Abstract
Objective
We performed a meta-analysis to determine whether the addition of probiotics to the bismuth quadruple therapy (BQT) for Helicobacter pylori would improve the incidence of eradication and reduce that of side effects.
Methods
Randomized controlled trials matching the inclusion criteria were collected from PubMed, Embase, Web of Science, and The Cochrane Central Register of Controlled Trials. A Mantel–Haenszel random-effects model was used to calculate pooled risk ratios (RRs) and 95% confidence intervals (CIs) for the incidences of eradication rate, side effects as a whole, diarrhea, and other side effects.
Results
Ten studies were selected for inclusion in the meta-analysis. The pooled RRs for the eradication rates in intention-to-treat and per-protocol analyses of the probiotic group vs. the control group were 1.07 (95% CI: 1.02–1.11) and 1.04 (95% CI: 1.00–1.07), respectively. Probiotic supplementation reduced the incidences of side effects (RR 0.58, 95% CI: 0.37–0.91), diarrhea (RR 0.41, 95% CI: 0.25–0.67), and bitter taste (RR 0.63, 95% CI: 0.40–0.99).
Conclusions
The results of this meta-analysis support the use of probiotics in combination with BQT in the clinical management of patients with H. pylori infection.
Introduction
Helicobacter pylori (H. pylori) is a widespread Gram-negative pathogen that causes chronic bacterial infections in humans. It has colonized approximately 50% of the world’s population. 1 The ability of H. pylori to resist gastric acid and to quickly create a pH-neutral environment principally depends on its production of urease, which hydrolyzes urea to produce carbon dioxide and ammonia. 2 Outer membrane proteins of the bacterium, such as blood group antigen-binding adhesin and sialic acid-binding adhesin, can bind to recognition sites on gastric epithelial cells, providing a basis for long-term colonization. 3 Biofilm formation and the antioxidant enzyme system can also help the bacterium achieve colonization. 4 Various virulence factors, such as cytotoxin-associated gene A protein and vacuolating cytotoxin protein, play a key role in the injury of host tissues and in inducing gastrointestinal disease. 5
H. pylori infection can induce many gastroduodenal diseases, such as gastritis, gastroduodenal ulcer, gastric cancer, and mucosa-associated lymphoid tissue lymphoma. 6 A study by Shatila and Thomas showed that H. pylori infection can be found in 90% of patients with stomach carcinoma and mucosa-associated lymphoid tissue lymphoma. 7 There is also a close relationship between H. pylori infection and duodenal ulceration, which occurs in 80% of cases, and with stomach ulceration, which occurs in up to 80% of cases. 8 The eradication of H. pylori has been demonstrated to reduce the risk of gastric cancer in all risk groups.9,10 Therefore, the Kyoto global consensus report on H. pylori gastritis advocates the treatment of all patients with H. pylori infection unless contraindications are present. 11
Increasing resistance to antibiotics worldwide has had negative effects on the effectiveness of polypharmacy-based regimens for the eradication of H. pylori infection, according to the Maastricht VI/Florence consensus report, which reviewed the potential applications of various empirical polypharmacy-based regimens for H. pylori infection in regions with differing resistance profiles. 12 The classic bismuth quadruple therapy (BQT) and a modified BQT are suitable options that are recommended in several guidelines and consensus reports.12–16 A meta-analysis by Bang et al. showed similar success of eradication for a modified BQT (rabeprazole 20 mg bid, amoxicillin 1 g bid, metronidazole 500 mg tid, and bismuth subcitrate 300 mg qid) and the classic BQT (rabeprazole 20 mg bid, bismuth subcitrate 300 mg qid, metronidazole 500 mg tid, and tetracycline 500 mg qid) over 14 days in regions with a high level of clarithromycin and metronidazole resistance. Both regimens yielded a high incidence of eradication (96.2% vs. 96.9%, respectively). 17 Although both the classic and modified BQT are effective approaches to the eradication of infection, poor tolerance and adverse reactions are common and are frequently reported to result in the discontinuation of BQT.18–20 Adverse reactions may be attributable to changes in gut microbial diversity/abundance because eradication therapies, such as BQT, can alter the gut microecology. 6
Regimens with higher incidences of eradication and lower incidences of side effects rate are being researched. Probiotics might improve the incidence of eradication and improve compliance by reducing the incidence of side effects. To date, several trials have shown that certain strains of probiotic may be useful as a complement to eradication therapy.21,22 Therefore, we performed a meta-analysis of randomized controlled trials (RCTs) that have assessed the incidences of eradication and side effects associated with probiotic supplementation of BQT.
