Abstract
Background:
The treatment for Crohn’s disease (CD) has increasingly required the use of biological agents. Safe and affordable tests have led to the active implementation of therapeutic drug monitoring (TDM) in clinical practice, which, although not yet widely available across all health services, has been proven effective.
Objective:
To analyze serum infliximab (IFX) and antidrug antibody (ADA) levels in CD patients, compare two tests, as well as construct a prediction of neural network using a combination of clinical, epidemiological, and laboratory variables.
Design:
Cross-sectional observational study.
Method:
A cross-sectional observational study was conducted on 75 CD patients in the maintenance phase of IFX treatment. The participants were allocated into two groups: CD in activity (CDA) and in remission (CDR). Disease activity was defined by endoscopic or radiological criteria. Serum IFX levels were measured by enzyme-linked immunosorbent assay (ELISA) and rapid lateral flow assay; ADA levels were measured by ELISA. A nonparametric test was used for statistical analysis; p value of ⩽0.05 was considered significant. Differences between ELISA and rapid lateral flow results within the measurement range were assessed by the Wilcoxon test, Passing–Bablok regression, and Bland–Altman method. Prediction models were created using four neural network sets. Neural networks and performance receiver operating characteristic curves were created using the Keras package in Python software.
Results:
Most participants exhibited supratherapeutic IFX levels (>7 mg/mL). Both tests showed no difference in IFX levels between the CDA and CDR groups (p > 0.05). The use of immunosuppressive therapy did not affect IFX levels (p > 0.05). Only 14.66% of patients had ADA levels >5 AU/mL, and all ADA-positive participants exhibited subtherapeutic IFX levels in both tests. The median results of both tests showed significant differences and moderate agreement (r = −0.6758, p < 0.001). Of the four neural networks developed, two showed excellent performance, with area under the curve (AUCs) of 82–92% and 100%.
Conclusion:
Most participants exhibited supratherapeutic IFX levels, with no significant serum level difference between the groups. There was moderate agreement between tests. Two neural network sets showed disease activity and the presence of ADA, noninvasively determined in patients using IFX by presenting an AUC of >80%.
Plain language summary
Crohn’s disease (CD) is a chronic condition that affects the gastrointestinal tract, with potential effects anywhere between the mouth and the anus. The primary treatment goal is symptom control and disease remission. The objective of this study was to analyze blood levels of infliximab (IFX), a commonly used medication for CD treatment. We also evaluated the level of antibodies that the body can produce against this medication to justify nonresponse to the drug. IFX levels were compared in 75 patients with CD in activity and in remission and using two different tests. The results showed that most patients had serum IFX above the recommended level (> 7 mg/mL). Neither of these tests showed differences in IFX levels when we evaluated disease activity or when the patients used immunosuppressants. Both tests showed antibodies against IFX in 14.66% of patients, all of whom had IFX levels below the therapeutic level. We compared two tests, ELISA and rapid test, and observed a difference between them, with moderate agreement. Normal serum IFX levels were higher with the rapid test than with the ELISA; however, they presented linear relationship. We also created prediction models using neural networks (artificial intelligence), which demonstrated excellent performance in noninvasively predicting disease activity and the presence of antibodies against IFX, achieving an area under the curve between 82% and 100%.
Introduction
Inflammatory bowel disease (IBD) is a chronic inflammation caused by the interaction of several factors, resulting in an inadequate immune response in a genetically susceptible population. It is represented by two conditions: ulcerative colitis (UC) and Crohn’s disease (CD). CD is a long-term condition that affects the patient’s social and productive life in the short, medium, as well as long term. It is characterized by chronic transmural inflammation of the gastrointestinal tract that can affect any segment from the mouth to the anus, that is, areas affected by the disease are interspersed with normal areas. CD mainly affects the ileal or ileocecal regions.1–5 Its etiology has not been fully understood; however, genetic and environmental factors, changes in gut microbiota, as well as alterations in the intestinal barrier have been implicated in disease onset. 6 CD is also characterized by intestinal dysbiosis, a condition that is increasingly understood, but distinguishing its causes and consequences remains a major challenge. 5
Infliximab (IFX) is a chimeric monoclonal antibody that inhibits the action of tumor necrosis factor-alpha (TNFα), a pro-inflammatory cytokine increased in IBD. It has been widely used in the intravenous treatment of CD, with good results. The induction dose is administered at weeks 0, 2, and 6, followed by maintenance doses every 8 weeks. 7 Anti-TNFα drugs relieve CD symptoms, promote sustained remission, induce mucosal healing, and reduce hospitalization as well as surgery rates. However, one of the main disadvantages of anti-TNFα therapy is primary nonresponse, which occurs in approximately 10–30% of patients with IBD, and secondary nonresponse occurs in 30–40% of patients over the follow-up period.8–10 The presence of antidrug antibodies (ADAs) is associated with a greater risk of clinical nonresponse to IFX and lower serum IFX levels.11,12 Thus, ADA and therapeutic IFX levels should be monitored in these patients.
