Abstract
Background:
Esophageal gastrointestinal stromal tumors (E-GISTs) are highly uncommon and have not been thoroughly examined.
Objectives:
The objective of this multi-center study was to assess the viability of endoscopic resection (ER) in the treatment of E-GISTs and to explore its clinical implications.
Design:
This was a multi-center retrospective study. Consecutive patients referred to the four participating centers.
Methods:
E-GISTs among the consecutive subepithelial tumors (SETs) treated by ER methods were enrolled from April 2019 to August 2022. Clinicopathological, endoscopic, and follow-up data were collected and analyzed.
Results:
A total of 23 patients with E-GISTs were included for analysis, accounting for 1.9% of all the esophageal SETs (1243 patients). The average size of the tumor lesions was 2.3 cm (range 1.0–4.0 cm). We observed that tumors larger than 2.0 cm were more likely to grow deeper, with a statistically significant difference (p < 0.001). End bloc resection was achieved in all 23 patients. The mean operation time was 53.6 min (range 25–111 min). One patient experienced significant intraoperative bleeding, which was promptly managed endoscopically without necessitating surgery. The average hospital stay was 4.5 days (range 3–8 days). The overall median follow-up period was 31 months (range 13–47 months). No tumor recurrence, residual tumor, distal metastasis, or death was observed during the follow-up period.
Conclusion:
Based on our limited data, our study indicates that ER may be a feasible and effective option for treating esophageal GISTs measuring 4 cm or less. We suggest submucosal tunnel endoscopic resection as the preferred approach, as all E-GISTs in our study were situated in the muscularis propria layer. Additionally, tumors larger than 2 cm were more prone to deeper growth or extraluminal extension.
Introduction
Subepithelial tumors (SETs) of the gastrointestinal (GI) tract are increasingly being detected with the popularity of endoscopic screening. Upper GI SETs primarily consist of leiomyomas and gastrointestinal stromal tumors (GIST). 1 The stomach is the most common site for GISTs to arise (60–70%), followed by the small intestine (20–33%), and the large intestine (5%).2,3 Esophageal GISTs (E-GISTs) are extremely rarer, accounting for not more than 1–2% of all GISTs. 4
Generally, current guidelines recommend endoscopic surveillance for SETs smaller than 2 cm as they will be of low risk and their significance remains unclear. 5 However, for E-GISTs, the natural history and unpredictable biological behavior changes are not fully investigated due to the rarity of these lesions. Additionally, distinguishing potential GISTs from benign leiomyomas based on features or endoscopic ultrasound (EUS) manifestations is challenging, and obtaining an accurate pathological diagnosis of SETs can be difficult. Endoscopic ultrasound-guided fine needle aspiration (EUS-FNA) is a good method for obtaining the pathology of SETs, but its accuracy varies depending on the size of the lesion target and other factors. The diagnostic accuracies of EUS-FNA for lesion sizes of 2 and 2–4 cm were 71% and 86%, respectively, and in lesions >4 cm in diameter the accuracies ranged from 95% to 100%.6,7 Some patients are not willing to undergo a procedure that does not guarantee a definitive diagnosis.
The factors mentioned above necessitate the removal of relatively small esophageal SETs in certain circumstances. Moreover, continuous endoscopic surveillance without resection may pose risks for certain patients. Complete resection of tumors with free margins is the standard treatment for GIST. Enucleation and esophagectomy were previously the primary surgical methods for resecting E-GISTs. 8 However, these invasive surgical methods may be questionable for small E-GISTs, especially in the present era where numerous advanced endoscopic resection (ER) techniques have emerged as more minimally invasive options for upper GI SETs. One notable technique is submucosal tunneling endoscopic resection (STER) which was initially introduced by us in 2012 and recommended by American Society for Gastrointestinal Endoscopy (ASGE) in 2019.9,10 Numerous reports on STER have demonstrated its feasibility and safety in treating esophageal SETs. However, studies specifically addressing STER for E-GISTs are still scarce, with most of the existing literature consisting of case reports or small single-center series studies. Hence, data regarding the management of these lesions remain controversial. The objective of this multi-center study, the largest endoscopic series to date, was to assess the feasibility of using STER to treat E-GISTs and to discuss their clinical significance based on our own experience. Our aim is to offer a valuable reference for physicians and researchers in selecting the optimal treatment approach for patients with E-GISTs. The study was approved by the Ethics Review Board of Shanghai East Hospital, Taizhou Municipal Hospital, People’s Hospital of Liaoning Province, and the second affiliated hospital of BaoTou medical college.
