Abstract
Background:
Irritable bowel syndrome (IBS) is a disorder of gut-brain interaction characterized by recurrent abdominal pain related to defecation and/or associated to a change in bowel habits. According to the stool type, four different IBS subtypes can be recognized, constipation predominant (IBS-C), diarrhea predominant (IBS-D), mixed (IBS-M), and undefined (IBS-U). Patients report that their IBS symptoms are exacerbated by food. Thus, it is important to find a nutritional approach that could be effective in reducing IBS symptoms.
Objective:
The present work is a post hoc analysis of the previously published DOMINO trial. It aimed to evaluate the effects of a self-instructed FODMAP-lowering diet smartphone application on symptoms and psychosocial aspects in primary care IBS stratifying the results for each IBS subtypes.
Design:
Post hoc analysis.
Methods:
Two hundred twenty-two primary care IBS patients followed a FODMAP-lowering diet for 8 weeks with the support of a smartphone application. Two follow-up visits were scheduled after 16 and 24 weeks. IBS–Symptoms Severity Score (IBS-SSS), quality of life (QoL), and adherence and dietary satisfaction were evaluated.
Results:
After 8 weeks, IBS-SSS improved in all IBS subtypes (p < 0.0001). Physician Health Questiionnaire (PHQ-15) improved only in IBS-D (p = 0.0006), whereas QoL improved both in IBS-D (p = 0.01) and IBS-M (p = 0.005).
Conclusion:
This post hoc analysis showed that the app is useful in all IBS subtypes; thus, it could be used as an effective tool by both general practitioners and patients to manage symptoms in primary care.
Trial registration:
Ethical Commission University Hospital of Leuven reference number: S59482. Clinicaltrial.gov reference number: NCT04270487.
Plain language summary
The low FODMAP (fermentable oligo-, di-, and monosaccharides and polyols) diet has shown efficacy for controlling IBS (irritable bowel syndrome) symptoms in small controlled trials in tertiary care patients. As this approach requires several visits with an experienced dietitian, it seems less suitable for primary care.
The benefit of the FODMAP lowering app was already present at 4 weeks and persisted during follow-up until 24 weeks.
Given its superiority to standard first-line pharmacotherapy, and its ease of use, a FODMAP lowering app has the potential to become the preferred first-line treatment for primary care IBS.
Introduction
According to Rome IV criteria, IBS is a disorder of gut-brain interaction 1 characterized by recurrent abdominal pain at least once a week in the previous 3 months related to defecation and/or associated to a change in the evacuation frequency or in the stool type. 2 The latter defines four different subtypes: IBS with constipation (IBS-C), IBS with diarrhea (IBS-D), mixed IBS (IBS-M), and undefined IBS (IBS-U). 2
Regarding epidemiology, a recent study conducted on more than 73,000 subjects, both through questionnaires via internet and household surveys, underlined that IBS affects about 4% of the global adult population, with a major prevalence among women. 1 Moreover, the prevalence of IBS subtypes is 32.4% IBS-C, 32.4% IBS-M, 28.7% IBS-D, and 6.5% IBS-U based on the survey. 1 IBS symptoms have a major impact on the patients’ quality of life (QoL). 3 In fact, this condition leads to absenteeism from work and to a higher number of general practitioners’ or specialists’ consultations.1,4
Although its etiology is not completely understood, 2 different therapeutic approaches are available, be it with limited efficacy. 5 More than 70% of IBS patients report that their symptoms are worsened by the meal,6,7 and therefore, lately, dietary interventions have gained more attention.8,9 The first-line approach for the treatment of IBS consists in encouraging healthy eating and lifestyle changes5,10 according to the National Institute for Health and Care Excellence (NICE) guidelines and the British Dietetic Association (BDA).11,12 Currently, the low FODMAP diet, a diet in which fermentable oligosaccharides, disaccharides, monosaccharides, and polyols (FODMAP) are excluded, has shown beneficial effect in clinical trials 13 and meta-analyses,14,15 therefore, gaining increasing interest for the treatment of tertiary care IBS. 16 However, not all experts support the efficacy of the low FODMAP diet, 17 and the available evidence is considered to be of low quality. 3 Moreover, the low FODMAP diet is very restrictive, needs the close follow-up of an experienced dietitian and the compliance is variable.18,19 In addition, it has been shown that it could negatively affect the microbiome by reducing levels of Bifidobacteria spp. and short-chain fatty acids (SCFAs) production.10,19,20
The recently published DIet Or Medication in Irritable bOwel syNdrOme (DOMINO) trial showed that in the primary care a FODMAP-lowering diet provided by a self-instructed mobile application was more effective than standard medication, otilonium bromide, after a 8-week treatment. 21 This diet does not exclude FODMAPs but only reduced them without determining nutritional deficiencies. The present work is a post hoc analysis of the DOMINO trial 21 that aimed to evaluate in detail the effect of the diet application on symptoms, psychosocial aspects, and QoL in the diet arm stratifying the results for each IBS subtype.