The ability to monitor serum concentrations during therapy allows for the adjustment or replacement of the administered dose of IFX, resulting in optimized and long-lasting therapy. 13 The increased need for monitoring biological drugs and the possibility of using safe and accessible tests promote the proactive use of therapeutic drug monitoring (TDM) to assess trough levels in clinical practice. TDM improves and intensifies the effectiveness of IFX, helping the patient maintain long-term remission. 14 Some studies correlated higher IFX levels with favorable short- and long-term outcomes in patients with IBD.15–18 Drobne et al. 15 reported that the maintenance of higher IFX levels of >7 μg/mL provided better IBD control, without increased risk of infection. Therefore, IFX TDM has been recommended for optimizing treatment, thereby benefiting patients. 19
Afif et al. identified that serum IFX levels underscore the importance of TDM, as adjustment of the drug dose was found to be more effective than opting for another anti-TNFα agent in patients with subtherapeutic IFX level (86% versus 33% response, respectively, p < 0.016). Serum IFX and ADA levels enhanced treatment by indicating the need to adjust the IFX dose or change the therapy. 20
Compared to CD patients who were not monitored, those under TDM exhibited increased mucosal healing and reduced unfavorable outcomes. 21 Therefore, TDM data should be assessed and incorporated into clinical practice to improve disease management. Fistula closure in perineal CD requires serum IFX levels of ⩾10 μg/mL. 17 Although higher levels (serum IFX levels of 6–10 μg/mL IFX) have been associated with mucosal healing, 18 the optimal therapeutic IFX levels range between 3.0 and 7.0 μg/mL.22–24 Valdés-Delgado et al. 25 reported that patients in remission had serum IFX levels between 4.26 and 8.26 μg/mL, compared to levels of 0.06–1.43 μg/mL in patients not in remission after induction. Thus, serum IFX levels above 4 μg/mL have been recommended for clinical remission in patients with CD. IFX monitoring assays are well correlated and can be used to monitor serum IFX levels. However, few studies have compared this variable to real-life patients. It is noteworthy that although these assays are interchangeable, they still report different serum levels. 26
Therefore, the objective of this study was to compare serum IFX levels and serum ADA levels between patients with CD in activity (CDA) and CD in remission (CDR) from a tertiary center cohort, as well as to establish prediction neural network models by combining noninvasive clinical, epidemiological, and laboratory variables.
Methods
Patient population and ethical considerations
Peripheral blood samples were collected from 75 patients with CD in the maintenance phase of IFX therapy; they were followed up at the IBD Outpatient Clinic, Gastrocenter, Unicamp, Campinas, São Paulo, Brazil, and they were consecutively included in the study after their acceptance and sign of the informed consent. The inclusion criteria were all patients more than 18 years old who had previously undergone induction therapy followed by maintenance therapy (5 mg/kg of IFX). Disease activity was determined by colonoscopy (defined as a Crohn’s disease Endoscopic Index of Severity score of ⩾5 or the presence of deep ulcers in at least one intestinal segment) or by magnetic resonance enterography (defined as the presence of ulcers, mucosa enhancement, or mesenteric changes in at least one intestinal segment). Patients with colonoscopy or magnetic resonance enterography up to 1 month before the date of peripheral blood collection were included. Participants were allocated into the CDA or CDR groups. The participants’ clinical and demographic characteristics and routine laboratory test results, such as C-reactive protein (CRP) and albumin levels, are shown in Table 1. Patients who did not have recent endoscopic or imaging exams, who did not regularly receive IFX injections, or who abandoned the proposed therapeutic regimen were excluded from the study.
Clinical and demographic characteristics of the participants.

Numerical variables are described as median [min–max range], and categorical variables as absolute frequencies.
Montreal classification.
CDEIS was calculated in 21 patients of the CDA group and 32 of the CDR group.
Presence of ulcers, mucosal enhancement, and/or mesenteric changes in at least one intestinal segment.
p < 0.0001 compared with the CDA group.
CDA, Crohn’s disease in activity; CDAI, Crohn’s disease Activity Index; CDEIS, Crohn’s disease Endoscopic Index of Severity; CDR, Crohn’s disease in remission; F, female; IFX, infliximab; IMS, immunosuppressive; M, male; MRI, nuclear magnetic resonance imaging; TNFα, tumor necrosis factor α.
This observational prospective cross-sectional study was conducted in accordance with the tenets of the Declaration of Helsinki and approved by the Unicamp Research Ethics Committee (CAAE no. 53097116.2.0000.5404). All participants signed an informed consent form. Laboratory analyses were carried out at the IBD Research Laboratory of the Unicamp School of Medical Sciences.
This study’s reporting conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 27
Serum IFX and ADA levels
Serum IFX and anti-IFX antibody levels were analyzed by enzyme-linked immunosorbent assays (ELISA) (Promonitor®, Grifols, Progenika Biopharma S.A., Spain). The lower and upper limits of quantification of serum IFX were ⩽0.035 and ⩾14.4 μg/mL, respectively. Antibody levels were considered positive when ⩾5 arbitrary units per milliliter (AU/mL). IFX was analyzed by ELISA in pre-coated 96-well microtiter plates, following the manufacturer’s recommendations.
Furthermore, serum IFX levels were determined by rapid lateral flow assay [BÜHLMANN Quantum Blue® Infliximab (QB-IFX), Bühlmann Laboratories, Basel, Switzerland]. The lower and upper limits of quantification of serum IFX were ⩽0.4 and ⩾20 μg/mL, respectively.
Statistical analysis
All results were reported as median ± standard error of the mean (SEM). Gaussian distribution was assessed by the Kolmogorov–Smirnov test (p > 0.1). Serum IFX levels according to disease activity, use of immunosuppressive (IMS), and presence of antibodies were analyzed by the non-parametric Mann–Whitney test, and categorical variables were compared by the chi-square test (χ2). The significance level was set at p < 0.05.