Methods
Search strategy
This study is reported in accordance with the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) statement. 23 The registration number is INPLASY202370051 and the related DOI is 10.37766/inplasy2023.7.0051. Trials were identified by systematically searching electronic databases (PubMed, Embase, Web of Science, and The Cochrane Central Register of Controlled Trials) up to May 2022. The search terms used for PubMed were: (Helicobacter pylori [MeSH Terms] OR Helicobacter pylori [Text Word] OR Helicobacter nemestrinae [Text Word] OR Campylobacter pylori [Text word] OR Campylobacter pylori subsp. pylori [Text word] OR Campylobacter pyloridis [Text word]) AND (probiotics [MeSH Terms] OR probiotics [Text Word] OR probiotic [Text word] OR yeast [Text word] OR yogurt [Text word] OR Lactobacillus [Text word] OR Bifidobacterium [Text word] OR Saccharomyces [Text word] OR Lactococcus [Text word] OR Streptococcus [Text word] OR Enterococcus [Text word]) AND (Bismuth [MeSH Terms] OR Bismuth [Text Word] OR quadruple [Text Word]). A manual search was also performed using the reference lists of the articles identified, all of which were published in English.
Inclusion criteria
The following were used as inclusion criteria. 1) Randomized controlled trial, in which infection with or the eradication of H. pylori was confirmed by at least one generally accepted method (urea breath test (UBT), rapid urea test (RUT), H. pylori culture, histology, or stool antigen test). 2) Comparison of at least two groups, consisting of test patients who underwent BQT in combination with probiotics and control patients who underwent BQT with placebo or no additional intervention. 3) Two types of antibiotics were used during BQT, which were not restricted to metronidazole and tetracycline, meaning that both classic BQT and modified BQT were eligible. 4) The incidence of eradication, with or without adverse effects, was reported. 5) Publication in English. 6) Human studies. 7) Full-text electronic version articles were available.
Because the analysis was based on previously published studies, the requirements for ethics approval and informed consent were waived.
Data extraction and quality assessment
Two authors (GY and XF) independently reviewed the study titles and abstracts, and studies that satisfied the inclusion criteria were retrieved for full-text assessment. The following data were extracted independently by the two authors from these studies: the total number of participants, the method used to diagnose H. pylori, the time taken for eradication, the treatment regimen for H. pylori, the types of probiotics used, the incidence of eradication, and the number of any side effects.
The assessment of risk of bias was conducted in accordance with the guidelines of the Cochrane Collaboration 24 . Random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias were assessed. The two authors categorized each of these as high, low, or unclear risk through discussion.
Statistical analysis
The effects of a combination of a probiotic and BQT on several outcomes were assessed: the incidences of eradication on an intention-to-treat (ITT) basis, eradication on a per-protocol (PP) basis, side effects as a whole, diarrhea, bitter taste, abdominal discomfort, nausea or vomiting, constipation, melena, rash, fatigue, and dizziness. For all these outcomes, the pooled risk ratio (RR) and the corresponding 95% confidence interval (CI) were calculated using the Mantel–Haenszel random-effects model for dichotomous variables and are presented as forest plots. The possibility of publication bias was assessed by constructing a funnel plot if more than 10 studies were included. Begg’s and Egger’s tests were also used to assess funnel plot asymmetry and a P-value <0.1 was accepted as indicating significant publication bias. We also used the Cochran Q and I2 tests to assess the magnitude of any heterogeneity. An I2 statistic >50% and a P-value <0.1 was taken to indicate the presence of significant heterogeneity. Sensitivity analyses were not performed because there was no significant heterogeneity among the studies with respect to the primary outcome of the incidence of eradication.
Results
Search results
We initially identified 315 potentially relevant studies, of which 10 (containing data for 1397 participants) were included in the analysis.25–34 Details of the selection protocol are presented in Figure 1. Nine studies were published between 2013 and 2022,25,27–34 and the earliest was published in 2004. 25 Eight studies were conducted in Asia (four in China, two in Thailand, and two in Iran),25,27–29,31–34 and two studies were conducted in Europe (one in Italy and the other in Spain).25,29 The time taken to eradicate H. pylori was 10 or 14 days. All 10 of the publications included in the meta-analysis reported the H. pylori incidence of eradication.25–34 Five publications reported the total incidence of side effects,26,27,31,32,34 six reported the incidence of diarrhea,26–29,32,34 four reported the incidence of bitter taste28,29,31,34, and a few of the 10 reported other side effects. The general characteristics of the studies are shown in Table 1.

