Abstract
Primary biliary cholangitis (PBC) is a chronic autoimmune cholestatic disease characterized by the destruction of the small intrahepatic bile ducts, which can progress to liver cirrhosis. The gold standard in the treatment of PBC is ursodeoxycholic acid (UDCA), which is indicated in all patients with PBC because it improves not only biochemical parameters but also patients’ survival. An important milestone in the identification of patients at risk is the assessment of biochemical response to UDCA. Patients who respond to treatment have a lower incidence of hepatic events and better prognosis than patients who do not. Several scoring systems can be used to assess the response and identify non-responders who will benefit from second-line treatment. Obeticholic acid (OCA) is currently the only approved second-line treatment for PBC, which is effective for non-responders to UDCA therapy or patients, who have not tolerated UDCA therapy. However, OCA is contraindicated in advanced liver cirrhosis and portal hypertension. Moreover, pruritus may be a limiting factor for the administration of OCA. Fibrates have shown promising data supporting their use in non-responders to UDCA because they improve the biochemical parameters and elastographic findings and have possible antipruritic effects. Therefore, the idea of a triple treatment seems interesting. Clinical research is focusing on several other groups of drugs: peroxisome proliferator-activated receptor (PPAR) δ- and α/δ agonists, non-steroidal farnesoid X receptor agonists, fibroblast growth factor 19 modulators, and inhibitors of nicotinamide adenine dinucleotide phosphate oxidase 1 and 4.
Introduction
Primary biliary cholangitis (PBC) is a chronic autoimmune, non-suppurative inflammatory disease of the liver characterized by the destruction of the small intrahepatic bile ducts and the development of liver fibrosis. In advanced stages, PBC can progress to liver cirrhosis.1,2 The disease is significantly more common in women and the prevalence of PBC increases with age. 3 Although the incidence of PBC in Europe and the United States has remained stable in recent years, the prevalence of the disease is increasing.1,2 Although the diagnosis of PBC has improved significantly in recent years, a large number of patients remain undiagnosed. 4 Genetics, epigenetics, immunological, and environmental factors play a key role in the pathophysiology of PBC (see Figure 1). The aim of PBC treatment is to improve laboratory parameters, stabilize or improve histological findings, prevent liver cirrhosis and its decompensation, prevent hepatocellular carcinoma, and improve quality of life.

Factors involved in PBC pathophysiology.
As part of non-pharmacologic treatment, patients with PBC should follow similar precautions as patients with other liver diseases. 5 Patients with PBC should be physically active and follow a diet without consuming alcoholic beverages. 6
In the past, the following were used in the treatment of PBC:
immunosuppressive drugs
drugs with an immunosuppressive and anti-inflammatory effect
drugs with anti-inflammatory and anti-fibrotic effects.
The clinical effect of these drugs was poor, and the side effects were considerable. 7 Ursodeoxycholic acid (UDCA) presents a breakthrough in the treatment of PBC. 8 A milestone in the treatment of PBC that does not respond to UDCA was the introduction of second-line therapy, which is used in addition to UDCA.
UDCA in the treatment of PBC
UDCA is the 7-β-epimer of chenodeoxycholic acid. It has a hepatoprotective and choleretic effect. 9 UDCA reduces the absorption of toxic hydrophobic endogenous bile acids, stabilizes the hepatocyte membrane against toxic bile acid salts, and suppresses interleukin-2 (IL-2) production. During treatment, UDCA replaces potentially hepatotoxic bile acids: cholic, chenodeoxycholic, deoxycholic, and lithocholic acid. A correlation has been described between the proportion of UDCA in bile on the one hand and the change in biochemical parameters on the other. 7 Mechanism of UDCA action is shown in Figure 2. 10 UDCA delays the progression of fibrosis in patients with PBC. 11

Mechanism of UDCA action.
The first clinical study confirmed that UDCA improved the laboratory parameters in patients with PBC. 8 A meta-analysis showed that treatment with UDCA in PBC lowers both total cholesterol and low-density lipoprotein cholesterol, while high-density lipoprotein cholesterol (HDL-C) and triglyceride levels remain unchanged. 12
UDCA is a drug that is very well tolerated by patients. A small proportion of patients experience pruritus, diarrhea, and pain in the right abdominal quadrant. 13 Only a very small proportion of patients do not tolerate treatment with UDCA. 4
Effect of UDCA treatment in patients with PBC on survival and hepatic decompensation
In the early years of PBC treatment with UDCA, mainly patients in advanced stages of the disease were treated. It is therefore not surprising that two previously published meta-analyses found no significant difference in mortality between PBC patients with or without UDCA.14,15
A final decision on the impact of UDCA on patient survival was only made by an international cohort study using the Global PBC Study Group database. In all, 3902 patients with PBC were included in the analysis. 90.4% of patients were treated with UDCA. The mean duration of follow-up was 7.8 years (4.1–12.1 years). UDCA significantly improved survival without transplantation compared to untreated patients (hazard ratio (HR): 0.46; 95%CI 0.40 to 0.52; p < 0.001). In addition, patients who had an inadequate response to UDCA treatment after 1 year of treatment had a significantly better survival rate without liver transplantation than untreated patients (HR: 0.56; 95%CI 0.45 to 0.69; p < 0.001). The differences in survival without liver transplantation between treated and untreated patients are displayed in Chart 1. 16

Comparison of transplant-free survival in PBC patients treated and untreated with UDCA.
