Abstract
Background:
Endoscopic ultrasound-guided biliary drainage (EUS-BD), classified as choledochoduodenostomy (CDS) and hepaticogastrostomy (HGS), is a feasible and effective alternative for distal malignant biliary obstruction (MBO) in failed endoscopic retrograde cholangiopancreatography. However, the preferred technique for better outcomes has not yet been evaluated.
Objectives:
We compared the long-term outcomes between the techniques.
Design:
Retrospective comparative study.
Methods:
We reviewed consecutive patients who underwent EUS-CDS or EUS-HGS with transmural stent placement for distal MBO between 2009 and 2022. The primary outcome was the stent patency. The secondary outcomes were technical and clinical success, adverse events (AEs) of each technique, and independent risk factors for stent dysfunction.
Results:
In all, 115 patients were divided into EUS-CDS (n = 56) and EUS-HGS (n = 59) groups. Among them, technical success was achieved in 98.2% of EUS-CDS and 96.6% of EUS-HGS groups. Furthermore, clinical success was 96.4% in EUS-CDS and 88.1% in EUS-HGS groups, without significant difference (p = 0.200). The mean duration of stent patency for EUS-CDS was 770.3 days while that for EUS-HGS was 164.9 days (p = 0.010). In addition, the only independent risk factor for stent dysfunction was systematic treatment after EUS-BD [hazard ratio and 95% confidence interval 0.238 (0.066–0.863), p = 0.029]. The incidence of stent dysfunction of EUS-HGS was higher than EUS-CDS (35.1% versus 18.2%, 0.071), despite no significant differences even in late AEs.
Conclusion:
In distal MBO, EUS-CDS may be better than EUS-HGS with longer stent patency and fewer AEs. Furthermore, systematic treatment after EUS-BD is recommended for the improvement of stent patency.
Introduction
Since the first announcement of endoscopic ultrasound-guided biliary drainage (EUS-BD) 1 in 2001, its indication has been expanding from rescue modality in cases of failed endoscopic retrograde cholangiopancreatography (ERCP) to primary drainage for distal malignant biliary obstruction (MBO).2–5 Among various techniques, EUS-BD with transmural stent placement can be classified into choledochoduodenostomy (CDS) through the duodenal route and hepaticogastrostomy (HGS) through the gastric route. In EUS-CDS, the extrahepatic duct (EHD) and duodenum are anastomosed, whereas a fistula between the intrahepatic bile duct (IHD) and the stomach is created in EUS-HGS. Although a recent meta-analysis 6 reported similar efficacy and safety with high technical and clinical success in both the approaches and these two approaches may theoretically have different indications, most experts consider EUS-CDS to be easier and safer and thus recommend it as the first approach for EUS-BD especially in distal MBO.
The greatest advantage of EUS-CDS is the anatomical approximation between EHD and duodenum and the larger diameter of the target duct for easy puncture, which can result in higher technical success.1,7 In a recent meta-analysis, Amato et al. 8 demonstrated that the pooled rates of technical success were 94.8% for lumen-apposing metal stents and 92.7% for self-expandable metal stents (SEMS), respectively. 8 Contrarily, EUS-HGS can boost its advantages in cases, which underwent failed ERCP because of surgically altered anatomy or gastric outlet obstruction (GOO).9,10 However, despite the theoretical benefits, echoendoscopists can encounter difficulty in the puncture of non-dilated IHD. In addition, the long distance between the stomach and the IHD through hepatic parenchyma can lead to difficulty in tract dilation and even procedure-related adverse events (AEs). 11 Another big issue is the risk of stent migration into the peritoneal cavity due to movement of the liver during respiration and frequent peristalsis of the stomach. 12
To date, despite the unique theoretical advantages of both approaches with similar technical success, there are only a few reports comparing the clinical efficacy and safety of EUS-CDS and EUS-HGS in distal MBO. Given these circumstances, we aimed to compare the clinical outcomes and patency between the two approaches and determine which technique is associated with better outcomes.
