild IBD [20/46 patients (43.5%)] for a median follow-up of 16 months (IQR: 7–28). In 9/46 patients (19.5%), IBD deterioration was noted compared with the status at the index visit (PD versus IA, p = NS); of these, five patients were switched from an anti-TNF to ustekinumab (corresponding to 35% of all patients who were switched to ustekinumab in this cohort).
Discussion
In this study, we have shown that clinical decisions taken by a dedicated team of an integrated IBD-DERMA clinic provided effective treatment strategies for over 80% of patients with anti-TNF-induced skin eruptions while maintaining good control of the IBD.
IBD is a complex disease frequently involving multiple organ systems, requiring treatment by different subspecialties. This complexity may lead to poor treatment integration, as each provider might have diminished insight into disease management decisions that do not fall under a single area of expertise. Such fragmentation of care frequently leads to contradictory recommendations by different providers. A multidisciplinary clinic has the advantage of treating IBD in a more holistic approach. Experts from various fields can provide coherent management recommendations while utilizing an expanded toolbox that different specialists bring to IBD patients’ care. By adopting such an approach to an IBD-DERMA clinic and setting an explicit schema for intervention strategies and treatment targets (though not rigid), we could provide practical therapeutic recommendations to a complicated cohort of patients with IBD with significant skin eruptions. We achieved good results both in treating the dermatological complications while maintaining good control of the IBD. To the best of our knowledge, this is also the largest cohort of patients with paradoxical skin eruption switched to ustekinumab and the most extensive series of patients with IA. This debilitating condition may develop secondary to anti-TNF therapy.
In our cohort, both anti-TNF-induced PD and IA were frequent, affecting almost 40% of the patients referred. Although PD is a well-established phenomenon,3,12,15,16 IA is a less well-defined entity with only a few reports in the literature.17,18 Interestingly, while psoriasis and especially palmoplantar pustular psoriasis are relatively common among rheumatologic patients treated with anti-TNF, IA is almost exclusively described among patients with IBD but still considered rare.15,19 In our cohort, IA was relatively common, affecting a third of patients with TNF antagonist induced dermatologic complications. A quarter of the subgroup with IA responded to discontinuation of the offending agent plus scalp injections with steroids, with or without systemic methotrexate. Almost half required a switch out of class and responded to ustekinumab. This course is in line with previous case reports stating that in IA, discontinuation of anti-TNF does not result in IA improvement and that switching to a different TNF inhibitor is rarely successful. 19 In fact, in most of these IA cases, therapies such as topical steroids, intralesional steroids, or systemic agents (mainly methotrexate or cyclosporin) were required to regain hair growth and to avoid scarring alopecia. 19
In 2018 ustekinumab, a human monoclonal antibody against the p40 subunit of interleukins-12 and -23, which was previously licensed to treat psoriasis, was first approved for CD 20 and later for UC. 21 However, its effectiveness for paradoxical skin eruption induced by anti-TNF data is still scarce.22,23
In paradoxical psoriasis, TNF inhibition precludes dendritic cell maturation, thus leading to sustained interferon (IFN) α induced acute inflammation in genetically predisposed individuals. 24 As paradoxical psoriasis is IFN α driven, withdrawal of anti-TNF leads to the resolution of skin eruption in a significant proportion of patients. As shown here, in patients without resolution of psoriasis despite discontinuation of the offending anti-TNF, ustekinumab might be a good treatment option.
Similarly, Tillack et al. 24 reported good response rates to ustekinumab for psoriasiform lesions secondary to TNF treatment, including alopecia in a small patient cohort. Skin lesions were histologically characterized by infiltration of IL-17A-IL-22 secreting TH17 cells and IFN α -expressing cells; IL-17A expression was significantly stronger in patients requiring ustekinumab than in patients responding to topical therapy. Moreover, IL23R-specific genotypes suggested disease-modifying effects in patients requiring ustekinumab. Correspondingly, in our cohort, almost half of the group with IA (7/15 patients) required a switch out of class and responded to ustekinumab.