In the analysis of the Global PBC Study Group database, the authors found in a multivariate analysis that patients who were not treated with UDCA had more frequent disease progression and a higher risk of developing a serious clinical event compared to treated patients. 17
The effect of UDCA treatment on the histological findings of patients with PBC
There are few studies with a small number of patients in which paired liver biopsies were performed during UDCA. Angulo et al. described paired biopsies in 16 patients with PBC who received UDCA and compared them with 51 patients who received ineffective treatment (
Evaluation of therapeutic response in the treatment of PBC with UDCA
UDCA monotherapy at a dose of 13–15 mg/kg body weight per day is the gold standard in the first-line treatment of PBC.1,2,21 To evaluate the efficacy of the treatment, the criteria for therapeutic response needed to be established. Several authors use different criteria to evaluate the efficacy of UDCA treatment in PBC patients. Qualitative binary scoring systems evaluate simple laboratory parameters after UDCA treatment; continuous scoring systems combine the results of some laboratory parameters or age before treatment with laboratory parameters after UDCA treatment.
The first major scoring system was the Rochester model. It defined alkaline phosphatase (ALP) < 2× upper limit of norm (ULN) and Mayo Risk Score < 4.5 after 6 months of UDCA treatment in patients with PBC as a therapeutic response to UDCA. Responders, after half a year, maintained their therapeutic response even after 2 years of therapy. 22
The Barcelona criteria evaluated a therapeutic response as a decrease in ALP levels after 1 year of UDCA treatment of >40% or a normalization of its level. The estimated survival time of responders was similar to the general population, while the estimated survival time of non-responders was shorter than that of the general population (Chart 2). 23

Comparison of survival between PBC responders to treatment with UDCA and the general population.
The French authors defined a response to treatment as ALP < 3× ULN, aspartate aminotransferase (AST) < 2× ULN and bilirubin ⩽1 mg/dL after 12 months of UDCA treatment. These criteria are known as the Paris-I criteria. Patients who met them had a 10-year liver transplant-free survival rate of 90%; patients who did not meet them had a survival rate of only 51% (p < 0.001). 24
Corpechot et al. modified these criteria 3 years later and created the Paris-II criteria: AST ⩽1.5 × ULN; ALP ⩽ 1.5 × ULN; and normal bilirubin level after annual UDCA treatment. Paris-II responders had a 5-, 10-, and 15-year event-free survival of 100%, while the 5-, 10-, and 15-year survival of non-responders was 93%, 87%, and 74%, respectively. 25
The Rotterdam criteria evaluate the therapeutic response without the use of liver tests. Patients who respond therapeutically to UDCA have a better survival rate after 1 year of treatment than non-responders. 26
The EHIME score was determined in asymptomatic patients with PBC. After 6 months of UDCA treatment, UDCA responders have a decrease in gamma-glutamyltransferase (GGT) levels of ⩾70% or a GGT level in the normal range. Responders were less likely to have clinical manifestations such as pruritus, ascites, esophageal varices, jaundice, and encephalopathy compared to non-responders to UDCA. 27
The most widely used criteria for assessing therapeutic response to UDCA treatment in PBC patients are the Toronto criteria. UDCA responders have an ALP ⩽ 1.67 × ULN after 24 months of UDCA treatment. Patients who achieved the response had better histological findings compared to non-responders. 20 It should be noted that in real practice it is quite illusory to evaluate therapeutic response after 2 years of treatment. Virtually all patients who achieved a therapeutic response according to the modified Toronto criteria after 6 months of treatment maintained it during the next treatment period. 28
Chinese authors have recently developed the so-called Xi’an criteria, which document the therapeutic response to UDCA in PBC after only 1 month of treatment and evaluate it as ALP ⩽ 2.5 × ULN, AST ⩽ 2 × ULN, and total bilirubin ⩽ 1 × ULN. The 5-year survival rate without serious hepatic events was 97% in responders and 64% in non-responders (p < 0.001). 29 An analysis of the Global PBC Study Group database found that the 10-year survival rate of PBC patients is best in patients who have a normal ALP and bilirubin ⩽ 0.6 × ULN after 12 months of UDCA treatment. 30
Continuous scoring systems can predict patient prognosis better than binary models. The British authors analyzed the data of 1916 treated patients from the UK-PBC Research Cohort database and designed a Cox model in which they included the values of albumin and platelets before treatment and the values of bilirubin, transaminases, and ALP after 12 months of UDCA treatment. They validated the model in 1249 patients with PBC who were treated with UDCA. The total area under the receiver operating characteristic curve (AUROC) score was 0.96, 0.95, and 0.94 for 5-, 10-, and 15-year survival without major hepatic complications. The UK-PBC Risk Score is excellent for predicting survival and severe liver complications in patients with PBC. 31
A similar scoring system is the GLOBE score, which was calculated using the Global PBC Study Group database. The GLOBE score was calculated using the age before the start of treatment and the values of bilirubin, ALP, platelets, and albumin after 1 year of UDCA treatment. Patients with a GLOBE score ⩽0.3 have a comparable survival to the general population; patients with a GLOBE score >0.3 have a shorter survival than the general population (p < 0.001). 32
Carbone et al. created a UDCA response score, which is based on pre-treatment parameters. The derivation cohort consisted of patients from the UK-PBC Re-search Group, and the validation cohort consisted of patients from the Italian PBC Study Group. Pre-treatment parameters associated with a lower probability of response to UDCA were later included in the calculation of the UDCA response score: higher ALP and bilirubin levels, lower aminotransferase levels, younger age, longer interval between diagnosis and initiation of UDCA treatment, and worsening ALP levels in the interval between diagnosis and initiation of treatment. The response to the treatment was defined as an ALP <1.67 × ULN after 1 year of UDCA treatment. The AUROC for UDCA response score after 1 year of treatment was 0.83 (95%CI 0.79 to 0.87). 33 It is lower than the UK-PBC risk score, but its advantage is that we can determine the achievement of a therapeutic response to UDCA treatment based on the parameters before starting treatment. A summary of the binary and continuous scoring systems is shown in Table 1.