Flow chart for the study selection.
General characteristics of the included studies.

Test, test group; Cont, control group; HP, Helicobacter pylori; UBT, urea breath test; RUT, rapid urease test; ITT, intention-to-treat; PP, per-protocol; TVC, total viable count; CFU, colony-forming units.
Quality and risk of bias
All 10 RCTs involved adequate randomization and none was stopped early. Four studies were categorized as having a high risk of bias with respect to allocation concealment owing to the lack of a placebo. There was a low risk of bias with respect to performance bias, detection bias, attrition bias, reporting bias, and with respect to other bias in five, three, nine, seven, and nine studies, respectively. The results of the assessment of the risk of bias is shown in Figure 2a and Figure 2b.

Risk of bias in the included studies. (a) Graph and (b) risk of bias summary.
Results of the meta-analysis of the incidence of eradication
The pooled RRs for the incidence of eradication in the ITT and PP analyses of the probiotic group vs. the control group were 1.07 (95% CI: 1.02–1.11) (Figure 3a) and 1.04 (95% CI: 1.00–1.07) (Figure 3b), respectively. There was no significant heterogeneity among the studies in either analysis (I2 = 6.00%, P = 0.39 and I2 = 0, P = 0.51; respectively).
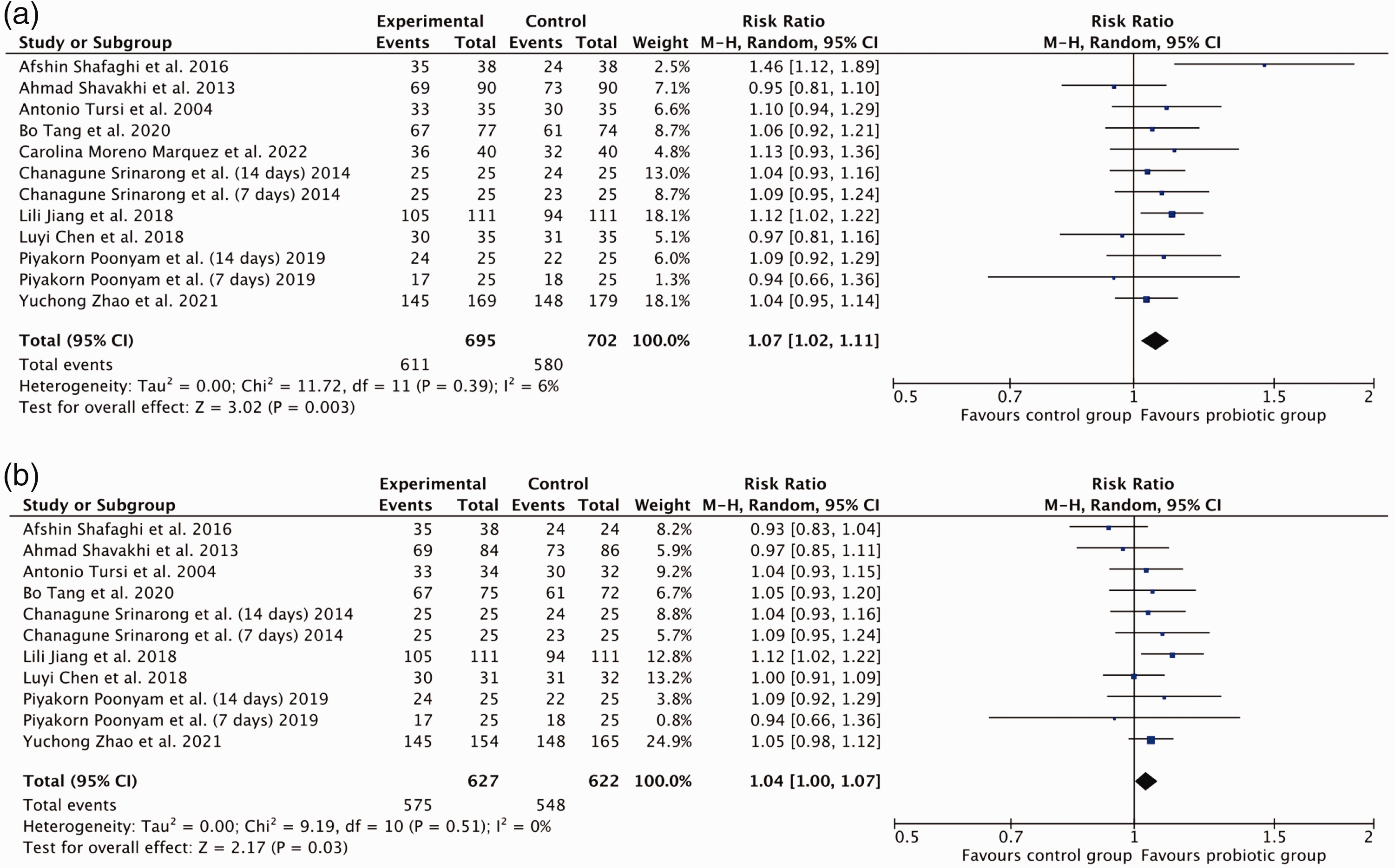
Effects of probiotic supplementation on the incidence of eradication on (a) an intention-to-treat basis and (b) a per-protocol basis.