Methods
Study design and patient selection
This was a multi-center retrospective study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 11 Consecutive patients referred to the four participating centers (Shanghai East Hospital, Taizhou Municipal Hospital, People’s Hospital of Liaoning Province, and the Second Affiliated Hospital of BaoTou Medical College) and presenting with esophageal Subepithelial leisons (SELs) underwent ER between April 2019 and August 2022. The inclusion criteria for this study were as follows: (1) lesions larger than or equal to 1.0 cm but smaller than 4.0 cm (maximum diameter ⩽4.0 cm); (2) patients expressing a strong desire for endoscopic treatment; (3) patients showing no signs of invasion of metastasis outside the digestive tract; and (4) GIST diagnosis confirmed by pathology following resection. Prior to the procedures, all patients provided informed consent. Those identified with E-GIST based on final pathological and immunohistochemical examinations were included in the study.
The exclusion criteria were as follows: (1) GISTs located within 3 cm of the esophageal inlet; (2) GISTs originating in the gastroesophageal junction; (3) patients with severe cardiovascular disease or blood coagulation disorders; and (4) patients with coagulation abnormalities (platelet count <50,000 and/or international normalized ratio >1.5).
STER procedures
EUS examinations were routinely conducted to assess the tumor’s location, echogenicity, size, shape, potential diagnosis, and depth, as well as to rule out invasion and metastasis beyond the GI tract. Computed tomography (CT) with three-dimensional reconstruction was requested for patients with larger tumors (>2.0 cm) to assess complete tumor images and their relationship with neighboring structures such as the aorta. 12 All procedures were carried out by experts with extensive experience in endoscopic submucosal dissection (ESD) or STER, each having performed over 500 cases. All patients were fasted for 12 h before the procedure. A single-accessory channel endoscope with a water jet system (GIF-Q260J; Olympus, Tokyo, Japan) attached with a transparent cap (D-201-11804; Olympus) was used to perform STER on patients under general anesthesia with endotracheal intubation. Carbon dioxide (CO2) insufflation was applied by a CO2 insufflator (UCR; Olympus). A high-frequency generator (ICC-200; ERBE, Tübingen, Germany) were used during the procedures. The detailed steps were almost the same as we described before 13 (Figure 1), except for ensuring complete resection of the GIST without disrupting the tumor capsule (using the ‘no-touch’ method) as a particularly important note. After exposing the tumor through the submucosal tunnel to create a working space, we performed the resection by excising the tumor along with its capsule and some surrounding normal tissues, ensuring no damage to the tumor. This was facilitated by using a cap attached to the front of the endoscope. If the tumor was dissected from the muscularis demanding full thickness incision, we consider it was originated from the deep muscularis propria (MP). Before closure of the tunnel incision site, we would routinely flush plenty of sterile water into the tunnel to avoid any invisible tumor seeding.

STER for a typical esophageal GIST. (a) Endoscopic view of the tumor. (b) The tumor was exposed after establishing the mucosal entry established. (c) Removal of the resected tumor after complete resection. (d) Endoscopic view of the submucosal tunnel after the tumor was removed. (e) The incision site was closed by clips. (f) The resected specimen.
Postoperative management
Prophylactic intravenous antibiotics were administered 30 min before general anesthesia. Intravenous proton pump inhibitor and antibiotics were routinely given as postoperative medications. Typically, unless specified otherwise, patients were instructed to fast for 2 days, transition to a liquid diet for 3 days, and then gradually resume a normal diet over a period of 2 weeks following ER. Postoperative observations encompassed monitoring for dyspnea, fever, chest pain, mediastinal emphysema, subcutaneous emphysema, pneumothorax, abdominal pain or distention, hematemesis or melena, and cyanosis or signs of peritonitis. Patients suffered from severe chest and/or abdominal pain would receive an abdominal/chest X-ray or CT scan examination. Thoracic paracentesis or closed drainage was performed as necessary in cases of pneumothorax. Patients without abnormal symptoms were discharged upon regaining the ability to consume oral intake.