Evaluation of therapeutic response to UDCA treatment in patients with PBC and prognostic scoring systems.

ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamaglutamyl transferase; PBC, primary biliary cholangitis; UDCA, ursodeoxycholic acid; ULN, upper limit of norm.
The relationship between biochemical response and histological findings is also important. Patients with biochemical response to UDCA treatment are less likely to progress in histological findings (Chart 3). 20 In a study investigating the UDCA response score in patients with PBC, a liver biopsy was performed prior to UDCA treatment. Some histological changes, for example, the extent of fibrosis or ductular response, correlate with this scoring system. 33

Changes in the stage of fibrosis in PBC responders and non-responders to UDCA treatment.
Prognostic aspects of PBC in UDCA treatment
Logically, patients who respond to registered PBC treatment can expect a better prognosis than patients who do not respond. In recent years, several studies have been published looking at the prognosis of PBC patients treated with UDCA, evaluating standard laboratory parameters prior to the initiation of PBC treatment28,34–37 Chinese authors found that a low albumin level and a higher IgG level as well as all liver tests (alanine aminotransferase (ALT), AST, GGT, and ALP) before starting UDCA treatment predicted a lack of response to UDCA. 36 High ALP prior to UDCA treatment is associated with an incomplete response to treatment. 35 High ALP prior to UDCA treatment correlated with an inadequate response to treatment in multivariate analysis, while young age at diagnosis of PBC, presence of pruritus prior to treatment, low albumin prior to treatment, and high ALT, GGT, and ALP predicted disease progression. 37 Low AST and ALT levels prior to UDCA treatment correlate with the achievement of a biochemical response. 34
Both the albumin–bilirubin score and the neutrophil-to-lymphocyte ratio predict liver mortality.38,39 The best predictive scoring system for pre-treatment response to UDCA remains the UDCA response score, with an AUROC value >0.8 in the original paper and in the validation publications.33,40–42 In addition, the UDCA response score correlates very well with the histological changes according to the Nakanuma classification. 43
Histopathological findings in PBC predict the achievement of a therapeutic response to UDCA. 20 The extent of liver fibrosis and the presence of liver cirrhosis are associated with a poor patient prognosis. 44 Elastographic findings, including the dynamics of values measured on repeated elastography, correlate with the prognosis of patients with PBC. 45 The degree of liver fibrosis in PBC is a prognostic factor that is independent of achieving a therapeutic response to UDCA. 46 Biomarkers in liver tissue can also be associated with the achievement of a therapeutic response. Both a decrease in CD4+ T lymphocytes and a decrease in T-beta positively expressed Th1 lymphocytes in the liver parenchyma after 1 year of UDCA treatment were associated with the achievement of a therapeutic response in patients with PBC. 47 Both serum IL-2 and sCD163 levels correlate with the progression of PBC.48,49 Serum proteome and gut microbiome assays may predict therapeutic response to UDCA treatment in PBC patients.50,51
The presence of anti-Sp100 antibodies is an independent predictor of the progression of PBC to liver failure. 52 Patients with PBC and the presence of anti-gp210 antibodies have a higher risk of liver mortality or liver transplantation. 49 PBC patients aged 15–39 years have almost thirteenfold higher risk of death compared to older patients. 53 Men with PBC are less likely to have clinical manifestations of PBC, the disease is diagnosed later than in women, and have higher mortality than women.54,55 Patients with PBC who have an overlap with autoimmune hepatitis have a significantly lower response to treatment after 1 year of UDCA monotherapy than PBC patients. 36
Second-line treatment for PBC
About 30% of PBC patients do not respond adequately to UDCA treatment, and another third have an incomplete response. 4 In these patients, UDCA should be continued and supplemented with a second-line treatment. The GLOBE score and the UK-PBC score are best suited for the selection of patients requiring second-line therapy. 56
The discovery of nuclear receptors and the understanding of their role in controlling the genes regulating bile acids homeostasis, inflammation, and fibrosis led to the development of nuclear receptor-targeted treatment. The nuclear receptors of main importance for PBC are the farnesoid X receptor (FXR), peroxisome proliferator-activated receptors (PPARs), and glucocorticoid receptor. The following can currently be administered in second-line PBC treatment:
obeticholic acid (OCA)
bezafibrate, possibly other fibrates
budesonide. 57
OCA in the treatment of PBC
OCA is a synthetic derivative of chenodeoxycholic acid; it acts as an FXR agonist. 58 FXR is mainly expressed in the gastrointestinal tract and the liver. It is a key receptor that ensures the homeostasis of bile acids via a complex signaling pathway. OCA has a ~100-fold stronger effect on FXR than endogenous bile acids. 59 In patients with PBC, the enterohepatic circulation is disturbed. By modulating FXR activation, OCA regulates the synthesis and secretion of bile acids and alters their composition. 60 OCA has an anti-inflammatory and anti-fibrotic effect by activating the sinusoidal cells of the liver endothelium and Kupffer cells. 61 The activation of these cells and the reduction in the production of proinflammatory cytokines reduce the activation of stellate cells, which are responsible for fibrogenesis. 60 OCA modulates fibroblast growth factor 19 (FGF-19) activity, resulting in a hepatoprotective effect of OCA that is greater than that of UDCA. 62 The activation of FGF-19 also contributes to the anti-cholestatic mechanism of OCA. 60 Mechanism of OCA action is shown in Figure 3. 63

Mechanism of OCA action.