Methods
Patients
This was a multicenter retrospective comparative study conducted at seven medical centers as members of the Research Group for Endoscopic Ultrasound in The Korean Society of Gastrointestinal Endoscopy (KSGE). Inclusion criteria were consecutive inpatients who underwent EUS-BD with transmural stent placement for distal MBO. Patients who underwent primary EUS-BD without any attempt at ERCP were also included, as well as those who underwent rescue intervention after failed ERCP. However, patients who underwent EUS-HGS with antegrade stent placement using the rendezvous approach combined with transmural stenting were excluded. Demographic, clinical, and endoscopic data were extracted from the computerized clinical information system for the previous 13 years (from October 2009 through August 2022) according to the two groups, EUS-CDS and EUS-HGS. Retrospective data collection for all included patients was performed from the initial procedure until death or the final follow-up. As a retrospective analysis, the study was conducted in accordance with the protocol of the Declaration of Helsinki and was approved by the Institutional Review Board of the Ethics Committee of the representative hospital (2022-08-012-001 for Hallym University Dongtan Sacred Heart Hospital), and the need for informed consent was waived. In addition, the reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 13
Endoscopic procedures for EUS-CDS and EUS-HGS
All procedures were performed by eight expert endosonographers according to standard protocols. As the diagnosis of distal MBO was established, treatment with appropriate antibiotics, bowel rest, and intravenous fluids was administered to all patients. 14 EUS-BD was also performed under sedation using intravenous administration of midazolam, propofol, and meperidine. 15
EUS-CDS
After identification of EHD in the duodenal bulb using a linear echoendoscope, puncture was performed using a 19-gauge standard fine-needle aspiration needle unmounted with a stylet to avoid puncturing the cystic duct. Thereafter, a contrast medium was injected to visualize the EHD under fluoroscopy. Then, a 0.025- or 0.035-inch guidewire was inserted through the needle into the bile duct. After the withdrawal of the needle, a standard catheter was inserted over the guidewire for track dilation. Then, a 4-mm balloon catheter was inserted over the guidewire, and track dilation was performed. The balloon catheter could be used initially without prior attempts using a standard catheter at the discretion of the endoscopist. If the catheter could not be inserted easily, a cystotome or needle-type knife was applied. 16 Finally, a metal or plastic stent was inserted into the EHD from the duodenum.
EUS-HGS
For EUS-HGS, the left IHD was punctured from the stomach, whereas the right IHD was punctured from the duodenum. After the injection of the contrast medium, the guidewire was inserted into the IHD with a maximal effort to pass across the biliary stricture to stabilize the guidewire. After withdrawal of the needle, tract dilation was performed using a standard catheter first and a sequential balloon catheter, or eventually a needle-type knife or cystotome if resistance to advancement of the standard catheter or balloon catheter was apparent. When sufficient tract dilation was achieved, a plastic or metal stent was inserted and deployed between the IHD and stomach. A conventional fully covered and modified partially covered SEMS with a bidirectional anti-migrating system was used in our study to prevent leakage alongside the stent, as well as to enable easy removal and prevention of tissue growth. The length of the stent was determined by approximating the distance between the IHD and bowel with an extra length (approximately 20 mm) on both sides.
Definitions of outcomes
The main outcomes evaluated in the study included (1) stent patency according to EUS-BD technique; (2) technical and clinical success rates of each technique for EUS-BD; (3) independent risk factors for stent dysfunction; (4) early AEs within 2 weeks after EUS-BD including hemorrhage, perforation, pneumoperitoneum, or bile peritonitis; and (5) late AEs 2 weeks after EUS-BD, including bile peritonitis, abscess, or infected biloma. 17
Technical success was defined as the successful placement of a transmural stent into the BD, as determined by smooth bile drainage endoscopically accompanied by appropriate positioning of the stent radiologically. Clinical success was defined as a decrease in total bilirubin level either to normal or by ⩾50% within 2 weeks of EUS-BD. 16 Following the procedure, patients were monitored until follow-up loss or death, with evaluations focused on AEs and the patency of the stent. Assessments were conducted on the next day, as well as at 7, 14, 30, 60, and 90 days post-procedure, during scheduled consultations for clinical assessment and laboratory analysis. Early and late AEs were those occurring within 2 weeks of stent placement and later, respectively. Bile peritonitis after EUS-BD refers to the leakage of bile juice into the peritoneal cavity, resulting in inflammation and infection. This condition may manifest as signs of peritoneal irritation, accompanied by the identification of newly developed ascites or localized fluid collection through imaging modalities such as CT scans. 18 Stent patency was defined as the interval from the index procedure of EUS-BD to the first stent dysfunction with recurrent obstructive symptoms and signs accompanied by biliary obstruction and dilatation of the bile duct on imaging modalities. 5 Patients who died without stent dysfunction were considered censored. The survival period was calculated as the interval between EUS-BD stent placement and death.