Materials and methods
Study design
This pragmatic study represents a sub-analysis of the DOMINO trial 21 and provides a stratification of the diet arm according to stool types. Patients were divided in four subtypes (IBS-C, IBS-D, IBS-M, and IBS-U) according to the evaluation of general practitioners during the screening phase.
The study protocol lasted 6 months and was divided in five study visits (Figure 1). All study visits were conducted by general practitioners in a pragmatic study setting. Before every study visit patients had to complete online questionnaires. During, the ‘patient alone’ visit (after 4 weeks of treatment), patients completed online questionnaires at home without visiting the physician.
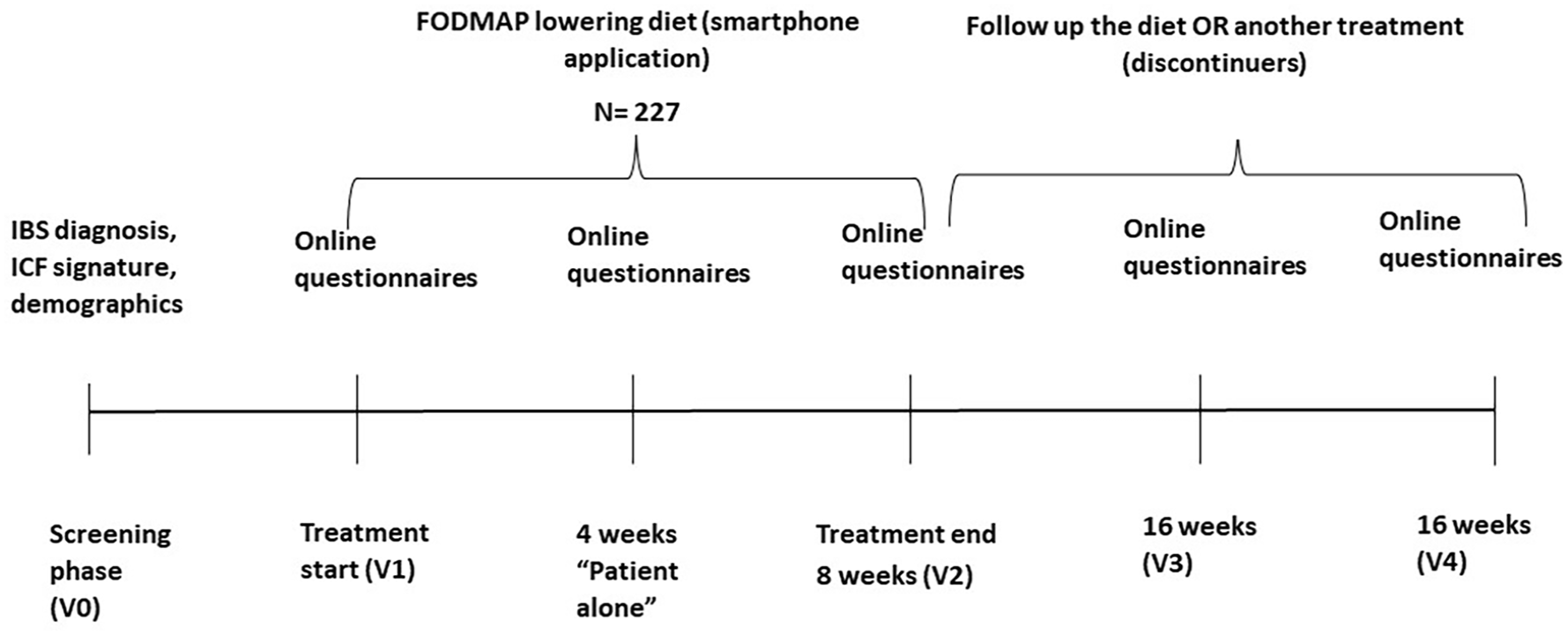
Study protocol.