Histology
The ER specimens were promptly immersed and fixed in a neutral 10% formalin solution within 30 min after the procedures. Standard hematoxylin and eosin (H&E) staining was performed. Specimens were fixed for 12–48 h to facilitate immunohistochemistry and molecular biology analysis accurately. Factors such as tumor size, mitotic rate, tumor primary site, and intraoperative integrity can all impact the prognosis of GISTs. Immunohistochemical staining of CD117, CD34, DOG1, as well as gene detection for KIT and PDGFRA if necessary were performed to confirm GIST diagnosis. Mitoses per 50 high-power field (HPFs) were counted and recorded to classify the risk group of GISTs. A widely used risk classification was proposed by the National Institutes of Health (NIH 2008 modified version), which categorizes tumors into low/very low risk, intermediate risk, and high risk according to the primary mitotic count, tumor size, and tumor site. 14 High-risk patients would receive an adequate Imatinib therapy (400 mg/day) for 3 years after procedures as the European Society for Medical Oncology (ESMO) guidelines committee proposed. 15
Follow-up
Follow-up gastroscopy and/or EUS were conducted at 6 and 12 months post-procedure, followed by annual check-ups. Patients classified as intermediate or high risk underwent contrast-enhanced CT scans every 6–12 months to assess for distant metastasis.
Outcome measures
We conducted a retrospective data collection, gathering information on chief complaints, age, gender, tumor location, EUS evaluation (tumor size, layers, echo signs), operation duration, complications, tumor mitotic index (<5 or >5 per 50 HPF), risk classification (NIH 2008 modified version), usage of pre- and/or post-surgical Imatinib therapy, and follow-up duration. The en bloc resection rate, complete resection rate, residual rate, recurrence rate, and mortality were analyzed to evaluate the effectiveness of ER procedures for E-GISTs, and the complication rate was documented to assess the safety of the procedures. In a prior study, minor intraoperative bleeding and mild emphysema were not classified as procedure-related adverse events due to their quick resolution during the procedures and minimal clinical impact. 12
Data collection and statistical analysis
Clinical, endoscopic, and pathological data were collected and analyzed. For the continuous variables, the statistical description was in the form of the median and range, and for the categorical variables, the counts and percentages were used for statistical description. The statistical analysis between groups was performed by using the Pearson’s Chi-squared test or the Fisher exact test as appropriate. p Values were two-sided, and p < 0.05 was considered statistically significant. Commercial software (IBM SPSS Statistics 20; SPSS, Chicago, IL, USA) was used for the statistical analysis.
Results
Clinical characteristics
From April 2019 to August 2022, a total of 1243 patients with esophageal SETs underwent ER at four centers. Among them, 23 patients were diagnosed with GIST and included in the analysis based on final histological findings, representing 1.9% of all esophageal SETs (see Supplemental Figure 1). The male-to-female ratio was 0.92 (11/12), and the median age of the patients was 61 years (range 43–72 years). Most patients were asymptomatic and incidentally identified through endoscopic screening, while six patients presented with atypical symptoms such as heartburn, regurgitation, and retrosternal or epigastric discomfort. Preoperative EUS indicated that all 23 lesions originated from the MP layer. The tumors were predominantly regular in shape, with an average size of 2.3 cm (range 1.0–4.0 cm). Six tumors exhibited extraluminal growth during resection, all of which were larger than 2 cm (refer to Table 1). A comparison between tumors ⩽2 and >2.0 cm revealed that tumors larger than 2.0 cm were more likely to grow deeper, and this difference was statistically significant (p < 0.001) (Table 2).
Clinicopathological characteristics of 23 E-GISTs patients.

E-GIST, esophageal gastrointestinal stromal tumors; MP, muscularis propria.
Comparison between E-GISTs smaller than or equal to 2 cm and larger than 2 cm.