Statistical analysis
Data are presented as frequency with proportion for categorical variables or means with standard deviation (SD) and medians with interquartile range (IQR) for continuous variables. For the comparison of categorical data, the χ2 test was used as indicated, while the Student’s t-test was used to compare continuous variables. Stent patency and patient survival were calculated using the Cox proportional hazards model, and the log-rank test was used to compare stent patency and survival curves, accounting for censored data. All reported p values were two-sided, and p values <0.05 were considered statistically significant. All statistical analyses were conducted using R statistical software (version 4.0.2; R Foundation for Statistical Computing, Vienna, Austria).
Results
Study population and baseline characteristics
During the study period, 187 patients who underwent EUS-BD (EUS-CDS or EUS-HGS) with transmural stent placement for MBO were initially assessed for eligibility. Among these, 16 patients who underwent only antegrade stent placement using the rendezvous approach through EUS-HGS were excluded. Furthermore, 55 patients who underwent EUS-HGS for hilar MBO were also excluded. Finally, one patient who underwent EUS-HGS with antegrade stent placement using the rendezvous approach combined with transmural stenting was excluded. As a result, the remaining 115 patients with EUS-BD with transmural stent placement were included in the analyses and divided into the EUS-CDS (n = 56) and EUS-HGS (n = 59) groups according to the EUS-BD technique (Figure 1).
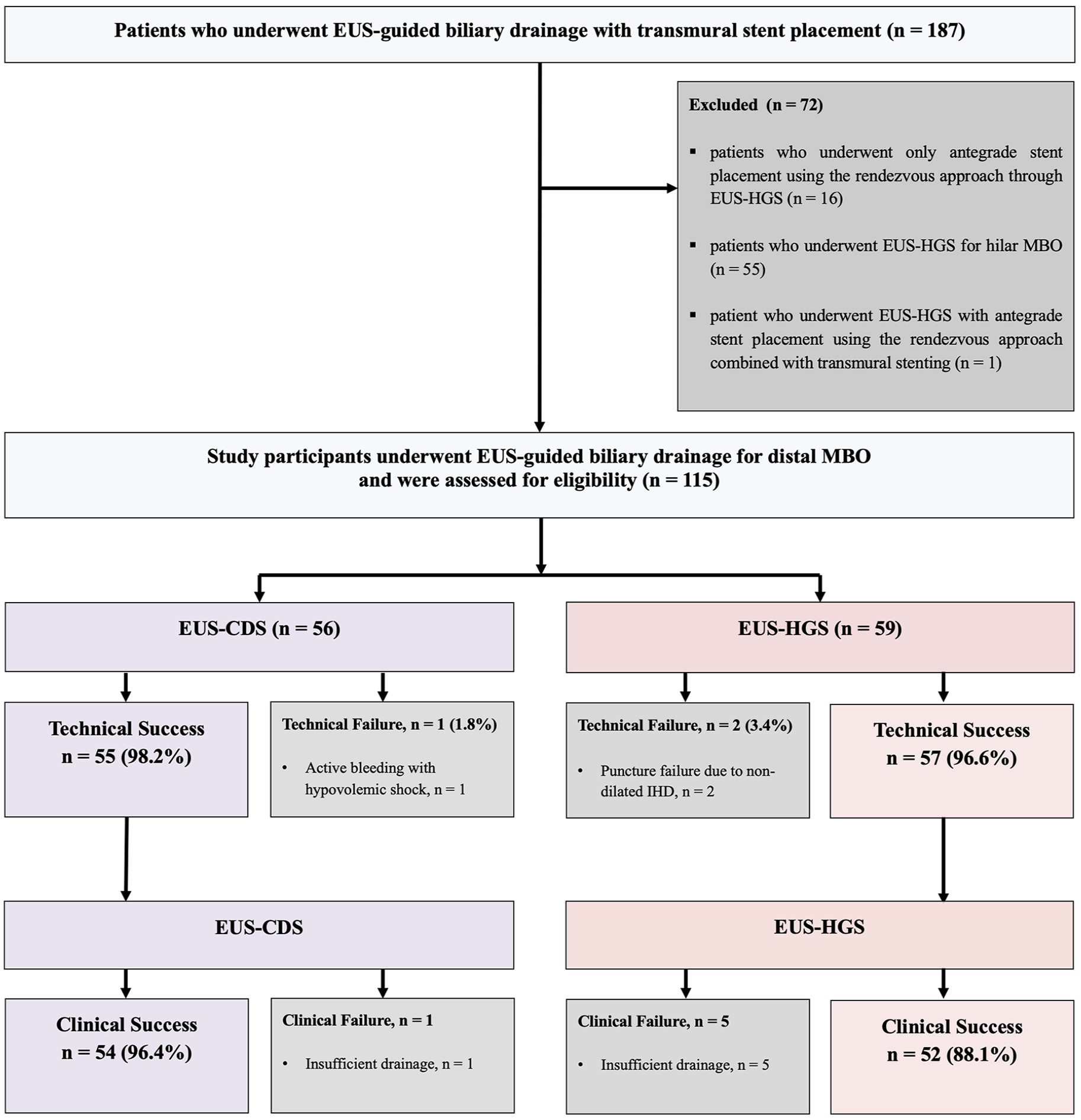
Study flow diagram.
Table 1 presents the baseline characteristics of the enrolled patients according to group. The mean age and proportion of men were 72.5 years and 39.3% in the EUS-CDS group, and 72.0 years and 55.0% in the EUS-HGS group, respectively. In both groups, pancreatic cancer was the most common cause of distal MBO (EUS-CDS, 75.0%; EUS-HGS, 67.8%), while cholangiocarcinoma and gallbladder cancer were more common in EUS-HGS than EUS-CDS group. The