Study population
In this study, 227 subjects were randomized to the diet arm in accordance to the inclusion and exclusion criteria. 21 Patients with a concurrent Gastrointestinal (GI) disease or a history of a major abdominal surgery, diabetes, or uncontrolled coexisting diseases (thyroid dysfunction, cancer, active endometriosis, psychiatric disorders), or women who were pregnant, lactating, or planning a pregnancy were excluded. Moreover, patients who had previously followed a FODMAP-lowering diet or any other elimination diets were also excluded. 21
The population was divided according to IBS subtypes as diagnosed by the general practitioners at visit 0. The majority of the patients were IBS-M (38%), followed by IBS-D (29%), IBS-C (20%), and IBS-U (13%). In accordance with a pragmatic trial, patients were allowed to change treatment in dialog together with their GP after the 8 weeks treatment period. Patients who changed treatment were defined as ‘discontinuers’. Discontinuers’ data are presented in the Supplemental Material. The ‘whole population’ includes the participants that have continued the diet throughout the entire study without changing their treatment (hence, the study population without discontinuers).
Dietary intervention
The prescribed diet was a FODMAP-lowering diet, based on NICE and BDA dietary recommendations11,12 of what food to avoid or reduce intake of without caloric restriction. The diet was self-administered and the patients were given information about the diet through an electronic application for smartphone and/or tablet (developed by EverywhereIM, Amsterdam, the Netherlands). If by any means the participants were not able to download the app or to use the application, they were provided a booklet with all information and dietary instructions (2%). 21 In total, 105 recipes were provided for breakfast, lunch, snacks, and dinner. Patients were also able to create their own menu or a shopping list via the app. The diet needed to be self-instructed, for patients to understand and perform by themselves. GPs were therefore instructed to not give additional information or instructions to their patients, other than on how to download the app.
Questionnaires
Participants were requested to complete questionnaires during the study before each study visit (Figure 1). They had to complete the IBS–Symptom Severity Scale (IBS-SSS) to evaluate symptom changes during the study. The IBS-SSS was composed by seven questions about abdominal pain intensity and frequency, abdominal distention and bloating, bowel habits satisfaction and impact of symptoms on daily life. The IBS-SSS ranges from 0 to 500, whereas higher scores indicate more severe symptoms. Patients with IBS-SSS score <175 are defined as mild IBS, 175–300 as moderate IBS, and scores >300 as severe IBS. 22 Responders were defined as patients who had decreased 50 or more points on the IBS-SSS compared to baseline.
Moreover, patients completed questionnaires to evaluate anxiety Generalised Anxiety Disorder questionnaire (GAD-7), depression (PHQ-9), and somatization (PHQ-15). 23 QoL was evaluated through a questionnaire composed of 34 items, whereas the overall QoL score and its eight subscales (dysphoria, interference with activity, body image, health worry, food avoidance, social reaction, sexual activities, and relationship) can be studied. 24
Participants were also asked to complete adherence and satisfaction questionnaires. Adherence was evaluated with the question: ‘How often have you forgotten to follow the prescribed diet for IBS?’ and patients could select the number of time on a Likert scale (from 0, never, to 7, several/most of the time or constantly). Patients who forgot at least 2 days/week to follow the diet, were considered ‘non-adherent’. Treatment satisfaction was recorded by the question: ‘Thinking about the past week, how are your irritable bowel symptoms compared to what they were before receiving the treatment?’. The score was calculated on a Likert scale (from 0, extremely worse, to 9, extremely better).
Statistical analysis
The statistical program Graphpad Prism 9.4.1. Graphpad Company, Boston MA, U.S.SA. version was used to analyze the data. A one-way Analysis of Variance (ANOVA) with Dunnett’s post hoc test was used to analyze changes between each time point and baseline. A chi-square test was used to evaluate percentages of responders rates, adherence and treatment satisfaction as well as some sub-questions of the IBS-SSS questionnaire. A p value <0.05 was considered to be statistically significant.
Results
Study participants
Of the 227 patients, 5 patients withdrew their consent and dropped out of the trial (V1). In total, 222 patients (85 IBS-M, 62 IBS-D, 44 IBS-C, and 31 IBS-U) were randomized and started the intervention phase of the study (Table 1). No statistically significant differences among subtypes for age, height, weight, and Body Mass Index (BMI) were observed at baseline (Figure 2).
Demographical parameters of the IBS subtypes at V1 (average ± SD).