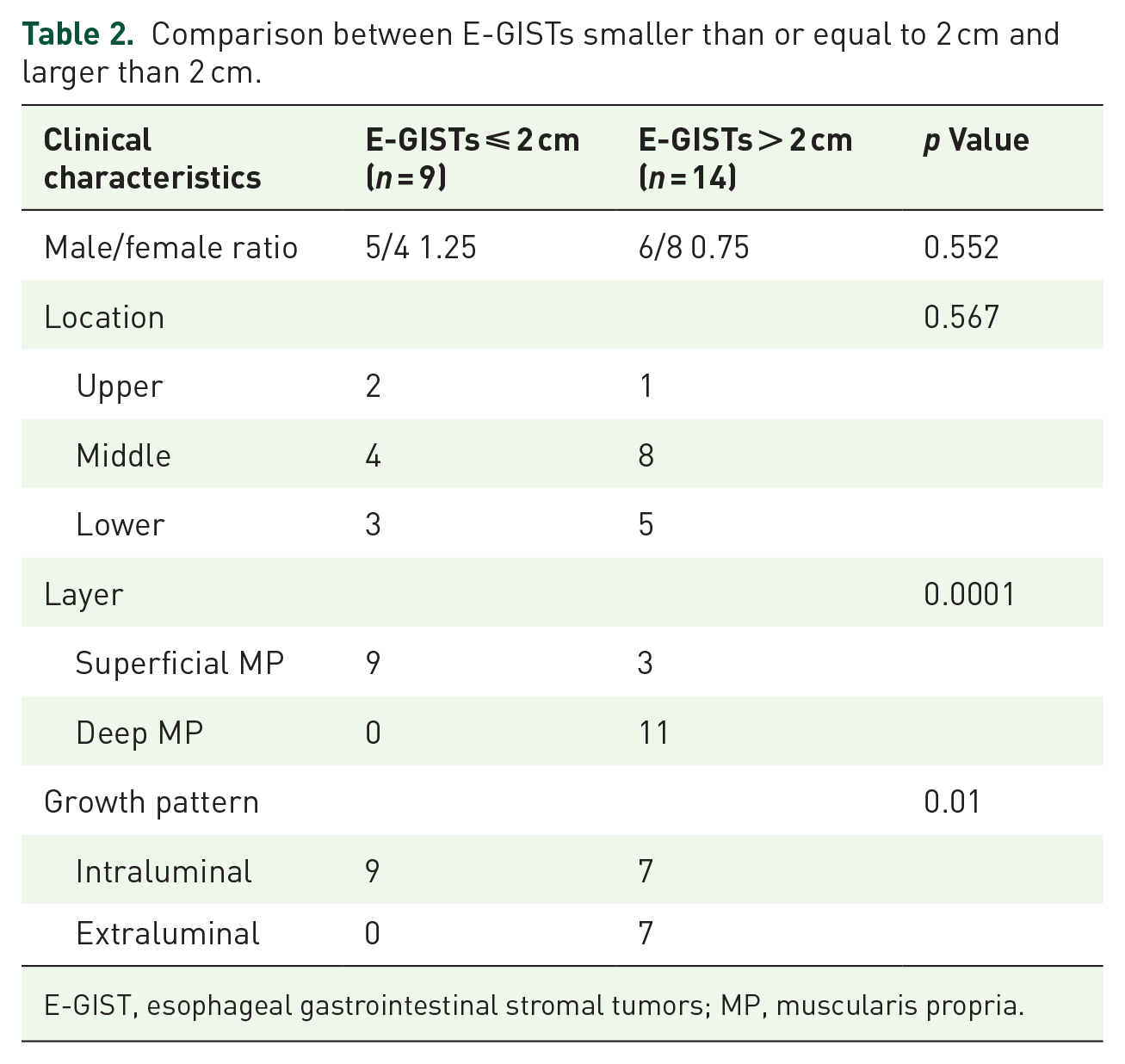
E-GIST, esophageal gastrointestinal stromal tumors; MP, muscularis propria.
Endoscopic and pathological outcomes
All 23 patients achieved en bloc resection successfully. The removed tumors were extracted using a snare or a net basket to grasp the tumor, followed by removal through the mucosal entry. For two tumors measuring 3.5 cm and larger, the tunnel entries were enlarged using a cutting knife to facilitate their extraction. The mean operation time was 53.6 min (range 25–111 min). Apart from actively enlarging the mucosal entry during STER procedures, no other mucosal injuries were observed. Closure of the tunnel incision sites involved the use of four to six clips (mean 5).