proportion of primary EUS-BD without any attempt at ERCP did not differ between the groups (EUS-CDS versus EUS-HGS: 25.0% versus 37.3%, p = 0.223). The most common reason for EUS-CDS was cases of failed ERCP (57.1%), while the most common indication for EUS-HGS was GOO (49.2%). In particular, the proportion of acute cholangitis in the index procedure was significantly higher in the EUS-HGS group than in the EUS-CDS group. However, the proportion of enteral stent placement for GOO, presence of ascites, and systematic treatment after EUS-BD did not differ between groups.
Baseline characteristics of enrolled patients.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; BMI, body mass index; ERCP, endoscopic retrograde cholangiopancreatography; EUS-CDS, endoscopic ultrasound-guided choledochoduodenostomy; EUS-HGS, endoscopic ultrasound-guided hepaticogastrostomy; IQR, interquartile range; SD, standard deviation; WBC, white blood cell.
Endoscopic findings between the groups
Supplemental Table S1 shows the endoscopic findings of EUS-BD in the patients with technical success. SEMS was used in the majority of the EUS-CDS group (90.9%), while it was used in 53.4% of the EUS-HGS group. The diameter of the targeted duct was smaller in the EUS-HGS group than in the EUS-CDS group (EUS-HGS versus EUS-CDS, 6.8 versus 15.7 mm, p < 0.001) while the distance from needle tip to targeted duct was significantly shorter in the EUS-CDS group (1.1 versus 2.3 cm, p < 0.001). The most common target duct in the EUS-HGS group was B3. The balloon catheter was mainly used for tract dilation in the EUS-HGS group, while various devices were used in the EUS-CDS group. In addition, the stent length was significantly longer in the EUS-HGS than in the EUS-CDS group (10.8 versus 5.5 cm, p < 0.001). Also, the total procedure time was shorter in the EUS-CDS than in the EUS-HGS group with a significant difference (15.7 ± 8.3 min versus 19.1 ± 9.1 min, p = 0.045). In the EUS-HGS group, one patient underwent simultaneous antegrade stent placement through the tract for EUS-HGS and transmural stent placement.