One-way ANOVA with Dunnett’s post hoc test was conducted.
IBS-C, IBS with constipation; IBS-D, IBS with diarrhea; IBS-M, mixed IBS; IBS-U, undefined IBS.

Study participants.
In total, 219 patients completed the 8 weeks of treatment and 176 patients continued with the allocated treatment for an additional 16 weeks. As this study was pragmatic, 59 patients decided to change treatment during the follow-up period. Details on the discontinuers are shown in the Supplemental Figure S1. At the end of the follow-up period (24 weeks), 152 subjects continued the treatment with the lowering FODMAP diet mobile application (59 IBD-M, 39 IBS-D, 30 IBS-C, and 24 IBS-U).
Body weight
Body weight did not change significantly through all study visits for all subtypes (Table 2). No significant changes were observed from each visit to the baseline.
Body weight of the IBS subtypes at each time point (average ± SD).
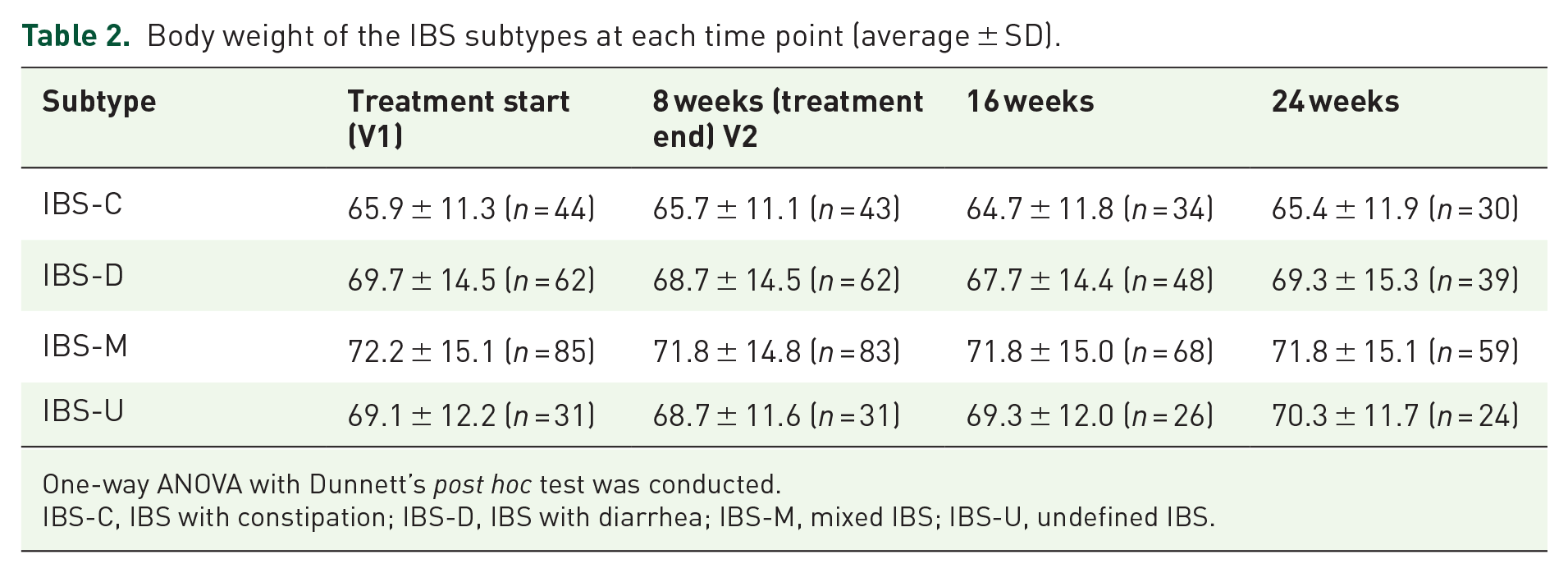
One-way ANOVA with Dunnett’s post hoc test was conducted.
IBS-C, IBS with constipation; IBS-D, IBS with diarrhea; IBS-M, mixed IBS; IBS-U, undefined IBS.
Symptom severity
At the end of the dietary treatment (8 weeks), all subgroups, showedsignificant improvement in the IBS-SSS score from baseline (all p < 0.0001). Improvement was maintained at week 16 and 24 for all subtypes (all p < 0.0001). Table 3 summarizes the IBS-SSS scores in the four subtypes for each time point.