The phase III study of obeticholic acid in patients with primary biliary cirrhosis (POISE) registration study involved 216 patients who did not respond adequately to UDCA treatment at standard dose or who could not tolerate UDCA treatment. The patients were divided into three groups: the first group received OCA at a dose of 10 mg/day (OCA 10 mg), the second group received OCA at a dose of 5 mg/day with the possibility of an increase to 10 mg/day (OCA 5–10 mg), and the third group received placebo. Randomization was performed based on the Paris-I criteria. 24 The primary aim of the study was to achieve a therapeutic response to OCA after 12 months of treatment, which was assessed as follows: a reduction in ALP <1.67× ULN and a concomitant reduction in ALP of at least 15% from pre-treatment levels and normalization of serum bilirubin. The primary aim was achieved by 46% of patients in the OCA 5–10 mg group, 47% of patients in the OCA 10 mg group, and 10% of patients in the placebo group (p < 0.001). Repeated elastographic examination and assessment of quality of life using the PBC-40 questionnaire revealed no significant differences between all three groups. The most serious side effect of OCA treatment was pruritus, which was described in 56% of patients in the OCA 5–10 mg group, in 68% of patients in the OCA 10 mg group, and 38% of patients in the placebo group. Ninety-seven percent of patients who completed 12 months of treatment continued open-label clinical follow-up, while patients in the placebo group switched to OCA. The primary endpoint remained in both OCA groups after 24 months of treatment; patients in the placebo group achieved comparable results after switching to OCA treatment as both OCA groups after 24 months of treatment. 64
Three years later, data on the long-term efficacy of OCA in patients with PBC were published. More than half of the patients achieved a therapeutic response after 2, 3, and 4 years of treatment. The results of this study show that OCA treatment is effective in the long term and relatively well tolerated by patients. 65
A paired biopsy was performed in 17 patients after 3 years. The Nakanuma fibrosis score improved in 24% of patients, remained stable in 47% of patients, and worsened in 29% of patients. The Nakanuma bile duct loss score improved in 41% of patients, remained stable in 35% of patients, and worsened in 24% of patients. Interface hepatitis improved or stabilized in 100% of patients, and lobular hepatitis improved or stabilized in 94% of patients after OCA treatment. Patients can benefit from OCA treatment not only through the adjustment of biochemical but also histological findings. 66
In another study, the authors investigated whether the use of OCA in PBC would improve patient’s survival. Propensity scores from the POISE study were calculated and compared with data from two registers: the Global PBC Study Group and the UK-PBC Research Cohort. The mean follow-up was 6 years; during follow-up, 2.4% of patients from the POISE study, 10% of patients from the Global PBC Study Group, and 13.2% of patients from the UK-PBC Research Cohort died. The mortality differences between the POISE study on the one hand and the registries on the other are significant and are shown in Chart 4. The results of this analysis suggest that the addition of OCA to treatment improves the survival of patients with PBC. 67
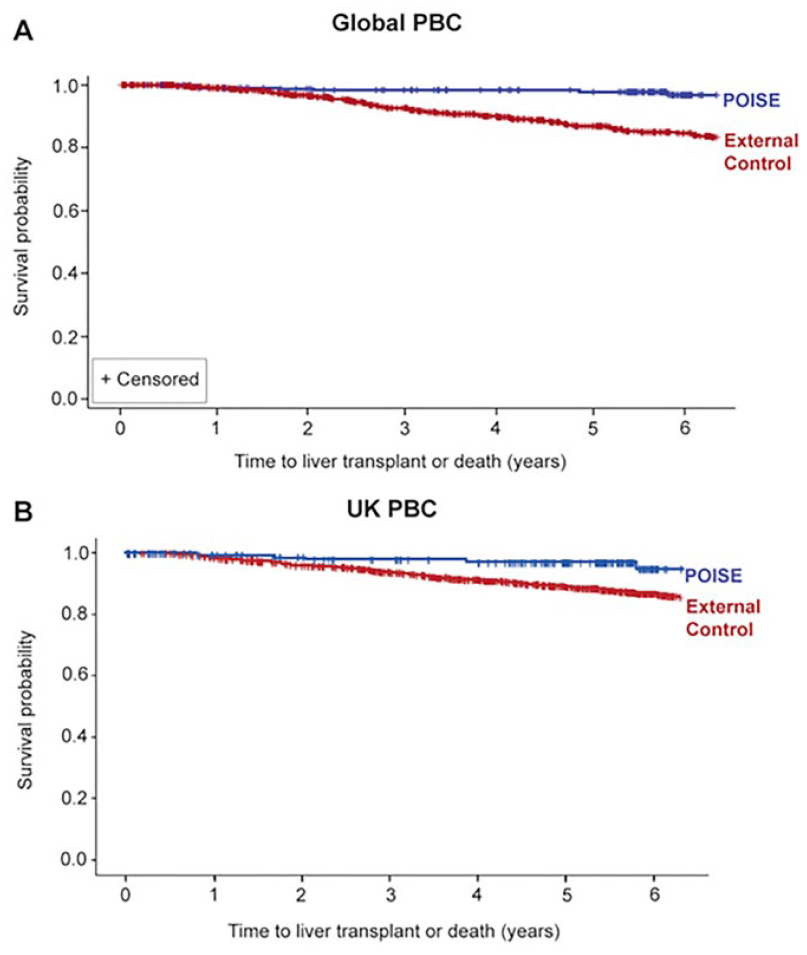
Comparison of survival in PBC patients from the Global PBC Study Group and UK-PBC Research Cohort POISE registries.