Differences between ELISA and Quantum Blue results within the measurement range were tested using the Wilcoxon matched-pairs signed-ranks test. We used nonparametric approaches as data did not adhere to normal distribution. The Passing–Bablok regression analysis 28 and the Bland–Altman determined the agreement between the ELISA and rapid tests. The Passing–Bablok regression analysis tests for a linear relationship between X and Y (ELISA and the rapid test), and provides confidence limits for the slope (B) and the intercept (A). These are used to determine whether there is only a chance difference between B and 1 as well as between A and 0. If the slope of the 95% confidence interval (95% CI) contains the value 1 and the intercept’s 95% CI contains the value 0, it indicates no proportional and no constant difference between the methods, respectively. 28 Patients whose IFX values were within the detection range in both tests were included in the analysis. The CUSUM (Cumulative sum) linearity test was used to determine if residuals are randomly distributed above and below the regression line, that is if there is linearity in the relationship between the methods and if the regression is applicable. 29 For this statistical analysis, Stata software version 14, StataCorp, Texas, USA was used.
Neural networks were used to create prediction models and to associate study variables. They are mathematical models that predict output variables using input variables.30–32 The code snippet defines a neural network model using the Keras Sequential API, which is a linear stack of layers. Here is a brief description of the neural network architecture: (a) Input layer: the first layer in the network is a dense layer with 32 units (neurons). It uses the rectified linear unit (ReLU) activation function, which introduces non-linearity into the model. The input shape is specified as (len(fact),), indicating that the input data have a shape determined by the length of the fact variable. (b) Hidden layer: the second layer is another dense layer with 32 units and ReLU activation. This layer further processes the features extracted by the previous layer, allowing the model to learn more complex patterns from the data. (c) Output layer: the final layer is a dense layer with a single unit, which produces the output of the model. It uses the sigmoid activation function, which squashes the output values between 0 and 1, suitable for binary classification tasks where the model predicts probabilities.
Two steps were made before obtaining neural network classification of the data: (a) The train–test split procedure: In the realm of machine learning, including neural networks, randomly selecting a train–test split is a common practice for preventing overfitting. Research has shown that random train–test splits help in achieving a more unbiased estimate of the model’s performance by providing a realistic evaluation of its generalization capability. In particular, in the reference ‘Pattern Recognition and Machine Learning’ by Christopher M. Bishop, 33 the author emphasizes the significance of random sampling for