IBS-SSS score in the whole population (average ± SD).

Four weeks: it was a ‘patient alone’ time point, there was no visit, thus some patients did completed the questionnaires; one-way ANOVA with Dunnett’s post hoc test was conducted.
p < 0.05. **p < 0.0001.
IBS-C, IBS with constipation; IBS-D, IBS with diarrhoea; IBS-M, mixed IBS; IBS-SSS, IBS-Symptoms Severity Score; IBS-U, undefined IBS.
According to IBS-SSS score, 65% of the patients were defined as responders the diet after 4 weeks and 72% after 8 weeks. This value remained high (70%) until the end of follow-up period (24 weeks). While evaluating subgroups, IBS-C showed the highest benefit of the diet after 8 weeks with a responder rate of 79%, followed by IBS-U (74%), IBS-D (70%), and IBS-M (67%). After 6 months of treatment, the responder rate of the IBS-C group remained higher than 75% (Figure 3 below and Supplemental Table S2).

IBS-SSS responders rate.
Evaluation of the individual questions of the IBS-SSS (Table 4) showed a decrease in abdominal pain intensity from 53.4 ± 23.5 at V1 to 36.5 ± 25.2 at 8 weeks in IBS-C (p < 0.05), from 51.9 ± 26.5 to 32.9 ± 24.8 for IBS-D (p < 0.05), and from 49.8 ± 24.9 to 38.9 ± 25.4 (p < 0.05) but not in IBS-U. In addition, intensity of bloating decreased significantly from 61.8 ± 21.7 at V1 to 39.1 ± 29.3 (p < 0.05) at 8 weeks in IBS-C, from 50.6 ± 28.4 to 29.8 ± 26.0 (p < 0.05) in IBS-D, from 52.9 ± 27.9 to 37.1 ± 28.5 (p < 0.05) in IBS-M, and from 46.1 ± 24.2 to 24.8 ± 25.0 in IBS-U. Abdominal pain and bloating was even more decreased after 16 weeks of treatment in all subgroups (p < 0.05), except for IBS-U and the reduction was higher in IBS-M group (p < 0.0001) for abdominal pain. In IBS-U subgroup we observed only a significant reduction in bloating at 16 weeks (p < 0.05).
IBS-SSS questions in the whole population.

p Value Baseline versus W4, 8, 16, 24; *p < 0.05, **p < 0.0001.
average, one-way ANOVA; %, chi-square test; IBS-C, IBS with constipation; IBS-D, IBS with diarrhoea; IBS-M, mixed IBS; IBS-SSS, IBS-Symptoms Severity Score; IBS-U, undefined IBS.
The percentage of participants who observed a reduction in their abdominal pain decreased significantly (p < 0.05) of 14% in IBS-C, of 15% in IBS-D, of 10% in IBS-M while for bloating it decreased of 17% (p < 0.05) in both IBS-C and IBS-D at 8 weeks. No significant changes have been seen for IBS-M and IBS-U.
The number of days in which participants observed abdominal pain in the last 10 days also decreased significantly in all subgroups (p < 0.05 for IBS-M and IBS-U and p < 0.0001 for IBS-D) except for IBS-C.
Finally, a significant reduction was observed for dissatisfaction for bowel habits in IBS-C, IBS-D, IBS-M (p < 0.001 for all), and in IBS-U (p < 0.05) from 8 weeks compared to the baseline. This result was maintained at 16 and 24 weeks. Patients also reported a significant reduction on how the abdominal symptoms interfered with the daily life. This was in IBS-C (p < 0.0001), IBS-D (p < 0.0001), IBS-M (p < 0.05), and IBS-U (p < 0.05) and this result was maintained also during the follow-up period. Individual IBS-SSS question analysis is reported in Supplemental Table S3.
Quality of life
At 8 weeks QoL improved only in the IBS-D (p < 0.05) and in the IBS-M (p < 0.05) subtypes compared to baseline (Table 5). Regarding QoL subscales we noticed significant variations in the IBS-D and IBS-M subtypes for dysphoria (p < 0.05 and p < 0.0001, respectively); interference with activity (p < 0.05, in both cases); body image (p < 0.05, in both cases), social reaction (p < 0.05, in both cases). Only dysphoria changed significantly also in the IBS-C subgroup (p < 0.05) at 8 weeks. No significant changes were seen in every subtype for health worrying, food avoidance, sexual activities, and relationship.