Results of the meta-analysis of the adverse effects as whole
Probiotic supplementation reduced the incidence of adverse effects as a whole, and the pooled RR was 0.58 (95% CI: 0.37–0.91) (Figure 4a). However, there was significant heterogeneity among the studies (I2 = 59.00%, P = 0.04).

Effect of probiotic supplementation on the incidences of (a) side effects as a whole, (b) bitter taste, and (c) diarrhea.
Results of the meta-analysis of bitter taste
Probiotic supplementation reduced the incidence of bitter taste, and the pooled RR was 0.63 (95% CI: 0.40–0.99) (Figure 4b). There was no significant heterogeneity among the studies (I2 = 29.00%, P = 0.22).
Results of the meta-analysis of diarrhea
Probiotic supplementation reduced the incidence of diarrhea, and the pooled RR was 0.41 (95% CI: 0.25–0.67) (Figure 4c). There was no significant heterogeneity among the studies (I2 = 27.00%, P = 0.22).
Results of the meta-analysis of other side effects
Probiotic supplementation did not affect the incidences of abdominal discomfort (RR = 0.60, 95% CI: 0.21–1.74), nausea or vomiting (RR = 0.91, 95% CI: 0.53–1.57), constipation (RR = 0.73, 95% CI: 0.27–1.97), melena (RR = 1.03, 95% CI: 0.86–1.22), rash (RR = 0.85, 95% CI: 0.30–2.42), fatigue (RR = 1.15, 95% CI: 0.33–4.06), or dizziness (RR = 1.00, 95% CI: 0.44–2.27).
Publication bias
Funnel plots were used to assess the potential for publication bias with respect to the incidence of eradication for the ITT and PP analyses. Although slight asymmetry was identified in the plots for both parameters, neither Begg’s test nor Egger’s test revealed significant publication bias regarding the included studies (Figure 5).