evaluating machine learning models effectively. Random train–test splits are often employed in various studies across different domains, from image classification to natural language processing, to ensure robust model evaluation and prevent overfitting. (b) Normalization technique: to enhance training efficiency and improve model performance. Some of the benefits are as follows: (b1) Robustness to input variations: normalization helps in making neural networks more robust to variations in input data, including differences in scale, distribution, or variance. This allows the model to generalize better across different datasets and input conditions. 34 (b2) Regularization effect: normalization acts as a form of implicit regularization by adding noise to the activations, which can help prevent overfitting and improve the generalization performance of the model. 35 Therefore, prediction models were created using four neural network sets for the following outcomes (output variables): disease activity, presence of ADA, serum IFX levels by ELISA, and serum IFX levels by lateral flow assay. Clinical, epidemiological, and laboratory data were used as input variables for the neural networks. Receiver operating characteristic (ROC) curves were used to determine the accuracy of each prediction neural network model. These neural networks were created using the Keras package in the Python software, a programming language used for software development and machine learning. The software developed with these models was registered at the Brazilian Institute of Industrial Property through the Unicamp Innovation Agency, number 512023003692-7, on 30 November 2023. Neural network sets may be used and validated by inserting input and output variables into this specific software.
Results
Serum IFX and ADA levels in patients with CDA and CDR
We used two assays (ELISA and lateral flow) to assess serum IFX levels in the CDA and CDR groups. The assays showed no differences in IFX levels between the two groups (p > 0.05) [Figure 1(a) and (b) and Table 2]. Combined therapy was prescribed to 50 (66.6%) participants, but neither of the tests showed a significant difference in IFX levels according to IMS use (p > 0.05) [Figure 1(c) and (d)].
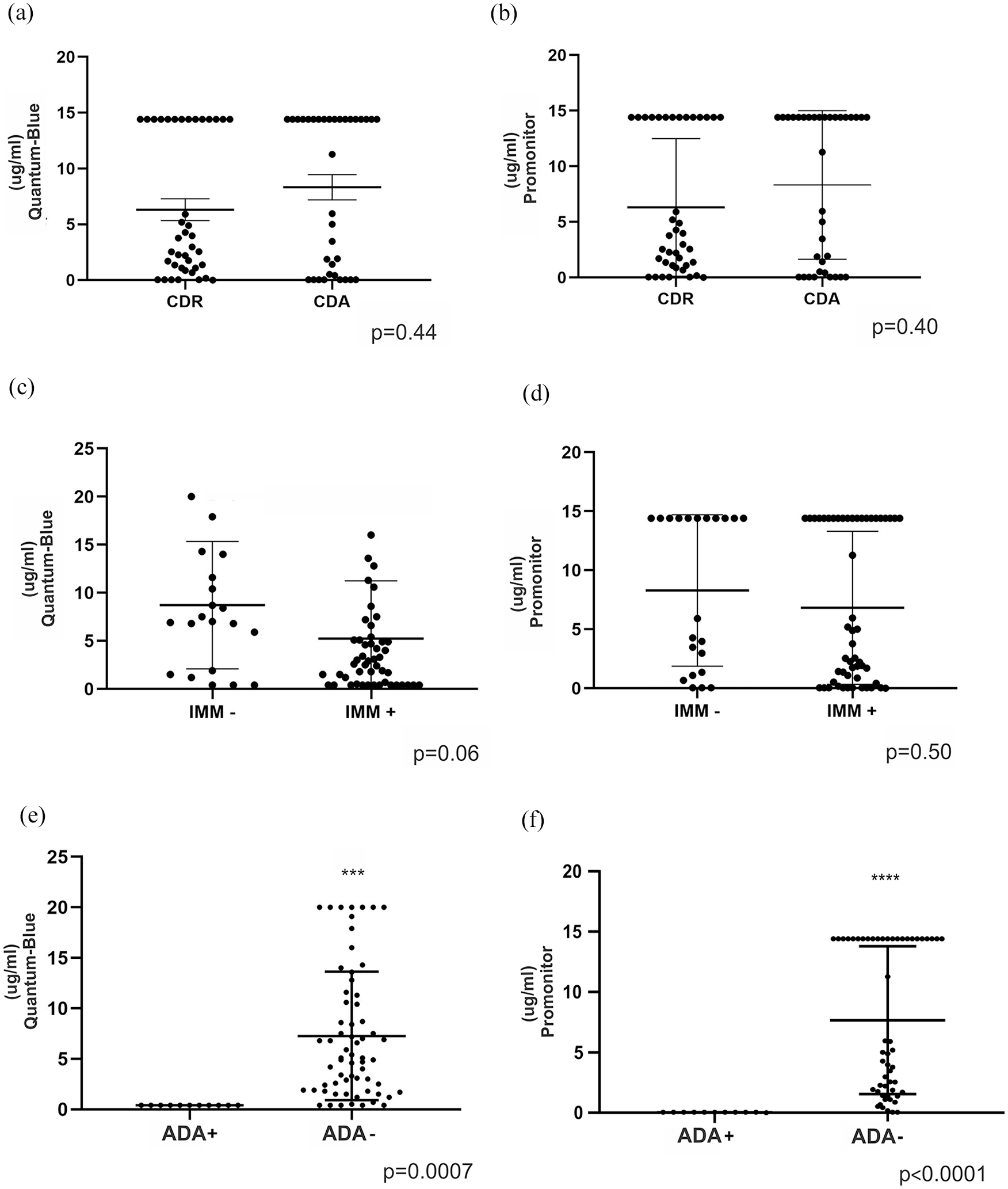
IFX trough level drug monitoring in active and remission Crohn’s disease. IFX serum levels (μg/mL) comparing patients with active and remission Crohn’s disease as measured by Quantum Blue® rapid test (a) and Promonitor® ELISA assay (b). IFX serum levels comparing the use versus no-use of concomitant immunosuppressant as measured by Quantum Blue® rapid test (c) and Promonitor® ELISA assay (d). IFX serum levels comparing the detection versus non-detection of ADA as measured by Quantum Blue® rapid test (e) and Promonitor® ELISA assay (f).
Number of participants by Crohn’s disease activity and serum IFX level by ELISA (Promonitor®) and lateral flow assay (Quantum Blue®).
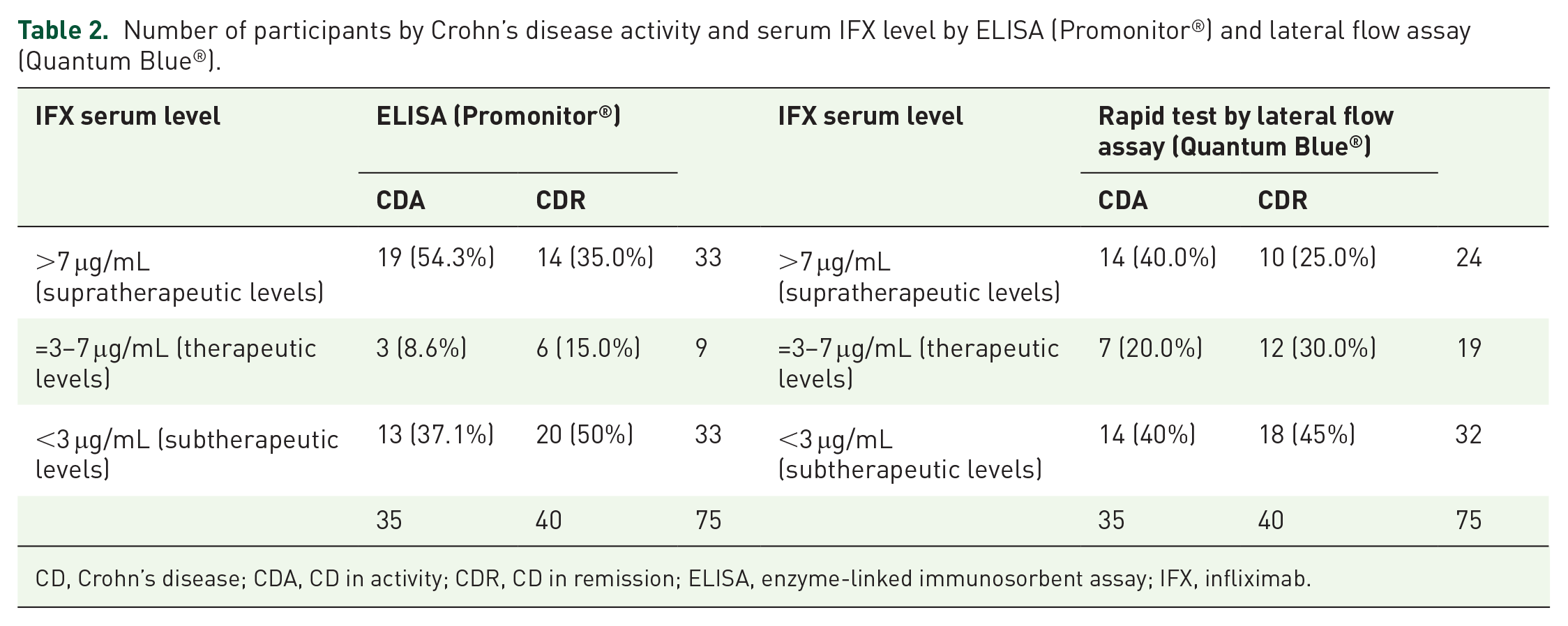
CD, Crohn’s disease; CDA, CD in activity; CDR, CD in remission; ELISA, enzyme-linked immunosorbent assay; IFX, infliximab.