Subscales of QoL questionnaires in the IBS subtypes in the whole population (average ± SD).

One-way ANOVA with Dunnett’s post hoc test was conducted.
p < 0.05, **p < 0.0001.
IBS-C, IBS with constipation; IBS-D, IBS with diarrhoea; IBS-M, mixed IBS; IBS-U, undefined IBS.
Somatization status (PHQ-15), PHQ-9 depression scale, and GAD-7 anxiety severity
In IBS-D, a significant reduction was observed after 4, 8, 16, and 24 weeks (p = 0.0006, p = 0.0006, p = 0.0004, and p < 0.0001, respectively), whereasIBS-M decreased significantly after 16 weeks (p = 0.03). In the IBS-C and IBS-U subtypes, no significant changes were observed. Depression scale (PHQ-9) decreased only in the IBS-D subtype after 16 (p = 0.03) and 24 weeks (p = 0.02), whereasnxious severity scale (GAD-7) significantly changed after 24 weeks (p = 0.02) (Table 6).
PHQ-15, PHQ-9, and GAD-7 questionnaire in the IBS subtypes in the whole population (average ± SD).

One-way ANOVA with Dunnett’s post hoc test was conducted.
p < 0.05, **p < 0.0001.
IBS-C, IBS with constipation; IBS-D, IBS with diarrhea; IBS-M, mixed IBS; IBS-U, undefined IBS.
Treatment adherence and satisfaction
Participants who forgot more than twice/week to follow the diet were considered as non-adherent. Thus, we observed that adherence values were high in all subtypes (70% in IBS-D, 67% in IBS-C and IBS-M, and 55% in IBS-U). After 8 weeks IBS-D had the higher adherence rate (72%) followed by IBS-C (67%), IBS-M (65%), and IBS-U (53%). At the end of the follow-up period, the IBS-M subgroup was the one who remained more adherent to the protocol (77%), followed by IBS-D (66%), IBS-U (56%), and IBS-C (54%) (Figure 4 and Supplemental Table S4).

Adherence in the IBS subtypes in the whole population.
Regarding treatment satisfaction, the mean values ranged from 45% to 53%. After 16 and 24 weeks, the satisfaction scores increased more than after the first 8 weeks (>50%). This was significant for IBS-U (58%) and IBS-M (56%) after 16 weeks and for IBS-C and IBS-M (57% in both cases) after 24 weeks (Figure 5 and Supplemental Table S5).
Satisfaction in the IBS subtypes in the whole population.
Discussion
To date, it is well established that a low FODMAP diet improves symptoms in IBS patients,25,26 but there is a lack of evidence in the literature of large randomized controlled trials on the effects of this diet both in the primary care setting and for the different subtypes of IBS. The DOMINO study provided nutritional recommendations that merge the indication of a lowering FODMAP diet and traditional dietary advice for IBS with the support of a smartphone application. The diet therapy was not a strict FODMAP diet and, in fact, resulted in high adherence (between 55% and 70% in a pragmatic approach). Furthermore, as the mobile application was self-instructed, the patients could self-manage their symptom control through avoidance of listed food items.
In this specific, post hoc analysis, we focused on the therapeutic impact of the DOMINO diet application in the different IBS stooltypes. Due to the heterogeneity of IBS symptoms, 5 it is difficult to determine a single nutritional protocol for the treatment of IBS. The diet suggested did not cause important weight loss during the study in any of the IBS subgroups. In fact, the diet protocol was aimed not to decrease the amount of calories ingested, and thus it helped to maintain the participants’ body weight until 8 weeks and also during the follow-up period. Symptom relief was observed in all IBS subtypes with the use of the FODMAP-lowering diet application at every study visit for all different IBS subtypes. IBS-C, IBS-U, and IBS-D showed a higher response rate, and IBS-C still remained more than 70% of response also after 24 weeks. The reduction in symptoms also resulted in a high satisfaction rate, which was almost 50% among groups.