Three clinical trials were included in a meta-analysis comparing OCA with placebo in the treatment of PBC non-responders to UDCA.64,68,69 OCA treatment was more effective than placebo and also led to a reduction in cholesterol and HDL-C levels. 70
Results of randomized controlled trials evaluating combination therapy (OCA + UDCA) compared with UDCA monotherapy and results of monotherapy with OCA compared to placebo are presented in Table 2.
Randomized controlled trials evaluating combination therapy (OCA + UDCA) compared with UDCA monotherapy and monotherapy with OCA compared to placebo.

ALP, alkaline phosphatase; CI, confidence interval; N, number; OCA, obeticholic acid; Pts, patients; RCT, randomized controlled trial; UDCA, ursodeoxycholic acid; ULN, upper limit of norm.
However, the data from real clinical studies showed slightly worse results than in the approval study. In a Canadian study (24% of patients with liver cirrhosis), only 18% of patients achieved a therapeutic response after 1 year; after 19 months, the number of patients with a therapeutic response increased to 43%. 71
In a study from Spain and Portugal, 22% of patients had liver cirrhosis and 27% were taking fibrates before the treatment. Seventy-eight patients completed the 1-year treatment, of whom 30% achieved a therapeutic response. 72 In an Italian study, 31% of patients had liver cirrhosis. Forty-three percent of patients achieved a therapeutic response. Patients with liver cirrhosis discontinued OCA treatment more frequently (30% vs 12%; p = 0004) and were less likely to achieve a therapeutic response (30% vs 49%; p = 0.01) compared to PBC patients without liver cirrhosis. 73 The same group investigated the efficacy of OCA treatment in patients with PBC in the cirrhosis stage, the most of them were in Child-Pugh stage A. Sixty-five percent of the patients received OCA at a dose of 5 mg/day, 15% at a reduced dose; in 20% of the patients, the dose was titrated from 5 to 10 mg/day. A therapeutic response was achieved in 32% of all cirrhotics and in 41% of cirrhotics who had completed 1 year of treatment. The history of ascites, low prothrombin time and albumin, high bilirubin, and a higher Child-Pugh score were associated with serious adverse events. These occurred mainly in patients with a Child-Pugh score ⩾6 points. 74
OCA is contraindicated in patients with advanced liver cirrhosis. This conclusion was drawn based on the observations in 25 cirrhotic patients. In 10 cirrhotic patients with portal hypertension, decompensation of cirrhosis occurred; in the remaining patients who had decompensation of liver cirrhosis before starting OCA treatment, liver failure occurred during treatment. Four patients had to undergo liver transplantation, two of whom died. 75 In the updated recommendations for the treatment of PBC, the FDA states that OCA treatment is contraindicated in patients with advanced cirrhosis. This is defined by evidence of decompensated cirrhosis, currently or in the past, or if signs of portal hypertension are detected. 76 These patients should not be treated, even at reduced doses.
Possible side effects may also be a limiting factor for the administration of OCA. A meta-analysis of eight studies of patients with PBC and non-alcoholic steatohepatitis treated with OCA showed that OCA increased the risk of pruritus by about 75% and the risk of constipation by about 88%. 77
OCA is an effective treatment for patients with PBC who have not responded to UDCA therapy or who have not tolerated UDCA therapy. It is currently the only approved second-line treatment for PBC. OCA is contraindicated in advanced liver cirrhosis.