ADA levels were measured for all participants included in the study, and positive values were observed in only 11 (14.6%; CDA, n = 6; CDR, n = 5) participants. Concomitant therapy was prescribed for five of the six participants of the CDA group and three of the five participants of the CDR group. Thus, of all ADA-positive participants, only three (27%) were not prescribed combination therapy with IMS. All ADA-positive participants presented undetectable IFX levels both by ELISA [Figure 1(e)] and lateral flow assay [Figure 1(f)], regardless of disease activity or remission. The three patients with near-positive ADA levels (2.7, 2.8, and 3.0 UA/mL) also presented undetectable IFX levels in both tests. Thus, there was a significant association between serum IFX levels and the presence of ADA (p < 0.0001). No participants with therapeutic IFX levels exhibited positive ADA levels.
Comparison between ELISA and rapid lateral flow assays in assessing serum IFX levels in patients with CD
We observed a significant difference (p < 0.001) in median results between the two tests [2.27 for Promonitor (Interquartile range (IQR): 1.36–4.27) versus 4.0 for Quantum Blue (IQR: 1.7–6.9)], as presented in Figure 2(a). The CUSUM test 36 showed a linear relationship between the two measurements; therefore, the Passing–Bablok method was applicable (p > 0.20). In our results, we observed concentration values below the lower limit of quantification (LLOQ) and the upper limit of quantification (ULOQ) for both tests. For the ELISA test (Promonitor®), 32 patients were found above the ULOQ and 13 patients below the LLOQ. In the Rapid test by lateral flow assay (Quantum Blue®), 7 patients were found above the ULOQ and 16 patients below the LLOQ. These patients who presented LLOQ or ULOQ in at least one test were excluded from the analysis. Therefore, IFX plasma concentrations, within the measuring range, evaluated in 27 CD patients for each method, showed higher values for the Quantum Blue® rapid test than the Promonitor® ELISA assay. The two IFX level tests had a moderate agreement (r = −0.6758; p < 0.001) [Figure 2(b)]. The Passing–Bablok regression intercept did not differ significantly from 0 (−0.018; 95% CI: −0.489 to 0.420), but the slope was lower than 1 (0.589; 95% CI: 0.454–0.766), that is, there is a proportional difference between the two tests [Figure 2(b)]. In addition, we performed a Bland–Altman test to complete the comparison and establish the level of agreement between the two assays. This analysis revealed a moderate agreement between the two assays [Figure 2(c)], and just one value was outside the CI.

Comparison between ELISA (Promonitor®) and lateral flow (Quantum Blue®) assays. (a) Box plot comparing IFX concentrations (mg/L), the horizontal line in the middle represents the median (*p = 0.0001). (b) Passing–Bablok linear regression of IFX levels (r = −0.6758; p < 0.001). (c) Bland–Altman graph: the difference between the two measurements (mg/L) is plotted on the y-axis, and the average of the two measurements (mg/L) is on the x-axis. Dashed lines are the 95% limits of agreement of the bias. Bias (in solid line) was calculated according to the Bland–Altman method (−42.1%).
Neural network prediction models
The first two neural network sets were developed with two output variables: serum IFX levels measured by the Promonitor® and by the Quantum Blue® tests. However, the ROC curves created to determine the accuracy of each neural network prediction model found no combination of variables that could result in a neural network with good performance [area under the curve (AUC) < 70%]. Thus, we developed two new neural network sets with ROC curves designed to determine the prediction accuracy of each neural network and to find the best model. The output variables used for these new models were ‘presence or absence of disease activity’, determined by objective endoscopic or radiological factors; and ‘presence or absence of ADA’. Input variables included a combination of clinical, epidemiological, and laboratory variables for each set of neural networks: 12 variables to determine disease activity, distributed in 4 neural networks (Figure 3), and 14 variables to determine ADA presence, distributed in 8 neural networks (Figure 4). The two sets of new neural networks showed excellent performance, with AUCs of 82–92% and 100%. Thus, this noninvasive method determined two output variables using clinical, epidemiological, and laboratory data collected in clinical practice.

Prediction models via neural network using a combination of clinical, laboratory, and epidemiological variables, with Crohn’s disease activity as the output variable. (a) Dimensionless percentage of participation of the variables for the prediction model, considering the combination of the following variables: drug monitoring classification according to IFX serum levels (μg/mL) measured by Quantum Blue rapid test (QB-IFX) [therapeutic (3–7 μg/mL), supra-therapeutic (>7 μg/mL), infra-therapeutic (<3 μg/mL)]; ADA; ESR; disease duration; gender. (b) Drug monitoring classification according to IFX serum levels (μg/mL) measured by Promonitor assay [therapeutic (3–7 μg/mL), supra-therapeutic (>7 μg/mL), infra-therapeutic (<3 μg/mL)]; CRP; disease duration; body weight; CDAI. (b and c) Drug monitoring classification according to IFX serum levels (μg/mL) measured by Promonitor assay [therapeutic (3–7 μg/mL), supra-therapeutic (>7 μg/mL), infra-therapeutic (<3 μg/mL)]; CRP; disease duration; body weight; CDAI. (d) Drug monitoring classification according to IFX serum levels (μg/mL) measured by Promonitor assay [therapeutic (3–7 μg/mL), supra-therapeutic (>7 μg/mL), infra-therapeutic (<3 μg/mL)]; IFX treatment duration; smoking; IFX dosage (mg/kg); age (months). (e) Performance (accuracy) of the four NN to predict disease activity.