There is lack of literature evaluating diet therapies in the IBS stooltypes. The study by Nybacka et al. 27 showed that FODMAP intake was generally similar among the various subtypes of IBS and that higher FODMAP intake correlated with greater symptom severity in all subtypes. Ong et al. conducted a single-blind crossover study to evaluate FODMAP restriction on symptoms in 15 healthy volunteers and 15 subjects with IBS (7 IBD-C, 4 IBS-D, 2 IBS-M, and 2 IBS-U). They followed a FODMAP-restricted diet (9 g/day) or a high FODMAP diet (50 g/day) for 2 days. Different IBS symptoms decreased significantly in the IBS group compared to healthy controls, although IBS subjects produced more gas during both nutritional protocols. Methane production was more associated with IBS-C subjects possibly due to the slower transit. 28 In a study of Halmos et al., a single-blind, placebo-controlled, crossover study, 30 patients with IBS were randomized to a low FODMAP diet or a traditional Australian diet. The low FODMAP group showed greater symptom improvement after 3 weeks, but they noted that after the first week, all subtypes of IBS showed symptom improvement with no statistical differences between them, although the groups were limited in size. 26 Our study, through a FODMAP-lowering diet, determined an improvement in symptoms in all IBS subtypes, in a large primary care IBS patient group.
Generally, IBS patients have higher levels of anxiety and depression compared to healthy controls and they seem to be more associated to IBS-M, even if the prevalence seems to be higher in IBS-C subtype, followed by IBS-M and IBS-D (check references).29,30 In the DOMINO study, the results showed that even if IBS-D started with the lowest depression scores, it was the only subgroup that had a significant improvement during the follow-up period. Although a similar trend was noted, we did not observe any significant changes for the constipation and mixed types in anxiety and depression scores. Regarding the somatization status (PHQ-15), the IBS-D and IBS-M subtypes showed an improvement from week 4 to week 24 and after 16 weeks, respectively. QoL showed to be significantly improved for IBS-D and IBS-M. However, in IBS-C and IBS-U the improvement was only observed after 24 weeks.
A limitation of our study is that, as this study was organized in a pragmatic setting, the use of daily diaries for symptoms and food ingestion was restricted; therefore, there is uncertainty to the actual amount of FODMAP ingested by the patients. Nevertheless, as the overall result was favorable, the diet can still be considered a viable alternative treatment option. To date, the application device is not available worldwide, but only in Belgium. A future perspective is to make it available internationally with traditional country-related recipes.
The low-FODMAP diet as regularly used in a tertiary centers, is too strict and needs the supervision of an experienced dietitian.31–34 The DOMINO trial app suggests a less strict diet that can be self-instructed and for which patients can play an active role in the management of their disease. Dietitians are relatively expensive and are not always available to guide patients in follow-up of the low-FODMAP diet. In this case also, the DOMINO app provides a useful solution. The main key point of the present study is that it was conducted in a large primary care cohort in a pragmatic setting, and it is therefore promoting the availability of a novel and beneficial therapeutic approach (the DOMINO diet app) that is also feasible to use in a primary care setting.
Even though there exist a number of dietary mobile applications providing dietary advise for IBS, no large-scales randomized clinical trials have been reported with these application as the actual therapeutic strategy. Previous studies have used electronic devices, such as smartphone applications, but they were mainly focused on symptom monitoring and diet adherence (generally for low FODMAP).35,36
Overall it can be concluded that the DOMINO dietary treatment should be considered the first-line therapy in all subtypes IBS. 37 This study showed that the app works for all IBS subtypes in reducing symptoms and psychological factors and QoL in a long-term period of 24 weeks. The nutritional protocol is effective and feasible for patients and clinicians and there are no risks for weight loss.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848241255296 – Supplemental material for DOMINO trial post hoc analysis: evaluation of the diet effects on symptoms in IBS subtypes
Supplemental material, sj-pdf-1-tag-10.1177_17562848241255296 for DOMINO trial post hoc analysis: evaluation of the diet effects on symptoms in IBS subtypes by Claudia Di Rosa, Karen Van den Houte, Annamaria Altomare, Michele Pier Luca Guarino, Linde Besard, Joris Arts, Philip Caenepeel, Hubert Piessevaux, Alain Vandenberghe, Cristophe Matthys, Jessica R. Biesiekierski, Luc Capiau, Steven Ceulemans, Olivier Gernay, Lydia Jones, Sophie Maes, Christian Peetermans, Willem Raat, Jeroen Stubbe, Rudy Van Boxstael, Olivia Vandeput, Sophie Van Steenbergen, Lukas Van Oudenhove, Tim Vanuytsel, Mike Jones, Jan Tack and Florencia Carbone in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
The DOMINO application was developed by EverywhereIM (Amsterdam, The Netherlands).
We would like to thank and acknowledge the 105 Belgian primary care physicians for their good performance and commitment in this trial as well as the 472 participating patients
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