One patient with a 3 cm tumor experienced significant intraoperative bleeding, resulting in an estimated blood loss of 600 ml (based on intraoperative suction volume). Hemostasis was successfully achieved using hot biopsy forceps without the need for surgical intervention. Another patient developed chest pain and fever but recovered within 3 days with the help of antibiotics. There were no cases of pneumothorax, GI tract leakage, delayed bleeding, or secondary peritoneal/abdominal infections reported among the patients. In general, a full fluid diet is usually started 2–3 days after the operation, unless there are special circumstances. The average hospital stay was 4.5 days (range 3–8 days).
Pathology showed that the tumor mitoses were <5 per 50 huff in 22 patients, and >5 per 50 huff in one patient. Nine patients were evaluated as very low risk, 13 were low risk, and 1 was high risk according to the NIH 2008 modified version (4 cm, >5 per 50 huff). Molecular testing was performed on the high-risk tumor and showed mutations of KIT exon 11. The patient received postoperative Imatinib for 3 years as the ESMO guidelines committee proposed. 15 The overall median follow-up period was 31 months (range 13–47 months). No tumor recurrence, residual tumor, distal metastasis, or death was observed during the follow-up period (Table 3).
Effectiveness and pathological outcomes of endoscopic resection for E-GISTs n (%).
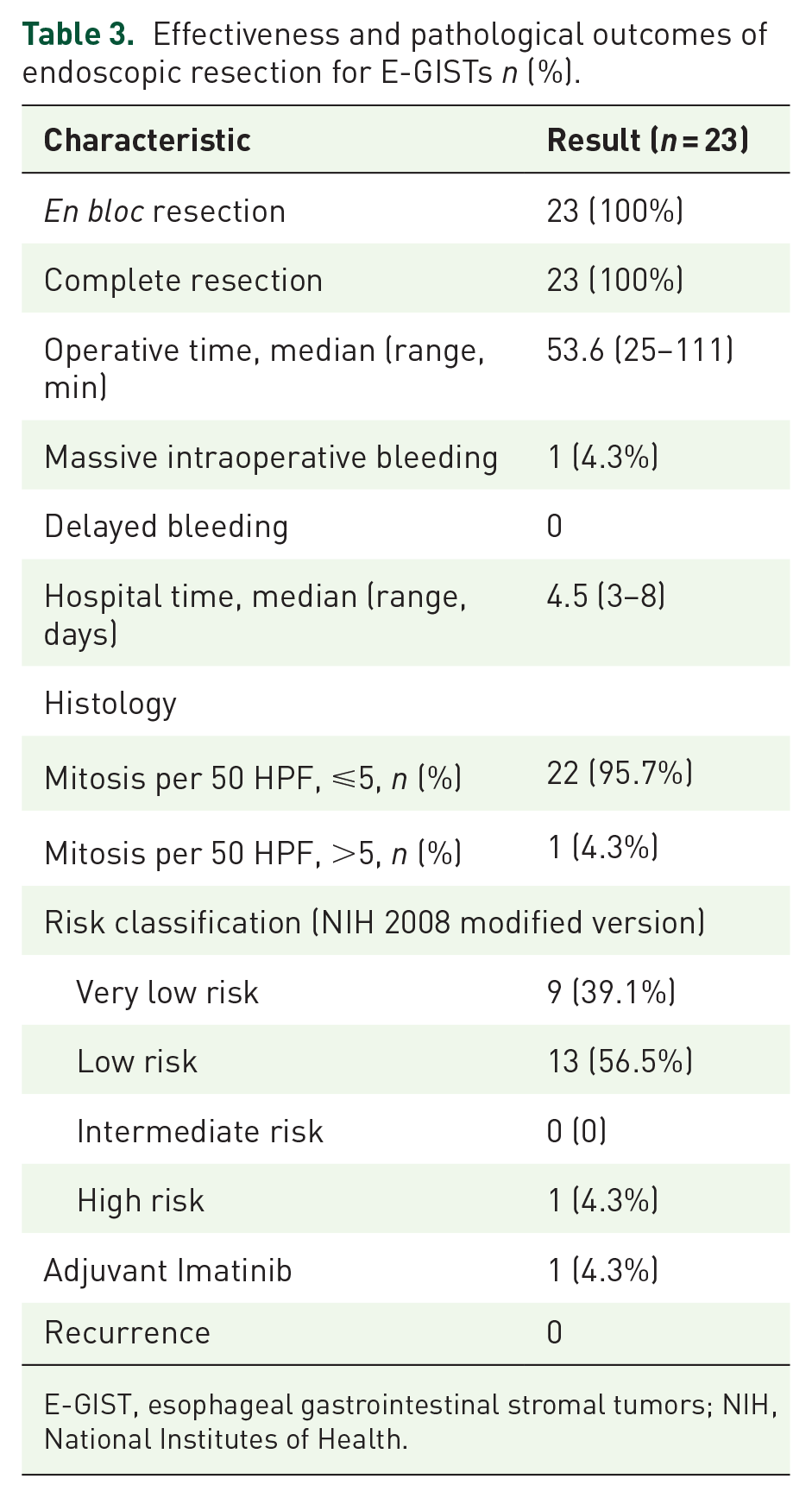
E-GIST, esophageal gastrointestinal stromal tumors; NIH, National Institutes of Health.