Prediction models via neural network using a combination of clinical, laboratory, and epidemiological variables, with ADA detection as the output variable. (a) Dimensionless percentage of participation of the variables for the prediction model, considering the combination of the following variables: ESR; CRP; body weight; CDAI. (b) Platelets; CRP; IFX treatment duration (months); disease duration; body weight. (c) ESR; previous appendicectomy; gender. (d) CRP; smoking; age (months); CDAI; gender. (e) Albumin; platelets; previous appendicectomy; age (months); gender. (f) CRP; hematocrit; IFX dosing intervals (2, 4, and 6 weeks); age (months); gender. (g) ESR; CRP; IFX dosage (mg/kg); gender. (h) Platelets; ESR; CRP; gender. (i) Performance (accuracy) of the eight NN to predict the presence of ADA.
Neural network results show that it is possible to use complex knowledge structures to efficiently extract data on endoscopic/radiological activity and ADA presence from patients with CD under IFX treatment. The criterion for selecting neural network output variables (outcomes) was the direct clinical applicability of the model in patient management. These data may improve clinical decision-making for each patient by prospecting a possible combination of neural networks for CD patients under IFX for the assessment of disease activity and the presence of ADA through demographic, clinical, and laboratory data (Figure 5). Chronic diseases require noninvasive methods to detect disease activity. We found variables to determine ADA levels as an outcome using noninvasive prediction based on routinely available clinical–biological parameters, but no good models to determine serum IFX levels, which still shows the practical importance of measuring IFX serum levels. However, drug immunogenicity may be established by a neural network, with no need for testing in patient follow-up.

Illustrative prospection of the possible use of neural network combinations for patients with CD using IFX in clinical practice. (a) Assessment of disease activity. (b) Assessment of the presence of ADA. Created with BioRender.com.
Considering the input combinations in which each of these variables affects the output variables, the contribution of each variable included in the prediction model is dimensionless. For the neural networks applied to the problem, the train–test split procedure was used to ensure that (a) Prevention of overfitting: training a neural network with all available data may lead to overfitting, where the model learns to memorize the training data rather than generalize. By holding out a portion of the data for testing, we can detect overfitting and adjust the model accordingly. (b) Assessment of generalization ability: the test set provides a means to assess how well the trained neural network generalizes to new, unseen data.
Moreover, an ROC curve, as the ones presented in Figures 3 and 4, is a graphical representation that illustrates the diagnostic ability of a binary classifier system as its discrimination threshold is varied. It plots the true-positive rate (TPR) against the false-positive rate (FPR) at various threshold settings. A typical ROC curve plots TPR against FPR for different threshold values. The curve visualizes the trade-off between sensitivity and specificity.
A diagonal line from the bottom left to the top right represents random guessing, while a curve closer to the top left corner indicates better performance, where the classifier achieves high sensitivity while keeping the FPR low. The area under the ROC curve (AUC-ROC) is a metric commonly used to quantify the performance of a binary classifier. A higher AUC-ROC value indicates better discrimination ability, with a perfect classifier having an AUC-ROC value of 1. The ROC curve presented in Figures 3 and 4 attests to the adequacy of the neural network configuration and the precision of its model. Furthermore, the same database and procedures served to generate both models with AUC lower than 1 (Figure 3) and equal to 1 (Figure 4).
Discussion
Herein, we compared serum IFX levels and serum ADA levels between patients with CDA and CDR as well as established prediction neural network models by combining noninvasive clinical, epidemiological, and laboratory variables. Our key findings were as follows: (i) IFX levels showed no differences between CDA and CDR groups, and with the use of IMS, which was prescribed to 50 patients (66.6%). (ii) Serum IFX levels measured by Quantum Blue® presented significantly higher values than the Promonitor® assay (p < 0.001). There was also a linear relationship between the two measurements, with moderate agreement. (iii) Four neural network sets were established to assess parameters available in clinical practice for noninvasive prediction of CD activity. Two of the four sets used in this study had an AUC of >80%, demonstrating high specificity and sensitivity performance.
Biological agents are a relevant tool in the treatment of IBD; however, nonresponse is still a paradigm that needs to be overcome. 36 IBD is a chronic disease; therefore, the pharmacokinetic potential of the few medications available to treat CD in clinical practice should be fully used. Primary and secondary nonresponse to treatment is a main challenge of anti-TNFα therapy, and therefore, anti-TNFα TDM has improved patient management targeted at sustained clinical, endoscopic, as well as laboratory remission. 37 TDM has been recommended by several international guidelines and is increasingly becoming the standard of care.10,38–41 However, the prolonged turnaround time for laboratories to deliver serum anti-TNF results can be a challenge. Thus, commercial quantitative rapid tests have been used to optimize pre-infusion treatment. 42
This study considered the therapeutic level recommended in the literature (3–7 µg/mL)14,16,18,22; thus, we had many patients with IFX levels exceeding the therapeutic limit. Some TDM tests on IFX levels reported outcomes ranging from clinical to biological remission with different cutoff values. A prospective study suggested appropriate therapeutic levels ranging between 3 and 7 µg/mL; however, some authors reported that healing and fistula closure require a minimum level of 10 µg/mL.16,18,22
A study by Lim et al. 43 reported that the Buhlmann lateral flow assay had excellent sensitivity and specificity for IFX levels <2 as established by ELISA, thereby making it a viable option for real-time dosing decisions.