Comparison of clinical outcomes and AEs between the groups
Technical success rates were 98.2% (55/56) in the EUS-CDS group and 96.6% (57/59) in the EUS-HGS groups (Table 2). The reason for technical failure in two patients from the EUS-HGS group was non-dilated IHD, while one case in the EUS-CDS group failed due to active bleeding at the puncture site. All patients with technical failures underwent rescue percutaneous transhepatic BD. A total of 107 patients showed clinical success rates of 96.4% and 88.1% in the EUS-CDS and EUS-HGS groups with significant differences (p = 0.200), respectively.
Clinical outcomes and adverse events in patients.

CI, confidence interval; EUS-CDS, endoscopic ultrasound-guided choledochoduodenostomy; EUS-HGS, endoscopic ultrasound-guided hepaticogastrostomy; IQR, interquartile range.
Regarding early AE, one case of stent dysfunction was noted in the EUS-CDS group, whereas two cases were noted in the EUS-HGS group. Three cases of bile peritonitis, one case of bleeding, and seven cases of pneumoperitoneum occurred in the EUS-CDS group. In the EUS-HGS group, four cases of bile peritonitis and three cases of pneumoperitoneum occurred. In terms of late AEs, one case of delayed bleeding was noted in the EUS-CDS group, while two cases of bile peritonitis and two cases of infected biloma due to stent-related occlusion of the other IHD beside the target duct occurred in the EUS-HGS group. Multivariate analysis using a logistic regression model was performed for overall AEs except stent dysfunction (Supplemental Table S2). The only independent risk factor for overall AEs in the population with technical success was the previous drainage procedure [odds ratio and 95% confidence interval (CI) 0.23 (0.03–0.89), p = 0.049].
Overall, 10 cases of stent dysfunction were noted in the EUS-CDS group, and 18 cases were noted in the EUS-HGS group. Among these, complete stent migration occurred in one patient in the EUS-CDS group and three patients in the EUS-HGS group. Furthermore, stent obstruction and sludge/food scraps were more common in the EUS-HGS group than EUS-CDS group (EUS-CDS versus EUS-HGS: 14.3% versus 18.6%, p = 0.794 and 3.6% versus 8.5%, p = 0.529, respectively). The revision for any stent dysfunction was performed in 10 patients (18.2%) in the EUS-CDS group and 20 patients (35.1%) in the EUS-HGS group, without significant difference (p = 0.071). The median number of revisions for stent dysfunction was higher in the EUS-HGS than in the EUS-CDS group [median 1 IQR (1–1) versus median 1 IQR (1–2), p = 0.133] without significant difference. The mean duration of stent patency for EUS-CDS was mean 770.3 days (IQR 475.8–1064.7 days) while that of EUS-HGS was mean 164.9 days (IQR 124.9–205.2 days, p = 0.010).
A multivariate analysis using the Cox proportional hazards model was performed for stent dysfunction (Table 3). The only independent risk factor for stent dysfunction in the population with technical success was systematic treatment after EUS-BD [hazard ratio (HR) and 95% CI: 0.238 (0.066–0.863), p = 0.0290; Figure 2]. In the Kaplan–Meier plot for patients with technical success, the EUS-BD technique did not show a significant difference in stent dysfunction between EUS-CDS and EUS-HGS groups in univariate and multivariate analyses [HR: 2.023, 95% CI: (0.559–7.320), p = 0.2828] (Figure 2).
Cox proportional hazard model for stent dysfunction among patients with technical success.

CI, confidence interval; DPPS, double pigtail plastic stent; EUS-BD, endoscopic ultrasound-guided biliary drainage; EUS-CDS, endoscopic ultrasound-guided choledochoduodenostomy; EUS-HGS, endoscopic ultrasound-guided hepaticogastrostomy; HR, hazard ratio; SEMS, self-expandable metal stent.