Discussion
GISTs occurring in the esophagus are uncommon, constituting a small percentage of all GIST cases. Our study found that out of 1243 patients with SETs, only 23 individuals (1.9%) were diagnosed with GISTs, consistent with the 2% incidence rate reported in existing literature. 5 While the fundamental pathophysiology is similar between esophageal and gastric GISTs, the natural course of GIST in the esophagus tends to be more aggressive compared to gastric GIST. Lott et al. 3 analyzed data of 55 patients with E-GIST from the literature and noted that, the overall survival rates were notably lower in E-GIST compared to gastric GIST. This difference may be due to the tendency for E-GISTs to exhibit higher mitotic rates and larger sizes at the time of diagnosis. 3 GISTs in other locations are smaller at the time of resection, leading to a more favorable prognosis. Therefore, it would make significant sense to perform early-stage resection for E-GISTs with small sizes. Numerous studies in the last decade have reported on ER as a minimally invasive approach for treating and accurately diagnosing esophageal SETs. However, investigations into endoscopic strategies for E-GISTs have been restricted to case studies and case series conducted at individual academic centers,16,17 making it challenging to draw any conclusions on their safety, feasibility, indication, and prognosis.
We enrolled the largest number of E-GISTs treated with ER therapy, with an average tumor size of 2.3 cm (range 1.0–4.0 cm), which was larger than in previously reported series where the mean size was 11.56 mm (range 6–21 mm). 16 All the tumors originated from the MP layer, and most of them were removed via STER. In the past, enucleation used to be preferred when treating smaller E-GISTs (2–5 cm). Although we did not compare the ER method with enucleation in this study, our previous research concluded that STER is more minimally invasive, with shorter procedure times and hospital stays compared to thoracic enucleation. 18
We recommended STER as the optimal ER strategy, as we observed that all the E-GISTs were located in the MP layer, and when tumors larger than 2 cm were more likely to grow deeper or even extraluminally. STER offers advantages over conventional ESD in terms of maintaining mucosal integrity and reducing the consequent risk of postoperative GI leaks. In our study, no mucosa tears or leaks occurred and only one patient experienced massive bleeding, which was successfully controlled without the need for surgical conversion or other severe consequence. For tumors growing extraluminally, transient subcutaneous emphysema were observed during the procedures but CO2 could be rapidly without the need for any intervention. Gas-related complications were used to be regarded as the most common complications for STER. 19 The use of endoscopic carbon dioxide insufflation, the proficient skills of the operators, and careful postoperative observations helped reduce the risk of gas-related complications. In our study, no severe complications requiring surgical treatment were reported.