According to Vande Casteele et al., 24 comparing trials is significantly relevant for understanding and interpreting clinical study data. Also, the comparison of four assays showed linearity, with a correlation coefficient ranging between 0.947 and 0.978. It should be emphasized that because of the variability of assays for monitoring therapeutic concentrations of IFX, it is ideal to consistently use the same assay. 44 A study comparing the Promonitor® and Quantum Blue® tests for quantifying serum IFX levels in patients with on-maintenance therapy reported a strong correlation between the methods, especially at lower IFX levels. Samples stratified by therapeutic range (<3, 3–7, and >7 μg/mL) demonstrated near-perfect agreement between the tests (κ = 0.81). 45 An advantage of rapid tests is the immediate availability of results post-infusion, eliminating the need to collect samples from different patients for testing in 96-well microtiter plates, which occurs requirement with ELISA. This implies that the dosage can be adjusted or the drug can be changed before the subsequent infusion. 46
In this study, the median values showed a significant difference (p < 0.001) between the two assays. Serum IFX levels within the measurement range were assessed in 27 patients with CD using each method, with the Quantum Blue® presenting higher values than the Promonitor® assay. There was also a linear relationship between the two measurements, with moderate agreement. The literature reports both quantitative and qualitative differences between assays in the IFX level measurement. Although the tests exhibit a significant statistical association, they may not be considered interchangeable, especially at high IFX levels. Thus, patients should be ideally monitored by the same TDM assay. 47 Furthermore, ELISA does not provide real-time results, and comparisons by therapeutic range with other rapid point-of-care tests demonstrated good agreement between assays, which represents a viable option for TDM. Rapid assays have the advantage of quantifying single serum samples, contrary to ELISA, which requires several samples to be tested together. As for response time testing, ELISA presents a response of 4–8 h, while rapid tests provide results in 15–20 min. Rapid tests also have disadvantages, such as no quality control and greater difficulty in tracking and monitoring measurement errors. 48
Although there are several TDM options, Bertin et al. demonstrated that the results of three tests were not interchangeable due to significant result variations (up or down), which can lead to divergent therapeutic decisions in some cases. IFX trough levels and different concentration range categories based on the therapeutic window showed that subsequent therapeutic decisions may differ according to the assay used. 49 Our results showed that category changes were frequent and different between the compared tests, with some variations not being explained by the variability found between assays. This variability was also reported in other studies, suggesting that the same assay should be used throughout the treatment, with no changes during follow-up.50–52
TDM involves measuring serum drug and ADA levels, proving to be an extremely useful tool in providing early and scheduled disease assessment to maintain IBD remission. 53 We found only 11 (14.66%) ADA-positive and three near-positive patients, all of them exhibiting subtherapeutic IFX levels. A similar study reported few ADA-positive patients. 49 A possible reason for the occurrence of ADA is the formation of immune complexes. ADA can be found both in free form or bound to immune complexes. 54
Proactive TDM helps improve IBD outcomes in patients initiating anti-TNF therapy; however, some data are conflicting. Patients at higher risk for increased drug clearance and immunogenicity are more likely to benefit from proactive TDM. 55 Although therapeutic IFX monitoring can prevent over- or underdosing, proactive TDM can prevent disease worsening. Also, proactive TDM can be scheduled regardless of symptom onset. 24 A study assessed 102 patients with IBD on IFX maintenance therapy and compared long-term results between patients undergoing proactive monitoring. Over 90% of patients in the proactive group presented an IFX trough level of >5 μg/mL, with a higher rate of treatment persistence and fewer IBD-related hospitalizations compared to the reactive test group. 56 Compared to reactive TDM, the proactive approach presented fewer disease activity events and no adverse reactions. 57 IFX levels above 5 mg/mL 18 were associated with mucosal healing, levels below 3 mg/mL after induction were associated with disease relapse, and levels above 7 µg/mL were associated with sustained remission. 58 The lack of patient assessment during IFX induction may be considered a limitation of our study.
Patients with trough, undetectable, or subtherapeutic IFX levels should be assessed according to ADA levels. In this group, therapy optimization (by increasing the dosage, decreasing the interval between infusions, or adding IMS) should be considered when the assay shows no anti-IFX antibodies. Also, ADA levels can vary, and they can even disappear after IFX therapy intensification. Low serum IFX and positive ADA levels have been associated with nonresponse,23,59 and a change in drug class should be considered in cases of high anti-IFX antibody levels. 53
We also emphasize the possibility of combined treatment with IMS after anti-TNF therapy initiation to decrease immunogenicity. 60 ADA may be a transient finding, not always leading to a worse clinical outcome. However, high serum ADA levels result in nonresponse, and IMS therapy can help reduce immunogenicity to IFX. 61 The use of combined IFX and IMS therapy has progressively increased over the last 20 years. 62 In the present study, IFX levels showed no differences with the use of IMS, which was prescribed to 50 patients (66.6%). This may be explained by the large percentage of patients with supratherapeutic IFX levels in our cohort. A study by Yao et al. 62 reported that combined IMS and IFX (12.0%) therapy represented almost half of the initial treatment strategies (within the first month after diagnosis) for patients with CD. Combination therapy is more effective than monotherapy with azathioprine or IFX in achieving remission rates, leading to clinical and endoscopic remission in most early-stage patients with CD. 63
Another relevant aspect is the detection of endoscopic or radiological activity in patients with CD during follow-up to diagnose nonresponse to the biological agent. Noninvasive solutions have been increasingly searched to monitor CD; fecal calprotectin is used as a fecal biomarker to assess disease activity. It shows a good correlation with endoscopic findings of CD activity. 64 The results of a neural network prediction model can reproduce the predictor-outcome association after adjusting the variables included in the model. Takenaka et al. identified patients with UC in endoscopic remission using endoscopic images through a neural network and reported an accuracy of 90.1% as well as a histological remission accuracy of 92.9%. Thus, those authors identified patients in remission without biopsy collection and analysis, 65 showing that artificial intelligence can be used to develop potential predictive models in IBD, although in a very incipient and initial phase.