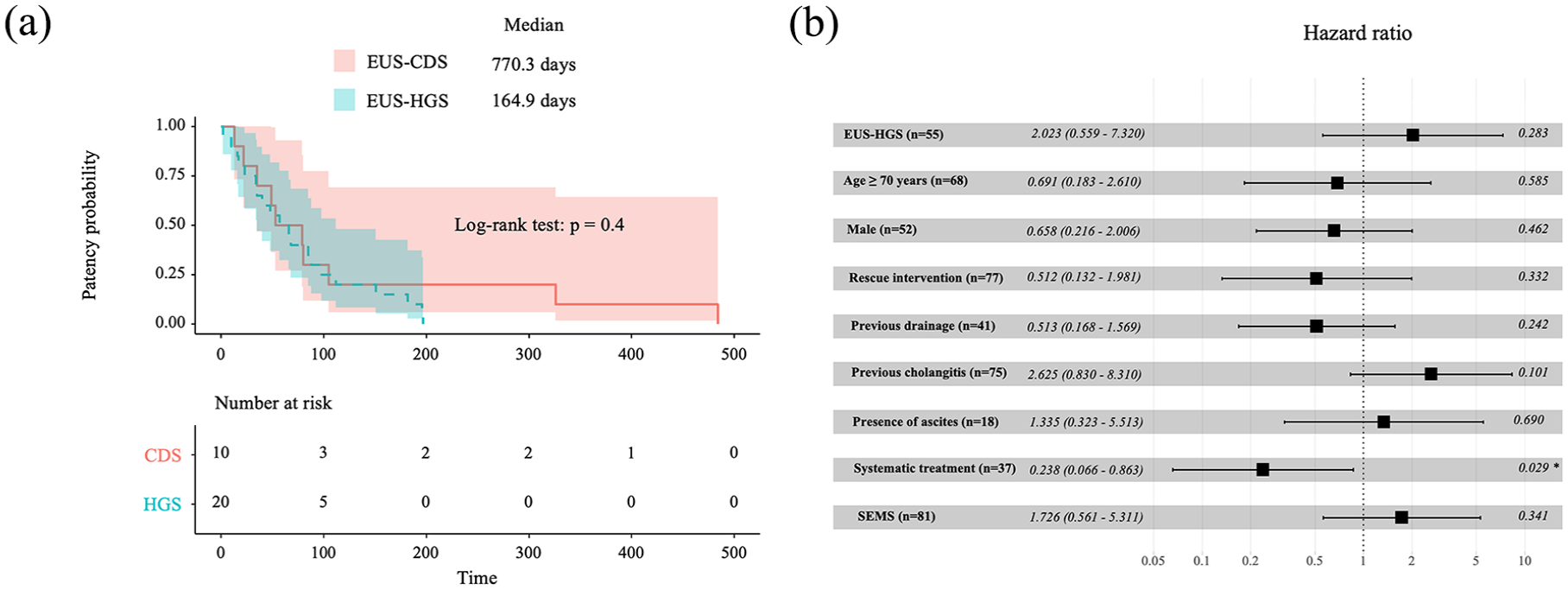
Forest plot according to the Cox proportional hazard model for stent patency in the population with technical success. (a) Kaplan–Meier curve analysis revealed that the EUS-BD technique did not show a significant difference in stent dysfunction between EUS-CDS and EUS-HGS groups in univariate and multivariate analyses [HR 1.427, 95% CI (0.623–3.271), p = 0.4004]. (b) A multivariate analysis using the Cox proportional hazards model for stent dysfunction revealed that the only independent risk factor in the population with technical success was systematic treatment after EUS-BD [HR 0.238, 95% CI (0.066–0.863), p = 0.0290].
More than half of the patients (51.3%, 59/115) died by the end of the study period (Table 2). Furthermore, the median survival times were 165 (IQR: 71.9–258.1) and 162 (IQR: 108.6–215.4) days in the EUS-CDS and EUS-HGS groups, respectively, as shown by per-protocol analysis.