In our preliminary study, we found that implementing STER for submucosal tumors with a long diameter ⩽5.0 cm and a transverse diameter ⩽3.5 cm could facilitate a high en bloc resection rate. Additionally, an irregular tumor shape was identified as a factor contributing to the likelihood of piecemeal resection. 20 Tumors with an oval or globular shape were considered regular in our observations. All the E-GISTs we encountered were regular in shape, leading us to speculate that the regular shape could be a key distinguishing characteristic between GISTs and leiomyomas. This suggests that large irregular tumors are more likely to be leiomyomas rather than GISTs. We achieved a 100% en bloc resection rate within a relatively short procedural time (median 56 min), including for the two large tumors measuring 3.5 and 4 cm in maximum diameters. Complete en bloc resection is crucial for GISTs due to their potential malignancy, and intraoperative tumor rupture is strictly prohibited. However, piecemeal resection of other benign SETs such as leiomyomas did not impact long-term outcomes. The key technique in this study revolved around ensuring the integrity of the tumor throughout the procedure. Our experience has shown that it is crucial to create a wide enough submucosal tunnel to provide ample dissection space for exposing the tumor boundary. Subsequently, the tumor should be separated along with the surrounding normal tissues to maintain a no-touch approach with the tumor capsule. It was crucial to make a significant incision at the tunnel entry to ensure the complete removal of a tumor as large as 4 cm. Additional clips were necessary to close the incision site in this instance. Our conclusion suggests that STER may be considered a viable option for treating E-GISTs measuring 4 cm or smaller. The latest cohort study noted that the risk of progression in E-GISTs is correlated with elevated mitotic activity (>5/50 HPF) and a size exceeding 5.0 cm. 21 Therefore, it is indeed sensible to remove these tumors at an early stage using minimally invasive and acceptable methods. In our study, only a 4 cm tumor was proved to have a mitotic index >5/50 HPF and was classified as a high-risk GIST. This female patient was recommended to accept Imatinib as an adjuvant therapy for 36 months to prevent recurrence. She was followed-up for 44 months without any recurrence or metastasis. The long-term outcomes of E-GISTs in the present study were excellent, no any other patient had signs of disease progression during a median follow-up of 31 months (range 13–47 months). Our study seems to be the first multi-center report about ER for E-GISTs. Nevertheless, there are some limitations to this research: firstly, the study design was retrospective with small population; secondly, the outcomes of ER were not compared with other conventional treatments; thirdly, the median follow-up time was relatively short. Therefore, a prospective, randomized controlled study with longer-term follow-up period is essential in the future to validate the observed results.
Conclusion
Based on our limited data, our study indicates that ER may be a feasible and effective option for treating E-GISTs measuring 4 cm or less. We suggest STER as the preferred approach, as all E-GISTs in our study were situated in the MP layer. Additionally, tumors larger than 2 cm were more prone to deeper growth or extraluminal extension.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241255304 – Supplemental material for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study
Supplemental material, sj-docx-1-tag-10.1177_17562848241255304 for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study by Jingjing Lian, Yingjie Ji, Tao Chen, Guoxiang Wang, Mizhu Wang, Shengxi Li, Jia Cao, Li Shen, Wei Lu and Meidong Xu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-docx-2-tag-10.1177_17562848241255304 – Supplemental material for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study
Supplemental material, sj-docx-2-tag-10.1177_17562848241255304 for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study by Jingjing Lian, Yingjie Ji, Tao Chen, Guoxiang Wang, Mizhu Wang, Shengxi Li, Jia Cao, Li Shen, Wei Lu and Meidong Xu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-3-tag-10.1177_17562848241255304 – Supplemental material for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study
Supplemental material, sj-jpg-3-tag-10.1177_17562848241255304 for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study by Jingjing Lian, Yingjie Ji, Tao Chen, Guoxiang Wang, Mizhu Wang, Shengxi Li, Jia Cao, Li Shen, Wei Lu and Meidong Xu in Therapeutic Advances in Gastroenterology
Supplemental Material
sj-jpg-4-tag-10.1177_17562848241255304 – Supplemental material for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study
Supplemental material, sj-jpg-4-tag-10.1177_17562848241255304 for Endoscopic resection for esophageal gastrointestinal stromal tumors: a multi-center feasibility study by Jingjing Lian, Yingjie Ji, Tao Chen, Guoxiang Wang, Mizhu Wang, Shengxi Li, Jia Cao, Li Shen, Wei Lu and Meidong Xu in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
We would like to thank Dr Yuan Zhao for her contribution to language editing of our manuscript. Special thanks to Professor Li Zhang for his expertise in diagnosing and interpreting our postoperative pathology reports. We also acknowledge the contributions of all team members involved in the study and data analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