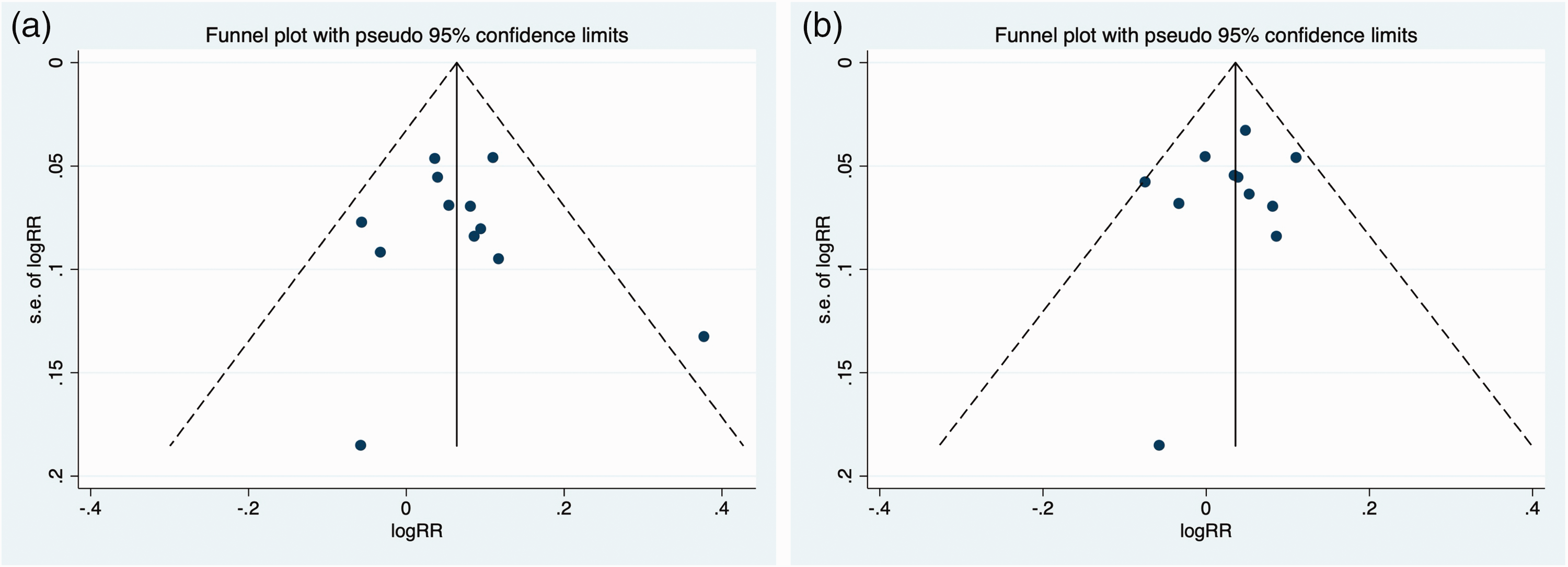
Assessment of publication bias for the included studies. Funnel plots for the incidences of eradication on (a) an intention-to-treat basis and (b) a per-protocol basis.
Discussion
A probiotic is defined as a living microbial species that may have a positive effect on the bowel microecology and improve health. Probiotics may be useful for the treatment of H. pylori infection. 35 A systematic review by Losurdo et al. yielded an OR of 7.91 for probiotic monotherapy vs. placebo for the eradication of H. pylori (95% CI: 2.97–21.05, P < 0.001). Thus, probiotics alone promote the clearance of H. pylori, 36 but at present we can only speculate regarding the mechanism underpinning this beneficial effect. Probiotics may secrete antibacterial substances, such as lactic acid, short-chain fatty acids, hydrogen peroxide, or bacteriocin. 37 They may also inhibit colonization by H. pylori by inhibiting adhesion or competing for binding sites.38,39 In addition, they may strengthen the mucous barrier, thereby inhibiting H. pylori colonization. 40 Furthermore, probiotics have been shown to reduce the local immune response and suppress the gastric inflammatory response.41,42 However, probiotic monotherapy is not recommended in the Maastricht VI/Florence consensus report. 12 A study by Yuan et al. showed that probiotic supplementation with a mixture of Bifidobacterium infantis, Lactobacillus acidophilus, Enterococcus faecalis, and Bacillus cereus ameliorates the gastric dysbiosis caused by eradication therapy. However, in another study, probiotic monotherapy significantly altered the gastric microbiota and did not reduce the H. pylori population. 43 Therefore, more evidence is required regarding the efficacy of probiotics for the therapy of H. pylori.
The Maastricht VI/Florence consensus report recommended that probiotics are used alongside antibiotic-based eradication therapy. 12 The present meta-analysis shows that, compared with BQT alone, BQT plus probiotics can increase the likelihood of eradicating H. pylori and reduce the incidences of side effects as a whole, diarrhea, and bitter taste. These findings are largely consistent with the results of previous meta-analyses. A meta-analysis by Zhang et al. showed that supplementation with probiotics increases the incidence of eradication achieved using anti-H. pylori therapy (OR = 1.94, 95% CI: 1.70–2.22), and reduces the incidence of side effects (RR = 0.56, 95% CI: 0.45–0.70). 44 Another by Zhu et al. yielded pooled ORs for the incidences of eradication and adverse effects in the probiotic group vs. the control group of 1.67 (95% CI: 1.38–2.02) and 0.49 (95% CI: 0.26–0.94), respectively. 45 We also found that probiotic supplementation reduced the incidence of diarrhea, consistent with the results of the studies by Zhang et al. and Zhu et al.44,45 Furthermore, the incidence of bitter taste was reduced by probiotic supplementation in the present study, but not in the other two studies.44,45 However, probiotic supplementation did not reduce the incidence of nausea and vomiting in our study or in the study by Zhu et al., but did so in the study by Zhang et al.44,45 Furthermore, the incidence of constipation was reduced in the study by Zhang et al., but not in the present study. 44 Heterogeneity in the probiotic or polypharmacy-based regimen used may lead to differing findings. In addition, the relatively low number of studies included in the present and previous meta-analyses with respect to each side effect may explain the differences. Finally, differing definitions of each symptom in these studies might also have had an effect. For example, epigastric pain, abdominal pain, abdominal distension, and flatus were analyzed separately in the study by Zhang et al., whereas these symptoms were analyzed together as “abdominal discomfort” in the present meta-analysis.