Discussion
The current study reported the long-term outcomes of EUS-BD in patients with distal MBO according to the interventional techniques received during the index procedure. This is the largest cohort study designed to compare EUS-CDS, which can be approached through the transduodenal tract, to EUS-HGS, which can be approached through the transgastric tract. Although both techniques were comparable with equivalent technical success, EUS-CDS showed higher clinical success accompanied by an improvement in cholestasis in the majority of patients. Furthermore, the overall late stent dysfunction occurred at a lower incidence of 17.9% in the EUS-CDS group and 28.8% in the EUS-HGS group; therefore, overall stent patency in EUS-CDS was significantly longer than that of EUS-HGS. These results are contrary to previous results from recent trials12,16,19 and our belief that the patency of EUS-HGS is comparable with that of EUS-CDS.
The median duration of stent patency via EUS-CDS was longer than that via EUS-HGS in distal MBO with significant differences (p = 0.010). There is a plausible explanation that the smaller diameter of the IHD may not allow placement of large-diameter stents in EUS-HGS, whereas a larger diameter of the EHD may allow placement of a larger-diameter stent in EUS-CDS. Differences in the stent diameter may affect stent patency. Although the use of a larger covered SEMS can prevent bile leakage by complete sealing of the iatrogenic bilioenteric tract and provide longer stent patency than plastic stents, it is currently debatable whether plastic or metal stents should be placed during EUS-BD. Khashab et al. 12 reported that EUS-BD with a plastic stent showed a significantly higher incidence of cholangitis, resulting in shorter stent patency. Considering that the SEMS was placed in the majority of patients (91%) in the EUS-CDS group, the differences in stent patency between EUS-CDS and EUS-HGS may be due to the stent type, although in our study Cox proportional hazard model revealed that stent type (SEMS versus plastic stent) was not associated with stent patency. Contrary to ours, da Silva et al. 20 reported that there was no significant difference in terms of effectiveness and safety based on the stent type in EUS-CDS.
Our study showed similar results to those of previous single-center reports12,16,19,21 in which technical success was not statistically different between both the groups (EUS-CDS: 98.2% versus EUS-HGS: 96.6%, p > 0.999). As opposed to the EHD for the puncture in EUS-CDS, the IHD is not always dilated enough for puncture, so it may be theoretically challenging to perform EUS-HGS in patients without bile duct dilation. 22 Along with the difficulty in puncture for non-dilated IHD, guidewire manipulation is more difficult and requires much more time than EUS-CDS. Although the technical success rate of EUS-HGS in the present study was 97%, we encountered two cases of technical failure in the EUS-HGS group as a result of difficulties in guidewire manipulation at non-dilated IHD. Contrary to theoretical belief, the initial prospective study 23 comparing these two approaches revealed that the technical success rate of EUS-HGS (96%) was higher than that of EUS-CDS (91%), although the difference was not statistically significant, suggesting the safety of both drainage routes when performed by experienced operators. Furthermore, they observed a slight trend favoring EUS-HGS (91.6%) over EUS-CDS (77.2%) in terms of clinical success. However, recent meta-analyses demonstrated that technical success was similar between both methods.24–26 Our results were similar to those of other reports. This may be explained by the refinement of the procedure and the increase in procedural competency as experience accumulates. 6
In terms of AEs, conflicting data remains.6,19,21,24,27 In two recent meta-analyses, the pooled AE of EUS-CDS was lower than that of EUS-HGS.24,28 These results can be explained by possible theories that the tract of EUS-CDS is thinner and less resistant and has a close approximation between EHD and duodenum. In addition, the EHD and duodenum are relatively fixed in the retroperitoneum with less influence from respiration and peristalsis while excessive movement of the liver during respiration and peristalsis of the stomach can lead to bidirectional stent migration, resulting in fatal AEs, such as bilomas or bile peritonitis. By contrast, AE was not different between the two approaches in several randomized studies.16,27,29 Similarly, a recent network meta-analysis revealed that there was no discernible difference in the AE rates across various comparisons. 30 In the present study, there were also no significant differences in both early and late AE between the two approaches. Different types of AE were reported between EUS-CDS and EUS-HGS, relating to technical differences in each technique. Immediate stent migration, which could lead to bile peritonitis, occurred in the EUS-HGS group in one case but not in the EUS-CDS group. Among the late AE in the present study, two cases of focal infected biloma, which occurred after EUS-HGS, were of particular interest. A focal infected biloma may be induced by branch duct obstruction by covering it during SEMS placement in IHD. Furthermore, contrary to what was expected, bile peritonitis was noted in two patients in the EUS-HGS group but not in the EUS-CDS group.