Artificial neural networks are promising, especially particularly those utilizing artificial intelligence technology and deep learning, as well as are promising tools that are accelerating scientific discoveries. Future patient cohorts must validate the classifiers determined in this study as biomarkers capable of replacing endoscopies and biopsies in the future, thus reducing the risks of invasive procedures. 66 Two of the four neural network sets used in this study had an AUC of >80%, demonstrating high specificity and sensitivity performance. Noninvasive clinical, epidemiological, and laboratory variables were used to determine disease activity/remission besides immunogenicity to IFX.
We used an artificial intelligence system based on neural network models to assess parameters available in clinical practice for noninvasive prediction of CD activity. This set of neural network models had an accuracy greater than 82% in determining CD activity or remission. Another study used a neural network model to predict endoscopic remission/activity, reporting 79% and 82% accuracy for the UC endoscopic index of severity. 66 That system distinguished histological activity/remission, with a sensitivity of 89% and a specificity of 85% by the PICaSSO Histological remission Index, 94% and 76% by the Robarts histopathological index, as well as 89% and 79% by the Nancy Histological Index. In addition to developing and validating the artificial intelligence model, the distinction between remission and activity can predict disease outbreaks. 67 Studies on the use of artificial intelligence in IBD have been conducted.68–70 Although humans will not be replaced in clinical practice, some uses of artificial intelligence are extremely promising in improving the care of patients with IBD.
Thus, two of our neural network sets had excellent performance, with AUCs of 82–92% and 100%, corroborating the performance reported in other studies. This was also seen for histological evaluation results with an artificial intelligence system, which highly agreed with manual testing results, demonstrating high sensitivity (89%) and specificity (85%).67,71
Furthermore, a deep learning diagnostic system trained on a large number of colonoscopy images to distinguish UC and CD was developed and validated in a recent study. The authors developed a deep learning model based on a deep neural network, whose identification accuracy was found to be higher than that of experienced endoscopists (deep neural model versus intern endoscopist, 99.1% versus 78.0%; versus competent endoscopist, 99.1% versus 92.2%, p < 0.001) and by lesion (deep neural model versus intern endoscopist, 90.4% versus 59.7%; versus competent endoscopist 90.4% versus 69.9%, p < 0.001). In addition to the reduced average reading time and the creation of a model to aid in the clinical diagnosis of IBD, this artificial intelligence has provided a device for medical education to improve the efficiency of diagnosis and treatment. 72
Despite artificial neural networks’ enormous potential, we are still far from practical daily implementation, given the need for learning strategies and input and output data sets, in addition to the need for external validations of these prognostic models. In the literature, computational models may already accurately determine endoscopic remission and predict activity or remission based on images alone without the need for biopsies, but this has not yet been implemented in clinical practice. 65
Concerning the limitations of our study, the measurement of ADA levels in a single test, which did not allow the assessment of inter-assay variability throughout a follow-up study, can be considered a limitation. Furthermore, the small sample size and heterogeneity of CD patients with different durations of IFX maintenance therapy may be a limiting factor. However, even with the small number of patients included, our cohort enabled reliable and strong results.
Another consideration is that we have not found robust models to determine serum IFX levels. Therefore, our study highlights the importance of continuing to measure IFX serum levels in clinical practice, considering the identified combination of variables that may only determine antibody levels. Our model uses noninvasive clinical, epidemiological, and laboratory variables that may predict antibody levels that show immunogenicity to the drug. The presence of ADA reduces clinical response in patients with CD. Neural network estimates should not replace the expert’s opinion but rather support and corroborate their decision, improving the decision-making process. Although neural network hypotheses may be feasible, applicable, and accurate, they require additional confirmation through the analysis of patients’ characteristics, symptoms, and clinical history.
A larger study cohort is, therefore, essential to validate our findings and strengthen the robustness as well as reliability of the results presented. Furthermore, a large sample size reduces the possibility of bias and increases the generalizability of findings, significantly contributing to the knowledge in the area.
Supplemental Material
sj-pdf-1-tag-10.1177_17562848241251949 – Supplemental material for Infliximab monitoring in Crohn’s disease: a neural network approach for evaluating disease activity and immunogenicity
Supplemental material, sj-pdf-1-tag-10.1177_17562848241251949 for Infliximab monitoring in Crohn’s disease: a neural network approach for evaluating disease activity and immunogenicity by Luis Eduardo Miani Gomes, Livia Moreira Genaro, Marina Moreira de Castro, Renato Lazarin Ricci, Livia Bitencourt Pascoal, Filipe Botto Crispim Silva, Pedro Henrique Leite Bonfitto, Michel Gardere Camargo, Ligiana Pires Corona, Maria de Lourdes Setsuko Ayrizono, Anibal Tavares de Azevedo and Raquel Franco Leal in Therapeutic Advances in Gastroenterology