A meta-analysis performed by McFarland et al. showed that not all mixtures of probiotics improve the incidence of eradication of H. pylori or minimize the incidence of side effects. For example, a mixture of Lactobacillus acidophilus and Bifidobacterium bifidum was not effective at improving H. pylori eradiation; and a mixture of Lactobacillus acidophilus, Lactobacillus casei, and Bifidobacterium longum mix did not significantly reduce the incidence of adverse events”. 46 In addition, McNicholl et al. reported that Lactobacillus Plantarum and Pediococcus acidilactici neither improved the incidence of eradication nor reduced the incidence of side effects during H. pylori treatment. 47 These conflicting results might be at least in part explained by the heterogeneity of probiotics, in terms of their species and strains, the doses administered, and the duration of treatment. 48 The optimum interval between probiotic and antibiotic administration remains unclear, and may be another cause of conflicting results. Indeed, previous publications have not described the most suitable time for probiotic addition. 49 Although the co-administration of probiotics and antibiotic-based therapies can improve the treatment of H. pylori, it should be noted that such regimens have certain drawbacks. One major issue is that the antibiotics may restrain the growth of the probiotics. 50 In addition, the potential hazards associated with some probiotic strains should not be ignored. For example, Lactobacillus species can delay or limit the post-antibiotic recovery of the normal host–microbiota balance, resulting in long-term gut dysbiosis. 51 Therefore, further large-scale, well-designed RCTs are needed to clarify these issues.
The present study is the first meta-analysis of the benefits of probiotic supplementation alongside BQT for the treatment of H. pylori infection. Diarrhea and bitter taste are frequently reported side effects of BQT,52,53 and the results of the present meta-analysis suggest that probiotics alleviate these side effects. A rigorous search strategy, tight inclusion criteria, and appropriate statistical analyses were used in the present study. However, a limitation of the present meta-analysis is that the included studies had relatively few participants, which limited the quality of the clinical evidence obtained. Second, most of the included studies (8/10) were performed in Asia, which limits the generalizability of the findings to populations in other parts of the world. Third, the included studies differed with respect to the type and dosage of probiotic and the BQT regimen used, which limits the comparability of the results of these studies. Subgroup analyses aimed at identifying the most efficient probiotic strains were not performed because of this heterogeneity in the regimens used. Fourth, the possibility of publication bias was not assessed with respect to the incidence of side effects because of the small number of studies involved.
Conclusions
The popularity of probiotics notwithstanding, the findings of studies performed during recent decades regarding the efficacy of probiotics as part of the treatment of H. pylori have been contradictory. The results of the present meta-analysis supports the use of probiotics in combination with BQT in the clinical management of patients with H. pylori infection. However, further studies are needed to establish the optimal approach to the use of probiotic supplementation during the treatment of H. pylori infection.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231203841 - Supplemental material for Efficacy and safety of probiotic-supplemented bismuth quadruple therapy for the treatment of Helicobacter pylori infection: a systematic review and meta-analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605231203841 for Efficacy and safety of probiotic-supplemented bismuth quadruple therapy for the treatment of Helicobacter pylori infection: a systematic review and meta-analysis by Gaoyan Yao, Xiaoyuan Fan and Dewen Lu in Journal of International Medical Research
Footnotes
Acknowledgement
We acknowledge Ting Weng for critically reading the manuscript.
Author contributions
G.Y. conceived the study. G.Y. and X.F. selected the studies for inclusion and extracted the data. D.L. performed the statistical analyses. G.Y. interpreted the results and wrote the first draft of the manuscript. X.F. and D.L. conducted a critical review of the manuscript. All the authors approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