In a recent guideline, EUS-CDS has been recommended over EUS-HGS in distal MBO owing to its lower procedural AEs. 31 However, currently, there is no clear consensus on which method is optimal in distal MBO.19,23,25,32 Based on previous reports and our experience, the therapeutic approach will be dependent on the patient’s anatomy, GOO, or the presence of IHD dilatation. In patients with distal MBO, EUS-CDS can be considered prior to EUS-HGS if IHDs are not dilated. By contrast, EUS-HGS should be considered in patients with surgically altered anatomy or dilated IHD. Considering that there is no difference in the technical success and safety between the two techniques, these approaches will be helpful for the standardization of EUS-BD, although the determination of which technique to employ, whether one over the other, the decision to use one technique over the other should be left to the discretion of the endoscopist. 23
Although our study evaluated the long-term outcomes of the two techniques for EUS-BD in a large cohort, it had several limitations that might have influenced our conclusions. As this was not a randomized controlled study, the type of technique used in each patient was based on the presence of GOO, anatomical variations of the bile duct, or even the clinicians’ discretion. For example, only EUS-HGS can be available in cases of failed ERCP because of surgically altered anatomy or GOO, or hilar MBO without communication between right and left IHD. Furthermore, the EUS-BD protocol was not standardized across patients. Regarding the heterogeneity in baseline characteristics, several parameters, including previous drainage, presence of cholangitis, and presence of ascites (with marginal significance), were found to be significantly different between the groups. These differences could potentially lead to misunderstandings, although it is important to note that we have adjusted for all these variables in the Cox proportional hazard model as well as logistic regression analysis. In addition, there is the possibility of incomplete data collection and potential selection bias, although great effort was made to collect accurate data according to the full medical chart review. Moreover, all procedures were performed by highly experienced endosongraphers at highly specialized facilities; therefore, the results from our study cannot be generalized to facilities that are less experienced with EUS-BD. Lastly, it is essential to acknowledge that therapeutic strategies or procedural protocols may lack homogeneity over time due to ongoing technical and instrumental innovation. This stems from the fact that the present study utilized extensive historical data spanning the previous 13 years. Nevertheless, we believe that our retrospective study demonstrates the potential for real-world procedural protocols during EUS-BD.
Contrary to our belief that EUS-CDS and EUS-HGS showed similar clinical outcomes based on previous reports, EUS-CDS presented more favorable outcomes in distal MBO, including improved stent patency. Therefore, for patients eligible for EUS-CDS, it may be a more effective treatment modality in terms of long-term effects. Further prospective randomized studies are required to compare the clinical outcomes between EUS-CDS and HGS.
Supplemental Material
sj-docx-1-tag-10.1177_17562848241239551 – Supplemental material for Comparison of long-term outcomes of endoscopic ultrasound-guided hepaticogastrostomy and choledochoduodenostomy for distal malignant biliary obstruction: a multicenter retrospective study
Supplemental material, sj-docx-1-tag-10.1177_17562848241239551 for Comparison of long-term outcomes of endoscopic ultrasound-guided hepaticogastrostomy and choledochoduodenostomy for distal malignant biliary obstruction: a multicenter retrospective study by Dongwook Oh, Sung Yong Han, Sang Hyub Lee, Seong-Hun Kim, Woo Hyun Paik, Hyung-Ku Chon, Tae Jun Song, Se Woo Park and Jae Hee Cho in Therapeutic Advances in Gastroenterology
Footnotes
Acknowledgements
This was implemented as part of the research projects conducted by the ‘EUS-New Medical Technology Research Group’ in the Korean Pancreatobiliary Association and the ‘EUS Research Group’ in the Korean Society of Gastrointestinal Endoscopy.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