Fibrates in the treatment of PBC
Fibrates act via nuclear PPARs. The different types of fibrates have a different affinity for the PPARs, which affects their pharmacotherapeutic effect. Fenofibrate acts only on PPARα. Its key role is to regulate cholesterol and bile acid homeostasis: it inhibits the enzymatic activity of cholesterol 7 alpha-hydroxylase (CYP7A1), resulting in reduced synthesis of bile acids, regulates the detoxification of bile acids and facilitates the export of phospholipids. Fenofibrate reduces the pro-inflammatory response via nuclear factor κB. Bezafibrate acts on PPARα, PPARβ/δ, and PPARγ, thus has further possible effects. Activation of PPARγ affects lipoprotein metabolism and provides the anti-inflammatory and anti-fibrotic effects of bezafibrate. 78
PBC patients who did not achieve a therapeutic response to UDCA treatment according to the Paris-II criteria were included in the double-blind, randomized BEZURSO study. In the first group, patients received UDCA at a standard dose and bezafibrate 400 mg/day; in the second group, patients received UDCA in combination with a placebo. The primary aim was to achieve a complete response: normal levels of ALP, bilirubin, aminotransferases, albumin, and prothrombin time after 24 months of treatment. A complete therapeutic response was achieved after 6 months of treatment by 18%, after 12 months of treatment by 23%, and after 24 months of treatment by 31% of patients in the bezafibrate group, compared to none patients in the placebo group (p < 0.001). At the end of treatment, a normal ALP value was registered in 67% of patients in the bezafibrate group and only in 2% of patients in the placebo group. Liver stiffness, measured by elastography, decreased by 15% in the bezafibrate group and increased by 22% in the placebo group. By contrast, in patients who underwent paired liver biopsy, no significant differences were found between the two groups of patients. The incidence of adverse events and serious adverse events was comparable between the two groups of patients. The addition of bezafibrate to UDCA in non-responders to UDCA improves laboratory and elastographic findings, but not histological findings, compared to UDCA monotherapy. 79
Recently, a meta-analysis of seven studies was published in which treatment with UDCA + bezafibrate was compared with monotherapy with UDCA. In addition to the BEZURSO study, several Japanese studies were included in the meta-analysis. The treatment of UDCA with bezafibrate led to a significant decrease in ALP and GGT, but not in bilirubin and immunoglobulin M (IgM) compared to UDCA monotherapy. The authors did not have sufficient data to evaluate the mortality. 80 Results of randomized controlled trials evaluating combination therapy (bezafibrate + UDCA) compared with UDCA monotherapy included in meta-analysis are shown in Table 3.
Randomized controlled trials evaluating combination therapy (bezafibrate + UDCA) compared with UDCA monotherapy included in meta-analysis.

Source: Khakoo et al. 80
ALP, alkaline phosphatase; CI, confidence interval; MD, mean difference; N, number; Pts, patients; RCT, randomized controlled trial; TB, total bilirubin; UDCA, ursodeoxycholic acid.
The Japanese authors studied 150 antimitochondrial antibody-positive PBC patients who did not respond to UDCA monotherapy and were treated with UDCA and bezafibrate. The patients were followed for an average of 15 years. Sixteen percent of patients developed ascites and/or jaundice during follow-up. These patients were less likely to achieve a therapeutic response to treatment with UDCA and bezafibrate compared to patients without liver complications. High IgM levels before treatment and during the first year of treatment were associated with shorter survival and the occurrence of liver complications. Normalization of IgM levels during treatment predicts therapeutic response. 86
In an analysis of 3908 patients from the Japanese National PBC Database, 19% of patients received UDCA in combination with bezafibrate, the rest received UDCA as monotherapy. Combined treatment with UDCA and bezafibrate reduced all-cause mortality by approximately threefold and liver-associated mortality by more than threefold compared to patients who were treated with UDCA monotherapy. 87
Another fibrate that can be used in the treatment of PBC is fenofibrate. Chinese authors compared the therapeutic response of UDCA in combination with fenofibrate (200 mg daily) with UDCA monotherapy in UDCA non-responders. Therapeutic response was defined as normal ALP, bilirubin, and GGT after 12 months of treatment. Twenty-one percent of patients in the fenofibrate group achieved a therapeutic response after 1 year of treatment compared to none patients treated with UDCA monotherapy. Fifty-four percent of patients in the fenofibrate group and 4% in the placebo group had normal ALP levels at the end of the study. 88
A small study found that in patients with asymptomatic PBC, 48 weeks of treatment with low-dose fibrate (80 mg daily) and UDCA resulted in a comparable biochemical response to bezafibrate (200 mg daily) and UDCA. 89
Since the treatment with fibrates is inexpensive and has acceptable side effects, the idea of combining UDCA and fibrates from the start of PBC treatment could be interesting. Chinese authors have recently published a study with naïve PBC patients. Response to treatment was assessed using the Barcelona criteria. 23 A therapeutic response after 12 months was achieved in 81.4% of patients treated with UDCA and fenofibrate (200 mg/day) and in 64.3% of patients on UDCA monotherapy (p = 0.048). 90
It should be noted that fibrates significantly reduce the intensity of pruritus in the treatment of PBC. This was confirmed in a meta-analysis that evaluated the therapeutic effect of fibrates in the treatment of patients with PBC. 91
The combined treatment with UDCA and fibrate improves the biochemical parameters and elastographic findings. The mortality data from Japan are optimistic. However, fibrates are not approved for the treatment of PBC yet, so this is an “off-label” therapy.
Combination treatment with fibrate and OCA in PBC
Combined treatment of UDCA with OCA or fibrates improves the prognosis of PBC patients who do not respond to UDCA; however, only some patients achieve a therapeutic response to dual treatment. Both OCA and fibrates have different mechanisms of action. The idea of treating non-responders with a combined treatment of UDCA + OCA + fibrate is therefore logical.