Notably, despite ustekinumab’s effectiveness in eradicating psoriasiform skin eruptions and IA, about a third of these patients had IBD exacerbation after switching, requiring an alternative strategy to control the inflammation. For such cases, more specific IL-23 treatments targeting the p19 subunit25 –27 are expected approval in the near coming future and might be a good solution.
In summary, we have shown that in an integrated IBD-DERMA clinic using a personalized set of treatment strategies and targets, we provided effective therapy to a complicated cohort of patients with IBD with paradoxical skin eruptions even after being assessed by IBD-oriented experts. To the best of our knowledge, this is the largest reported cohort of complicated patients with IBD with paradoxical skin eruption treated with ustekinumab and the largest cohort of patients with paradoxical IA.
Our study has several limitations linked to its retrospective nature, the inherent risk of bias, incomplete data for some patients, lack of objective assessments to evaluate disease activity, and small size. Also, treatments were not rigidly standardized; there was no control group. Finally, patients were also treated by expert physicians in a large referral center, thus impacting the generalizability of our findings.
In conclusion, we have shown that implementing an MDT IBD-DERMA clinic provides patients with safe and effective treatments for dermatological complications of IBD therapy while maintaining IBD control. This was also true in patients with severe dermatological side effects, such as the previously sparsely reported TNF-induced IA. Further studies are needed to identify molecular and/or genetic markers in prone individuals and biomarkers that will allow for tailoring the most optimal therapeutic approach for these complicated cases.
Footnotes
Author contributions
HY: Steering committee, design of the study, acquisition of data, analysis and interpretation of the data, drafting the article; LP: Steering committee, design of the study, acquisition of data, analysis, and interpretation of the data; HAB: acquisition of data; KEO: drafting the article; IAB: drafting the article; IG: drafting the article; all authors reviewed and approved the final version of the manuscript.
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Henit Yanai: reports institutional research grants from Pfizer; consulting fees from Abbvie, Ferring, Janssen, Neopharm Ltd., Pfizer, Takeda; honoraria for lectures from Abbvie, Janssen, Pfizer, Takeda; participation on a Data Safety Monitoring Board or Advisory Board from Abbvie, Neopharm Ltd., Pfizer, Takeda. Hadar Amir Barak: none. Jacob E Ollech: none. Irit Avni Biron: none. Idan Goren: reports institutional research grants from Pfizer. Yifat Snir: none. Hagar Banai Eran: none. Yelena Broitman: none. Maya Aharoni Golan: none. Elena Didkovsky: none. Iris Amitay-Laish: none. Ayelet Ollech: none. Emmilia Hodak: none relevant. Iris Dotan: research grants from Altman Research, Pfizer; Advisory board/consulting fees: Pfizer, Janssen, Abbvie, Takeda, Genentech/Roche, Arena, Neopharm, Gilead, Galapagos, Celltrion, Rafa Laboratories, Ferring, DSM, Cambridge Healthcare, Sublimity, Sangamo, Wild Biotech, Food industries organization, Integra Holdings, Celgene/BMS, Abbott, 89 Bio, Alimentiv; Speakers Bureau: Roche/Genentech, Falk Pharma, Abbvie, Janssen, Pfizer, Takeda Neopharm, Celltrion, Ferring, Nestle, Celgene/BMS. Lev Pavlovsky has served as an investigator for Abbvie, Coherus, Novartis Pharmaceuticals Corporation, Janssen Biotech, Eli Lilly, Bristol Myers Squibb and as an advisor, consultant, and/or invited lecturer for Abbvie, Janssen Biotech, Novartis Pharmaceuticals Corporation, Pfizer Inc., Dexcel Pharma, Eli Lilly, and Boehringer Ingelheim.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conference presentation
This study was presented at the 27th United European Gastroenterology Week (UEGW), Spain, Barcelona, October 2019.