A multicenter study investigating the efficacy of the triple combination UDCA + OCA +fibrate in non-responders to dual treatment with UDCA + OCA or fibrate had an interesting design. Patients were initially treated with UDCA at a standard dose. After at least 1 year of treatment, the efficacy was evaluated according to the Paris-II criteria. 25 If the patients did not achieve a therapeutic response, the treatment was combined with OCA at a dose of 5–10 mg/day or a fibrate (bezafibrate 200–400 mg/day or fenofibrate 100–200 mg/day). After at least 3 months of dual treatment, the therapeutic response was reassessed according to the Paris-II criteria. In case of non-response to UDCA and OCA, fibrate was added to the treatment (OCA-fibrate group), while in case of non-response to UDCA and fibrate, OCA was added to the treatment (fibrate-OCA group). The average decrease in ALP in the OCA-fibrate group was 44% per year (p < 0.001); in the fibrate-OCA group, the annual decrease in ALP was 11% (p = 0.1). Normalization of ALP levels occurred significantly more frequently in the OCA-fibrate group. The triple combination therapy also increased the number of patients who achieved a therapeutic response compared to the dual combination treatments. Pruritus was reduced in the OCA-fibrate group, however, not in the fibrate-OCA group. 92
The study presented above has shown the benefits of the triple combination of UDCA + OCA + fibrate. The future will show whether it is more beneficial to administer the triple combination therapy to high-risk patients immediately or sequentially after UDCA failure.
Budesonide in the treatment of PBC
Budesonide is a 16-α-hydroxyprednisolone with significant hepatic first-pass metabolism and little systemic effect. Budesonide is a combined glucocorticoid receptor and pregnane X receptor agonist with 15–20 times higher affinity for the glucocorticoid receptor than prednisolone. Due to its low systemic effect, it has fewer side effects than corticosteroids. 93
In a randomized study published in 2021, non-responders to UDCA (ALP > 1.5 × ULN) after 6 months of therapy with histologically confirmed hepatic inflammatory activity were evaluated. The patients continued UDCA treatment at the original dose; two-thirds of the patients additionally took budesonide at a dose of 9 mg/day; the remaining third of the patients received a placebo. The treatment lasted 36 months. The primary aim defined as improving inflammatory activity without worsening fibrosis in liver biopsy was not achieved. Patients treated with the combined treatment of UDCA + budesonide were more likely to achieve a biochemical response by the modified Toronto criteria. 94
A recently published meta-analysis has shown that combination therapy with UDCA and budesonide is more effective than UDCA monotherapy in patients with an overlap of PBC and autoimmune hepatitis. Budesonide has fewer side effects than prednisone. 95
Perspectives on the treatment of PBC
Although the treatment of PBC patients non-responders to UDCA has improved significantly in recent times, not all patients benefit from the treatment. Therefore, new therapeutic options for the treatment of PBC are being sought. Clinical research is focusing on several groups of drugs:
PPAR agonists
non-steroidal FXR agonists
FGF-19 modulators
inhibition of nicotinamide adenine dinucleotide phosphate oxidase (NOX)1 and 4
nor-UDCA. 96
The identification of three isoforms of PPAR and their different places of expression led to the development of non-fibrate PPAR agonists as a selective ligand targeting each isoform of PPAR.
Seladelpar is a selective PPARδ-agonist. PPARδ in contrast to PPARα is expressed not only in hepatocytes but also in cholangiocytes, hepatic stellate cells, and Kupffer cells, which could potentiate the anti-inflammatory effect of seladelpar. Seladelpar at a dose of 2–200 mg/day, the ALP value decreases by more than half. Hepatotoxicity has been observed at doses of 50 and 200 mg; doses of 2, 5, and 10 mg/day are safe.97,98 Treatment with seladelpar results in a composite response (decrease in ALP < 1.67× ULN and a concomitant decrease in ALP by at least 15% compared to pre-treatment levels; and normalization of serum bilirubin) after 1 year of treatment in 57.1% of patients on 5 mg daily, 78.2% of patients on 10 mg daily, and only 12.5% of patients on placebo (p < 0.0001); normalization of ALP occurred in 5.4% of patients on the lower dose and 27.3% of patients on the higher dose, the difference was statistically significant for seladelpar 10 mg daily compared to placebo. Treatment with seladelpar led to a decrease in aminotransferases. 99 After 24 months of treatment, the number of patients responding to seladelpar increased from 66% to 79% and the number of patients with normalization of ALP levels increased from 26% to 42%. 100 Seladelpar relieves itching and lowers bile acid and IL-31 levels. 101 Seladelpar at a dose of 5/10 mg also improves sleep and fatigue in patients with PBC. 102 Seladelpar 10 mg daily was superior to placebo in achieving biochemical response after 1 year of treatment (61.7% vs 20.0%; p < 0.001) in a phase III clinical trial. ALP normalization was seen in 25% of patients in the seladelpar group but in none in the placebo group. The reduction in pruritus was greater in patients treated with seladelpar compared to placebo. 103
Elafibranor is a PPARα/δ agonist; at a dose of 80 mg daily, it led to a 48% decrease in ALP, and at a dose of 120 mg to a 41% decrease in ALP after 12 weeks of treatment. Therapeutic response at the end of follow-up was defined as in the previous study: after 12 weeks, 67% of patients with a lower dose of elafibranor and 79% of patients with a higher dose achieved this. Treatment with elafibranor resulted in a reduction in the intensity of pruritus. 104 In the phase III clinical trial, 51% of patients treated with elafibranor and only 4% of patients in the placebo group achieved a biochemical response after 1 year of treatment (p < 0.001). Normalization of ALP was found in 15% of patients in the elafibranor group but in no patients in the placebo group (p = 0.002). Greater pruritus reduction was seen in the elafibranor group compared to placebo. 105
Saroglitazar is a PPARα/γ agonist. PPARγ exerts anti-inflammatory activity in Kupffer cells and anti-fibrotic activity in hepatic stellate cells; however, in hepatocytes, its activation is steatogenic. 106 In a phase II study, it led at a dose of 2 or 4 mg daily to a prompt decrease in ALP after 16 weeks of treatment. One limitation of the study was hepatotoxicity, which resolved after discontinuation of treatment. 107
The non-steroidal FXR agonists cilofexor and tropifexor are being developed to increase FXR potency and selectivity and to overcome the OCA-associated clinical side effects, especially pruritus and atherogenic dyslipidemia. Cilofexor and tropifexor led to an improvement in cholestasis; however, the mentioned side effects appear to be dose-dependent class effects of all FXR agonists. 96
Aldafermin modulates FGF-19 activity. It acts on two receptor complexes—FGFR1c-KLB and FGFR4-KLB. Activation of the FGFR1c-KLB receptor leads to a reduction in hepatic steatosis and a reduction in insulin resistance. Activation of the FGFR4-KLB receptor strongly suppresses the expression of CYP7A1, which encodes the first and rate-limiting enzyme in the de novo synthesis of bile acids. 108 A 4-week treatment with Aldafermin at a dose of 0.3 mg and at a dose of 3 mg/day led to a decrease in ALP levels of at least 15% in about half of the patients. No worsening of pruritus was observed in patients; the limiting factor of treatment was gastrointestinal discomfort. 109
NOXs are transmembrane enzymes that transfer electrons from NADPH in the cytoplasm across the cell membrane, leading to the formation of reactive oxygen species (ROS). The NOX family consists of seven members, NOX1–5 and DUOX1–2. These enzymes differ in terms of enzyme complex composition, tissue, cellular and subcellular distribution, activation mechanisms, and the subtype of ROS they produce. 110 Setanaxib is the first dual NOX1/4 inhibitor in its class to block NOX1/4 activity, thereby reducing the production of ROS and blocking their harmful fibrotic effects. It is currently in a phase IIb/III clinical trial in patients with PBC. 111 It improves not only cholestasis but also fatigue in patients with PBC.96,112
Nor-UDCA is a derivate of UDCA, which repairs the defective HCO3− secretion. 109
A summary of targets for PBC treatment is shown in Figure 4. Drugs in clinical development for PBC are presented in Table 4.

Targets for PBC treatment.
The drugs in clinical development for PBC.

ALP, alkaline phosphatase; FGF-19, fibroblast growth factor 19; FXR, farnesoid X receptor; GGT, gamma glutamyl transferase; NADPH, nicotinamide adenine dinucleotide phosphatase; NOX, nicotinamide adenine dinucleotide phosphate oxidase; PBC, primary biliary cholangitis; PPAR, peroxisome proliferator-activated receptor; UDCA, ursodeoxycholic acid.
Optimistically, other drugs, apart from non-steroidal FXR agonists, do not cause or exacerbate pruritus. However, the limiting factor is hepatotoxicity or gastrointestinal distress, which forces clinical researchers to find the optimal dose of the drug.
PPAR agonists were more effective than other drugs in a meta-analysis of 23 clinical trials evaluating second-line treatment of UDCA-refractory PBC. 115
Treatment of PBC in special groups of patients
UDCA treatment in PBC patients does not need to be interrupted during pregnancy; it is safe during pregnancy and after delivery. 116
Treatment of PBC in patients with advanced liver cirrhosis is not effective; the only chance for these patients is liver transplantation.117,118 Cyclosporine A and azathioprine reduce; however, tacrolimus and mycophenolate mofetil increase the risk of PBC recurrence after liver transplantation. Patients with PBC recurrence in the transplanted liver have worse survival compared to patients without PBC recurrence. 119 Long-term prophylactic administration of UDCA after liver transplantation more than two times reduces the risk of PBC recurrence, approximately three times reduces the risk of graft loss, and more than two times decreases the risk of liver-associated mortality. It also reduces the risk of general mortality. 120
Conclusion
UDCA treatment should be initiated immediately after the diagnosis of PBC. Almost three-fourths of patients achieve a therapeutic response; about one-third of patients have a complete therapeutic response. Patients who respond to UDCA have a better prognosis than patients who do not respond; however, in some patients, the histological findings worsen despite the biochemical response. In non-responders, second-line treatment of PBC should be considered in addition to UDCA. The only approved second-line treatment is OCA; treatment with fibrates is effective but not approved for PBC. Second-line therapy improves the prognosis of PBC patients who do not respond to UDCA. In advanced cirrhosis, liver transplantation is required.
